Abstract
Objective
Medication prescription errors represent a significant and persistent challenge within healthcare systems globally, constituting a primary focus for clinical pharmacy practice. Additional complexities involve the optimization of drug dosing and the implementation of personalized medicine. This review aims to synthesize the current advancements in artificial intelligence (AI) applications within clinical pharmacy and to discuss future directions for the field.
Methods
To present this narration, 30 articles were reviewed in total. The literature search was done using electronic databases, for example, PubMed, Medline, and Google Scholar, with the help of some keywords. Only articles published in peer-reviewed journals were included.
Results
A total of 30 articles that demonstrated the utility of AI-based applications in clinical pharmacy were included for further analysis. Across all included studies, AI was utilized primarily for the detection of adverse drug events, clinical decision support, verification of prescription accuracy, and pharmacometrics. Secondary applications included providing recommendations to pharmacists for medication therapy management and, importantly, predicting the therapeutic response to a given treatment in conjunction with its cost-effectiveness.
Conclusion
Artificial intelligence-based algorithms have been identified as applicable tools for the early detection of adverse drug events and prescription errors, the prediction of individual drug response, and the design of patient-specific treatment plans. Prior to broad clinical implementation, future multicenter, prospective studies employing standardized clinical endpoints, external validation, and cost-effectiveness analyses are required.
Keywords
Introduction
John McCarthy, an American computer scientist renowned as the father of artificial intelligence (AI), first introduced the term “Artificial Intelligence” during a conference at Dartmouth College, New Hampshire, United States, in 1956. 1 This landmark event is widely regarded as the foundational moment of AI as a formal discipline. Subsequently, the field advanced at an accelerated pace. In its initial stages, scientists and researchers employed AI primarily for mathematical computations, solving algebraic problems, developing algorithms, and constructing logic-based systems to address challenges traditionally requiring human cognitive capabilities. 2
A significant innovation occurred in 2006 when Geoffrey Hinton and his collaborators proposed the development of neural networks designed to preserve information and prevent the vanishing gradient problem during data training. 3 Among the various algorithms applied in AI, deep learning (DL) techniques have demonstrated exceptional utility. These methods utilize multiple layers of artificial neural networks to automatically generate abstract representations and derive insights from input data, thereby minimizing the need for human intervention. 4
Recent advancements in technology have enabled the integration of multiple features alongside vast quantities of data, which can now be effectively utilized by AI systems. 2 Contemporary DL algorithms are capable of remarkably emulating human cognitive abilities through the replication and abstraction of complex datasets. 3 Initially developed as a tool for solving mathematical problems and executing complex algorithms, AI has evolved into a system of advanced intelligence capable of surpassing human problem-solving efficiency in certain domains.. 1
Clinical pharmacy is a specialized field dedicated to the optimization of medication use to promote health, wellness, and disease prevention. 1 It represents a pharmacist-driven approach to patient care. A central responsibility of the clinical pharmacist is to advance safe, effective, and economically efficient drug therapy to enhance patient outcomes. This goal of optimal patient care is achieved through a range of services, including the design of pharmaceutical care plans, resolution of drug therapy problems, participation in interdisciplinary rounds and consultations, patient education during hospitalization and after discharge, as well as thorough documentation and data management. 2
The evolutionary advancement in the application of AI within healthcare has enabled the utilization of vast, accumulated datasets, presenting significant opportunities to enhance patient care across all domains of clinical practice including pharmacy and allied health disciplines. In response, clinical pharmacy has begun to integrate these innovative technologies to address and advance its core objectives: improving patient access to care, reducing healthcare costs, and optimizing clinician satisfaction. 3
In recent years, numerous studies have investigated the utilization of AI in clinical pharmacy. ChatGPT-based systems have shown to be successful in detecting drug–drug interactions (DDIs) and recommending alternate therapies in medication management. 5 Artificial intelligence-driven clinical decision support systems have been utilized to minimize prescribing errors and enhance dosage for patients with intricate medication regimens. 6 Machine learning algorithms have been employed to identify potentially inappropriate medications (PIMs) in older populations, enhancing prescription accuracy and safety. 7 Moreover, robotic dispensing systems are currently being implemented in hospital pharmacies to enhance workflow efficiency, minimize human error, and elevate satisfaction among patients. 8
Artificial intelligence is a broad term used for the computational technique which is capable of performing the tasks which otherwise need human brain cognition such as learning, understanding, language, planning, and problem-solving. 4
Although the current surge in AI applications within healthcare services and research is a relatively recent phenomenon, its origins trace back to the 1980s with the development and commercialization of speech recognition software, which relied on statistical predictive modeling. 9 During this same period, the automation of hospital pharmacies began, primarily to support drug distribution, manage inventory, and generate financial reports. By the late 2000s, applications of informatics in clinical pharmacy started to emerge, contributing to the formal definition of the pharmacist informaticist role. 10 However, limited datasets and technical resources hindered progress, resulting in a prolonged period of stagnation in the practical application of AI in healthcare. 11
Now, in last decade, the recent advances in data processing techniques and availability of huge electronic healthcare records have created this resurgence of association of AI with medicine. 12 The applications of AI in healthcare can be observed in the fields of risk assessments, prediction of hospital readmissions, clinical decision-making, and individual patient's data management. These all applications are supported by the algorithms generated through the process of machine learning that further based on the techniques of Deep Neural Networking (DNN), Generative Adversarial Networks, Reinforcement learning, and Knowledge graphs. 13
The integration of these applications with medicine has progressed rapidly and is evident across three distinct levels within the healthcare industry:
at the clinician level, primarily through the rapid and accurate interpretation of complex information, at the patient level, by empowering individuals to process and manage their personal health data, and at the health system level, by optimizing workforce capabilities and available resources to minimize medical errors and enhance the efficiency of patient care management
14
Saudi Arabia's Vision 2030 seeks to modernize healthcare via digital transformation, with AI designated as a critical instrument for enhancing care delivery and efficiency. Although AI projects have been extensively used in diagnostics and telemedicine, their incorporation into clinical pharmacy is currently developing. However, extensive deployment throughout the Kingdom is still restricted highlighting the necessity for additional research and policy assistance aimed at integrating AI into clinical pharmacy practice.
15
The technical systems associated with AI are already providing good assistance in routine, manual, and repetitive tasks of medical industry. In addition to that, many researchers have been putting their efforts in observing the effects of implementation of different AI or machine learning models on various aspects of clinical pharmacy. 13 For example, the automation of drug dispensing and pharmacy administration has been started at various hospital settings. In Jeddah city, Saudi Arabia, Ahmed et al. evaluated the drug dispensing and administration process in central pharmacies across the city. They found 28.6% hospitals were using automated dispensing cabinets. 16 Recently, in an ongoing study, in Saudi Arabia, the efficacy of robotic pharmacy solution was studied. In a 21-month long study, the investigators found 22% increase in patients’ satisfaction regarding the pharmacy services; pharmacist productivity was increased by 33% with zero observed dispensing errors. 17
Data, information, and knowledge management; optimal dosing; identification of over- or underprescribed medications; and prediction of drug interaction effects represent key competencies within clinical pharmacy to which AI methods have been applied. Accordingly, this article aims to review published research on the implications of AI in the field of clinical pharmacy. The broader objective is to contribute knowledge that clinical pharmacists can apply to advance both research initiatives and patient care delivery.
Material and methods
Search strategy
A systematic review was conducted in accordance with the PRISMA 2020 guidelines to identify relevant studies on the application of AI in clinical pharmacy. A comprehensive search was performed across seven databases: ScienceDirect, PubMed, Web of Science, Scopus, Directory of Open Access Journals (DOAJ), ProQuest, and Google Scholar.
To ensure specificity and reproducibility, we developed detailed search strings using Boolean operators (AND, OR) and truncation where applicable. Examples of the search strings used are as follows:
PubMed: (“Artificial Intelligence"[MeSH Terms] OR “AI” OR “Machine Learning” OR “Deep Learning”) AND (“Clinical Pharmacy” OR “Pharmaceutical Care” OR “Pharmacy Practice”) AND (“Current Scenario” OR “Future Perspectives”) Scopus/Web of Science: TITLE-ABS-KEY(“Artificial Intelligence” OR “AI” OR “Machine Learning” OR “Deep Learning”) AND TITLE-ABS-KEY(“Clinical Pharmacy” OR “Pharmacy Practice” OR “Pharmaceutical Care”) AND TITLE-ABS-KEY(“Current Scenario” OR “Future Perspectives”) Google Scholar/DOAJ (broad exploratory): “Artificial Intelligence” AND “Clinical Pharmacy” AND (“Current Scenario” OR “Future Perspectives”)
Filters were applied to limit results to peer-reviewed articles published in English. Search strategies were tailored to the syntax of each database but were conceptually aligned.
Inclusion and exclusion criteria
Studies were included if they met the following criteria:
Addressed the application of AI in clinical pharmacy. Discussed the current scenarios or future perspectives. Were original research articles, reviews, or case studies published in peer-reviewed journals.
Exclusion criteria included
Studies not related to clinical pharmacy.
Articles lacking full text or not in English.
Conference proceedings and case studies not directly relevant to the objectives of the review.
Study selection
A total of 1875 records were retrieved. After removal of duplicates (n = 592) and renounced records (n = 23), 1260 records were screened based on titles and abstracts. Of these, 1098 were excluded for not meeting inclusion criteria.
Two reviewers independently screened the titles, abstracts, and full texts of all retrieved records. Disagreements regarding eligibility were first resolved through discussion, and when consensus could not be achieved, a third reviewer was consulted to make the final decision. The selection process followed the PRISMA 2020 guidelines, and the flow of studies is illustrated in Figure 1.

PRISMA flowchart of the systematic review.
Retrieval and eligibility assessment
Full-text retrieval was attempted for 162 studies, which included 47 conference proceedings and 29 case studies. Of these, 76 were unavailable. The remaining 86 studies underwent full-text screening for eligibility, resulting in the exclusion of 56 studies. Exclusions were due to not meeting the inclusion criteria (n = 37) or continued unavailability of the full text (n = 19).
Quality assessment
In accordance with PRISMA guidelines, we assessed the methodological quality of the included studies to evaluate the strength of the evidence and potential for bias. Given the heterogeneity in study designs, the Mixed Methods Appraisal Tool (MMAT, version 2018) was employed for this purpose. The MMAT evaluation consists of two parts: an initial screening section and a subsequent appraisal with criteria specific to five categories of study designs. All items were scored as “Yes,” “No,” or “Can’t tell.” Consistent with the tool's guidelines, studies that received a “No” on both screening questions or a “Can’t tell” on one or more were deemed potentially unsuitable for full MMAT appraisal. A “Yes” rating signifies that the study met the methodological standard for that item, while “Can’t tell” indicates insufficient reporting to make a judgment. Two reviewers conducted the assessments independently; any discrepancies were resolved through consensus or by consulting a third reviewer. In keeping with MMAT guidance, we present the individual item-level assessments and have refrained from calculating an overall summary score.
Final selection
Following systematic screening and eligibility assessment, 30 studies were included in the final review for detailed analysis. Two reviewers independently screened the titles and abstracts of all identified records for relevance and subsequently assessed the full-text articles against the predefined inclusion and exclusion criteria. Discrepancies were resolved through discussion, and when consensus was not reached, a third reviewer adjudicated the decision. The included studies offered important insights into the application of AI in clinical pharmacy, highlighting both current practices and future directions.
Registration and protocol
Registration Information: This review was registered in the PROSPERO International Prospective Register of Systematic Reviews under registration number CRD420251058239.
Available from: https://www.crd.york.ac.uk/PROSPERO/view/CRD420251058239
Protocol Access: A full protocol was submitted to PROSPERO at the time of registration. The protocol will be made publicly available after the completion of the review as per PROSPERO policy.
Amendments to Protocol: No amendments have been made to the registered protocol since its initial submission.
Results
The PRISMA diagram (Figure 1) illustrates the process of study identification, screening, and inclusion. In total, 1875 records were retrieved from various databases. Following the removal of 592 duplicates and 23 renounced records, 1260 records remained for screening. Of these, 1098 were excluded. Full texts of 162 articles were sought, but 76 could not be retrieved (47 conference proceedings and 29 case studies). After assessing 86 articles for eligibility, 56 were excluded (37 not meeting the inclusion criteria and 19 without accessible full text). Ultimately, 30 studies were included in the final review.
Characteristics of the study
Of the 30 studies included in the analysis, the majority were conducted in the United States (n = 12), followed by China (n = 3). Contributions were also made by researchers from Israel, France, the Netherlands, Japan, Portugal, Nigeria, Saudi Arabia, Switzerland, Russia, the United Kingdom, and Australia. As summarized in Table 1, the reviewed articles encompassed a range of methodological approaches, including observational, exploratory, experimental, and retrospective designs, reflecting the methodological diversity within this field of research. The analysis synthesized key details from each study, including authorship, geographical context, research design, study population and sample size, research objectives, forms of AI employed, principal findings, conclusions, and recommendations.
Study characteristics of the included studies.
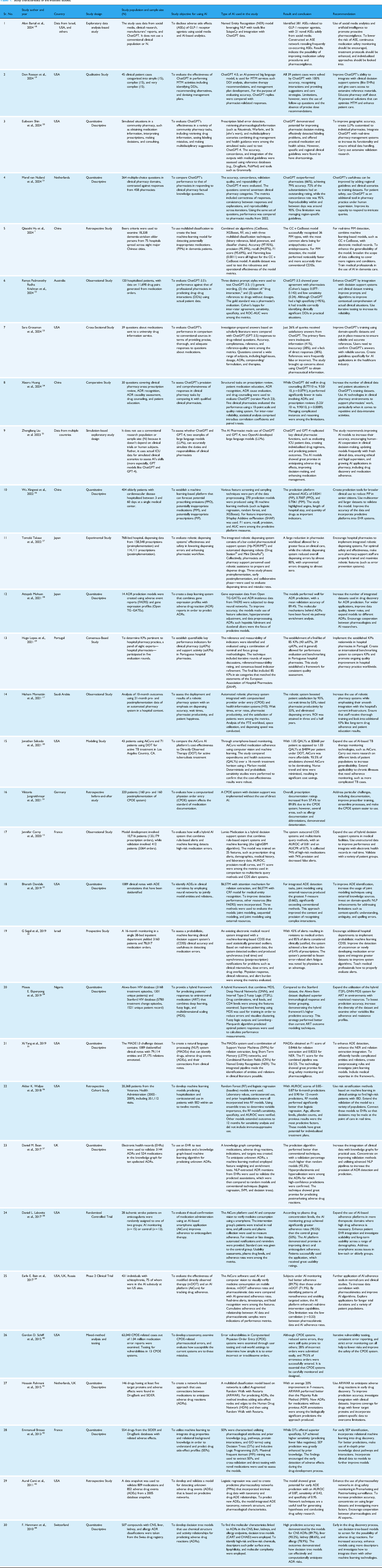
Research indicates that automated robotic dispensing systems, deployed in nations such as Saudi Arabia and Japan, significantly reduce medication dispensing errors. These technologies enhance the precision and efficiency of pharmacy operations, thereby improving overall patient satisfaction. Concurrently, digital health platforms, exemplified by AiCure, have proven effective in augmenting medication adherence, with notable success in managing complex treatment regimens for conditions including anticoagulation therapy and tuberculosis.
Furthermore, predictive analytics has emerged as a critical component in proactively identifying prescription inaccuracies and adverse drug reactions (ADRs), substantially augmenting patient safety and supporting clinical decision-making. Investigations from China have demonstrated the efficacy of sophisticated machine learning algorithms, including CatBoost and hybrid models, in the accurate detection of PIMs and the prediction of ADRs.
The findings from the included studies underscore the transformative potential of AI in advancing healthcare delivery and enhancing patient outcomes. Collectively, the evidence suggests that AI can serve as a valuable adjunct in clinical pharmacy, supporting healthcare professionals in providing more precise, effective, and patient-centered interventions. By aligning AI applications with the specific needs of diverse healthcare settings, these technologies may contribute to more consistent, reliable, and high-quality patient care on a global scale.
Comparative outcomes analysis
Across included studies, reported outcomes included: (i) DDI detection accuracy ranging 24–100% in ChatGPT-based medication therapy management (MTM) evaluations; (ii) machine learning models achieving AUROC up to 0.90 for hospitalization risk and PIM detection; and (iii) robotic dispensing associated with ≈80% lower dispensing errors and >30% higher pharmacist productivity. Reporting of patient-level clinical outcomes and long-term implementation effects was limited.
Quality assessment
Among the 30 included studies, the majority were quantitative descriptive in nature (n = 24), followed by quantitative nonrandomized designs (n = 3), mixed-methods studies (n = 2), and a single randomized controlled trial (n = 1). This variation reflects the diverse methodological approaches used to investigate AI applications in clinical pharmacy. The results of quality assessment are presented in Table 2.
Mixed methods appraisal tool (MMAT), version 2018.

The MMAT appraisal highlighted three common concerns across nonrandomized and descriptive studies: (i) sample representativeness was often unclear or low, (ii) confounders were frequently not accounted for, and (iii) completeness of outcome data was sometimes unclear. By contrast, measurements and statistical analyses were usually appropriate. Overall, the evidence quality ranged from average to good, with no study rated as low quality.
Using the MMAT (2018), all included studies clearly stated their research questions and collected data that appropriately addressed those questions. Among quantitative descriptive studies, most applied relevant sampling strategies (e.g., Alon Bartal et al., Don Roosan et al., Euibeom Shin et al., Merel van Nuland et al., Qiaozhi Hu et al., Ramya Padmavathy Radha Krishnan et al., Sara Grossman et al., Xiaoru Huang et al., Zhengliang Liu et al., Wu Xingwei et al., Attayeb Mohsen et al., and Jonathan Salcedo et al.), although several had limitations in representativeness of the target population, which was frequently rated as “can’t tell” or “no.” Measurements were generally appropriate across these studies, and statistical analyses were consistently suitable. However, nonresponse bias was noted in a few studies (e.g., Alon Bartal et al.).
For quantitative nonrandomized designs, outcome measurement and fidelity of intervention administration were reported adequately (e.g., Tomoki Takase et al. and Hisham Momattin et al.), but control of confounding variables was generally lacking. Only one randomized controlled trial (Daniel L. Labovitz et al.) provided sufficient information on randomization, group comparability, completeness of outcome data, and blinding of outcome assessors.
The single mixed-methods study (Hugo Lopes et al.) demonstrated a clear rationale for the mixed-methods design, effective integration of components, and adequate interpretation of findings, while also addressing divergences between qualitative and quantitative results.
Overall, while methodological quality was acceptable across the included studies, the most common limitations related to representativeness of study samples and insufficient consideration of confounding factors.
Discussion
This review synthesizes current evidence on the application of AI across various domains of clinical pharmacy, with particular emphasis on its impact on medication safety, clinical decision support, and patient adherence. While several AI-driven tools demonstrate strong predictive performance in retrospective analyses or simulated environments, their external validation, integration into routine workflows, and demonstration of patient-level outcomes remain limited and inconsistent.
Artificial intelligence holds significant promise for enhancing clinical pharmacy services by improving precision, efficiency, and decision-making. As healthcare systems become increasingly complex, AI-based solutions are being applied to support pharmacists in diverse tasks, including the detection of ADRs at an individual patient level, optimization of medication management, identification of PIMs, automated prescription verification, and pharmacometric modeling. By leveraging machine learning algorithms, natural language processing, and predictive analytics, these tools have the potential to improve patient outcomes, reduce medication errors, and optimize resource utilization.
Despite these advancements, several challenges remain. Many AI applications are developed and validated in controlled or single-center settings, limiting their generalizability. Moreover, the integration of AI tools into existing clinical workflows is often insufficiently addressed, which can hinder adoption and limit measurable impact on clinical practice. Importantly, few studies report outcomes at the patient level, underscoring the need for prospective evaluations and real-world implementation studies.
In this review, AI applications were categorized based on prevalent themes and areas of clinical significance identified across 30 studies, including adverse drug event (ADE) detection, clinical decision support, and pharmacometrics. These categories were selected based on their frequency in the literature, documented clinical relevance, and potential impact on pharmacy practice. The sorting of AI applications in clinical pharmacy was based on prevalent themes and focal points identified in the 30 papers taken into account. These categories such as ADEs, clinical decision support, and pharmacometrics were selected based on their frequency, clinical significance, and documented impact in the literature:
Adverse drug events and side effects
In this review, the term “ADEs” is used as a broad umbrella term that includes ADRs and adverse side effects (ASEs), unless otherwise specified. The majority of medical errors during hospitalization are the result of invasive procedures, hospital-acquired infections, and the use of drugs and medical devices. However, adverse events can manifest as mistakes during any stage of medical treatment. Prescribing drugs and their administration are more frequently occurring sources of errors. 45 However, this part of medication errors can be potentially prevented by clinical pharmacists’ medication review, which is currently a Gold standard for drug verification. On the other hand, the process is time-consuming and has consistency issues.
In the present review, analysis of the 30 selected studies highlighted the primary roles of AI in clinical pharmacy, including the identification of DDIs, prediction of adverse drug effects based on patient demographics, and detection of side effects (SEs). A recent study by Bartal et al. underscores the transformative potential of AI in detecting ADEs and SEs by leveraging diverse public data sources, including social media platforms and large language models (LLMs) such as ChatGPT. Utilizing a Named Entity Recognition (NER) model, AI was able to identify 21 previously unreported ASEs associated with GLP-1 receptor agonists, including irritability and numbness. This AI-driven methodology not only enhances drug safety monitoring but also facilitates rapid identification of latent risks, supporting proactive post-market pharmacovigilance and ultimately improving patient safety. 18
A recent study by Roosan et al. highlights the pivotal role of AI in enhancing clinical pharmacy, particularly in the context of MTM. The study evaluated the performance of ChatGPT 4.0 in detecting DDIs, recommending alternative therapies, and developing comprehensive management strategies. Across 45 patient cases categorized as easy, complex, and very complex, ChatGPT demonstrated 100% accuracy in identifying DDIs, providing actionable recommendations, and formulating detailed care plans. These findings underscore the potential of AI-driven tools to support pharmacists in optimizing therapy, reducing medication errors, and improving patient outcomes. 19
The study highlights AI's potential to improve clinical decision-making, but certain limitations, like the inability to make accurate dose recommendations or ask follow-up questions. Artificial intelligence-driven approaches like ChatGPT may contribute to improvements in patient safety and care standards in clinical pharmacy practice; however, real-world evidence and further evaluation are required. 19
A study by Radha Krishnan et al. highlights the limitations of AI models, such as ChatGPT-3.5, in clinical pharmacy applications. The study found that while the model exhibited high specificity, it demonstrated gaps in sensitivity and consistency when identifying clinically significant DDIs compared to pharmacist evaluations. These findings emphasize the need to enhance AI tools through comprehensive training on clinically relevant datasets, optimization of prompt strategies, and integration into structured clinical decision support systems. Such improvements are critical to increasing contextual awareness, reliability, and safe application of AI in real-world clinical settings. 22
A study by Mohsen et al. demonstrates the potential of DL to predict ADRs by integrating gene expression profiles with adverse event data. Using advanced neural network models, the study elucidated the molecular mechanisms underlying ADRs through pathway enrichment analysis and achieved a high predictive accuracy of 89.4%. These findings underscore the importance of developing comprehensive, high-quality integrated datasets, and designing models capable of handling diverse ADRs. Furthermore, the study highlights the need for close collaboration between pharmacologists and AI researchers to refine these systems, thereby enhancing their applicability in drug safety monitoring and pharmacological discovery. 28
Corny et al. conducted a study to assess the accuracy of an algorithm-based medication review system. A total of 10716 (with 133179 prescription orders) patients’ data were included in the study for medication review. After reviewing, 20% of patients’ prescriptions were found with the risk of a potential medication error which was either overdosing or underdosing or noncompliance with the drug formulary. Simultaneously, the data were used to generate an algorithm with a rule based expert system. In the process of validation of their model, 412 patients (with 3364 prescription orders) dataset was randomly selected. In 42% of patients, pharmaceutical intervention was suggested by the algorithm while demonstrating 74% accuracy. 15
In a similar study, Segal et al. developed a machine learning based computerized decision support system (CDSS), named MedAware, for finding potential medication based errors and generating the real time alerts. Out of total alerts generated, 89% were found accurate, 85% were clinically valid, and 80% were clinically useful. It was observed with low alert burden and low false alarm rate as compared to existing CDS (Clinical Decision Support) system. Hence, the prescribing doctor could focus on the alerts and change their behavior of prescribing medication after following 43% of errors. Another benefit of using this system was its ability to do surveillance of post prescribing ADEs. Over 60% of alerts were generated after the medication was already prescribed to the patients. 33
A 2019 study by Bharath Dandala highlights the advancements of AI in detecting ADEs from clinical narratives using neural network-based approaches. The study employed joint modeling strategies, combining BiLSTM-CRF for medical entity recognition with BiLSTM incorporating attention mechanisms for relation extraction. The performance of these models was further enhanced by integrating external resources such as the FDA Adverse Event Reporting System (FAERS), achieving the highest F-measure of 0.662 for ADE identification. These findings underscore the importance of domain-specific natural language processing (NLP) enhancements to address challenges such as contextual understanding, ambiguity, and spelling variations, paving the way for more accurate and reliable recognition of ADEs from clinical texts. 32
Yang and his fellow researchers in 2019 demonstrate how well NLP systems such as MADEx can identify ADEs from clinical notes, obtaining strong F1 scores for connection extraction and NER. To increase ADE identification in clinical practice, nevertheless, integration issues decreased pipeline performance overall, highlighting the necessity of better entity–relation integration, joint learning models, and the integration of domain-specific expertise. 35
A study by Bean et al. illustrates the application of knowledge graph-based machine learning algorithms to predict previously unreported ADRs using electronic health record (EHR) data. This approach outperformed traditional methods, achieving a validation accuracy of 92.3% and successfully predicting high-confidence ADRs, including hyperprolactinemia and hypersalivation. The findings highlight the potential of integrating clinical data with knowledge graphs, alongside advancements in natural language processing (NLP) techniques, to enhance the detection and prediction of ADRs, thereby supporting more effective pharmacovigilance and patient safety strategies. 37
In contrast to a study by Rahmani et al. presents a network-based approach, Augmented Random Walk with Restarts (ARWAR), for predicting ADRs. By enhancing the Human Drug Network with side-effect nodes and edges, ARWAR achieved a 20% improvement in F-measure over traditional methods. For medications without previous notes, it also predicted biologically important ADRs. In order to improve prediction accuracy and application in early drug discovery, future research should concentrate on integrating clinical datasets, expanding coverage for medications with fewer target proteins, and incorporating patient-specific data. 41
Bresso and his fellow researchers in 2013 emphasize how machine learning can be used to predict drug side-effect profiles (SEPs) by combining relational information and pharmacological characteristics. Decision Trees (DTs) provided higher specificity, while Inductive Logic Programming (ILP) offered better sensitivity, with prior knowledge significantly improving SEP prediction. With future improvements concentrating on utilizing extensive pathway or interaction data and including clinical datasets to further refine predictions, the strategy exhibits potential for early adverse effect detection. 42
Predictive pharmacosafety networks (PPNs) offer significant potential for the early detection of previously unrecognized ADEs, as demonstrated by a study by Cami et al. By integrating pharmacological data with ADE taxonomies and relational information, the model achieved high specificity (0.95), moderate sensitivity (0.42), and an area under the receiver operating characteristic curve (AUROC) of 0.87. These findings underscore the promise of network-based approaches in drug safety research while highlighting the need for future improvements, including the utilization of larger datasets, incorporation of additional predictive variables, and closer collaboration between pharmacologists and AI specialists to enhance prediction accuracy and clinical applicability. 43
The application of decision tree models (CART and CHAID) for predicting ADR hazards based on molecular properties including polar surface area, lipophilicity, and chemical complexity has been highlighted in another research article by Hammann et al. As computational techniques for early ADR screening, the models demonstrated good prediction accuracy for CNS (89.7%), liver (90.2%), kidney (88.6%), and allergy (78.9%) ADRs. To further improve predicted accuracy, future research should concentrate on adding more descriptors and combining these models with other machine learning approaches. 44
Despite the promising results reported, many studies are limited by small sample sizes and restricted external validation, which constrain the generalizability of their findings. ChatGPT-based evaluations, in particular, often rely on simulated patient scenarios rather than real-world clinical environments. Additionally, numerous AI models have been assessed primarily in retrospective or experimental settings, frequently without longitudinal follow-up, raising concerns about their true clinical effectiveness. This reliance on limited datasets or the absence of pharmacist-based comparative standards increases the risk of overestimating the readiness and reliability of AI tools for routine clinical practice.
Clinical decision support and personalized therapeutic strategies
Several studies have evaluated clinical decision support systems alongside therapy prediction tools, and therefore, these themes are discussed together to highlight their complementary roles and combined significance in enhancing clinical decision-making. There have been positive advancements in the application of AI to treatment response prediction and clinical decision support. According to studies such as Segal et al. 33 and Hu et al., 7 machine learning can help detect PIMs and prevent medication errors. Segal et al. demonstrated the effectiveness of a probabilistic CDSS in identifying real-time medication issues with a minimal alert burden, whereas Qiaozhi Hu used a CatBoost-based multilabel classification model to achieve excellent predictive accuracy.
Additionally, with AUROC scores of 0.85–0.90, the study by Waljee et al. shows how machine learning models, such as Random Forests, can predict hospitalizations and corticosteroid use in patients with inflammatory bowel disease (IBD). These models provide up the possibility to individualized treatment regimens in addition to helping with risk stratification. 36
A study by Labovitz et al. demonstrated that AI-based systems, such as AiCure, significantly improved adherence to anticoagulant therapy compared with standard care, highlighting the potential of AI-driven interventions in optimizing medication adherence monitoring. This demonstrates how AI may improve patient engagement and increase therapeutic efficacy. 38
Although decision support algorithms have demonstrated impressive performance metrics, many studies were conducted using controlled datasets with limited demographic diversity. Additionally, the integration of these systems into existing hospital infrastructure was not consistently addressed, raising concerns about their practical applicability and compatibility with real-world clinical workflows.
Personalized medication and optimal dosing suggestions
The potential for personalized medication and optimal dosing strategies has greatly increased with the introduction of AI into clinical practice. Artificial intelligence-powered tools and models demonstrated potential in customizing treatments to meet the needs of each patient, improving therapeutic results and adherence.
The 2017 study by Labovitz and his fellow researchers emphasizes how AI-based tools such as AiCure can help patients adhere to their anticoagulant treatment. The platform used personalized interventions, reminders, and real-time tracking to achieve adherence rates of 90.5% in the intervention group and 50% in the control group. These results highlight AI's capacity to modify medication regimens in response to unique behavioral patterns, encouraging adherence and ensuring the best possible treatment outcomes. 38
Similarly, a study by Waljee et al. demonstrated that machine learning models, specifically Random Forests, can accurately predict key clinical outcomes in patients with IBD, including hospitalizations and corticosteroid use. By incorporating patient-specific variables such as age, albumin levels, and platelet counts, these models achieved AUROC scores of up to 0.90, illustrating their potential to optimize dosing, enable risk stratification, and support data-driven clinical decision-making. 36
Additional advancements are highlighted in the study by Liu et al., where LLMs, including ChatGPT and GPT-4, were shown to replicate key clinical pharmacist functions. These models were capable of predicting therapeutic outcomes, designing individualized medication regimens, and analyzing data from intensive care unit patients. Such applications illustrate the potential of AI to optimize drug therapy using real-time clinical data, thereby supporting precision medicine and personalized pharmacotherapy. 25
Although many studies discussed in this section report high predictive accuracy, only a limited number assess downstream clinical outcomes, such as actual therapeutic efficacy or the reduction of adverse events. Sample sizes are often small, and tools like ChatGPT have not been rigorously compared against standard clinical judgment. Furthermore, the reliance on retrospective datasets, absence of randomized clinical trials, and frequent exclusion of high-risk populations highlight the need for caution when interpreting these findings and underscore the importance of prospective validation in diverse patient cohorts.
Accuracy in prescription
Patient safety critically depends on the accuracy of prescriptions, and the integration of AI into clinical workflows has demonstrated promising advances in this domain. Several studies have explored the use of AI and machine learning techniques to enhance clinical decision-making, reduce medication errors, and improve prescribing practices. In a study by Radha Krishnan et al., ChatGPT-3.5 was evaluated for its ability to predict DDIs using real patient data. While the model exhibited high specificity (>95%), its sensitivity (0.24) and concordance with pharmacist assessments were modest. These findings underscore the current limitations of AI in managing complex prescription scenarios and highlight the need for more robust algorithms to ensure comprehensive DDI detection in clinical practice. 22
In contrast, van Nuland et al. demonstrated that ChatGPT-4 outperformed pharmacists in answering standard clinical pharmacy questions, achieving an overall accuracy of 79% compared with 66% for pharmacists. However, the study also highlighted challenges in addressing complex prescription scenarios and managing region-specific recommendations, indicating that AI systems must be adapted to local clinical practices and regulatory frameworks to achieve optimal performance and practical applicability. 21
Platforms based on machine learning have also demonstrated an enormous amount of potential. A probabilistic CDSS coupled with an EHR was evaluated by Segal et al. The system obtained 85% clinical justification for alerts, with 43% of these resulting in prescription changes. 33 Similarly Xingwei et al. created a machine learning technology that achieved an AUC of 0.8341 for PIP detection in order to identify prescription errors, inappropriate medications, and prescriptions in elderly cardiovascular patients. 26
To identify high-risk medication errors, the hybrid decision support system Lumio Medication (Corny et al.) integrated machine learning with rule-based alerts. It identified 74% of high-risk medications with 74% precision, achieving an AUROC of 0.81. This method improved the precision of prescription error detection and reduced false alerts by incorporating advanced analytics. 15
Although AI systems such as ChatGPT-4 outperform pharmacists in certain tasks, their limitations in regional adaptation and context-specific clinical knowledge remain inadequately addressed. Furthermore, many models have evaluated DDI detection in isolation rather than within the context of complex polypharmacy, limiting their external validity. Several tools also fail to report false negatives transparently, which poses potential safety risks if these systems are implemented in clinical practice prematurely.
Pharmacometrics
The application of AI and machine learning models has significantly advanced pharmacometrics, the quantitative assessment of drug efficacy and safety. Studies have demonstrated that these approaches can enhance patient-specific outcomes, optimize dosing regimens, and predict both therapeutic responses and potential adverse effects, thereby supporting more precise and individualized pharmacotherapy.
Random Forest models were used to predict hospitalization and corticosteroid use in patients with IBD, according to a 2018 study by Waljee et al. The models obtained AUROC scores as high as 0.90 by incorporating patient-specific variables such as lab values and previous hospital stays, offering important information when designing personalized treatment regimens. These results demonstrate how AI can maximize medication efficacy while reducing hazards. 36
Similarly, Ekpenyong et al. proposed a hybrid framework combining fuzzy logic with DNNs to predict outcomes of antiretroviral therapy. By incorporating patient-specific parameters such as viral load and CD4 counts, this approach demonstrated higher predictive accuracy compared with traditional methods. The integration of pharmacometric parameters in these models ensures greater precision in therapeutic decision-making and supports individualized treatment strategies. 34
Additionally, Labovitz et al. used AI platforms to track anticoagulant therapy adherence and correlate it with pharmacokinetic outcomes. These applications demonstrate how important it is to combine real-time monitoring with pharmacometric details to improve therapeutic results and adherence. 38
Despite the strong predictive potential of AI in pharmacometrics, many models have relied on narrowly restricted datasets or surrogate outcomes. Real-world integration remains limited, and evaluation in multiethnic or resource-constrained settings is scarce. Furthermore, insufficient transparency in methodological aspects such as variable selection and handling of missing data reduces confidence in the robustness and generalizability of these models across diverse patient populations.
Community and dispensing pharmacy
Although limited in number, the studies in this category reveal emerging trends that merit brief discussion. Automation and AI have revolutionized community and dispensing pharmacy procedures, improving accuracy and efficiency while assisting pharmacists in making decisions. The study by Shin et al. demonstrates how ChatGPT and other AI tools can help with tasks in community pharmacies. The requirement for geographically customized AI models that are adapted to community pharmacy settings is highlighted by limitations associated with region-specific guidelines. 20
Further developments are seen in studies such as Takase et al. and Momattin et al., which found that robotic systems greatly decreased dispensing errors and increased workflow efficiency, demonstrated further developments in pharmacy dispensing. These findings demonstrate the way automation might reduce human error and free up pharmacists to concentrate more on patient-centered activities.17,27
Despite these advancements, challenges remain in the seamless integration of robotic and AI technologies into community pharmacy practice. Successful adoption will require additional research focused on localized adaptations, rigorous validation, and comprehensive training for pharmacy staff. By addressing these gaps, AI and automation have the potential to enhance the quality of community and dispensing pharmacy services, promoting safer, more efficient, and patient-centered care. Additionally, most evaluations are conducted in controlled environments, and long-term effects on staffing needs and pharmacist roles remain underexplored.
Computerized prescriber order entry
This category includes fewer studies but provides important insights into how AI-enhanced computerized prescriber order entry (CPOE) systems can impact prescription quality and safety. The section remains concise due to the limited but relevant scope of available research. With the goal of reducing prescription errors and improving the overall standard of medication management, CPOE systems have emerged as a crucial part of modern healthcare. The present review provides important knowledge on CPOE's efficacy and points out areas that still require development.
A study by Jungreithmayr et al. evaluated the impact of a CPOE system on hospital medication documentation standards. The study reported a significant improvement in prescription documentation scores, increasing from 57.4% to 89.8%. However, persistent issues such as incomplete allergy documentation and frequent abbreviation errors underscore the need for ongoing prescriber training and continuous system optimization to ensure accurate and comprehensive medication records. 31
Similarly, Schiff et al. analyzed over one million prescription error reports to assess vulnerabilities in CPOE systems. The study found that, while CPOE implementation reduced certain types of errors, 28% of incorrect orders were still successfully recorded, indicating persistent system vulnerabilities. These findings underscore the importance of iterative vulnerability testing, consistent error reporting, and ongoing system optimization to enhance medication safety and address remaining gaps in CPOE performance. 40
Together, these studies show how CPOE systems can improve prescriber accuracy and reduce errors. However, in order to take full advantage of their benefits, ongoing issues including usability problems, insufficient data entries, and system vulnerabilities must be resolved. In order to ensure optimal efficiency and user compliance, future initiatives should concentrate on integrating real-time feedback mechanisms, advanced decision assistance tools, and frequent system audits.
Computerized prescriber order entry systems can improve patient safety in a variety of clinical settings, reduce errors, and improve prescribing workflows by addressing these limitations.
Alert fatigue, inadequate user training, and insufficient interface testing persist in undermining efficacy. Furthermore, numerous systems operate independently, without effective connection with comprehensive EHRs or decision support systems.
The cost effectiveness and other benefits
Artificial intelligence and automated systems have proven to be very cost-effective and operationally beneficial when used in pharmacy practice, especially when it comes to decreasing errors, streamlining processes, and enhancing patient outcomes. In the current review, a number of studies emphasize these features.
Salcedo et al. study showed how cost-effective the AiCure AI technology is for tracking drug adherence during TB treatment. AiCure maintained greater adherence rates (93.5%) while lowering expenses per patient from $4894 to $2668 when compared to Directly Observed Therapy. This demonstrates that by eliminating the need for time-consuming monitoring, AI technologies not only enhance health results but also result in significant cost savings. 30
Likewise, Momattin et al. investigated the implementation of a robotic pharmacy system in a hospital setting. The system not only reduced dispensing errors but also increased pharmacist productivity by 33%, decreased patient wait times by 53%, and improved patient satisfaction by 93%. The achievement of return on investment within just three and a half years further highlights the cost-effectiveness and operational benefits of integrating robotic technologies into pharmacy practice. 17
The benefits go beyond cost savings. For example, robotic dispensing devices save dispensing errors by 80%, freeing up pharmacists to concentrate on clinical treatment in a study conducted by Takase et al. By reallocating resources, pharmacy staff burnout is decreased and the quality of care is improved. 27
Despite these advantages, the integration of AI and robotic systems can be costly and often requires extensive training as well as modifications to existing workflows. To optimize their impact, future research should explore strategies to reduce implementation costs, evaluate long-term benefits across diverse healthcare settings, and enhance training programs for pharmacy staff to ensure effective and safe utilization.
Overall, automation and AI technologies provide cost-effective solutions with multiple benefits, including enhanced productivity, improved patient safety, and more efficient resource utilization. Their integration into pharmacy practice has the potential to transform healthcare delivery while maintaining financial sustainability and supporting high-quality patient care.
Critical appraisal and generalizability
The body of evidence in this review shows promising performance for several AI tools; however, methodological limitations and external-validity concerns restrict how confidently the findings can be generalized. First, many evaluations were retrospective or based on simulated or convenience scenarios (e.g., LLM/ChatGPT tasks), which are vulnerable to spectrum and selection biases and may overestimate clinical readiness compared with prospective, real-world use. Several studies also reported small samples or lacked longitudinal follow-up, limiting the stability of estimates and the assessment of sustained effectiveness.
The MMAT appraisal further indicates recurring risks: unclear representativeness, incomplete outcome data in some nonrandomized designs, and limited control of confounding all of which can bias effect estimates. In contrast, measurement approaches and statistical analyses were generally appropriate, and no included study was categorized as low quality overall.
Chronological Evolution of AI Applications
The application of AI in clinical pharmacy has demonstrated a distinct evolution throughout time. Initial research 2010–2015 concentrated on rule-based systems and computerized order entries. From 2016 to 2020, machine learning started to emerge in medication safety surveillance and risk prediction models. Since 2021, big language models such as ChatGPT and GPT-4 have been progressively investigated for clinical decision support, drug evaluation, and individualized treatment. This timeline demonstrates a transition from static automation to dynamic, real-time decision-making tools, highlighting AI's development and adaptation to intricate pharmacy settings.
Future directions perspectives
As AI continues to evolve, emerging trends such as explainable AI, real-time predictive analytics, and integration with wearable technologies are gaining traction. However, challenges remain in ensuring data transparency, minimizing algorithmic bias, and developing ethical guidelines for AI deployment in pharmacy settings. Policymakers and academic institutions must collaboratively define frameworks for safe, equitable, and evidence-based AI implementation in clinical pharmacy.
Conclusion
This study investigated the multifaceted applications of AI within clinical pharmacy, including ADE detection, clinical decision support, personalized medicine, pharmacometrics, and operational automation. The reviewed literature indicates that AI has significant potential to augment pharmacist-led care by improving accuracy, efficiency, and patient outcomes.
However, several limitations were identified across the current body of evidence. Many studies relied on retrospective or simulated datasets, lacked external validation, or were conducted within narrowly defined populations. The integration of real-world data into pharmaceutical workflows was often limited or unassessed, and the long-term therapeutic impacts of AI interventions remain largely unquantified. These shortcomings constrain the generalizability and translational potential of many existing AI models.
This review itself has limitations, including heterogeneity in the methodological quality of included studies, a restriction to English-language publications, and the potential for publication bias. Future research should prioritize large-scale, prospective clinical trials in diverse real-world settings, improve model transparency and interpretability, and focus on the seamless integration of AI within existing EHR systems. The establishment of clear regulatory frameworks, dedicated training for healthcare professionals, and robust ethical oversight will be critical to ensuring the safe and effective adoption of AI in pharmacy practice.
In conclusion, while AI is not a substitute for clinical pharmacist expertise, it represents a powerful adjunct tool. Its successful implementation to advance patient care is contingent upon rigorous validation, responsible deployment, and continuous evaluation.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251388145 - Supplemental material for Artificial intelligence in clinical pharmacy—A systematic review of current scenario and future perspectives
Supplemental material, sj-docx-1-dhj-10.1177_20552076251388145 for Artificial intelligence in clinical pharmacy—A systematic review of current scenario and future perspectives by Saad S. Alqahtani, Santhosh Joseph Menachery, Ali Alshahrani, Bander Albalkhi, Dhfer Alshayban and Muhammad Zahid Iqbal in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251388145 - Supplemental material for Artificial intelligence in clinical pharmacy—A systematic review of current scenario and future perspectives
Supplemental material, sj-docx-2-dhj-10.1177_20552076251388145 for Artificial intelligence in clinical pharmacy—A systematic review of current scenario and future perspectives by Saad S. Alqahtani, Santhosh Joseph Menachery, Ali Alshahrani, Bander Albalkhi, Dhfer Alshayban and Muhammad Zahid Iqbal in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors would like to thank Deanship of Scientific Research at King Khalid University and colleagues who provided valuable input, feedback, and support during the preparation of this manuscript.
Contributorship
SSA contributed to conceptualization, methodology, supervision, review, and correspondence with the journal. SJM contributed to literature search, data curation, and drafting of the manuscript. AA contributed to data extraction, synthesis of results, drafting, and review. BA contributed to critical revision of the manuscript and validation of data interpretation. DA contributed to formal analysis, manuscript structuring, and review. MZI contributed to methodology, literature review, final proofing, and editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is a systematic review and did not involve the recruitment of human participants or animals; therefore, ethical approval was not required.
Funding
The authors thank the Deanship of Scientific Research at King Khalid University for funding this work through the large Groups Project Under grant number (RGP.2/578/45).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
