Abstract
Objective
To summarise the effectiveness of digital health maintenance interventions for subjectively and objectively measured physical activity level (PA) and physical function, fitness and health-related quality of life (HRQoL) after completion of phase II cardiac rehabilitation (CR).
Methods
We conducted a search for studies in MEDLINE, Embase, CENTRAL, and CINAHL (inception to May 2024). Independent reviewers selected and included randomised controlled trials (RCTs) using digital health interventions to maintain PA in patients with cardiovascular disease after phase II CR. Independent reviewers conducted data extraction, assessed the risk of bias using the Cochrane Risk of Bias 2 tool and rated the certainty of evidence using Grading of Recommendations Assessment, Development and Evaluation – registered at PROSPERO: CRD42023396629.
Results
From 17,455 hits, 20 RCTs with 1801 participants were included. Risk of bias for most studies reporting on PA outcomes was rated as ‘some concerns’. Comparing digital health intervention with minimal intervention/usual care showed no effect of objective PA (standardised mean difference (SMD) 0.85, 95% CI: −0.07 to 1.77) and a small effect of subjective PA (SMD 0.37, 95% CI: 0.05 to 0.69) at the end of intervention both graded as very low certainty of evidence. We found very low certainty of evidence for moderate effects on physical function (SMD 0.63, 95% CI: 0.03 to 1.24), and low certainty of evidence for no effect on physical fitness (SMD 0.19, 95% CI: 0.05 to 0.34) and HRQoL (SMD 0.13, 95% CI: −0.02 to 0.28), I2= 0.00%) at the end of intervention.
Conclusion
Digital health interventions showed a small effect on subjectively measured PA, and no effect on objective PA, physical fitness and HRQoL, but may increase physical function after CR completion. Yet, the certainty of evidence is low and higher quality studies with longer follow-up duration are needed to guide this area further.
Introduction
Regular physical activity (PA) is vital for the secondary prevention of cardiovascular disease.1,2 PA plays a key role in exercise-based cardiac rehabilitation (CR), to improve aerobic fitness and health-related quality of life (HRQoL), and reduce symptoms and risk of post-cardiac events.3,4
CR typically consists of three phases 5 : Phase I CR begins after an acute cardiovascular event and takes place in an inpatient setting aiming to stabilise and treat the patient. Phase II CR provides structured exercise-based rehabilitation, assessing risk factors and treating patients through exercise. A plan is initiated towards the end to prepare for a physically active, independent life at home. Globally, phase II CR consists of a median of three sessions per week with a session median of 60 minutes for a median of 8 weeks. 6 CR III is the lifelong maintenance phase, during which the patient independently maintains exercise recommendations from CR II by self-monitoring their PA levels.
Unfortunately, many patients do not maintain the recommended level of PA in their daily lives after completing CR phase II.7–10 Lack of PA engagement could be attributed to personal, environmental and organisational barriers, such as lack of time and lack of motivation.11,12 There are no standardised definitions for maintenance.13–15 Hence, we define maintenance as a period of at least 6 months with the goal of keeping the benefits gained and preventing relapse. 16
The use of digital health has increased rapidly in recent years.17,18 Digital health, often referred to as eHealth, serves as an umbrella term for eHealth, mHealth and telehealth, which all cover the use of various types of technology in healthcare. 18 eHealth involves internet-based solutions such as websites and emails. mHealth includes phone calls, text messages and apps. Digital devices encompass wearables (with accelerometer or pedometer capabilities), activity trackers or Bluetooth-connected devices. Digital health interventions are easily accessible to patients and can be tailored to the individual patient, encouraging them to become more physically active, thereby potentially reducing barriers and to improve their disease management and improve PA.19–21
Of previous systematic reviews22–25 investigating maintenance interventions after CR II, only Hannan et al. 24 and Heimer et al. 25 investigated the effectiveness of digital health interventions exclusively. Yet, Hannan et al. 24 only assessed the effects of wearables after CR II and found a small effect on step count. Heimer et al. 25 found a small overall effect on physical exercise and activity that favoured digital health interventions on maintenance after CR phase II (standardised mean difference (SMD): 0.35 (95% CI: 0.02 to 0.70), I2 = 72.3%). However, both reviews did not distinguish between subjective and objective PA outcomes, and both excluded interventions that used phone calls or emails exclusively. Subjective (self-reported) and objective assessments of PA capture different aspects, emphasising the need for separate analyses. 26 Objective measurements provide a more precise evaluation of PA levels and intensity compared to subjective measures, 26 while subjective measurements are not affected by the type of activity or placement of a wearable. 27
Hence, an up-to-date overview is needed to assess the effectiveness across all digital health interventions, separating subjective and objective PA assessments and contributing knowledge to support patients after CR II completion.
Objective
The purpose of this systematic review and meta-analysis is to summarise the effectiveness of digital health interventions in maintaining objectively and subjectively measured PA (primary outcomes) and physical function, physical fitness, HRQoL and adverse events after completion of phase II CR compared to usual care or minimal intervention.
Methods
Study design
This study was conducted according to the recommendations from the Cochrane Handbook and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement.28,29 The completed PRISMA checklist is available in Appendix A in the online supplemental materials. The review was performed according to the protocol pre-registered with PROSPERO (CRD42023396629).
Eligibility criteria
Population
The study population of each study had to have a mean age of ≥18 years and had to have finished phase II CR after being medically diagnosed with cardiovascular disease or undergoing a heart-related procedure, e.g., coronary artery bypass graft or percutaneous coronary intervention.
Intervention
The studies had to evaluate the effectiveness of digital health interventions, either exclusively or as a multicomponent (e.g., in-person intervention + digital component). The intervention had to aim to increase, maintain or promote PA or physical fitness after CR II completion. Unlike previous reviews by Hannan et al. 24 and Heimer et al., 25 to be included as digital health interventions, interventions had to begin after CR II and not during CR II. This was done to ensure more consistency, homogeneity and comparability across studies.
Comparison
The intervention had to be compared to usual care or minimal intervention (e.g., blinded wearable, sham website or education).
Outcomes
For a study to be included, it had to have at least one outcome on the primary outcome of interest, PA (objectively or subjectively measured), or at least one outcome on secondary outcomes of interest, physical function (objectively or subjectively), physical fitness, HRQoL or adverse events (serious and non-serious). Eligible time points were at the end of intervention, 6 months, and 12-month follow-up after the digital health intervention.
Study types
Eligible study designs had to include randomisation, i.e., randomised controlled trials (RCTs), randomised pilot and feasibility studies, factorial design studies, cluster-randomised trials and cross-over studies (only the period until cross-over was included).
Search strategy, data sources and study selection
Embase (Ovid), MEDLINE (Ovid), CINAHL (Ebsco) and Cochrane Central Register of Controlled Trials (CENTRAL) were searched from inception to the 2nd of August 2023. ClinicalTrials.gov was searched for any ongoing studies. The search was updated 22nd of May 2024. References of eligible studies were hand-searched (‘pearl growing’) by two independent reviewers (AL and TGH). The ‘pearl growing’ approach was used to identify relevant studies, with each study (pearl) containing relevant keywords or citations, which were used to identify and track more relevant studies. 30 The search was validated against included studies from Graham et al. 22 and Martinello et al. 23
The search matrix comprised relevant keywords, MeSH and Thesaurus terms for cardiovascular disease, digital health, maintenance and PA. However, RCT filters31–33 were added to the search strings to reduce the number of non-randomised studies from the search. No limitations or restrictions in relation to the publication date or language were applied. The full search strategy for each database is available in Appendix B in the online supplemental materials.
Studies not reported in English were translated into English via Google Translate. 34 Two reviewers (AL reviewed all studies, while the studies were equally distributed between SRM, TGH and JRA) independently screened the studies. Reviewer disagreements were discussed, and if no consensus could be reached, then a third reviewer (LHT) was consulted. In addition, authors were contacted if full-text articles were inaccessible and had missing data, or if data from their analysis were not reported. A maximum of four email attempts were made: two emails to the corresponding author and two emails to the first, second, or last author. 35
Data extraction
Data extraction was done by the same reviewers as for study selection. The following data were extracted using a priory-defined and pilot-tested extraction template. Data reported only in graphs or figures were extracted using the pilot-tested Web-Based Plot Digitizer. 36 Extracted data included study, author, year of publication, the country where the trial took place, study design, duration of preceding CR, weekly frequency of preceding CR, mean age, the proportion of females, number of participants enrolled, number of participants completing the trial, heart conditions, non-serious adverse events and serious adverse events. Adverse events were extracted if reported explicitly in text or figures/tables, as recommended by The Consolidated Standards of Reporting Trials. 37 The adverse events reported in the flow diagram were not considered explicit, even if there were indications of adverse events. Flow diagrams often lack sufficient details to determine whether an event is directly linked to the intervention and was therefore deemed inappropriate for extraction. The Food and Drug Administration's definition of non-serious (e.g., pain, fatigue or falls) and serious adverse events (e.g., hospitalisation, disability or permanent damage) was used. 38 Primary digital health component (mHealth, eHealth, digital device or a combination), whether the type of digital health intervention was exclusive or combined with in-person intervention, was also extracted. We also extracted the duration, weekly frequency of the intervention, number of personal encounters during the intervention, use of theoretical model or framework, and usual care or minimal intervention (e.g., sham website). Mean and standard deviations (SDs) were extracted for outcomes in the following prioritised order: the primary outcome of interest was PA objectively (e.g., measurements by using an accelerometer) or subjectively (e.g., International Physical Activity Questionnaire) measured. Physical function objectively (e.g., 6-minute walk test) or subjectively (Short-Form 36 physical function score) measured, physical fitness (e.g., peak oxygen uptake (VO2 peak)) and HRQoL (e.g., European Quality of life - 5 Dimensions) were secondary outcomes of interest. If reported, both objective and subjective outcomes were extracted and sought. A pre-specified hierarchy for outcomes was used for data extraction (Table 1).
Outcome prioritisation for data extraction.
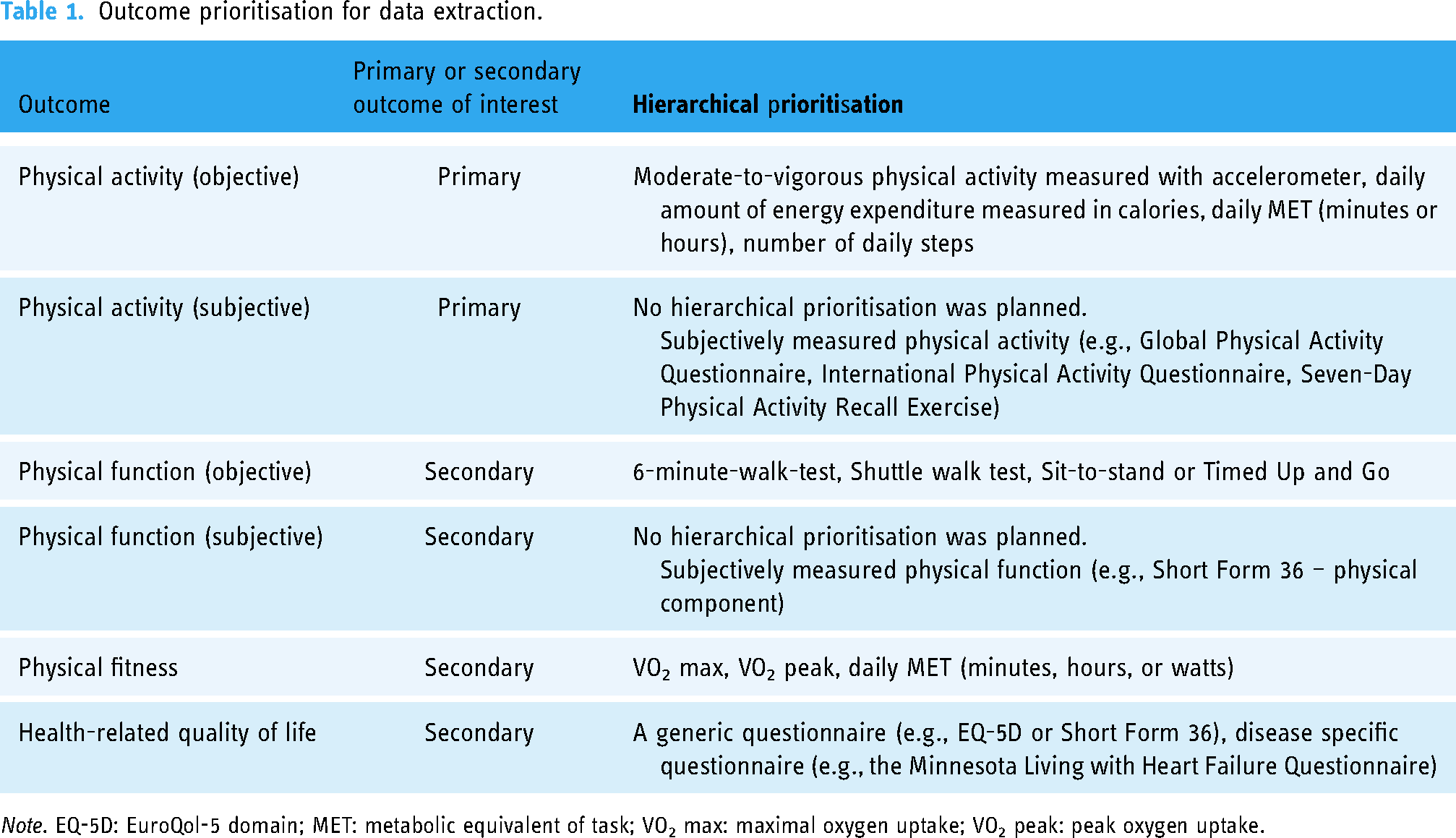
Note. EQ-5D: EuroQol-5 domain; MET: metabolic equivalent of task; VO2 max: maximal oxygen uptake; VO2 peak: peak oxygen uptake.
In addition, data were extracted for each outcome of interest at each assessment point, end-of-intervention, closest to 6-month follow-up and closest to 12-month follow-up. Furthermore, the type of instrument used to measure the outcome and the placement were also extracted. The data extraction form and data are available in Appendix C in the online supplemental materials.
Quality assessment
Risk of bias was assessed for each outcome of interest using the Cochrane Risk of Bias Tool version 2 (RoB 2) for each study. 39 All assessments were made in Excel (RoB 2, 22nd of August 2019, version 9). 40 Studies were assessed by two independent reviewers (AL assessed all studies, while the studies were equally distributed between SRM, TGH and JRA). A third reviewer (LHT) was consulted if a consensus was not reached.
Certainty of evidence
Grading of Recommendations Assessment, Development and Evaluation (GRADE) was used to assess all outcomes at the end of the intervention (assessed by AL and JRA). A summary of findings table was used for reporting the results, along with a separate evaluation of the certainty in evidence for each outcome. Assessments and the summary of findings were made using the online software GRADEpro GDT. 41
Statistical analysis
The effectiveness of the interventions on objectively measured PA, subjectively measured PA, objectively measured physical function, subjectively measured physical function, physical fitness, and HRQoL was evaluated separately. The priory-defined prioritisation was used if more than one relevant outcome was reported within a trial (Table 1). Missing SDs were calculated using reported 95% confidence intervals, p-values or standard errors. 42 If two or more studies reported data on the same outcome of interest using different outcome measures, a random-effects model, adjusting to Hedges’ g using endpoint scores, was used to calculate the effect size. If more than one study reported the same outcome using the same measures, an effect size was calculated as the weighted mean difference (WMD) of the final scores using a random-effects model meta-analysis adjusting to Hedges’ g. If only one study reported data for a specific outcome, a narrative approach was used in accordance with the guidelines from the Cochrane Handbook. 42
Standardised mean difference (SMD) with a 95% confidence interval was calculated for outcome measures of continuous data. Effect sizes pooled as SMD were interpreted as a small effect (0.2), moderate effect (0.5) or large effect (0.8). 43 WMD was calculated for the outcome reported in the same unit. Dichotomous outcomes, e.g., dropouts and adverse events, were analysed and expressed as risk ratios with 95% confidence intervals.
The heterogeneity of the results was examined using the Cochrane Q test and quantified as I2 values and the between-study variance τ2. Arbitrary interpretation of I2: 0% to 40% represents that it might not be important, 30% to 60% may represent moderate heterogeneity, 50% to 90% may represent substantial heterogeneity and 75% to 90% represents considerable heterogeneity. 44 Small study bias was examined by using Eggers's test score (p ≤ 0.05 indicates a small study bias) and visually inspected funnel plots for each outcome.
The statistical software Stata 17 (StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC) was used for all statistical analyses.
As protocolised, heterogeneity was explored on nominal variables (cardiac diagnoses, primary digital health component, intervention type, type of comparator and risk of bias) by sub-group analyses. For continuous data, heterogeneity was explored by sub-analyses using univariate meta-regression on mean age, sex distribution, intervention length and mean baseline PA.
Results
Study characteristics
In 2023, the search identified 16,012 studies, ultimately including 19 studies.45–63 In 2024, the search was updated and identified 1443 studies, ultimately resulting in the inclusion of one study. 64 Among the included studies, there were 18 RCTs and two studies with secondary reporting,51,59 involving a total of 1801 unique participants for analysis. The study flow diagram is visually presented in Figure 1. A summary of the included studies is shown in Table 2. Excluded studies and ongoing studies are available in Appendix D in the online supplemental materials. The mean age of the study participants was 61.2 years, ranging from 57 63 to 66.8. 58 Two of the included studies only included participants with coronary heart disease.46,55 The rest of the studies included a mixed cardiac population.45,47–54,56–64 The length of CR II ranged from 4 weeks 54 to 16 weeks, 50 and the weekly frequency of CR sessions was reported as two times per week48–50,61 or three times per week,52,57,59 while the remaining studies did not report the length of CR II.
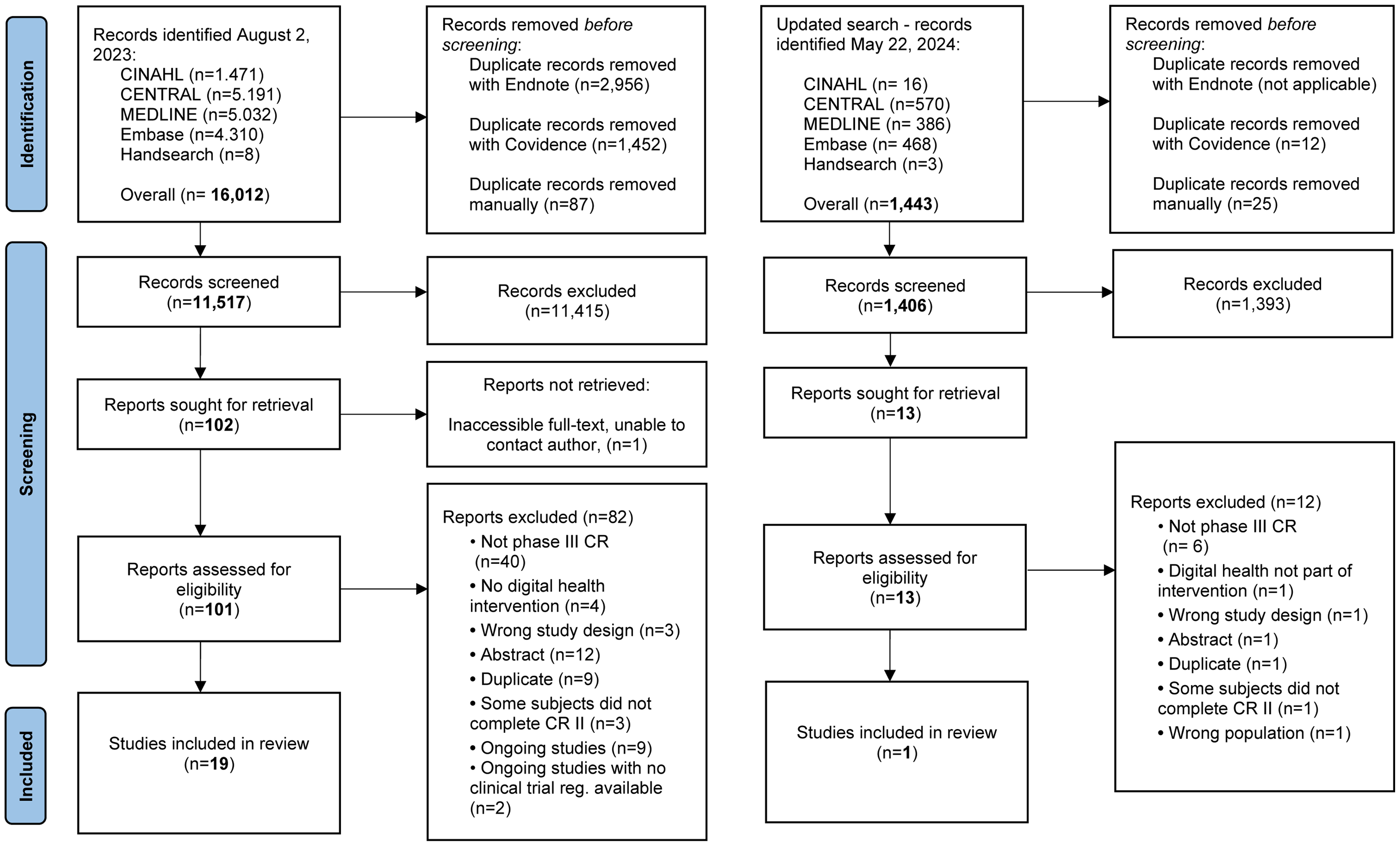
Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram.
Study characteristics of the included studies.

Note. ACS: acute coronary syndrome; AF: atrial fibrillation; AP: angina pectoris; CABG: coronary artery bypass graft surgery; CAD: coronary artery disease; CHD: coronary heart disease; CHF: congestive heart failure; CVD: cardiovascular disease; HF: heart failure; HRQoL: health-related quality of life; MET: metabolic equivalent; MI: myocardial infarction; MVPA: moderate–vigorous physical activity; Obj PA: objective physical activity; Obj PF: objective physical function; PA: physical activity; PCI: percutaneous coronary intervention; PFIT: physical fitness; PTCA: percutaneous transluminal coronary angioplasty; SF-36: Short Form 36; Subj PA: subjective physical activity; Subj PF: subjective physical function; UC: usual care; *: no available information or data.
The median digital health intervention duration was 26 weeks ranging from 6 weeks 48 to 208 weeks. 50 All studies included a mHealth component in their interventions, utilised through SMS,47,54,63,64 phone calls45,47–50,52,57–62 or phone apps.46,55,56,58,64 In the intervention by Avila et al.,51,53 it was not stated whether feedback given via phone utilised SMS or phone calls. The intervention was solely digital in 13 studies,45–49,54–59,61,62 where the interventions primarily involved apps,46,55,56,58,61 wearables (e.g., accelerometers)47,48,57–59,61,62 and web-based tools.54,61 The remaining seven studies50–53,60,63,64 integrated digital health interventions with in-person components, including face-to-face counselling,52,60,63 supervised exercise sessions50,51,53 and exercise booster sessions.50,64 Seven studies based their intervention on a model or a theory45,50,54,55,57,58,60 while the remainder did not. Only six studies reported follow-up after completion of the digital health intervention45,48,49,51,60,61; follow-up durations ranged from 2 weeks 60 to 39 weeks, 51 and no studies reported at the 12-month follow-up.
The primary outcome, objective PA, was measured using accelerometers47,49,51,60,62 and pedometers, 58 primarily reported as moderate PA, 51 moderate to high PA 47 and moderate to vigorous PA.49,60 Furthermore, subjective PA was measured using questionnaires,46,48,50,54,57,61,63,64 primarily reported as leisure PA50,64 and total PA.48,54,61 Of note, secondary outcomes for objective physical function46,51,64 was primarily measured using a 6-min-walk-test;58,64 physical fitness was primarily measured using a VO2 peak,47,51,53,55,56,60,61 and HRQoL was primarily measured using European Quality of life - 5 Dimensions.55,60
Study quality
Risk of bias assessments is shown in Figure 2.

Risk of bias on all included studies and categorised based on the assessed outcome.
Risk of bias assessments for objectively measured PA: 16.5% (1/6) 47 of the RCTs was rated as ‘low risk of bias’, 67% (4/6)49,51,60,62 as ‘some concerns’ and 16.5% (1/6) 58 as ‘high risk of bias’. Subjectively measured PA was rated as ‘some concerns’ in 75% (6/8) of the RCTs46,48,57,61,63,64 and ‘high risk of bias’ in 25% (2/8) of the RCTs.50,54 All (4/4)46,51,58,64 of the RCTs that measured objectively and subjectively (4/4):45,51,53,57 physical function were rated as ‘some concerns’. For physical fitness, 18% (2/11) of RCTs52,55 was rated as ‘high risk of bias’, while the remaining 82% (9/11)46,47,50,51,53,56,60,61,64 was rated as ‘some concerns’. For HRQoL, one RCT – 11% (1/9) 56 was rated as ‘low risk of bias’, 67% (6/9) as ‘some concerns’51,53,59–61,63 and 22% (2/9) as ‘high risk of bias’.50,55
The effects of digital health interventions
Objectively measured PA
Four studies (357 participants) assessed the effect of digital health interventions on PA.47,49,58,62 We found no effect on objective PA, when digital health interventions were compared to usual care or minimal intervention at the end of intervention (K = 4, SMD 0.85, 95% CI: −0.07 to 1.77, I2= 90.37%) (Figure 3). After completing the digital health intervention, at 6-month follow-up, no effect was found between groups (K = 2, SMD 0.02, 95% CI: −0.13 to 0.17, I2= 0.01%) (Fig. E.1).

Forest plots of standardised mean difference for (a) objectively measured and (b) subjectively measured physical activity in digital health interventions at the end of intervention.
Subjectively measured PA
Eight studies (711 participants) investigated the effect of digital health interventions on subjectively measured PA,.46,48,50,54,57,61,63,64 The results showed a small effect on subjective PA when comparing digital health interventions to usual care or minimal intervention at the end of intervention (K = 8, SMD 0.37, (95% CI: 0.05 to 0.69), I2= 73.42%) (Figure 3).
WMD for two studies (136 participants)54,61 using International Physical Activity Questionnaire (IPAQ) showed no effect between groups (K = 2, WMD 1384.55 MET-min/week, (95% CI: −1273.76 to 4042.85), I2= 77.81%) (Fig. E.2). After the digital health intervention, at 6-month follow-up, no effect was found between groups (K = 2, SMD 0.11, (95% CI: −0.25 to 0.48), I2= 43.65%) (Fig. E.3).
Effects of digital health on physical function
Objectively measured physical function
Three studies (161 participants)46,58,64 were included for analysis. A moderate effect for objectively measured physical function was found favouring digital health interventions compared to usual care or minimal intervention at the end of intervention (K = 3, SMD 0.63, (95% CI: 0.03 to 1.24), I2 = 70.73%) (Figure 4). WMD for two studies (131 participants)58,64 using a 6-minute walk test showed no effect between groups (K = 2, WMD 40.57 meter, (95% CI: −17.65 to 98.78), I2= 77.81%) (Fig. E.4). After the digital health intervention, one study 51 measured physical function objectively at 9-month follow-up, reporting no effect on the Sit and Rising test (intervention group: N = 16, Sit and Rising test score 7.9, SD 1.3; control group: N = 18, score 7.3, SD 1.3 (between group p = 0.53)).

Forest plot of standardised mean difference for (a) objectively measured and weighted mean difference for (b) subjectively measured physical function in digital health interventions at the end of intervention.
Subjectively measured physical function
Three studies (191 participants)45,53,57 assessed the effect of digital health interventions on subjective physical function all using the Short Form 36 physical function subscale, showing a WMD of 8.47 points (K = 3, (95% CI: 3.83 to 13.11), I2= 0.00%), favouring digital health interventions compared to usual care or minimal interventions at the end of intervention (Figure 4).
Effects of digital health on physical fitness
Nine studies (748 participants)46,47,50,52,53,55,56,61,64 assessed the effect of digital health interventions on physical fitness. Little to no effect for physical fitness was found, favouring digital health interventions compared to usual care or minimal intervention at the end of intervention (K = 9, SMD 0.19, (95% CI: 0.05 to 0.34), I2= 0.00%) (Figure 5). WMD for the VO2 peak at the end of intervention showed an effect favouring the digital health interventions (K = 5, WMD 1.75 ml/kg/min, (95% CI: 0.47 to 3.02), I2= 0.00%) (Fig. E.5). After the digital health intervention, at 6-month follow-up, this was not maintained (K = 2, WMD 0.67 ml/kg/min, (95% CI: −0.79 to 2.13), I2= 0.00%) (Fig. E.6). The same conclusion was found in one study [51] measuring the VO2 peak at 9-month follow-up (intervention group: N = 26, VO2 peak: 27.1, SD 6.5; control group: N = 25, VO2 peak: 26.2, SD 7.6 (between group p = 0.54)).
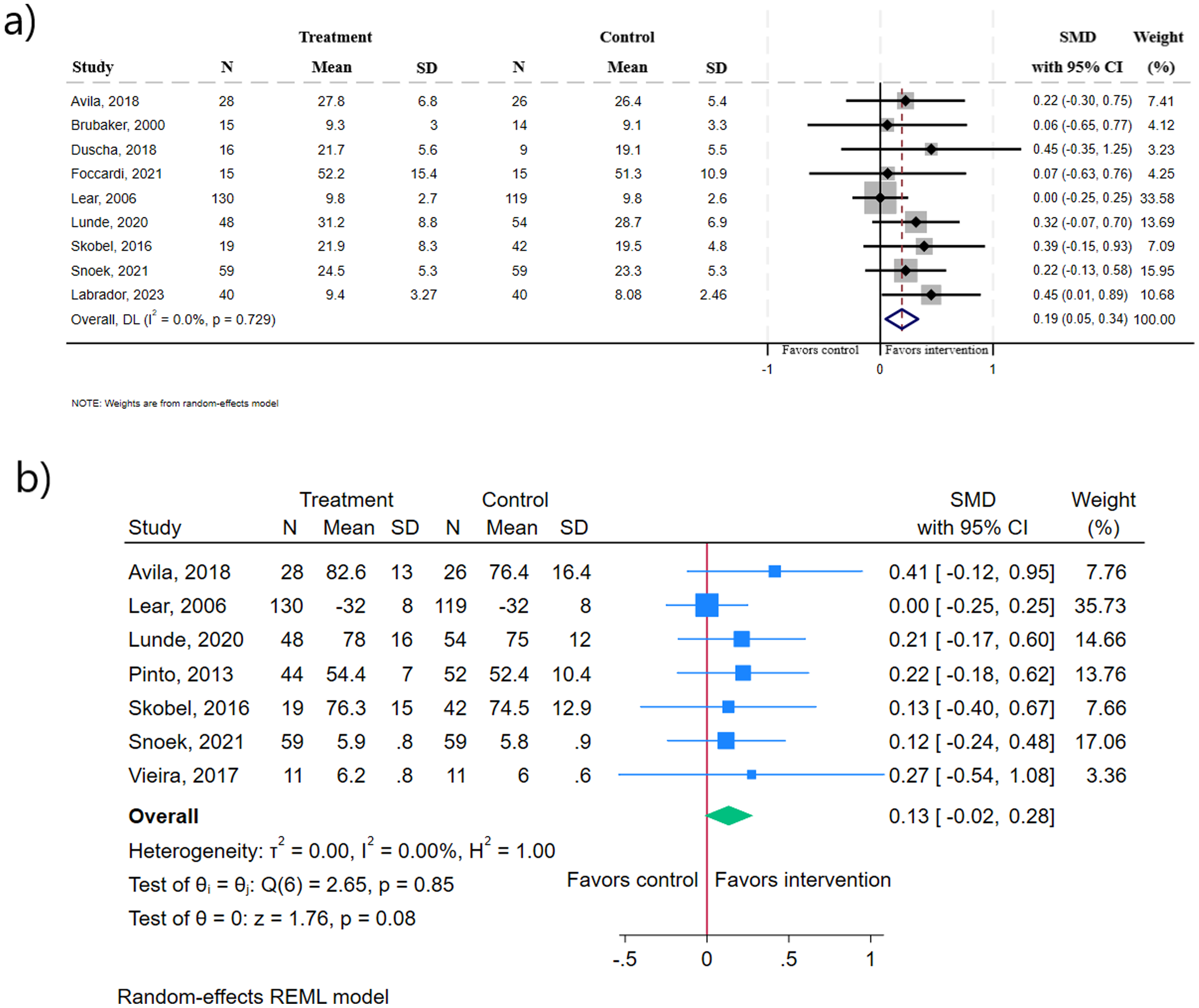
Forest plot of standardised mean difference for (a) physical fitness and (b) health-related quality of life in digital health interventions at the end of intervention. In (a), heterogeneity is reported as I2.
Effects of digital health on health-related quality of life
Seven studies (702 participants)50,53,55,56,59,61,63 investigated the effect on HRQoL. Little to no effect was found for digital health interventions compared to usual care or minimal intervention at the end of intervention (K = 7, SMD 0.13, (95% CI: −0.02 to 0.28), I2= 0.00%) (Figure 5). WMD for two studies (163 participants)55,56 using the EuroQol-5 VAS at the end of intervention showed no effect between groups (K = 2, WMD 2.57 VAS score, (95% CI: −1.81 to 6.95), I2= 00.0%) (Fig. E.7). Two studies60,61 (567 participants) reported data on HRQoL at 6-month follow-up (Fig. E.8), which showed no effect between groups (K = 2, SMD −0.04, (95% CI: −0.20 to 0.13), I2= 0.00%).
Adverse events
In total, seven studies46,53,55–57,61,64 reported on adverse events within their respective study periods, and adverse events were not reported solely in relation to the applied digital health solution. More details are shown in Table 2. The varied reporting methods for adverse events made quantitative analysis unfeasible. A narrative approach was therefore taken instead.
One study 55 reported that 24% (6/25 participants) in the digital health group experienced a serious adverse event (chest pain, infection after coronary artery bypass grafting and chest pain before training) compared to 7% (3/45) of the control group (atrial fibrillation, new angina and pseudo-aneurysm). One study 61 reported the number of study participants presenting with at least one serious adverse event for each of the following categorisations (the same patient could be present in several categories): cardiac-related hospitalisations (intervention: 7; control group: 10), procedures (intervention: 19; control group: 19) and adverse events (intervention: 13; control group: 16). Non-serious adverse events were reported as outpatient visits (intervention: 18; control group: 22) and general practitioner visits (intervention: 43; control group: 46). Lunde et al., 56 Foccardi et al. 46 and Avila et al. (2018) 53 reported adverse events but only if it occurred during or in relation to exercise 53 or exercise testing.46,56 Lunde et al. 56 and Pinto et al. (2011) 57 did not observe any adverse events; however, the flow diagrams mentioned a case of death (intervention: 1; control group: 0 56 ; intervention: 1; control group: 1 57 ) and medical issues (intervention: 8; control group: 6 57 ). Avila et al. (2018) 53 observed no serious adverse events but reported two dropouts in the control group due to percutaneous coronary intervention, indicating a serious adverse event. Labrador et al. 64 observed no adverse events and reported no dropouts.
Heterogeneity
Heterogeneity was only found for objectively, subjectively measured PA and objectively measured physical function. None of the meta-regressions may explain the heterogeneity for objectively or subjectively measured PA, but age and proportion female may explain the heterogeneity for the objectively measured physical function (p = 0.01) (Table F.1–3). Only the pre-specified sub-group analyses (Fig. F.1–15) for the primary digital health component (mobile, internet based or digital device) (p = 0.03) may explain some of the heterogeneity for objective PA, and the type of cardiac diagnosis (p = 0.01) and the type of digital health use (exclusively or as component) may explain some of the heterogeneity for subjective PA (p = 0.05). For objective physical function, only the primary digital health component (p = 0.01) may explain the heterogeneity.
Evidence level and small study bias
The certainty of evidence was very low for objective and subjective PA and objective physical function and low for subjective physical function, physical fitness and HRQoL. The certainty of evidence was (Table 3) evaluated accordingly to GRADE.
Summary of the findings – the effectiveness of digital health interventions after CR phase II completion compared to minimal intervention or usual care.

Note. CI: confidence interval; MD: mean difference; RCTs: randomised controlled trials; SMD: standardised mean difference.
Majority of studies had some concerns.
I2 is considerable >90%.
CI overlaps the line of no effect.
Indicated by the funnel plot, Egger's test score and the number of studies.
I2 is substantial >50%.
Important outcome for patients with cardiovascular disease.
Visual inspection of funnel plots (Fig. F.16–21) only indicated publication bias in the outcome domain for objective and subjective PA, which was confirmed by the p-values calculated by Egger's test: subjective PA (p = 0.0001); objective PA (p = 0.004).
Discussion
We assessed the effectiveness of digital health interventions on PA, other physical outcomes, HRQoL and adverse events after CR II completion. Digital health interventions supporting PA maintenance showed a small effect on subjectively assessed PA but no effect on objectively assessed PA at the end of the digital health intervention or at 6-month follow-up. However, the evidence was very uncertain. For physical function, digital health interventions showed a moderate effect, whereas digital health interventions may have little to no effect on physical fitness and HRQoL at the end of intervention. The quality of evidence was low to very low, with few studies reporting results at 6-month follow-up, and none at 12-month follow-up. Furthermore, adverse events were poorly reported. High-quality studies on digital health interventions with longer follow-up periods are needed to investigate which type of digital component can provide the most optimal support to maintain PA health behaviour.
PA is a core component in rehabilitation for patients with cardiovascular disease as it positively affects prognosis and HRQoL, and is crucial for reducing mortality.1,2,65 Furthermore, PA is an important secondary prevention component essential for managing and prolonging a healthier life.1,66 Based on the current evidence, we cannot confirm or deny findings from previous systematic reviews investigating the effect of digital health intervention to support patients’ maintenance of PA after CR II completion. Martinello et al. 23 (2019) reported that either in-person or digital health (i.e., pedometer, telemonitoring, online or mobile component) maintenance interventions had a large effect on PA levels. Unfortunately, they did not conduct a sub-analysis on the mode of delivery of digital health interventions and in-person interventions in their PA analysis, which is why a conclusion of an isolated effect on digital health interventions cannot be drawn from this study. 23 Heimer et al. 25 (2023) reported a small effect on PA levels from eHealth interventions that aimed to maintain PA after CR comparison to usual care. However, a direct comparison with Heimer et al. 25 is not appropriate, as they combined subjective and objective measurements in their analysis. In contrast to our review, all previously mentioned reviews either combined the subjective and objective outcome data in their PA analysis or reported exclusively on objective PA. Objective measurements provide a more precise evaluation of PA levels and intensity compared to subjective measures, which tend to overestimate these parameters. 19 Subjective measurements, however, capture the individual's self-perceived activity level. 26 Hence, subjective and objective assessment tools on PA assess two different aspects of PA, which is why it is important to analyse them separately. 26 Hence, by separating subjective and objective PA measurements and adding a broader spectrum of digital health interventions, our findings add important knowledge to the existing literature.
In our PA analysis, we observed a high level of heterogeneity, which affected our ability to conclude the overall effects. Despite our planned sub-analyses, the underlying cause remained unidentified. However, some of the heterogeneity could be explained by how PA was measured, e.g., type and placement of the PA assessment wearables, i.e., on the wrist 58 or the waist.49,58,62 In the study by Park et al., 58 PA was assessed using a Fitbit device for the intervention group (worn on the waist) and a pedometer for the control group (worn on the wrist), introducing measurement bias into the meta-analysis of objective PA. The placement of the wearable may affect the ability to detect certain activities. 27 Different wearables may have different cut-points for activity-related intensity levels, and may, therefore, have different abilities in measuring PA. 67 Some of the heterogeneity may also be explained by the type of cardiac diagnosis, type of primary used digital component (mobile, internet based or digital device), and type of intervention (exclusively digital health or digital health + in-person). Heterogeneity may also be explained by the complexity of the digital health interventions, which vary across studies. Some interventions only include phone calls, 45 while others incorporate virtual reality headsets. 63 The number of digital health components employed also varies; for instance, some studies include only a single digital health component, such as a phone call, 49 whereas others involve two components (wearable devices and phone calls),47,62 and some incorporate three components (wearable devices, apps and phone calls). 58 The impact of all these varying aspects should be considered and explored in future studies.
Our findings of little to no effect for physical fitness are similar to the overall findings by Heimer et al. 25 The WMD analysis for the VO2 peak illustrates an increase of 1.75 ml/kg/min favouring digital health interventions. Typically, an increase of 3.5 ml/kg/min (1 MET) is considered clinically relevant as it has been shown to improve the survival rate by 12% in patients with cardiovascular disease.68,69 However, an increase of 1 ml/kg/min has been associated with a 15% decrease in risk for all-cause and cardiovascular disease-specific mortality. 70 Therefore, the increase of the 1.75 ml/kg/min may have some clinical relevance. At 6-month follow-up, the effect was not sustained (WMD of 0.67 ml/kg/min).
We found three studies indicating a moderate effect of digital health intervention on objective physical function. In contrast, Hannan et al. 24 found only one study that assessed physical function using the 6-minute walk test, but they found no between-group difference at 6 weeks or 6-month follow-up. Thus, the limited evidence on digital health maintenance interventions makes the effectiveness of physical function uncertain. Qualitative research indicates that digital health could potentially support patients and may improve their HRQoL. 71 Yet, it is not supported by our findings. Improving well-being and quality of life for individuals with cardiovascular diseases is the overarching aim of cardiac rehabilitation. 5 Exploring various ways in which digital health interventions can contribute to enhancing patients’ HRQoL should be investigated.
For the control group, measurements of PA were only considered minimal intervention if participants in the comparator group were not blinded to their PA levels; e.g., Park et al. 58 was considered minimal intervention as the control group monitored PA using a pedometer and recorded their steps in a diary. If both groups were unaware of their PA levels, the Hawthorne effect 72 should be present in both groups. In some studies,47,48,51,53,62 intervention group participants were aware of their PA levels during measurements and received feedback on the prior week's performance. In the intervention group, this feedback or awareness of PA levels was expected to have an additional effect beyond the Hawthorne effect.
We define maintenance as a period of at least 6 months with the goal of keeping the benefits gained and preventing relapse. 16 Our analyses show the trajectory of effectiveness of digital health analysed at the end of the intervention, closest to 6-month follow-up, and closest to 12-month follow-up. However, our findings show that most of the included studies assessed outcomes only at the end of the digital health intervention.46,47,50,52,54–56,58,62–64 Despite this, all of the studies used the maintenance term, except two,55,63 to describe their intervention while having no follow-up46,47,50,52,54,56,58,62,64 or a short follow-up period (under 6 months).45,48,49,60,61 One could question whether the included studies truly investigated maintenance. No studies clearly defined when maintenance was considered successful. We recommend future studies to include longer follow-up periods and clearly describe their definition for maintenance.
An interesting observation was that in studies where exercise was prescribed at the start of the digital health intervention followed by weekly47,53,54,58,64 or bi-weekly 62 contact through text messages,47,53,54,64 messages in app, 58 phone calls47,62,64 or emails, 53 the effect size tends to be higher. This is contrary to studies where digital contact with the patients was only made through phone calls, averaging throughout the intervention duration every 2 weeks, 61 4–5 weeks, 48 8 weeks 49 and 11 weeks. 50 Hence, associations between intervals of digital interactions and patient-related outcomes are worth exploring more in depth.
Implications for clinical practice and future research
The aim of our review was not to understand the deeper effective mechanisms when applying digital health interventions to patients with cardiac diagnoses. However, to tailor effective future digital interventions and help explain some of the current heterogeneity, which is an important research area. Digital health is accessible for many and can be tailored to patients’ individual needs and preferences, encouraging healthy behaviour.19,20 Current evidence highlights that digital health interventions applied to support PA after phase II CR are heterogeneous in type and often combine several types of behavioural change techniques. 25 Incorporating behaviour change theory and behavioural change techniques into an intervention has been found to encourage changes in health-related behaviour, especially social support and action planning have been found effective in improving PA. 73 Still, the research field needs to explore the optimal dose and combination of digital health to promote or maintain PA, as numerous combinations and scant evidence in the field of digital health interventions compromise any solid clinical recommendation. Nevertheless, given that our data suggest a favourable effect on PA and physical function, and that digital health solutions were previously reported to be cost-effective for improving health-outcomes 74 and could improve accessibility in healthcare for rural or remote populations, 75 healthcare providers could consider to implement digital health into clinical practice. Yet, it is important to tailor interventions based on patients’ preferences and needs, and healthcare providers should consider that digital health might not be the optimal solution for all patients. 76 The evidence includes limited reporting on adverse events, and the possibility of adverse events during a digital health intervention cannot be dismissed. Still, given the risks associated with inactivity, the benefits of increasing or maintaining exercise seem to outweigh any potential associated risks.1,2 However, future studies need to enhance their reporting on adverse events following the recommendations presented by The Consolidated Standards of Reporting Trials. 37 Future studies should consider the complexity, the number of digital health components used, the intervals between digital interactions with the study participants, the duration of the digital health intervention and using a follow-up period of at least 6 months after completing the digital health intervention.
Limitations
The present systematic review has several strengths. First, our review was pre-registered at PROSPERO and adhered to PRISMA guidelines, ensuring transparency and completeness in reporting. Second, only RCTs were included. Third, the methodology followed the recommendations from Cochrane. 77 Last, our search was broader than previous systematic reviews, identifying seven additional relevant studies46,52,58–61,64 compared to existing reviews within the same field,22–25 and we divided outcomes in objective and subjective PA assessment types. 26
Nevertheless, our review holds some limitations. First, our PA analyses had considerable heterogeneity. The Cochrane Handbook advises against performing subgroup analyses, meta-regressions or funnel plots when the number of studies is fewer than ten. 44 Therefore, until further research explores the heterogeneity further, our results should be interpreted with caution. Second, digital health interventions were primarily investigated among males, and the effect was not reported separately for males and females. Third, although we tried to mitigate the risk by manually searching references; there is a possibility that eligible studies might have been missed due to the use of RCT filters31–33 in a search block. Fourth, the exclusion of studies that introduced digital health interventions during phase II CR limits our understanding of how early digital health integration affects patients during the transition between CR phases and its impact on long-term maintenance. Last, we only covered physical outcomes and HRQoL. Hence, the effectiveness of digital health maintenance interventions on other important outcomes such as return-to-work, anxiety, depression, re-hospitalisation and death were not investigated.
Conclusion and perspective
Current evidence shows that digital health interventions in patients with cardiovascular disease has a small effect on subjectively measured PA and a moderate effect on objective physical function but no effect on objectively measured PA and little to no effect on physical fitness and HRQoL compared to usual care or minimal intervention after completing phase II CR. However, the evidence is very uncertain across all outcomes, with few available studies, and potential harms poorly reported. Despite uncertain evidence, the health risks induced by physical inactivity, coupled with the potential health benefits of digital health interventions, may still justify the use of digital health in clinical practice to maintain PA until further evidence is available.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Supplemental Material
sj-xlsx-3-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-xlsx-3-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Supplemental Material
sj-docx-4-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-docx-4-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Supplemental Material
sj-docx-5-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-docx-5-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Supplemental Material
sj-docx-6-dhj-10.1177_20552076241286641 - Supplemental material for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis
Supplemental material, sj-docx-6-dhj-10.1177_20552076241286641 for The effectiveness of digital health interventions for the maintenance of physical activity following cardiac rehabilitation: A systematic review and meta-analysis by Alexander Luijk, Sofie Rath Mortensen, Trine Grønbek Hamborg, Graziella Zangger, Jonas Risum Ahler, Jan Christensen, Søren T Skou and Lars Hermann Tang in DIGITAL HEALTH
Footnotes
List of abbreviations
Acknowledgements
We want to express gratitude to Konstantinos Antypas, Sherry L. Grace and Jomme Claus for providing us with additional data and information. The authors used a large language model (ChatGPT, version 3.5) solely to improve grammar and structure. The authors verified the accuracy and appropriateness of any suggested corrections proposed by ChatGPT. All content, analyses, and conclusions presented in this review were solely the work of the authors and were not influenced by large language models or artificial intelligence.
Contributorship
All authors contributed to the development of the protocol. AL, GZ, JC, STS and LHT contributed to the design. AL performed the systematic literature search, and AL, SRM, TGH and JRA screened the studies, extracted the data and assessed the studies using RoB 2. AL and JRA assessed the certainty of evidence using GRADE. AL, GZ and JC performed the meta-analysis, and AL, JC, STS and LHT interpreted the results. AL and LHT wrote the manuscript. All authors contributed to the revision of the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
This study is a systematic review and meta-analysis of existing literature, and no ethics approval or consent to participate was required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Region Zealand funded the project (Exercise First research program), but was not involved in any part of designing, conducting or reporting the study. Luijk, Dr Tang and Dr Skou were funded by a grant from Region Zealand (Project: Exercise First). Dr Tang was further funded by The Danish Health Confederation through the Development and Research Fund (project no. 2703) and the Næstved-Slagelse-Ringsted Hospitals research fund, Denmark (project no. A1277). Dr Skou was further funded by two grants from the European Union's Horizon 2020 research and innovation program – one from the European Research Council (MOBILIZE, grant agreement no. 801790) and the other under grant agreement no. 945377 (ESCAPE).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
