Abstract
Objectives
Effective physician–patient communication is essential for improving health literacy and achieving optimal surgical outcomes. However, patients often struggle to interpret conventional two-dimensional (2D) imaging during preoperative counseling for thyroid surgery. This study assessed the feasibility of a ray-traced three-dimensional (3D) virtual reality (VR) educational model to enhance patient health literacy and satisfaction.
Methods
A patient-specific ray-traced 3D VR educational model was developed using deep learning–based multi-organ segmentation of CT scans and rendered on flat monitors commonly available in outpatient clinics. A structured two-round counseling protocol was conducted: in the first round, conventional 2D CT images were used, and in the second round, the 3D VR model was presented. After counseling, patients completed a survey assessing comprehension and satisfaction by comparing the 3D VR educational model with 2D CT images.
Results
All patients reported improved understanding of their condition after viewing the 3D VR educational model. Most participants (88.9%) expressed high satisfaction and willingness to use the model in future consultations.
Conclusions
Ray-traced 3D VR educational models are feasible for improving patient health literacy during preoperative counseling. This digital technology–based approach offers a practical and scalable tool to support more informed, meaningful discussions and enhance patient understanding.
Keywords
Introduction
Health literacy is a core competency that enables patients to understand medical information and promotes behavioral changes that improve health outcomes. 1 Good health literacy supports informed and shared decision-making (SDM), resulting in better treatment adherence, higher satisfaction, and stronger physician-patient rapport.2,3 However, complex medical information within a limited consultation time often hinders patient comprehension and contributes to low health literacy.2,4,5
For surgical patients, understanding intricate anatomical structures, accurately grasping the extent of disease, comparing treatment options, and anticipating possible complications and postoperative care—all within a brief consultation—can be particularly challenging. Low health literacy can lead to poor treatment adherence, delayed recovery, increased complications, and higher risk of readmission.6,7
In surgical settings, anatomical information is essential for patients to understand their condition and the planned procedure. However, interpreting two-dimensional imaging such as ultrasound, CT or MRI can be challenging without medical background knowledge. To address this issue, previous studies utilized three-dimensional (3D) anatomical models to enhance patient education and demonstrated their benefits for health literacy.8,9 Despite these advantages, high production costs, time constraints and lack of visual fidelity have limited their clinical application.7–10
Recent advances in virtual reality (VR) have been applied in various medical fields, enabling immersive educational experiences that allow patients to visualize and interact with 3D anatomical structures.11,12 Compared to 3D printing, VR is more cost-effective and can employ ray-tracing techniques to generate highly realistic visualizations.
In thyroid surgery, CT scans are essential for assessing disease status, including lesion location, degree of invasion, and lymph node metastasis.13,14 Given the importance of accurately conveying information derived from CT imaging, effective communication of anatomical details to patients is essential to improve health literacy. Previous studies have reported that utilizing 3D models can improve patients’ understanding during preoperative education.8,9 However, to date, no studies have applied VR–based models directly in patient consultations for thyroid surgery.
Therefore, in this study, we developed and implemented a patient-specific VR educational platform. Our approach combined a deep learning–based segmentation algorithm to automate the generation of 3D models with ray tracing techniques to achieve photorealistic visualization. Furthermore, the VR system provided interactive functions such as model zooming, layer selection, rotation, and boundary highlighting of anatomical structures. We aimed to evaluate the feasibility of VR-based 3D educational models to improve patients’ health literacy and satisfaction during preoperative counseling.
Materials and methods
Study design
This feasibility study was designed as a single-arm prospective study conducted at a tertiary hospital outpatient clinic between 2022 and 2023, following approval from the Institutional Review Board of Hospital A (IRB No. 2022-09-015-003). The study enrolled adult patients (≥ 18 years) who were scheduled for thyroidectomy and had undergone contrast-enhanced neck CT imaging. Patients with communication difficulties due to language impairments or intellectual disabilities that could hinder understanding of the counseling process or the expression of comprehension were excluded. All participants provided written informed consent, and their CT data anonymized as de-identified DICOM files.
Of the 11 participants who initially consented, 9 completed the study; one foreign patient was excluded due to participation restrictions and another patient withdrew consent (Figure 1). The characteristics of the participants are presented in Supplemental Table 1. The study was reported in accordance with the CONSORT guidelines. 15

Flow diagram of this study. The red-outlined box represents the feasibility survey study, and the blue-outlined box shows the development process of a deep learning–based automatic multi-organ segmentation model using thyroid CT scans.
Creation of ray-traced 3D VR educational models
To create patient-oriented 3D VR educational models, we developed a deep learning–based automatic multi-organ segmentation model using MEDIP PRO v2.4.0.0 (MEDICAL IP, Seoul, Korea), built upon the 3D U-Net framework (Appendix 1 and Figure 1). The model was designed to automatically segment anatomical structures from CT scans, including the thyroid gland, skin, muscle, thyroid cartilage, cricoid cartilage, trachea, bone, internal jugular veins, common carotid arteries, and anterior jugular veins. All segmented structures, except for the skin and anterior jugular vein, achieved Dice similarity coefficients above 95%, indicating clinically acceptable performance (Supplemental Table 2). 16
After patient-specific automatic multi-organ segmentation was completed, ray-traced 3D VR educational models were generated (Figure 2 and Video 1). The initial segmentation results were manually refined to ensure accuracy. The refined segmentation masks were then converted into 3D mesh files and smoothed to enhance visual clarity. Finally, the 3D meshes were exported in Universal Scene Description (USD) format.
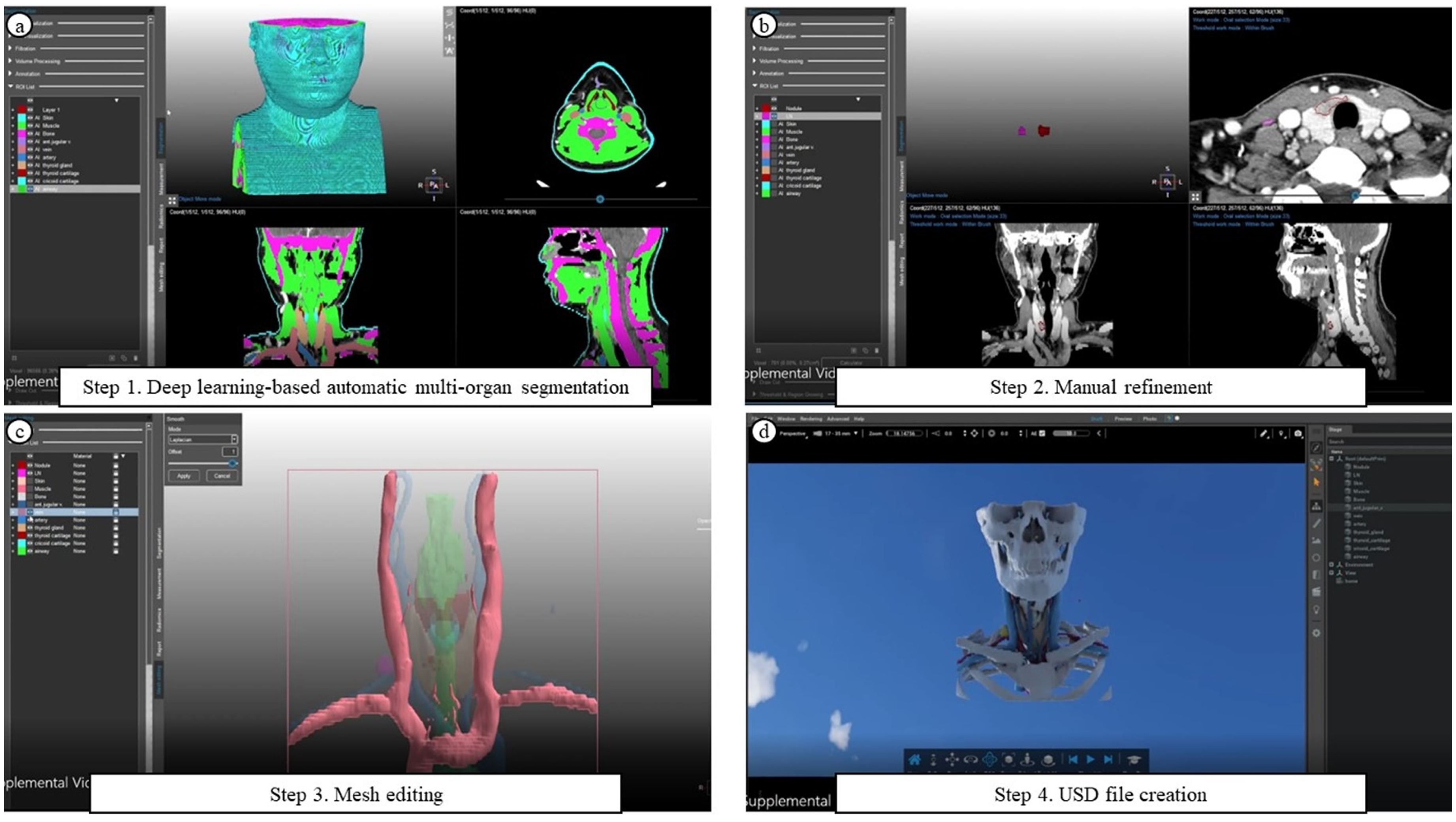
Creation of Ray-traced Three-dimensional Virtual Reality Educational Model from Two-dimensional CT Data. (a) Step 1. Deep learning-based automatic multi-organ segmentation. (b) Step 2. Manual refinement. (c) Step 3. Mesh editing. (d) Step 4. Universal Scene Description (USD) file creation.
The 3D VR educational model was integrated into the NVIDIA Omniverse platform (NVIDIA Corporation, Santa Clara, CA, USA). Previous reviews have highlighted that although virtual reality offers significant educational and therapeutic benefits, its adoption in healthcare remains limited due to barriers in usability, accessibility, and structured implementation. 17 To eliminate the potential confounding effect related to patients’ unfamiliarity with the VR platform, the clinician operated the system during preoperative counseling, demonstrating interactive functions such as zooming, rotation, and anatomical layer selection. This clinician-guided approach enhances the accessibility and effectiveness of VR educational models in routine clinical care.12,18 The model was deployed on flat displays of the laptop (ROG Zephyrus M GU502, ASUS, Taiwan) commonly available in clinical settings, eliminating the need for the head-mounted displays and the associated discomfort.3,5,19,20
Feasibility survey
To evaluate the feasibility of 3D VR educational models in improving patients’ health literacy, we implemented a structured two-round counseling protocol during preoperative outpatient visits (Video 2). In the first round, the researcher explained the patient's disease status and surgical plan using conventional CT images. In the second round, the same information was conveyed using a patient-specific, ray-traced 3D VR model via the NVIDIA Omniverse platform, which enabled interactive features such as zooming, rotation, and anatomical layer selection for intuitive counselling (Figure 3).

Representative ray-tracing 3D VR twin model implemented into NVIDIA Omniverse View. (a) A 3D model displays the skin level (yellow arrow: lymph nodes, green arrow: thyroid nodule). (b) Skin and muscles removed. (c) Bones removed. (d) Blood vessels removed. (e) Thyroid gland removed. The orange outlines indicate the patient's lesions (thyroid nodule and lymph node).
After counseling, patients completed a structured questionnaire. Since validated tools like the SDM Questionnaire (SDM-Q-9) are unsuitable for this study, as surgery had already been planned. 21 We developed a custom questionnaire comprising two domains focused on disease-specific understanding and model usability (Table 1). The first domain assessed health literacy through five open-ended questions on key clinical details—malignancy status, tumor size and location, and the presence of lymph node or distant metastasis. The second domain evaluated the perceived usefulness of the 3D VR educational model compared to CT images, using a five-point Likert scale to rate its helpfulness for understanding, overall satisfaction, willingness to use in future consultations, and perceived applicability to other conditions. A qualitative descriptive approach was used to analyze responses.
The questionnaire used in the prospective survey study of the ray-traced three-dimensional patient-specific virtual reality educational model.

Results
The results demonstrated the feasibility of the 3D VR educational model in enhancing patient health literacy and its high level of satisfaction among participants (Table 2). All participants (100%) demonstrated a high level of understanding and information recall by accurately identifying their pathological findings and the location of their nodules. One participant (11.1%) did not respond to some of the survey questions without providing a specific reason. Among those who did respond, only one participant (11.1%) provided an incorrect answer, indicating overall high patient health literacy.
Results of the feasibility survey on the impact and usefulness of ray-traced three-dimensional virtual reality patient educational models.

The responses to each item in the survey were rated on a five-point scale, ranging from the most negative to the most positive evaluation.
Regarding the perceived usefulness of the 3D VR educational model, all patients (100%) responded very positively when asked about its effectiveness in understanding their diseases and its potential applications to other clinical contexts. In terms of satisfaction with the platform and willingness to use it again for future consultations, eight patients (88.9%) provided very positive responses. One patient (11.1%), whose lesion measured less than 3 mm and therefore could not be visualized on either CT scans or the 3D VR educational model, rated their overall satisfaction as positive but provided a negative response (score 2) regarding future use.
Discussion and conclusion
Discussion
This feasibility study demonstrated that a digital technology–based approach integrating ray-traced 3D virtual reality visualization with deep learning–driven segmentation can meaningfully improve patient health literacy and satisfaction during preoperative counseling for thyroid surgery. All participants indicated improved comprehension of their disease status, with high satisfaction and a willingness to use the model in future counseling. These findings support the utility of the 3D VR educational model as a practical and effective tool for enhancing patient education and clinical communication. 3 From a digital health perspective, these findings highlight the potential of advanced visualization technologies to promote patient engagement in shared decision-making and facilitate comprehension of complex anatomical and procedural information. The integration of automated segmentation and interactive visualization enabled intuitive, patient-specific anatomical education, supporting more effective physician–patient communication. To our knowledge, this represents the first application of VR technology in preoperative counseling for thyroidectomy.
Compared with conventional 2D CT imaging, the 3D VR model provided a more spatially coherent and intuitive understanding of anatomical relationships, even for patients without medical training. 22 The combination of 3D educational visualization and ray-traced rendering yielded realistic and interactive displays that may promote deeper engagement and information retention. 23 Furthermore, the model was presented on flat monitors commonly available in outpatient settings, lowering hardware barriers to adoption and facilitating integration into clinical workflows.3,5,19,20
Operation of a VR platform generally requires prior user training; however, such familiarization was impractical within the limited timeframe of outpatient consultations. 24 Therefore, in this study, clinicians operated the VR system and presented the 3D educational model directly to patients on a flat monitor, effectively mitigating usability and accessibility challenges previously reported in VR implementation. 17
From a practical standpoint, a major advantage of this approach lies in its scalability. Once the segmentation and visualization pipeline is established, models can be rapidly generated and standardized for diverse surgical specialties and healthcare systems. This scalability is particularly valuable in resource-limited environments where 3D printing or immersive VR hardware may be impractical.
Several limitations should be acknowledged. This single-center feasibility study involved a small and relatively homogeneous cohort, which limits the generalizability of the findings. The study primarily addressed the functional dimension of health literacy and did not incorporate broader cultural or communicative aspects. 25 Moreover, standardized assessment tools and objective outcome measures were not employed. 26 Although the four explanatory elements selected were clinically relevant for thyroid cancer counseling, they may not encompass the full spectrum of patients’ informational needs, and excessive content delivery could lead to cognitive overload.27,28 Finally, the absence of a control group precludes quantification of the incremental benefit over conventional counseling. Future multicenter randomized studies using validated instruments are warranted to confirm these findings and evaluate broader clinical, behavioral, and educational outcomes.
Conclusion
This feasibility study demonstrated that a ray-traced 3D VR educational model implemented in a real clinical setting improved patient health literacy and increased satisfaction during preoperative counseling for thyroid surgery. Overall, this study provides early evidence that digital technology–based educational tools can enhance patient understanding and satisfaction in preoperative counseling. As healthcare increasingly embraces digital transformation, such solutions hold promise for improving the quality and accessibility of patient-centered communication and supporting more informed, shared decision-making in surgical care.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251394962 - Supplemental material for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery
Supplemental material, sj-docx-1-dhj-10.1177_20552076251394962 for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery by Jong-hyuk Ahn, Soon Ho Yoon, Jong-Min Kim, Sulgina Kim, Young Hoon Jeong, Hyeonuk Hwang, Sang Joon Park, Su-jin Kim and Kyu Eun Lee in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251394962 - Supplemental material for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery
Supplemental material, sj-docx-2-dhj-10.1177_20552076251394962 for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery by Jong-hyuk Ahn, Soon Ho Yoon, Jong-Min Kim, Sulgina Kim, Young Hoon Jeong, Hyeonuk Hwang, Sang Joon Park, Su-jin Kim and Kyu Eun Lee in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251394962 - Supplemental material for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery
Supplemental material, sj-docx-3-dhj-10.1177_20552076251394962 for Feasibility and initial clinical application of a digital health–enabled 3D VR education model to improve health literacy in thyroid surgery by Jong-hyuk Ahn, Soon Ho Yoon, Jong-Min Kim, Sulgina Kim, Young Hoon Jeong, Hyeonuk Hwang, Sang Joon Park, Su-jin Kim and Kyu Eun Lee in DIGITAL HEALTH
Supplemental Material
Supplemental Material
Footnotes
Acknowledgements
The authors have no acknowledgements to declare.
Ethical considerations
This study was approved by the Institutional Review Board of Hospital A (IRB No. 2022-09-015-003).
Consent to participate
All participants provided written informed consent, and their CT data were anonymized as de-identified DICOM files.
Consent for publication
Not applicable.
Authors contributions
(I) Conception and design: SHY and KEL.
(II) Acquisition and analysis of data: All authors.
(III) Interpretation of data: JHA, SHY, and KEL.
(IV) Drafting of the manuscript: JHA and SHY.
(V) Critical revision of the manuscript for important intellectual content: All authors.
(VI) Development of algorithm: JMK and SJP.
(VII) Obtained funding: JHA, SJP, and KEL.
(VIII) Administrative, technical, or material support: All authors.
(IX) Supervision: SHY and KEL.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Medical Device Development Fund, Seoul National University Hospital, Naver Corporation, (grant number RS-2020-KD000146, 0420202120, 3720230030).
Declaration of conflicting interest
Sang Joon Park (chief executive officer), Soon Ho Yoon (chief medical officer), Jong-Min Kim, and Sulgina Kim work in MEDICAL IP and have a stock option of the firm. The authors affiliated with MEDICAL IP had no role in data analysis, interpretation, or manuscript preparation. All analyses were independently performed and verified by the corresponding author.
The other authors declare that they have no competing financial interests or employment/personal relationships that have contributed to the production of this manuscript.
Data availability statement
The datasets generated and/or analyzed during the current study are not publicly available due to the inclusion of personally identifiable information. However, they are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
