Abstract
Background
Photoplethysmography (PPG) is an effective non-contact technique for measuring vital signs. This report aims to evaluate whether a non-contact PPG-based mobile application supports an appropriate level of accuracy in monitoring SpO2, HR, and BP, compared against clinically approved medical devices.
Methods
Adult volunteers (≥18 years, both male and female) were consecutively enrolled from September to November 2024. Remote photoplethysmography (rPPG) for data collection involves recording subjects’ faces using the front camera of mobile devices for ∼1.5 minutes. Simultaneously, vital signs will be continuously collected for about 1.5 minutes using the reference devices alongside data collected via the comestai application.
Results
A total of 562 participants (279 F/283 M) were enrolled. The app demonstrated excellent accuracy for HR (MAE 2.96, MAPE 4.14, and accuracy 99.1%) and SpO2 (MAE 2.10, MAPE 2.12, and accuracy 93.4%; no participants with SpO2 < 95% were not present). Results for systolic blood pressure (SBP) (MAE 14.24, MAPE 11.33, and accuracy 61.3%) and diastolic blood pressure (DBP) (MAE 9.83, MAPE 11.66, and accuracy 56.0%) were more moderate. The ROC curve analysis confirmed the app's robust detection capabilities for HR (AUC 0.976) and SpO2 (AUC 0.947), while showing fair performance for SBP (AUC 0.668) and weaker results for DBP (AUC 0.578). Bland-Altman plots revealed mean differences of −0.83 for HR, 1.84 for SpO2, −4.49 for SBP, and 2.54 for DBP compared to conventional methods.
Conclusions
The non-contact PPG-based mobile app holds promise as an accessible, user-friendly tool for wellness monitoring. Solutions like comestai.app present both new opportunities and challenges in the wellness sector, emphasizing the need for continued research in this field.
Introduction
The assessment of wellness involves a holistic evaluation of an individual's physical, mental, and emotional health. 1 This approach considers not only the absence of disease but also the factors that promote positive health and life satisfaction. Implementing standardized wellness measurement tools can enhance preventive care, overall patient management, and the prediction of long-term outcomes.2,3
The measurement of vital signs, such as heart rate (HR), blood pressure (BP), and oxygen saturation (SpO2), is crucial for assessing both wellness and health. These parameters offer key insights into physiological status and allow early detection of potential issues.
Traditional methods of measuring these signs through physical examination can be challenging, and the pandemic has accelerated the shift toward telehealth and remote monitoring. In the past few years, there has been growing interest in self-monitoring tools, which, thanks to advances in technology, have become increasingly user-friendly and widely accessible.4,5
Mobile health (mHealth) applications have proven to be a valuable resource for supporting self-monitoring and facilitating healthcare interventions.6,7 In particular, the development of non-contact methods for monitoring vital signs presents considerable promise for improving patient care in various environments.8,9
In this context, photoplethysmography (PPG) is an effective non-contact technique for measuring vital signs, utilizing a red, green, and blue (RGB) camera, and has gained attention in recent studies. 8
This method involves a transducer that emits infrared light through an LED into the skin's dermal layer. The light that is reflected back is detected by a photodetector and visualized as a line tracing. The intensity of this reflected light varies in response to changes in the volume of red blood cells within the dermal capillaries. 9 PPG captures facial color fluctuations that reflect changes in blood volume, linking these changes in red blood cell concentration, influenced by blood circulation, to the intensity of reflected light in each region. By analyzing shifts in the average color signals, the signal-to-noise ratio (SNR) from the camera can be improved.
This study aims to evaluate whether a non-contact PPG-based mobile application supports an appropriate level of accuracy in monitoring SpO2, HR, and BP, compared against clinically approved medical devices. Such innovations in mHealth could play a pivotal role in empowering individuals to monitor and maintain their wellness more effectively.
Materials and methods
Subjects
Adult volunteers (≥18 years, both male and female) were enrolled in an observational study and recruited from ASST-Fatebenefratelli Sacco-Buzzi Hospital (Milan, Italy), IRCCS MultiMedica (Milan, Italy), and IRCCS Istituto Auxologico Italiano (Milan, Italy). They were referred to outpatient clinics for obesity, diabetes, and/or hypertension between September and November 2024. All participants provided written consent after being fully informed about the study.
Participants will be excluded if they have impaired circulation, injuries, physical abnormalities, or tattoos in regions critical to the study, as these may interfere with the assessment. Severe allergies to materials used in medical sensors will also result in exclusion.
For all participants, gender, age, weight, height, and BMI (calculated as weight in kg/height in m2) were collected.
The comestai.app application was downloaded to a smartphone.
The accuracy of the app's measurements, compared to the reference devices, was assessed for HR, SpO2, and systolic and diastolic BP. Data from the investigational device were gathered by qualified personnel.
Vital signs data, both from the application and the reference devices, was securely stored on a cloud-based system. To prevent any data from being uploaded to the comestai cloud, the applications were only activated when the phone was in offline mode. As a result, the company did not have access to any data collected during the testing process.
Data collected using the reference devices were stored without any identifiable subject information and were only identifiable using the subject identification number provided during screening.
The study was approved by the Ethics Committee (Comitato Etico Territoriale Lombardia 1, approval code: CET 98-2024). The study was conducted in accordance with the Declaration of Helsinki guidelines.
Vital signs measurements
The participants were seated and allowed to stabilize for 2 minutes before measurements were taken. Subjects were positioned in front of the mobile phone, and the application automatically began measuring physiological parameters. Simultaneously, vital signs data (HR, SpO2, and BP) were collected continuously for ∼1.5 minutes using reference devices alongside data collected via the comestai application. Ambient lighting was standardized during remote-PPG (rPPG) measurement.
Blood pressure was measured twice using the reference devices (if the systolic value differed by ±10 mmHg and/or the diastolic value by ± 5 mmHg between the two measurements, a third measurement was taken). Measurements were taken with a one-minute interval using the Withings BPM Connect device.
SpO2 was measured using a Masimo MightySat Rx Finger Pulse Oximeter.
No medical decisions have been made based on the data obtained from the comestai application.
Application
Comestai.app is a mobile app that leverages an advanced integration of artificial intelligence (AI) technologies and signal processing techniques, along with a unique mathematical backend, to analyze the skin on the upper cheek using the smartphone's front camera. The app captures video frames (Figure 1) and uses AI-driven signal processing to analyze data through PPG techniques. This approach enables real-time tracking of several physiological metrics, such as HR, SpO2, and BP. More specifically the pipeline is as follows:
Signal acquisition: Vital signs were extracted using the comestai.app software development kit (SDK), which leverages a smartphone's front-facing camera. rPPG: Comestai.app utilizes a proprietary rPPG, which detects subtle color changes in the facial skin caused by blood flow. These variations are translated into raw pulse wave signals. Signal processing: The rPPG signal is then processed using filtering techniques to remove noise and motion artifacts. This includes bandpass filtering, normalization, and signal quality assessment. AI-based feature extraction: The processed signal is analyzed using a combination of AI (supervised machine learning, deep learning, computer vision) and signal analysis technologies to extract vital signs. Output validation: The application provides real-time feedback on signal quality and outputs results only when the confidence level of the prediction exceeds a predefined threshold.

Video frame captured by comestai.app.
Unlike other applications, the comestai.app uses ambient light RGB signals from real-time facial video without relying on cloud services or internet connectivity. Version 5.6.3 of the app and an iOS phone were used in this study. It is worth noting that version 5.6.3 is an initial version for the target population.
Statistical analysis
Numerical data are presented as minimum and maximum values, means, standard deviations (SD), medians, and quartiles, while categorical data are shown as absolute and relative frequencies.
The app's measurement accuracy was evaluated in comparison to reference devices and standard tests across all parameters. Confidence intervals were computed using a non-parametric method.
The effectiveness of the proposed method was assessed using several quality metrics, including mean error (ME), mean absolute error (MAE), root mean square error (RMSE), and mean absolute percentage error (MAPE).
To evaluate the application's accuracy in measuring vital parameters, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), accuracy, and likelihood ratios (LRs) were calculated. All calculations included the 95% confidence interval (CI).
For the validation of HR, SpO2, and BP measurements, the normality of the vital signs was considered, and the following reference thresholds were adopted: HR ≤ 101 bpm; SpO2 ≥ 95%; SBP ≤ 130 mmHg; DBP ≤ 85 mmHg, in accordance with recommendations from the American Heart Association, 10 the World Health Organization, 11 and the European Society of Cardiology. 12
The correlation between the app's measurements and those obtained from reference devices and standard tests was determined using the Pearson correlation coefficient. The results were graphically represented using regression plots, including the line of best fit and the coefficient of determination (
Agreement between the two sets of quantitative measurements (app vs. reference devices) was further evaluated using Bland-Altman plots.
All statistical analyses were performed using Microsoft Excel, including specific extensions (e.g. Solver add-in) and the Data Analysis ToolPak. The accuracy of the results was confirmed through manual double-checking and partial verification using R (version 4.3.3), with full concordance observed between outputs.
Results
A total of 562 participants (279F/283M) were enrolled. Participant characteristics are reported in Table 1.
Participant's characteristics.

BMI: body mass index.
Table 2 summarizes the descriptive data and the performance of the values recorded using the app compared to standard medical devices. The app achieved an MAE and MAPE of 2.96 and 4.14 for HR, 2.10 and 2.12 for SpO2, 14.24 and 11.33 for SBP, and 9.83 and 11.66 for DBP, respectively.
Descriptive and the effectiveness of values recorded via the mobile application (App) and reference medical devices.

ME: mean error; MAE: mean absolute error; MAPE: mean absolute percentage error; Q: quartil, RMSE: root mean square error; percentage error; SD: standard deviation.
In Table 3, sensitivity, specificity, PPV, NPV, positive likelihood ratios (+ LR), negative likelihood ratios (− LR), and accuracy were calculated to compare measurements obtained using the mobile application and the conventional method are reported. The results demonstrated high sensitivity (100%), specificity (99.1%), and accuracy (99.1%) for HR. For HR, the medical device correctly identified all “true positives” and “true negatives,” resulting in a sensitivity and NPV of 1 and a −LR of 0. The high sensitivity and specificity also contributed to a very high + LR for HR.
Accuracy of vital parameters using the mobile application and the conventional method.

The clinical reference values assumed as thresholds have been indicated in brackets.
SpO2: pulse oxygen saturation; SBP: systolic blood pressure; DBP: diastolic blood pressure; PPV: positive predictive value; NPV: negative predictive value; +LR: positive likelihood ratios; −LR: negative likelihood ratios.
For SpO2, the app exhibited high specificity (93.6%) and accuracy (93.4%). However, no patients recorded a SpO2 value below 95%, leading to sensitivity, PPV, and + LR being zero.
BP measurements showed more limited performance. For SBP, sensitivity and specificity were 63.5% and 60.0%, respectively, with an overall accuracy of 61.3%. For DBP, sensitivity was 37.7%, while specificity reached 67.4%, resulting in an overall accuracy of 56.0%.
The ROC curve was generated to evaluate the performance of measurements obtained via the mobile application in detecting patients with altered vital signs as compared to measurements taken using the conventional method. The area under the curve (AUC) was 0.976 (95% CI: 0.960–0.991) for HR, 0.947 (95% CI: 0.924–0.970) for SpO2, 0.668 (95% CI: 0.621–0.715) for SBP, and 0.578 (95% CI: 0.529–0.626) for DBP.
To examine the reproducibility of vital parameters, the Bland-Altman plots were constructed, Figure 2 (Panels a to c). Based on the graphs, it was noted that for all four parameters analyzed the majority of the statistical units fell within the limits of the 95% CI. The averages of the differences between the measurements of the two methods (device and app) are −0.83 for HR, 1.84 for SpO2, −4.49 for SBP, and 2.54 for DBP. It could also be observed that the dots clustered near the mean line and became thinner as the values on the ordinate axis grew: all the aspects highlighted above demonstrated good agreement between the two sets of measurements carried out.
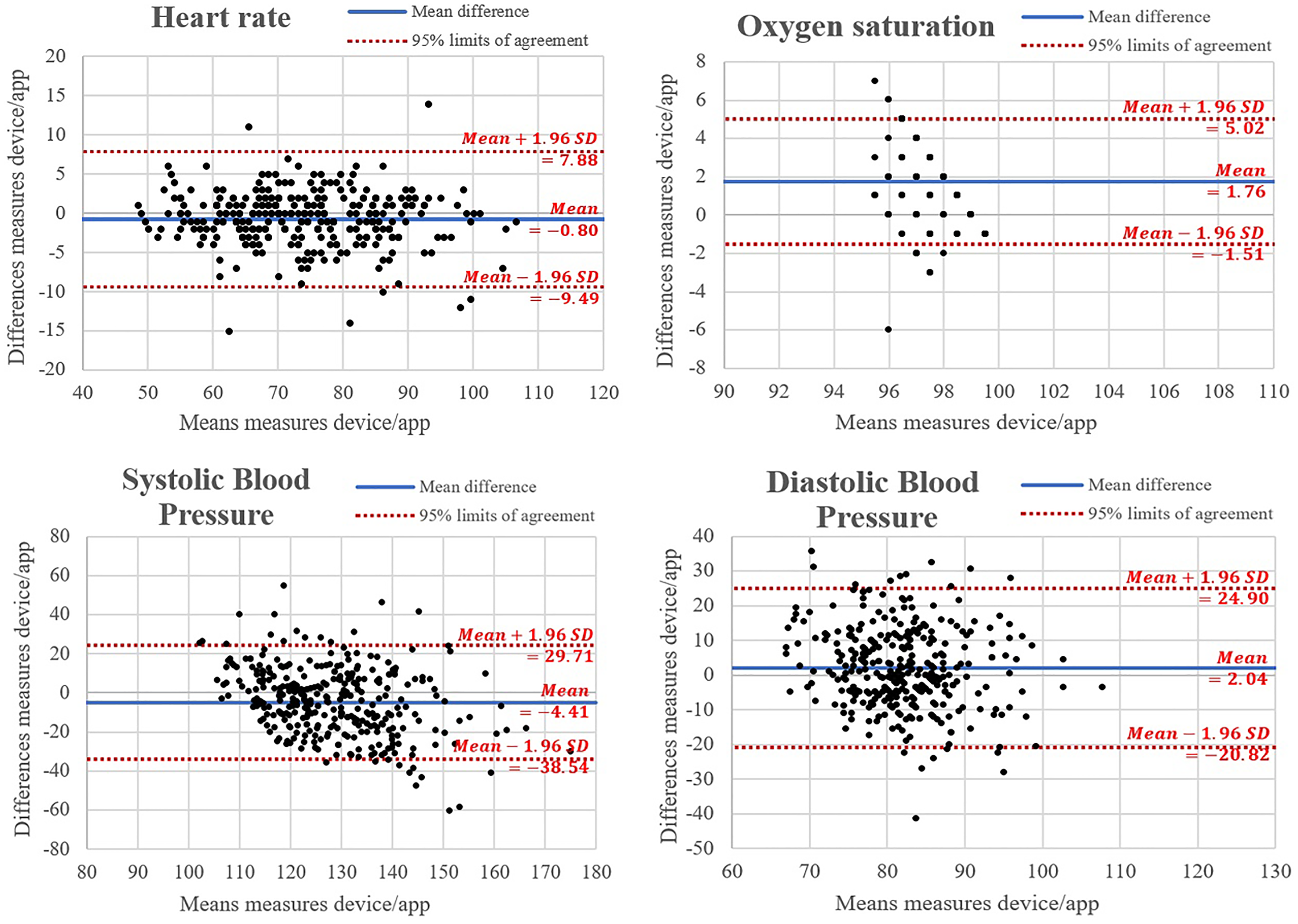
Bland-Altman plots of systolic and diastolic pressure (Panels a and b), heart rate (Panel c), and oxygen saturation (Panel c), measures made using the mobile application and the conventional method. For each one, the horizontal line at the center of the mean difference between the two methods is plotted and the other two horizontal lines represent the limits of agreement, calculated as the
Discussion
We reported on the accuracy of the comestai.app mobile application in monitoring vital signs such as HR, SpO2, and BP compared to standard reference devices. The results showed agreement between the two sets of measurements, with high accuracy in measuring HR and SpO2 and more limited performance in measuring BP. These preliminary results may support the app's potential as a promising and user-friendly tool for self-monitoring wellness.
As reported by the World Health Organization, there is a strong connection between personal wellness and overall health. 13 Both quality of life and individual well-being are universally acknowledged as essential components in preventing disease. The role of each individual is evolving, transitioning from a passive recipient of care to an active participant in managing their own health. Thus, self-monitoring has become the most commonly used strategy in wellness-promoting interventions. 14
In this context, emerging technologies and systems designed to support self-assessment are poised to play a pivotal role in personal health management. MHealth apps, in particular, stand out for their accessibility, affordability, and effectiveness in promoting wellness. 15 Moreover, recent advancements in non-contact methods for monitoring vital signs further highlight the transformative potential of these technologies in enhancing health and well-being. Among these methods, many researchers have proposed the use of PPG signals to measure HR, SpO2, and BP, offering a foundation for further development of non-invasive monitoring tools. 8
Particularly through rPPG, HR is currently the most accurately measured parameter, with an MAE between 0.23 and 5 bpm, depending on the dataset and methodology used, and exceptional values falling under 1 bpm in ideal conditions or with customized datasets.16–18 As for oxygen saturation, the results are encouraging, with error margins around 2% (1.64%–4.5%), but accuracy varies widely due to factors like skin type, ambient lighting, and signal quality, suggesting potential reliability even in non-critical clinical settings.19–21
On the other hand, estimating blood pressure using contactless methods presents clear limitations. Although some studies report errors below 7 mmHg,22–24 such values still fall short of international standards for high-precision medical devices, making the technology suitable only for preliminary screening and not for reliable diagnosis. This issue is compounded by the lack of well-annotated public datasets, which limits model training and generalization.
A summary of the literature data on the measurement of vital parameters using rPPG is provided in Table 4.
A summary of the literature data on the measurement of vital parameters using remote photoplethysmography.

MAE: mean absolute error; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Comestai.app has developed an innovative non-contact PPG-based mobile wellness application designed to measure multiple vital parameters through a quick real-time analysis of the patient's face using the smartphone's front camera. This study compared the comestai.app app to traditional in-person methods for monitoring HR, SpO2, and BP, showing that the app provides reliable measurements for detecting changes in vital signs. The findings highlight its potential as an accessible and user-friendly tool for vital sign monitoring.
Monitoring HR is essential for determining the heart's efficiency and identifying potential health concerns. By measuring HR regularly, valuable insights into cardiovascular health can be gained.
In line with previous literature data on non-contact vital sign monitoring,25–33 we recorded high performance in HR measurement, with an MAE of < 3 bpm and a high correlation between the two measurement methods, demonstrating high accuracy, sensitivity, and specificity. Certified or tested contact-based PPG typically achieves an accuracy close to 5 bpm using common datasets, 8 so our results can be considered highly satisfactory. Thus, as reported by Saikevičius et al., 8 we confirm non-contact PPG as a useful method for measuring HR to monitor personal well-being. The lack of a need for additional tools or wearable devices to record HR makes the application an innovative, cost-effective, and widely acceptable solution.
SpO2 monitoring provides valuable insights into an individual's respiratory and cardiovascular health, which are key aspects of overall well-being. For SpO2 monitoring, the comestai.app application showed good performance. SpO2 was measured with an MAE of around 2% and an accuracy exceeding 90%. Our accuracy results support data reported by Paladino et al., 33 confirming the usefulness of non-contact PPG. PPG in SpO2 monitoring, 8 with promising prospects in the context of wellness. However, no patients recorded a SpO2 value below 95%, influencing sensitivity results; further data are necessary to evaluate the performance in patients with altered SpO2 levels, as even small differences can significantly impact health. 33
Additionally, we observed a lower Pearson correlation coefficient between the SpO2 values measured by the app and those obtained through standard instruments. Although a low Pearson correlation does not necessarily indicate poor clinical reliability, the improvement of this parameter remains a crucial aspect. Casalino et al. 34 reported a significant enhancement in the Pearson correlation for SpO2 using the rPPG-new method, which is largely attributed to a more robust signal processing pipeline and the integration of multiple facial ROIs, helping to mitigate the impact of lighting variations and head movements. These aspects will be carefully reconsidered in future developments in order to further enhance the performance and reliability of our device in estimating oxygen saturation.
For BP findings, we observed more limited accuracy, with an MAE <15 mmHg for systolic BP and <10 mmHg for diastolic BP, along with limited sensitivity and specificity. Literature data on non-contact BP monitoring have shown performance results similar to ours 35 with some studies reporting better performance.27,36,37 The reported data are adequate for a device classified as having low accuracy according to the three-level system for medical devices, with more than a 50% chance of failure. Specifically, according to ISO 81060-2:2018 and AAMI/ANSI/ISO standards, clinically acceptable BP measurement devices should demonstrate a mean error of ≤ 5 mmHg and a standard deviation of ≤ 8 mmHg when compared to reference standards. Limited accuracy is acceptable and expected for a wellness application. As a tool designed to support the monitoring of user well-being, it is not intended for diagnostic or therapeutic purposes.
It is useful to remember that each year, high BP is responsible for ∼12.8% of deaths worldwide and 3.7% of permanent disabilities. 38 Detecting high BP early is critical, as it often shows no symptoms until it progresses to serious conditions such as heart disease, heart failure, or even fatal outcomes. Wellness devices can help identify trends or changes in parameters over time, even if the data lacks pinpoint accuracy. However, improving the accuracy of these tools remains essential, even when they are used exclusively in a wellness context. To address this, it is crucial to facilitate the collection of large datasets, including individuals with abnormal BP values, to enhance algorithm performance in detecting blood pressure variations. This approach is expected to accelerate advancements in measuring vital signs with these innovative instruments.
Given the widespread availability of smartphones, they offer an accessible platform to support better wellness management. Proposing a self-monitoring app on smartphones allows reaching the population in a widespread manner, with equal distribution and minimal barriers.15,22 Additionally, the advantages of the mobile application include the lack of need for additional devices to monitor multiple physiological parameters and the ability to provide immediate readings. The availability of low-cost and intelligent devices will open up new frontiers in wellness promotion.15,34
It is important to acknowledge the limitations of this study. Although subjects with comorbidities were included in the protocol to obtain pathological values, the study was conducted in controlled measurement environments. The use of mobile devices in real-world settings may introduce challenges not present under these controlled conditions, primarily due to increased parameter variability. Additionally, variability in ambient lighting and user movement can introduce noise and artifacts that reduce measurement accuracy. User compliance and ease of use are also critical factors affecting data quality and the overall user experience. These practical considerations underscore the need for robust signal processing algorithms and adaptive calibration methods to ensure reliable performance outside of laboratory conditions. Future work should prioritize extensive field testing and usability studies to address these challenges and verify the application's effectiveness across a variety of everyday scenarios. Furthermore, subgroup analyses based on BMI, age, or comorbidity status were not performed, which limits the generalizability of the findings across diverse population groups. Further research should aim to overcome these limitations by conducting larger-scale studies across varied settings and incorporating stratified analyses to better assess performance variability in clinically relevant subpopulations. The ROC curve analysis was included in this study as a complementary tool to evaluate the overall agreement between the app and the standard device in detecting variations in oxygen saturation. While the AUC can be influenced by dataset imbalance, such as a low prevalence of non-pathological cases, it remains a valuable indicator of the model's overall discriminative capacity. The ROC curve is especially useful in the early phases of tool development and validation, as it helps visualize the system's ability to correctly rank subjects by saturation levels. Although it should be supported by other clinical metrics for a comprehensive assessment, the AUC offers clear descriptive and comparative value at this stage. Finally, it is worth noting that version 5.6.3 represents a pilot release specifically designed for the target population. As such, it serves as an initial implementation. Updated versions are expected to incorporate algorithmic refinements based on user feedback, performance evaluations, and ongoing validation studies.
Conclusions
The non-contact, PPG-based mobile application shows considerable potential as an accessible and user-friendly tool for wellness monitoring. As mobile technologies continue to integrate more deeply into healthcare, individuals are increasingly expected to rely on such devices to monitor their well-being.
Solutions like comestai.app introduce both opportunities and challenges within the wellness domain, highlighting the importance of ongoing research to validate and enhance their effectiveness.
Footnotes
Acknowledgements
The authors thank Come Stai S.p.A. for providing the Comestai app and all materials used for the protocol free of charge for the entire duration of the project. The project was performed within 1) the MUSA—Multilayered Urban Sustainability Action—project, funded by the European Union—NextGenerationEU, under the National Recovery and Resilience Plan (NRRP) Mission 4 Component 2 Investment Line 1.5: Strengthening of research structures and creation of R&D “innovation ecosystems,” set up of “territorial leaders in R&D”; 2) National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.3—Call for tender No. 341 of 15 March 2022 of Italian Ministry of University and Research funded by the European Union—NextGenerationEU. Project code PE00000003, Concession Decree No. 1550 of 11 October 2022 adopted by the Italian Ministry of University and Research, CUP F13C22001210007, Project title “ON Foods—Research and innovation network on food and nutrition Sustainability, Safety and Security—Working ON Foods, supporting pubblication cost.
Ethical considerations
The study was approved by the Ethics Committee (Comitato Etico Territoriale Lombardia 1, approval code: CET 98-2024). The study was conducted in accordance with the Declaration of Helsinki guidelines.
Consent to participate
Respondents gave written consent for review and signature before starting the protocol.
Author contributions
GZ, POA, VC, and SDP: conceptualization, data curation, methodology, supervision, writing–original draft, and writing–review and editing. LL, EC, DB, SB, DM, MC, FL, AF, and MX: methodology, investigation, and writing–original draft. PF, SB, and CCFB: methodology, writing–original draft, and writing–review and editing.
Funding
The author(s) received no financial support for the research, authorship of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: POA and SDP are co-founders of Come Stai S.p.A.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. All data have been anonymized to protect participant privacy and comply with applicable ethical standards.
