Abstract
Objective
To evaluate the effectiveness of digital therapeutics for rehabilitation of urinary incontinence.
Methods
Seven electronic databases were searched for randomized controlled studies investigating the effectiveness of digital therapeutics on urinary incontinence, reporting outcomes on urinary incontinence severity, adherence, the patient global impression of improvement (PGI-I), quality of life, and pelvic floor muscle function. The literature was reviewed, and the risk of bias was assessed. Meta-analysis was then conducted for each outcome using RevMan 5.4 and Stata 18.0.
Results
A total of 18 studies were included, involving 2308 individuals with urinary incontinence. Meta-analysis showed significant difference in urinary incontinence severity (SMD = −0.30, 95%CI: −0.53 to −0.07, P = 0.010), adherence (SMD = 1.79, 95%CI: 0.74–2.84, P = 0.0008), and PGI-I (RR = 1.70, 95%CI: 1.12–2.58, P = 0.010) between digital therapeutics compared with any type of control. However, the results showed no significant difference in quality of life and pelvic floor muscle function (all P > 0.05).
Conclusion
This review suggests that digital therapeutics-based interventions may be significantly more effective than controls in alleviating urinary incontinence severity, enhancing adherence and improving PGI-I. However, due to the evidence quality, confidence in these findings is low. Therefore, future research should generate more robust evidence to validate the effectiveness of digital therapeutics on rehabilitation of urinary incontinence.
Keywords
Introduction
Urinary incontinence (UI) is defined as the complaint of any uncontrolled, involuntary loss or leakage of urine.1,2 Approximately 423 million people are affected by UI globally 3 and it has also been identified as a health priority by the World Health Organization. 4 Moreover, a meta-analysis of 29 studies found that the prevalence of UI in Asia, Europe, and United States was 45.1%, 43.8%, and 25.8%, respectively. 5 UI is usually associated with age, obesity, pregnancy or childbirth, smoking, hysterectomy, chronic cough, history of miscarriage, race, and surgery.6–8 It can adversely affect urinary tract infections, 9 sexual function, 10 productivity, 11 and quality of life. 12 In addition, UI can also negatively affect psychological well-being, 13 self-esteem, 14 and activities. 15 More importantly, it caused a serious financial burden on individuals and healthcare accounts. Previous review has shown that annual expenditures directly related to UI surpass $12 billion in the United States. 16
Currently, the major treatments for UI includes pelvic floor muscle training (PFMT), medication, lifestyle changes, behavioral therapy, surgery, biofeedback, and electrical stimulation. 17 Multiple clinical guidelines recommend PFMT as the first-line treatment for UI.18,19 However, some studies reported that 49% of individuals with UI can make a standardized and correct PFMT 20 and only 23% of patients have long-term adherence. 21 Hence, conventional treatments demonstrate constrained therapeutic efficacy in UI management. There is a growing need to provide an effective and feasible therapeutics to improve UI among individuals.
Digital therapeutics (DTx) have played an increasing potential to significantly enhance the management of diseases. 22 In 2019, DTx is defined as delivering evidence-based therapeutic interventions driven by high-quality software programs for the treatment, management, or prevention of medical conditions or diseases. 23 DTx is software-based and mediated by smartphones, computers, tablets, and smartwatches, which optimizes patient care by intervening in patient illnesses through information (e.g. text, images, and videos on the App), and physical factors (e.g. sound, light, electric current, and magnetic fields). 23 These can be used independently or in conjunction with medications, devices, or other therapies to optimize care and health outcomes of patients. DTx significantly emphasizes evidence-based therapeutic approaches and high-quality software programs to treat, manage, or prevent medical conditions or diseases, which can reduce the healthcare system's financial burden and therapist time. 24 There is some evidence to suggest that DTx is effective in the treatment and management of chronic diseases25,26 and mental illnesses.27–30
Some randomized controlled trials (RCTs) demonstrated the effectiveness of DTx on improving UI and quality of life, compared with conventional interventions.31–33 However, some studies have shown that the effectiveness of DTx on the symptom improvement of patients with UI is similar to that of the control group. 34 Even some studies have found that compared with DTx, the improvement of UI symptoms and quality of life in the booklet group is more significant. 35 It can be seen that the conclusions from single studies on the effectiveness of DTx for individuals with UI are inconsistent. Although numerous RCTs on the effectiveness of DTx for individuals with UI have been conducted worldwide, these primary studies have limited sample sizes, large variations in interventions, and different outcome indicator evaluations, which evaluate the effectiveness of DTx on patients with UI still lack comprehensive evidences. The specific impact of DTx on UI management is not yet clear, and there is no meta-analysis to assess the impact of DTx on patients with UI wordwilde. Therefore, the purpose of this systematic review and meta-analysis was to evaluate the effectiveness of DTx on UI rehabilitation, including UI severity, adherence, patient global impression of improvement (PGI-I), quality of life, and pelvic floor muscle function.
Methods
Study design
This systematic review and meta-analysis were performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 checklist. The protocol has been registered in PROSPERO with CRD42024617164.
Search strategy
In total, seven electronic databases were searched, including PubMed, EBSCO, Cochrane Library, Embase, Scopus, Web of Science, and the Grey Literature Report, from their establishment to December 2024. Our search terms encompassed a blend of medical subject headings (MESH) and free words, including (“digital technology” OR “digital health” OR “digital therapeutics” OR “digital therap*” OR “digital Electronics” OR “digital technolog*” OR “digital health technolog*” OR “digital” OR “digit*” OR “digital assistant”) AND (“urinary incontinence” OR “urinary incontinence, stress” OR “urinary incontinence, urge” OR “incontinence urinary” OR “urinary stress incontinence” OR “urgency urinary incontinence” OR “mixed urinary incontinence” OR “pelvic floor” OR “pelvic floor muscle” OR “pelvic floor training”). Moreover, other relevant literatures were also identified from the references list of previous reviews (Table S1).
Study selection
All search results from seven databases were managed using the Endnote X9 software. Two independent authors (QLJ and WSY) simultaneously looked through each article's abstract and title after removing any duplicates. Every article that might be relevant was included for a full-text review later on.
After that, two researchers (QLJ and WSY) independently examined the full-text based on the inclusion and exclusion criteria. Disagreements were resolved through discussion, and a third researcher (HSJ) was consulted to achieve consensus.
Inclusion and exclusion criteria
The inclusion criteria and exclusion criteria were established through the PICOS acronym.
The inclusion criteria were as follows: (i) P (population): adult patients (≥ 18 years) with the diagnosis of UI. (ii) I (intervention): DTx was used as an intervention (The criteria of DTx included the following: (a) the DTx needs to be software driving, such as mobile applications, platforms, and computer software; (b) the development of DTx products involves the participation of medical professionals). (iii) C (Comparator): comparison group receiving usual care, education or instructions only, postponed treatment, or only information app. (iv) O (outcomes): UI severity, adherence, quality of life, pelvic floor muscle function, and PGI-I. (v) S (study design): RCTs.
The exclusion criteria were as follows: (i) Studies were excluded if the telehealth interventions used were not specifically developed for UI patients, such as video, Wechat, text message, email, and recreational products. (ii) If intervention platforms or apps in studies are solely used for symptoms detection and reporting in patients with UI, or merely for health education, they are also not eligible. (iii) Reviews, meetings, conference abstracts, reports, protocols, guidebooks, and grants are also excluded.
Quality appraisal
The quality assessment criteria were standardized once all researchers agreed on them through discussion. Two researchers (QLJ and LK) then independently assessed the quality of all included studies using the Cochrane Collaboration's Risk of Bias tool 2.0. 36 Disagreements were resolved through discussion, and a third researcher (ZBB) was consulted to achieve consensus. This assessment is divided into six domains: the randomization process, deviations from the intended interventions, missing outcome data processing, outcome measurement, reported result selection, and overall risk of bias. Each signaling question is rated with five options: yes (Y), probably yes (PY), no (N), probably no (PN), and no information (NI). Every domain is classified in three levels: low risk, some concerns, and high risk.
Data abstraction
Two independent authors (QLJ and ZY) extracted data from all included studies using a prespecified, unified data extraction sheet, which included study year, author, country, sample size, mean age, the type of UI, setting, study duration, the intervention of the experimental and control groups, outcomes.
Data synthesis
Review Manager (RevMan) 5.4 and Stata 18 were utilized for statistical analysis. When data from two or more studies were accessible, the results were incorporated into the meta-analysis. To enhance the accuracy of the results, the analysis focused on the changes from baseline to final within the control and intervention groups. For continuous variables, if the outcomes were measured using the same scales, we calculated the mean difference (MD) with corresponding 95% confidence intervals (95%CI). Otherwise, standardized mean difference (SMD) with corresponding 95%CI was used. For dichotomous outcome data, effect measures were calculated using risk ratios (RRs) or odds ratios (ORs), with corresponding 95%CI. If the change of mean or standard deviations (SDs) was not provided in studies, we would calculate them as advised by the Cochrane Handbook. We evaluated the heterogeneity through I2 statistics. If studies show low heterogeneity (I2 ≤ 50%, P > 0.1), a fixed-effects model is applied. For high heterogeneity (I2 > 50%, P ≤ 0.1), sensitivity analysis is performed to identify causes. If heterogeneity remains, a random-effects model is used. The stability of the summary effect size was evaluated through sensitivity analyses, which involved sequentially excluding each study and reanalyzing the remaining data to determine if any single study significantly influenced the results. Publication bias was assessed by a funnel plot, and the Egger and Begg test assessed funnel plot symmetry, with P < 0.05 suggesting potential bias. To further explore the sources of heterogeneity, meta-regression and subgroup analyses will be conducted.
Results
Study selection and characteristics
A total of 24,962 records were searched through seven databases, and 2 records were identified by other previous reviews. So, 24,964 records were initially imported into Endnote X9. After removing 10,597 duplicate articles by Endnote, 14,367 records were screened by title and abstract. At this stage, 256 records were determined to be included for full-text screening, as they met the inclusion criteria; 14,111 records were removed. Two authors independently conducted the screening full-text of 256 records, and then compared their findings. Any disagreements were addressed through discussion. Finally, 18 studies met the inclusion and exclusion criteria and were included in the systematic review and meta-analysis. All studies included are RCTs. The study selection flow diagram is illustrated in Figure 1.

PRISMA flow diagram of study selection.
Table 1 presents the characteristics of the included studies. Fourteen studies31–35,37–45 were conducted between 2020 and 2024, 1 46 in 2017, 1 47 in 2015, 1 48 in 2013, and 1 49 in 2019. Three studies33,38,41 were undertaken in China, 332,43,44 in America, 331,35,37 in Brazil, 342,46,48 in Sweden, 234,40 in the Netherlands, 1 47 in Spain, 1 45 in Turkey, 1 39 in Thailand, and 1 49 in Canada. Six studies33,38,39,41,45,47 recruited participants from hospitals, 734,35,40,43,44,46,48 from websites, 331,37,49 from clinics, 1 42 from the community, and 1 32 from seven participating sites. The duration of DTx interventions ranged from 4 to 48 weeks. Fourteen studies31–34,38–40,42–46,48,49 reported the treatment effectiveness of UI symptoms, 13 studies31,32,34,35,37,38,40–42,44,46,47,49 reported the quality of life, 6 studies32,33,37,39,41,45 reported the adherence, 6 studies32,34,43,44,46,48 reported PGI-I, and 4 studies31,37,39,49 reported the relevant information of pelvic floor muscle. The mean age of participants across 9 studies31,32,34,39–44 exceeded 50 years, and in 8 studies33,35,38,45–49 were under 50 years. The number of sample sizes was 19 to 363. Besides, the study included 2308 patients with UI, of whom 9 were men and 2299 were women.
Brief summary of the characteristics of the included studies.
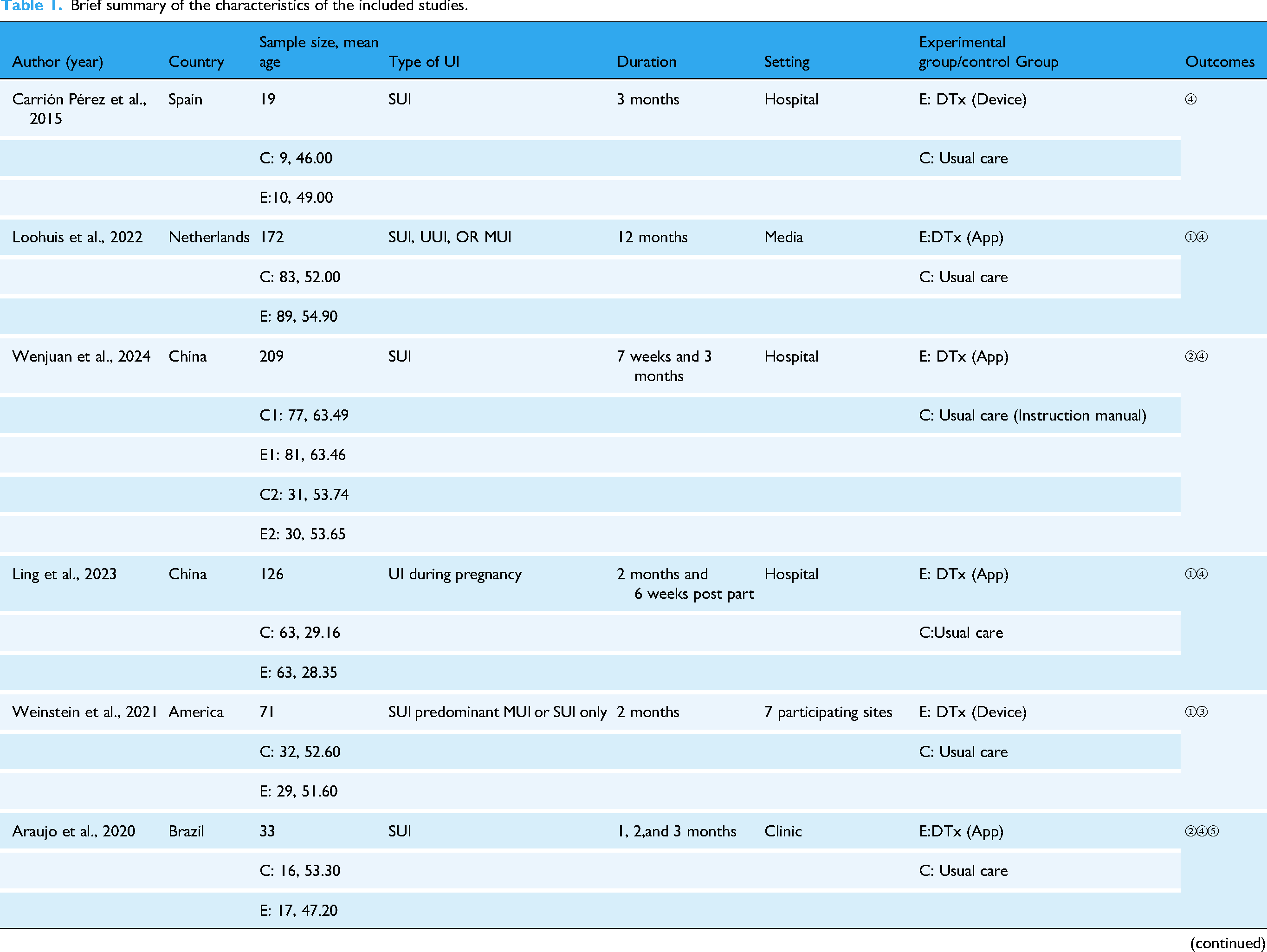
Abbreviations: ① Urinary incontinence symptom improvement ② Adherence ③Patient global impression of improvement (PGI-I) ④ Quality of life ⑤ Pelvic floor muscles function. E: Experimental Group; C: Control Group; UI, urinary incontinence; SUI, stress urinary incontinence; UUI, urge urinary incontinence; MUI, mixed urinary incontinence; DTx, digital therapeutics; PFMT, pelvic floor muscle training.
Eleven studies31,33–35,37–42,46 utilized APP as the main way for DTx interventions, 532,43,44,47,49 used DTx devices, 1 45 used videoconference (Skype), and 1 48 used program (the treatment program was internet-based and constructed on a secure platform, with all communications occurring asynchronously and via encrypted email). The specific intervention content of DTx interventions using apps includes the functions of guidelines with the visual or aural component, an alarm of exercise reminders, consultation, automatically recording exercise data, and offering information about the PFMT. The characteristics of DTx devices include: (a) the device consists of an intravaginal insert and mobile phone application; (b) the device can assess pelvic floor muscle motion in real-time and dynamically; (c) some provide interactive games or activities; (d) the device enables remote tracking of adherence and symptom information. One RCT 48 used a program incorporating multilevel training supported by a urotherapist, along with self-monitored evaluation, and cognitive behavioral therapy. The study 45 used videoconference (Skype) where a physiotherapist provides remote guidance and supervision through video conference.
Effects of DTx for individuals with UI
UI severity
Twelve studies31,32,34,38–40,42–44,46,48,49 involving 1780 participants measured UI severity and were included in the meta-analysis. We used the SMD because of the different measurement scales. Due to high heterogeneity (P < 0.01, I2 = 81%), a random-effect model is used. The pooled analysis demonstrated that DTx-based interventions had a statistically significant effect on the improvement of UI symptoms (SMD = −0.30, 95%CI: −0.53 to −0.07, P = 0.010) (Figure 2).

Forest plots of the effects of digital therapeutics for urinary incontinence on symptom improvement.
Additionally, Wang et al. 33 demonstrated that following DTx interventions, participants exhibited a significant improvement in the severity of UI symptoms. Yavas et al. 45 showed that compared to the control group, participants who underwent DTx interventions had significantly better scores on the symptom. However, there was no statistically significant difference between the groups in this 2 RCTs.33,45
Adherence
Three37,39,45 studies including 97 individuals with UI, were included in the meta-analysis of adherence. DTx-based interventions demonstrated a statistically significant effect on adherence (SMD = 1.79, 95%CI: 0.74–2.84, P = 0.0008), and 3 studies revealed high heterogeneity (P = 0.02, I2 = 75%) (Figure 3).

Forest plots of the effects of digital therapeutics for urinary incontinence on adherence.
Additionally, Mao et al. 41 showed that the adherence of DTx group was markedly superior to that of the control group. A significant difference in adherence was observed between the two groups after 12 weeks (P < 0.05). Wang et al. 33 used self-efficacy scale to predict adherence to the training and outcomes demonstrated that DTx group indicated greater adherence. Weinstein et al. 43 reported that at the 4-week, the adherence was 68% for DTx group and 60% for the control group; at the 8-week, the adherence was 97% for both groups.
PGI-I
Six studies32,34,43,44,46,48 investigating PGI-I were enrolled in the meta-analysis. As there was high heterogeneity across 6 studies (P < 0.01, I2 = 88%), the random-effect model was selected. It demonstrated that DTx-based interventions had a statistically significant effect on PGI-I (RR = 1.70, 95%CI: 1.12–2.58, P = 0.010) (Figure 4).

Forest plots of the effects of digital therapeutics for urinary incontinence on PGI-I.
Quality of life
Ten studies,31,32,35,37,40,42,44,46,47,49 including 1276 participants, were included in the meta-analysis of quality of life. As there was high heterogeneity across 10 studies (P < 0.01, I2 = 80%), the random-effect model was selected. We used the SMD because of the different measurement scales. The DTx-based interventions demonstrated no effect on quality of life (SMD = −0.12, 95%CI: −0.39 to 0.15, P = 0.39) (Figure 5).

Forest plots of the effects of digital therapeutics for urinary incontinence on quality of life.
Additionally, Mao et al. 41 showed that the score of DTx group was higher and there was a significant difference between DTx group and the control group on quality of life after 12 weeks of intervention (P < 0.01). Chen et al. 38 showed that participants in DTx group experienced a significant improvement in quality of life (P < 0.05). Loohuis et al. 34 merely reported that quality of life improved in both groups.
Pelvic floor muscle function
Two studies,31,37 including 180 individuals with UI, were included in the meta-analysis of pelvic floor muscles. We used the MD because of the same measurement scales. The DTx-based interventions demonstrated no effect on the power, endurance, repetition, and fast of pelvic floor muscles (power MD = 0.30, 95%CI: −0.25 to 0.85, P = 0.29; endurance MD = 0.20, 95%CI: −0.07 to 0.47, P = 0.15; repetition MD = 0.21, 95%CI: −0.11 to 0.53, P = 0.19; fast MD = 0.25, 95%CI: −0.16 to 0.66, P = 0.22) (Figure 6).

Forest plots of the effects of digital therapeutics for urinary incontinence on pelvic floor muscles function (power, endurance, repetition, and fast).
Additionally, Kijmanawat et al. 39 assessed the strength of the pelvic floor muscles using the Brink scale and showed that scores in both groups increased, but there was no statistically significant difference (P > 0.05). Dufour et al. 49 showed that the pelvic floor muscles function improved for both groups, but there was no statistically significant difference between pre- and post-intervention.
Subgroup analyses and meta-regression analysis
Due to the high heterogeneity, subgroup analyses were conducted based on duration, age, setting, DTx type, and year. The results are shown in Table 2. (a) Subgroup analysis based on duration, age, and setting: the heterogeneity within each subgroup was high. Specifically, for the subgroup with <3 months, ≥50 years, and from hospital settings consistently demonstrated significantly lower UI symptom scores in the intervention group compared to controls. Moreover, these subgroups also produced the largest effect sizes. (b) Subgroup analysis based on DTx type and publication year: the heterogeneity within each subgroup was high. Specifically, for the app group and the 2021–2024 group, the UI symptom scores in the intervention group were lower than those in the control group, with statistically significant differences, and both produced the largest effect sizes.
Results from subgroup analyses.

To further explore the sources of heterogeneity, meta-regression was conducted with duration, age, setting, DTx type, and year as covariates. The results showed that the P-values for all six covariates were greater than 0.05, indicating no statistical significance (Table 3). Further investigation into the sources of heterogeneity is needed.
Results from meta-regression analysis.

Sensitivity analysis and risk of bias results
The symmetry of the funnel plot for the effectiveness of DTx suggests no significant publication bias (Figure 7), as demonstrated by the Egger test (P = 0.723) and Begg test (P = 0.631). Given the significant heterogeneity observed in the included studies evaluating the effectiveness of DTx interventions, it was necessary to conduct a sensitivity analysis to ensure the reliability and confidence in the outcomes. The analysis revealed that the combined point estimates of the remaining studies when any single study was excluded, fell within the 95%CI of the effect size (Figure 8). This demonstrated a strong robustness in the results.

Funnel plot of symptom improvement in randomized controlled trials, plotted as net change in effect vs. SE of the net change.

Sensitivity analysis for effects of DTx on symptom improvement in randomized controlled trials.
Among the 18 included studies, 16 were rated as high risk, and 2 were rated as having some concerns. In terms of the measurement of study outcomes, 77.8% of the studies showed a high risk. During the randomization process, 83.3% of the studies were rated as low risk. Regarding deviations from intended interventions, 61.1% of the studies were rated as having some concerns. In terms of missing outcome data, 77.8% of the studies were rated as low risk. Regarding the selection of reported results, 83.3% were rated as having some concerns (Figures 9 and 10).

“Risk of bias” graph: review authors’ judgments about each risk of bias item presented as percentages across all included studies.
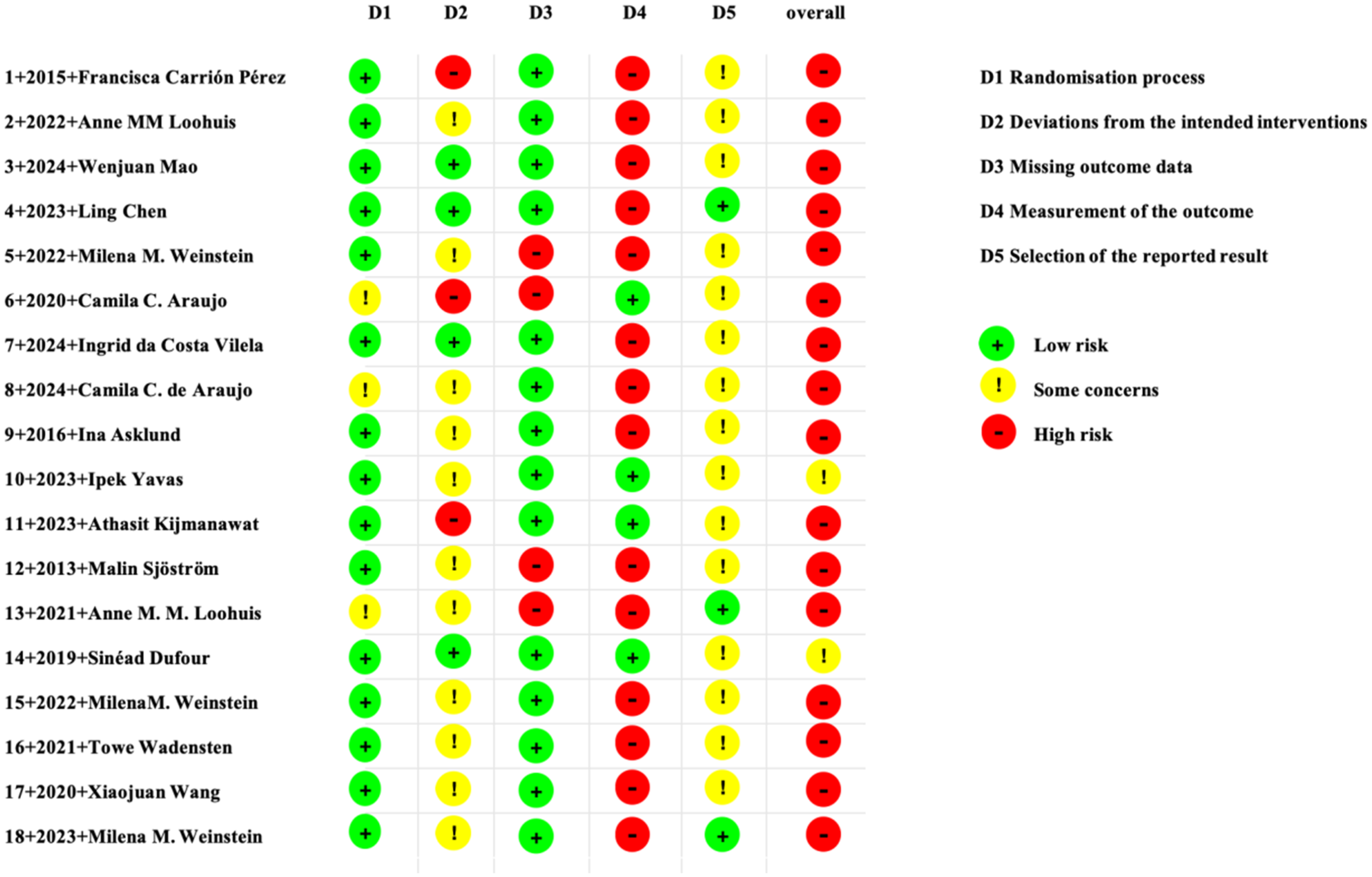
“Risk of bias” summary: review authors’ judgments about each risk of bias item for each included study.
Discussion
Methodological quality assessment of the included studies
Following a rigorous screening process in accordance with the specified inclusion and exclusion criteria, 18 RCTs were finally included in the systematic review. The quality assessment of the included studies revealed that 16 articles31–35,37–44,46–48 were at high risk, and 245,49 were rated as having some concerns. Among them, the primary reason for the high-risk rating of 14 studies31–35,38,40–44,46–48 was that the use of questionnaires that included subjective questions and were self-reported by participants aware of the interventions. This could affect the participants’ reporting of outcomes, thereby influencing the measurement of outcome indicators and introducing bias. Researchers are therefore advised to use objective outcome indicators whenever possible to minimize the risk of subjective judgments influencing outcome reporting, even when participants are aware of the interventions.
In the other 2 studies37,39 rated as high risk, the main issue was the inappropriate use of statistical methods. Specifically, the analysis only included participants who completed the follow-up, which could introduce bias. This serves as a reminder for researchers to adopt proper statistical methods when analyzing data. They need to focus not only on the results of participants who completed the follow-up but also analyze and explain the data of those lost to follow-up. In this way, the quality and credibility of a study can be enhanced.
In addition, most studies failed to develop a detailed and feasible research protocol and register it on a platform before implementation. This led to most studies being rated as having some concerns in the domain of selective reporting bias. This suggests that researchers should formulate a detailed and feasible research plan before the study, and try to be consistent with it when measuring and analyzing the results to avoid selective reporting.
Subgroup analyses and meta-regression
Due to the high heterogeneity, this study also conducted subgroup analyses and meta-regression based on duration, age, setting, DTx type, and year. However, the heterogeneity within each subgroup remained high after the subgroup analyses, and the meta-regression results were not statistically significant, failing to identify the source of heterogeneity from the meta-analysis. The high heterogeneity may be related to the following reasons. (a) Among the 18 studies included in this study, 16 were rated as high risk, and 2 were rated as having some concerns, indicating the overall quality of the primary studies was not high. (b) The tools used to assess the severity of UI symptoms contained many options with subjective judgments and lacked objective indicators. (c) There may be significant differences in the research protocols of each study, with variations in the interventions received by the experimental and control groups.
DTx can alleviate UI severity
The meta-analysis showed that DTx were more effective in alleviating UI severity compared to the control group, and with a statistically significant difference (SMD = −0.30, 95%CI: −0.53 to −0.07, P = 0.010). This is consistent with the meta-analysis results of Huang et al. 50 However, the study results of Dufour 49 and Kijmanawat 39 showed no difference in the effectiveness of symptom improvement between the experimental and control groups. This may be due to the limited sample size, which fails to detect the actual differences between the experimental and control groups. However, this study included 12 studies, with 877 participants in the intervention group and 903 participants in the control group.
DTx offers several advantages for UI patients. First, they provide patients with the correct methods to exercise their pelvic floor muscles in a scientific and proper way. Second, DTx devices can collect real-time feedback on patients’ physical data, enabling patients to quickly identify and address issues. Moreover, DTx are portable, allowing patients to exercise regularly. Additionally, DTx offers patients a wealth of health education resources, which can enhance their initiative in PFMT. Therefore, clinical medical personnel can integrate DTx with traditional treatment methods to manage UI. Specifically, physicians can employ DTx tools to devise personalized treatment plans, leveraging standardized exercise instructions and real-time feedback to replace traditional verbal guidance, thereby minimizing deviations and enabling the standardized and correct PFMT. In addition, clinical medical personnel can use DTx platforms to incorporate health education modules into the treatment process, continuously providing health education to patients with UI, thereby enhancing their disease awareness and self-management abilities.
DTx can enhance the adherence of PFMT
The meta-analysis showed that DTx enhance PFMT adherence more effectively than the control group, and the difference is statistically significant (SMD = 1.79, 95%CI: 0.74–2.84, P = 0.0008). This is consistent with the results of Widdison et al. 51 First, DTx allows patients to engage in training anytime and anywhere, eliminating the need for frequent hospital visits. This flexibility significantly enhances patient engagement and compliance, which in turn helps to improve health outcomes. 52 Second, by incorporating interactive and enjoyable elements that encourage patients to engage in physical activity, DTx helps improve patient compliance. Furthermore, DTx utilizes high-quality mobile applications that act as digital coaches, sending timely reminders to patients and encouraging them to consistently follow their prescribed exercise plans. Baumann et al. 53 also found that supervised PFMT is more effective in improving short-term UI after radical prostatectomy than unsupervised. This finding suggests that for nonadherent, busy patients, healthcare providers can prioritize DTx to create fragmented training plans tailored to patients’ routines and use the it's reminder function to help patients develop good exercise habits.
Although DTx can improve PGI-I, it's effect on improving the quality of life still remains unclear
The meta-analysis showed that DTx can improve PGI-I compared to the control group, and with a statistically significant difference (RR = 1.70, 95%CI: 1.12–2.58, P = 0.010). However, this review does not show a statistically significant improvement in quality of life (SMD = −0.12,95%CI: −0.39 to 0.15, P = 0.39), which is inconsistent with the findings of Huang et al. 50 and Leme Nagib et al. 54 The discrepancy of the results between PGI-I and quality of life may be attributable to numerous factors. First, the measurement content and focus of the two evaluation tools are different; the PGI-I is highly subjective and focuses on the improvement of UI symptoms; in contrast, the assessment of quality of life is broader and more comprehensive. Second, although the current studies use ICIQ-SF (International Consultation on Incontinence Modular Questionnaire-Short Form), ICIQ-LUTSqol (the ICIQ Lower Urinary Tract Symptoms Quality of Life), I-QOL (the Incontinence Quality of Life Questionnaire), and so on, to assess the quality of life of patients with UI, these evaluation indicators may not fully reflect the changes in patients’ quality of life. Additionally, quality of life is influenced by many factors, and DTx may have ignored the comprehensive intervention of patients from multiple aspects during the intervention of UI. Finally, the intervention time and follow-up time of DTx may be limited, leading to poor improvement in quality of life. This outcome suggests that it is crucial to report both the PGI-I and quality of life in the same study, as they offer complementary information. An improvement in the PGI-I is a prerequisite for enhanced quality of life. In addition, future research needs a longer follow-up period to observe the improvement in quality of life. More importantly, clinical professionals should integrate more comprehensive modules into DTx, such as psychosocial support, to improve the negative emotions caused by UI and strengthen the improvement of quality of life.
The effect of DTx on improving the pelvic floor muscle functions is still unclear
The meta-analysis showed that there was no statistically significant difference between DTx and the control group in improving pelvic floor muscle function. This is consistent with the results of Kijmanawat et al. 39 and Dufour et al. 49 This result may be related to following reasons. First, among the 18 included studies, only 2 were incorporated into the meta-analysis, and both were reported by the same author, with few studies reporting pelvic floor muscle function. Second, enhancing pelvic floor muscle function is a gradual process that requires sustained effort and time. However, the intervention and follow-up durations in DTx studies were insufficient to capture long-term outcomes. These findings indicate that future research should prioritize reporting changes in pelvic floor muscle function while incorporating extended intervention and follow-up periods. This would allow for a more comprehensive assessment of the long-term effects of DTx on pelvic floor muscle function.
Limitations
Our study has several limitations. First, this study exclusively covered English-language literature, which may have resulted in the absence of studies in other languages. Second, the quality of the included studies is low, with the majority assessed as high risk due to the potential influence of intervention knowledge on outcome assessment, which is associated with the use of subjective measurement methods. It is suggested that future research ought to improve experimental design and methodological quality in primary investigations. Third, the included studies are derived from a diversity of nations, which means the medical conditions and cultural backgrounds vary from one to another, and in the same vein, the inclusion and exclusion criteria for study subjects also display heterogeneity. Furthermore, the intervention measures for DTx and the control group in the literature are not standardized, with specific interventions, intervention times, frequencies, and follow-up durations varying significantly across studies. Additionally, the measurement tools employed for the same outcome indicators differ, introducing challenges in comparing and synthesizing results across studies. Finally, due to the limited number of included studies, it is difficult to conduct more subgroup analyses, which may lead to heterogeneity in the study.
Conclusion and implication
The meta-analysis results show that DTx can alleviate UI severity, enhance adherence and improve PGI-I. However, the effects on improving the quality of life and pelvic floor muscle function are still limited. This provides a potentially effective intervention strategy for clinical medical personnel to alleviate UI severity and enhance their compliance with pelvic floor muscle exercises and improve PGI-I. However, due to various reasons leading to high heterogeneity in this study, the reliability of the results may be reduced. Therefore, future high-quality studies should be conducted to further confirm the effectiveness of DTx on individuals with UI rehabilitation. Finally, this study predominantly included female patients with UI. Further research should explore the effectiveness of DTx for male with UI and its potential benefits.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251379339 - Supplemental material for The effectiveness of digital therapeutics for rehabilitation of urinary incontinence: A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076251379339 for The effectiveness of digital therapeutics for rehabilitation of urinary incontinence: A systematic review and meta-analysis by Lijuan Quan, Siyuan Wu, Kun Li, Ying Zhang, Sijia Hou, Binbin Zhu and Wei Wang in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251379339 - Supplemental material for The effectiveness of digital therapeutics for rehabilitation of urinary incontinence: A systematic review and meta-analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076251379339 for The effectiveness of digital therapeutics for rehabilitation of urinary incontinence: A systematic review and meta-analysis by Lijuan Quan, Siyuan Wu, Kun Li, Ying Zhang, Sijia Hou, Binbin Zhu and Wei Wang in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to express our sincere gratitude to all of the authors who generously shared their experiences and dedicated time to participate in the survey.
Contributorship
Conceptualization was done by QLJ, WSY, LK, ZY, WW. Methodology and formal analysis were done by QLJ, LK,WSY. Writing-original draft was done by QLJ, WSY, ZY. Writing-review and editing were done by QLJ, WSY, ZY, HSJ, ZBB, WW.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This systematic review and meta-analysis was registered with PROSPERO (CRD42024617164).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang University “Double First-class” Construction Advantageous Discipline Research Cultivation Fund, (grant number HL202402).
Informed consent
The informed consent of this systematic and meta-analysis is not applicable. All data is collected through database.
Supplemental material
Supplemental material for this article is available online.
