Abstract
Introduction
Low- and middle-income countries (LMICs) account for 99% of the global neonatal mortality. Limited access to advanced technology, such as bedside patient monitors contributes to disproportionately poor outcomes for critically ill newborns in LMICs. We designed a study to assess the feasibility, performance, and acceptability of a low-cost wireless wearable technology for continuous monitoring of sick newborns in resource-limited settings.
Methods
This was a mixed-methods implementation study conducted between March and April 2021 at two health facilities in Western Kenya. Inclusion criteria for newborns monitored included: age 0 to 28 days, birthweight ≥2.0 kg, low-to-moderate severity of illness at admission and the guardian's willingness to provide informed consent. Medical staff who participated in monitoring the newborns were surveyed about their experience with the technology. We used descriptive statistics to summarize our quantitative findings and qualitative data was coded and analyzed as an iterative process to summarize quotes on user acceptability.
Results
The results of the study demonstrated that adoption of neoGuard was feasible and acceptable in this setting. Medical staff described the technology as safe, user-friendly and efficient, after successfully monitoring 134 newborns. Despite the positive user experience, we did observe some notable technology performance issues such as a high percentage of missing vital signs data.
Conclusion
The results of this study were critical in informing the iterative process of refining and validating an innovative vital signs monitor for patients in resource-limited settings. Further research and development are underway to optimize neoGuard's performance and to examine its clinical impact and cost effectiveness.
Introduction
An estimated 2.4 million neonatal deaths occurred globally in 2020, with the majority (99%) occurring in low- and middle-income countries (LMICs). 1 Nearly 75% of these deaths are attributable to complications due to prematurity, asphyxia, and severe infections 2 —conditions which can be successfully managed through early detection, effective treatment and timely access to high quality care by skilled medical workers.3–5 Newborns in low-resource settings are therefore dying of largely preventable causes, at disproportionately higher rates than the rest of the world.
Over the past three decades, global efforts have achieved tremendous progress in reducing under-5 child mortality. 2 However, momentum in addressing neonatal mortality has continued to lag in developing countries, partly due to logistical 6 and human resource challenges 7 that negatively impact the delivery of specialized medical care to small and sick newborns in LMICs.
Small and sick newborns require timely, high-quality clinical care to subvert a number of risks, including hypothermia, apnea and infection, which they are particularly vulnerable to during the first 28 days of life. Vital signs like respiratory rate (RR), oxygen saturation (SpO2), pulse rate (PR), blood pressure (BP) and skin temperature often provide early warning scores of clinical deterioration and are therefore an essential part of monitoring hospitalized newborns.8,9
In high-income countries, continuous patient monitors are a basic tenet of care for all critically ill patients, including newborns. These equipments allow medical workers to observe patients who are at risk of deteriorating more closely, so they can intervene more quickly to administer effective treatment if and when the need arises. Such early action can help prevent further complications, improve recovery time, reduce treatment costs and ultimately saves lives.10,11
In LMICs like Kenya, which has one of the highest neonatal mortality rates in the world (at 19 deaths per 1000 live births), 1 conventional equipment for continuous vital signs monitoring are not readily accessible. This is often due to the prohibitive costs of purchasing and maintaining complex technology, inconducive infrastructure, and other systemic barriers. Health workers in low-resource settings often employ manual techniques and/or use spot-checking equipment like thermometers and pulse oximeters to capture intermittent vital signs for hospitalized newborns.12,13 Manual methods are not always effective for detecting early clinical deterioration because critically ill newborns are prone to being unstable and often experience rapid changes in their condition. Manual systems are further prone to human errors that can lead to incorrect or missed vital signs measurements. 14 Moreover, the ability of health workers to effectively observe, judge and respond to concerning vital sign changes is largely affected by their experience, training and patient load. 7
As the proportion of deliveries in health facilities in LMICs continues to rise, effective solutions to optimize supportive care for neonates are needed to improve the newborn survival rate. The adoption of innovative multi-parameter vital signs monitoring technologies can play an essential role in alleviating the need for manual operations and ensuring that the few health workers who are available can commit more time to performing other critical tasks as they care for newborns.
Currently, there exists a variety of wearable vital signs monitoring technologies that have been investigated for various populations and settings such as: newborns,15,16 pediatric and adult patients,17–19 pregnant women20,21 and post-surgical patients.11,22 However, there is need for more evidence of feasibility, acceptability and successful implementation of wearable vital signs monitors, 23 most especially in the context of resource-limited settings.
Objectives
The objective of this study was to assess the preliminary feasibility, performance and acceptability of an innovative vital signs monitoring technology known as the ‘neoGuard system’ for hospitalized newborns in resource-limited settings. Specifically, we sought to:
Characterize the population of newborns successfully recruited and monitored with neoGuard at two health facilities. Evaluate the performance of neoGuard technology with regard to safety and its ability to generate vital signs output for four parameters: RR, SpO2, PR and skin temperature. Assess the reliability of neoGuard as an electronic medical records system that stores patient information and vital signs data in one location. Examine insights from neonatal medical staff on the acceptability of using the neoGuard technology for monitoring of their patients.
Outcomes
Newborns monitored were characterized by demographic and clinical characteristics, in addition to morbidity factors and mortality outcomes. Performance measures for the neoGuard technology included incidence of adverse events, average duration of monitoring per patient per day (in hours), and frequency of vital signs output. To assess the reliability of neoGuard as an electronic medical records system we measured the level of agreement between the neoGuard database and patient medical charts. To examine user acceptability, we captured qualitative feedback from medical staff.
Methods
Design and setting
This was a mixed-methods implementation research conducted between March and April 2021 at Kakamega County Referral Hospital and Bungoma County Referral Hospital in Western Kenya. The study was implemented over an 8-week period and the two health care facilities were purposively selected based on high neonatal caseloads, low nurse-to-patient ratios, availability of a neonatal ward in the facility and a need for vital sign monitoring equipment. The selected facilities are located in western Kenya where neonatal morbidity and mortality rates are among the highest in Kenya. 24
Study population and sampling frame
Our study population involved two groups of participants: newborns admitted to the neonatal ward and medical staff who interacted with the technology. Newborns were sampled consecutively on admission to the neonatal ward using the following the criteria: age 0 to 28 days, birthweight ≥2.0 kg, admitted with low-to-moderate severity of illness (based on Bhal et al. severity of illness score for children) 25 and guardian's willingness to provide informed consent for use of the technology on their newborn. Newborns were excluded if they had a condition such as hydrocephalus, head laceration or conditions that required administration of treatment through a scalp vein cannulation since the neoGuard band is designed to be worn around the forehead. Neonatal medical staff (4 nurses and 4 doctors) were selected purposively to participate in using the neoGuard technology for monitoring ill newborns based on the following the criteria: role at the neonatal ward, level of experience in caring for newborns and willingness to participate in the study.
Data collection
Device
The neoGuard technology (Figure 1) is designed to measure and relay 4 vital sign readings (skin temperature, PR, RR and SpO2) to a centralized screen. The system is intended to aid in early detection of deterioration in patients by generating real-time audio and visual alerts to notify health care providers when patient vital signs drop below or exceed the expected normal range. The device consists of the following components: a wireless sensor, reusable wearable bands and a software application that receives, displays and stores data from the sensor devices. The neoGuard device uses reflectance pulse oximeter and temperature sensors to measure vital signs from the patient, and the readings are displayed on a portable tablet screen, which can show vital signs for up to 15 patients simultaneously. The operational range between the tablet and device is up to 30 meters. It is battery powered with a long-lasting rechargeable battery.

The NeoGuard vital signs monitoring system.
Procedure
Medical staff purposively selected to participate in using the neoGuard technology were trained on how to use it 1 week before the study kicked off. Over the 8-week implementation period, the trained medical staff worked with a research assistant to identify newborns that met the criteria for monitoring with neoGuard. The process involved screening of newborns for eligibility, and for those found eligible, guardians were approached and taken through study objectives and procedures. Guardians willing to take part in the study and have their newborns enrolled provided written informed consent before enrollment of their newborn into the study. Newborns were monitored by first adding their demographic and clinical data into the neoMonitor central monitoring application and then having the neoGuard device affixed to their foreheads. Newborns were monitored for the duration they were admitted within the 8-week study period. At the end of the study, medical staff who participated in the monitoring of newborns with the technology completed self-guided surveys about their experience with the neoGuard system and their perceptions of its acceptability and utility on the neonatal ward.
Data analysis
Descriptive statistics were used to characterize the patient population monitored based on demographic and clinical characteristics, morbidity factors and mortality outcomes. Patient data entered into the neoMonitor app was compared against each patient's medical chart to assess the level of agreement. Vital sign measurements were also extracted from the neoMonitor database and analyzed to describe the total duration of monitoring, frequency of device errors such as missing and invalid readings, and the continuity of monitoring in each patient.
The medical staff surveys included a broad array of questions in the form of a 5-point Likert scale, close-ended questions and open-ended questions grouped into four domains: acceptability, perceptions of performance, usability, and practicality. We derived frequencies for the Likert scale and close-ended questions, and used thematic text analysis to evaluate qualitative insights and perspectives obtained from open-ended questions. Medical staff perceptions will be explored more broadly in a separate publication, but some general highlights from our analysis have been included here to encapsulate the user experience.
Results
Patients monitored
All of the parents with eligible newborns that were approached to participate in the study consented to the monitoring of their newborns. Health providers successfully monitored 66 newborns at Kakamega County Referral Hospital and 68 newborns at Bungoma County Referral Hospital over a cumulative period of 61 days. An average of 3 newborns were monitored daily with an average monitoring time of 3.9 and 10.4 hours per patient per day for Kakamega County Referral hospital and Bungoma County Referral hospital, respectively. For the entire study period, there were no reported cases of adverse events and only 1 parent withdrew their consent to have their newborn monitored using the neoGuard device.
Table 1 shows descriptive statistics of the newborns monitored. Out of the total 134 newborns, more than half (55%) were male. The majority had a gestational age of 38 to 40 weeks (66%) and were born with a birth weight of 2.5 to 3.5 kg (61.2%).
Demographic and clinical characteristics of patients.

Table 2 shows morbidity factors, duration of monitoring and mortality outcomes for patients monitored with neoGuard. The most common reasons for admission among the newborns monitored were: birth asphyxia (31.1%), sepsis (22.4%), prematurity (11.7%) and respiratory distress (11.7%). The modal range for length of admission for newborns was 1 to 7 days, a majority of whom were discharged alive (96.3%). There were no device-related adverse events reported during the study.
Morbidity, monitoring duration and mortality outcomes for newborns on neoGuard.

Neomonitor data reliability
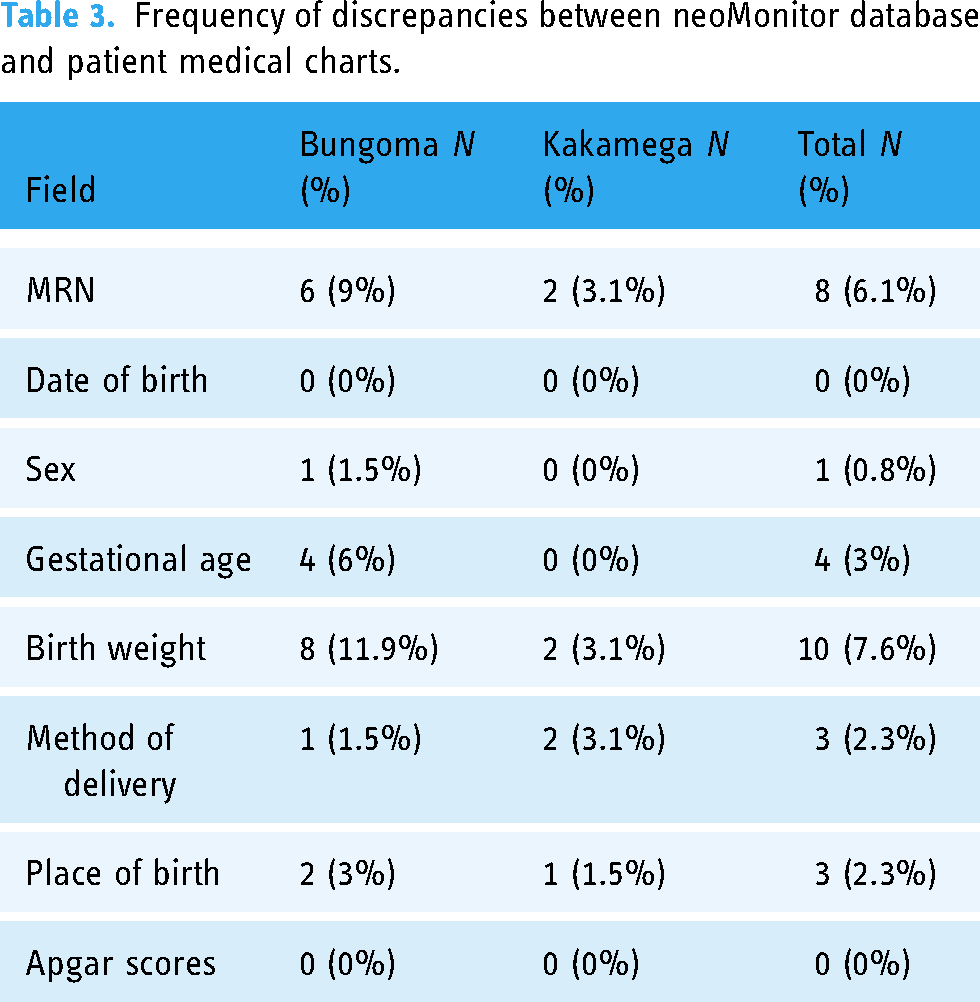
For each subject, we matched electronically captured patient information from the neoGuard database (neoMonitor) with patient information from the manually recorded medical charts used in routine provision of care. To assess the level of agreement between the two sources, we obtained the frequency of discrepancies that were observed across eight main fields: medical record number, date of birth, sex, gestational age, birth weight, method of delivery, place of birth and Apgar scores at 1 minute and 5 minutes. The results observed are summarized in Table 3.
Frequency of discrepancies between neoMonitor database and patient medical charts.
Vital signs data
The neoGuard system generated a total of 5,715,351 readings for PR, 5,069,867 readings for SpO2, 5,704,395 readings for RR and 5,804,947 readings for skin temperature. In the case of skin temperature, all the readings were excluded from analysis due to a manufacturing error that compromised the fidelity of the temperature sensor. While PR, SpO2 and RR measurements are all derived from the same pulse oximetry sensor, it is worth noting that the computations are calculated independently. Due to varying characteristics of the photoplethysmograph (PPG) waveform, the three readings are not always calculated in sync, leading to the observed difference in the total number of readings.
For PR, SpO2 and RR, we broke the readings into three categories: within the normal range, outside the normal range, and missing/blank outputs, as illustrated in Figure 2. Capturing alarm notifications and nurses’ response data was not within the scope of this study, so we were unable to determine if the vital sign readings that were outside the normal range were valid (consistent with a true physiological change or deterioration in clinical status) or invalid (due to motion artifacts or device error, and not representative of a true physiological change). This is a critical performance measure that will be explored in future studies.

Relative frequency of vital signs output by health facility.
Overall, PR had the highest proportion of readings within the expected range, with 73% and 65% of the readings from Kakamega and Bungoma, respectively, within the expected range of 70 to 190 beats per minute (bpm). SpO2 had approximately 31% of the readings within the expected range of 90% to 100%, and RR had <0.08% of the readings within the expected range of 30 to 60 breaths/respirations per minute (rpm). For the vital signs that were outside the expected range, readings were considered to be invalid if they were found to be physiologically incompatible with a clinically stable newborn, that is readings of 70% to 85% for SpO2, <70 bpm for PR and <30 breaths per minute for RR were considered invalid if the patient was stable and had not registered similar readings during intermittent measurement with standard-of-care equipment. All vital sign data sets contained a large percentage of missing/blank outputs (≥26%). Figure 2 shows a breakdown of the vital signs output by health facility.
User acceptability
The overall perception of the users was that the neoGuard product was convenient as it minimized time and improved on their monitoring capacity. Users mentioned they initially had difficulties using the device due to unfamiliarity but with practice and training, they gained the confidence and understanding to use the device comfortably. They also observed value in its alarm system, utility as an electronic medical record and noted its efficiency. “NeoGuard is fast, efficient and has real-time vital information… It saves a lot of time and energy and is positively received by mothers… Hands on training proved effective in getting familiar with the technology.”—User 2 (Medical Officer) at Kakamega County Referral Hospital “It has several uses apart from monitoring vitals. The app itself acts like a digital file… It saves on a lot of paper work which is very tiresome and alarm response is very effective hence saving time.”—User 3 (Nurse) at Kakamega County Referral Hospital.
“Concerns about RR accuracy; nurses need to constantly check that it is appropriately positioned.”—User 5 (Nurse) at Bungoma County Referral Hospital
“No cases reported with itching or inflammation.”—User 4 (Nurse) at Kakamega County Referral Hospital
Users also noted that neoGuard is not appropriate for use on convulsing patients or those with scalp lacerations as it is positioned on the forehead. These are contraindications that have been incorporated in user instructions. “… placement on forehead not appropriate for scalp lacerations and convulsing patients; does not read when patient is sweating or wrinkly; sensitive to patient movement.”—User 5 (Medical Officer) at Bungoma County Referral Hospital
“… patients may have myths concerning device placement.” User 6 (Nurse) at Bungoma County Referral Hospital
Discussion
From our early experience implementing neoGuard at two health facilities in Kenya, we were able to observe its performance in a real-world situation and identify some key challenges in adopting a wireless vital signs monitor worn on the forehead. While enrolment numbers were lower than anticipated (60% of our target), users successfully monitored 134 newborns with a wide range of indications and reported no device-related adverse events. The low recruitment numbers were partly due to contraindications of the product (e.g. patients receiving phototherapy treatment, patients on CPAP with masks that interfere with placement of the neoGuard device), as well as an abundance of caution with enrolling any newborns that received a low SICK score 25 since this was the first time implementing neoGuard in a neonatal unit.
Parents of newborns were generally receptive to the technology and only one parent withdrew consent to have their newborn monitored. Medical staff caring for the newborns also expressed a high level of acceptability of the product, which is consistent with similar studies on the feasibility, acceptability and implementation of other wearable vital signs monitors.20,21,26,27 Users surveyed highlighted neoGuard's simplicity to learn, ease of use and ability to measure multiple parameters concurrently as the most positive features of the technology, and they emphasized the importance of adequate user training for successful adoption. Conversely, respondents cited respiratory rate accuracy and device placement as parameters that could be improved upon to optimize performance and increase user preference for the neoGuard technology. The importance of training and dispelling myths and misconceptions to overcome barriers to acceptability has been highlighted by other studies.21,26
We observed a significant difference in the utilization of the neoGuard product between the two facilities; medical staff at Kakamega County Referral Hospital logged an average of 3.9 hours of monitoring per patient per day, while medical staff at Bungoma County Referral Hospital logged an average monitoring period of 10.4 hours per patient per day. The two facilities received the same number of devices, the same user training for medical staff, and had similar admission rates. There were no clear insights from the survey respondents to explain the large difference in utilization; however, innovators of the neoGuard product have also observed a wide divergence in utilization rates at several other facilities outside this study. The standard guideline for most critically sick newborns is to keep the patient monitor on at all times, unless its removal is necessary to provide other interventions or care. Future efforts to examine and understand utilization trends may require more direct observation and targeted questions on when and how decisions to discontinue or interrupt monitoring of the patient are made by medical staff.
From the vital signs data that was generated, we observed large sums of missing data and vital signs readings that were outside the expected range. Nurses also reported frequent deviations between the neoGuard readings and the readings they observed during intermittent measurement of vital signs with standard-of-care equipment. This is inconsistent with our prior validation studies on neoGuard and may be attributed in part to usability challenges. In preparation for CE mark, the neoGuard technology was tested on 20 healthy volunteers in a controlled setting at an independent lab (Clinimark, Boulder, CO). NeoGuard was found to meet the acceptable accuracy threshold for measurement of PR (±4 bpm), oxygen saturation (±3%) and respiratory rate (±5 breaths per minute) in comparison to gold standard methods. In a subsequent field study performed on adult patients in a hospital recovery ward in Uganda, neoGuard performed comparably well to a conventional bedside monitor with accuracy limits for PR (±6 bpm), oxygen saturation (±4%) and respiratory rate (±7 breaths per minute) meeting our expected threshold for a less controlled field trial. 19 In prior validation studies, we had significantly lower rates of missing data or invalid data (10%–15%).
The large amount of missing data and out of range readings can partly be explained by incorrect positioning of the device on the newborn, not cleaning the device sensor adequately before use, motion artifacts from the patient, or the device sensor coming into contact with too much sweat/perspiration from the patient, as moisture can interfere with its performance. In its design, the neoGuard system is equipped with notifications such as “check device” and “device disconnected” to alert users of some of the forementioned challenges, but perhaps more can be done around training to convey the purpose of these alerts and the necessary corrective actions that users should take. A similar study on technical performance also observed challenges with missing data from three wearable vital sign monitors evaluated on surgical patients. 28
An appealing aspect of the neoGuard system highlighted by users is its ability to store patient information, treatment history and vital signs data in one location. This has potential applications for both clinical practice and research, especially in settings where digitization of patient medical charts is low—making it difficult to investigate correlations between vital sign trends, response to interventions and clinical outcomes of interest. Our study found that the reliability of neoGuard as an electronic medical records system was high, with low rates of discrepancies (2%–8%) between the neoGuard database and paper-based medical charts.
The main limitation of this study was the small sample size of technology users. We recruited and trained only 8 medical staff to use the devices and provide monitoring to the patients. The qualitative feedback we generated on feasibility, performance and user acceptability may therefore not be representative of the frequency of major themes. The small sample of technology users also contributed to the limited hours of monitoring logged by the system. The second limitation was the readiness of the technology. We discovered deficiencies with the temperature sensor well after the study had been initiated and were unable to retrieve any valid skin temperature data from the database as a result of this.
Our study had several strengths as well, including: implementation across two sites to detect facility-related challenges and differences in utilization rates and patient profiles, a mixed-methods approach to generate deeper insights on feasibility, performance and acceptability, rigorous data quality procedures and prospective collection of data before newborns were discharged to prevent missing and erroneous data entry.
Conclusion
Continuous vital signs monitoring has the potential to reduce time to detect deteriorating patients, promote faster treatment initiation, reduce length of hospital-stay, lower readmission rates and mortality rates, and reduce healthcare costs to providers and patients. Owing to recent advances in wearable technologies more suited to low-resource settings are gradually being deployed and evaluated to meet these objectives. While early research indicates promising results in the feasibility, usability and acceptability of many of these devices, more evidence of optimal performance and clinical benefits under real-world use is necessary to scale adoption.
The neoGuard system is still undergoing improvements, an iterative process that has been informed by the findings from this study. The key lessons for innovators from this early stage implementation include: (1) ensuring that all aspects of the monitor are functional before deployment; this calls for more extensive performance testing in the intended settings, (2) developing more specific guidelines on operating conditions and how to use the technology for optimal results, (3) exploring hardware improvements to make the device less sensitive to motion artifacts and more robust to ward conditions that might contribute to sensor errors (e.g. high humidity and ambient temperature could cause the patient to sweat more), and (4) considering application of the device on other body parts like the arm or wrist, which would make it more convenient and acceptable for different groups of patients and users. Additionally, for wearable systems like neoGuard to be effective and reliable for use in life-threatening environments like a NICU, innovators must do more to guarantee accuracy and minimize missing data through continuous validation beyond the marketing approval milestone.
Future studies on neoGuard should include more high acuity patients and explore further validation of accuracy for different patient populations, considering the discrepancies observed between this neonatal study and previous adult studies. Additional research is also needed to further examine user and patient acceptability, barriers to utilization, clinical benefits, and cost effectiveness in real practice.
Footnotes
List of abbreviations
Acknowledgements
The authors dedicate their sincere appreciation to the Kakamega County Referral Hospital and Bungoma County Referral Hospital administration and medical staff for hosting this study.
Availability of data and materials
The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Contributorship
AN, SS, TC, DO, BN and BO collaborated on the conception and design of the study. AN, TC, SS and BAO analyzed the data and interpreted results. AN and BOA drafted the initial manuscript and made subsequent drafts based on co-authors’ comments. All authors were substantively involved in revising the manuscript for intellectual content and scientific integrity. All authors have read and approved this manuscript for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The research described here has been conducted in collaboration with the innovators of the neoGuard™ technology, Ms. Sona Shah and Ms. Teresa Cauvel. Sona Shah and Teresa Cauvel own the patent to the neoGuard™ product and are the founders of Neopenda, PBC. Sona Shah, Teresa Cauvel, Assumpta Nantume and Bertha Akinyi Oketch are employees of Neopenda, PBC, the manufacturer of the neoGuardTM technology. All other authors have no competing interests to declare.
Ethical approval
The study received ethical approval from Maseno University Ethics Review Committee (No. 864/20).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Neopenda is the recipient of a grant from the Efficiency for Access Coalition (EforA) Research and Development Fund (RD3006). The EforA Research and Development Fund is supported by UK aid and the IKEA Foundation.
Guarantor
Assumpta Nantume is the lead author and manuscript's guarantor.
