Abstract
Background and Aims
Prediabetes, characterized by elevated blood glucose levels below diabetes diagnostic thresholds, presents a significant global health challenge due to high prevalence and associated risks for developing type 2 diabetes. Traditional diabetes prevention programs (DPPs) often face challenges in scalability and accessibility, particularly in underserved communities. Digital therapeutics (DTx), like TH-001, offer potential scalable solutions requiring clinical validation. This randomized, open-label, controlled, parallel-group clinical trial aimed to evaluate the efficacy and safety of TH-001, a digital therapeutic application, reducing hemoglobin A1c (HbA1c) among adults with prediabetes over a 90-day intervention period.
Methods
68 eligible adults were randomly assigned to either standard care alone or standard care plus the TH-001 intervention. Primary outcomes measured were glycaemic parameters including HbA1c, fasting glucose, 2-h post-load glucose from an oral glucose tolerance test, and insulin levels, while secondary outcomes included anthropometric measures. User engagement metrics were also assessed.
Results
The intervention significantly reduced HbA1c levels (mean difference −0.22%, p = .023) and body weight (−2.53 kg, p < .001) compared to the control group. Although fasting glucose and insulin levels showed positive trends, differences were not statistically significant. User engagement was high, correlating strongly with HbA1c reduction (r = .95, p < .001), demonstrating the critical role of sustained interaction with TH-001.
Conclusions
TH-001 demonstrates significant effectiveness in HbA1c reduction and body weight, with strong user engagement suggesting it may effectively overcome traditional barriers of DPPs. Long-term studies are needed to confirm sustained benefits.
Efficacy and Safety of Digital Therapeutic in Adults with Prediabetes; https://clinicaltrials.gov/study/NCT05668819; ClinicalTrials.gov ID: NCT05668819
Keywords
Introduction
Prediabetes is a condition characterised by blood glucose (BG) levels that are elevated above normal but remain below the diagnostic threshold for type 2 diabetes. Prediabetes poses a significant global health challenge, with a high prevalence and increasing incidence, imposing a substantial burden on healthcare systems worldwide. Individuals with prediabetes are at significantly increased risk of progressing to type 2 diabetes, 1 a chronic metabolic disorder with numerous long-term complications, including cardiovascular disease, kidney disease, nerve damage, and blindness. 2 There is increasing evidence to suggest that diabetes also increases the risk of malignancy,3,4 stroke 5 and dementia. 6 Early intervention in prediabetes is crucial for preventing or delaying the onset of type 2 diabetes and its associated complications. 7
Previous studies have demonstrated that lifestyle changes, such as increased physical activity, dietary modifications, and behavioural support, can significantly reduce the incidence of diabetes in high-risk populations. Notably, the Finnish Diabetes Prevention Study (DPS) and the Diabetes Prevention Program (DPP) in the United States have shown that structured lifestyle interventions can lower diabetes onset by approximately 58% over a three-year period.8,9
As a result, health systems worldwide have implemented structured lifestyle change programs, such as the National Health Service Diabetes Prevention Program (NHS DPP) in the UK and similar efforts in other countries.10–12 These programs are delivered through in-person coaching, remote interventions, or digital platforms that require session-based human communication. Although traditional DPPs have shown effectiveness, they frequently rely on considerable human resource involvement, which can hinder accessibility and scalability. In addition, they may particularly struggle to reach individuals in underserved communities or those with limited resources. 13
Digital therapeutics (DTx) have emerged as a promising new tool to address these challenges and transform the management of chronic metabolic diseases. DTx are software-driven solutions to prevent, manage, or treat medical conditions. 14 Unlike traditional digital health applications, DTx solutions undergo rigorous clinical validation and regulatory approval processes to ensure their efficacy. Recent clinical trials have demonstrated the effectiveness of digital lifestyle interventions in improving glycaemic control and diabetes management.15–17 Block et al. reported a significant hemoglobin A1c (HbA1c) reduction of 0.26% (p = .002) using a digital intervention based on CDC DPP principles. 18 Similarly, Katula et al. observed improvements in glycaemic control with internet-based interventions delivered via mobile devices. 19
TH-001 is a DTx designed to support individuals with prediabetes through behavioural guidance, daily tasks, virtual scoreboards, gamification techniques, structured education, and self-monitoring tools to create lifestyle modification and behaviour change. In this study, we evaluate the impact of TH-001 on glycaemic control and metabolic parameters among adults with prediabetes.
Methods
Study design
This open-label, parallel group study randomized participants 1:1 to the intervention (TH-001 plus standard care) or control (standard care) group for 90 days using a computer-generated sequence with block randomization (block sizes 4, 6, and 8) to ensure balanced groups and allocation concealment. An independent statistician generated the sequence, concealed via sealed envelopes opened post-consent. Bias was minimized with objective endpoints (HbA1c and glucose), standardized procedures, and a centralized randomization system to prevent allocation foresight. The parallel design avoided carryover effects and contamination. Data were entered into a secure electronic database with automated validation to prevent data entry errors. The study adhered to consolidated standards of reporting trials guidelines, with a flow diagram in Figure 1 and a checklist in supplementary material.

Consolidated standards of reporting trials flow diagram illustrating the process of participant enrolment, allocation, follow-up, and analysis within a randomised clinical trial. Of the 119 individuals assessed for eligibility, 51 were excluded due to criteria such as HbA1c levels outside the specified range (n = 21), lack of smartphone or digital literacy (n = 9), presence of disqualifying comorbidities (n = 7), participation in a competing trial (n = 4), unwillingness to commit time (n = 7), or administrative/other issues (n = 3). The remaining 68 participants were randomly assigned to either the intervention group (n = 33), which received TH-001 in addition to standard care, or the control group (n = 35), which received standard care alone. During the follow-up period, five participants from the intervention group and seven from the control group were lost to follow-up, with reasons including withdrawal of consent, loss to follow-up, protocol violation, relocation, and medical events. Ultimately, 28 participants from each group successfully completed the trial.
The study has been reviewed and given a favourable opinion by the Istinye University Clinical Research Ethics Committee (3/2022-K-15, 3/2022.K-94, 11/2023.K-68), prospectively registered at www.clinicaltrials.gov (Identifier: NCT05668819) and conducted in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki. All participants provided written informed consent before enrolment.
Inclusion and exclusion criteria are presented in Table 1. Eligible participants met the American Diabetes Association (ADA) Prediabetes Diagnostic Criteria, including fasting glucose levels of 100–125 mg/dL, oral glucose tolerance test (OGTT) 2-h BG of 140–199 mg/dL, or HbA1c between 5.7% and 6.4% and being able to use a smartphone with an iOS or Android operating system. Major exclusion criteria included a prior diagnosis of diabetes mellitus, having a disease that could cause prediabetes, or taking medication that may induce prediabetes.
Inclusion and exclusion criteria.

Abbreviations: ADA: American Diabetes Association; FPG: fasting plasma glucose; OGTT: oral glucose tolerance test; HbA1c: hemoglobin A1c; iOS: iPhone operating system by Apple Inc.
Study endpoints
The primary endpoint of the study is the mean change in glycaemic parameters (fasting BG, 2-h post-load glucose from an OGTT, HbA1c, and fasting plasma insulin) from baseline to Day 90. The secondary endpoint is the mean change in anthropometric parameters (height, body weight, body mass index [BMI], waist circumference, and waist-to-hip ratio) from baseline to Day 90. Safety assessments included adverse events (AEs) and adverse device effects (ADEs).
TH-001; digital therapeutic intervention
TH-001 is a digital therapeutic software application intended to be used by adult patients diagnosed with prediabetes to develop lifestyle changes for the treatment of prediabetes. It is a stand-alone software that provides a 90-day treatment program consisting of six modules, with each module being activated upon completion of the previous module. The content of the software is aligned with a cognitive behavioural approach that helps users change negative thoughts, break unhealthy habits, and aids in establishing new habits that are to be gradually sustained in the long term. The gamification element of the software makes the journey engaging and motivating for the patients, and physician oversight allows doctors to monitor patient activity remotely in a real-time setting.
There are two daily tasks to be completed every day: a physical activity-related task and a nutrition game. While the physical activity task aims to accumulate at least 150 min per week of moderate-intensity physical activity (brisk walking), the nutrition game aims to teach the patient about healthy eating via a food-card swipe game, which asks the patient about their food intake over the course of the last day. The game creates a tracking list of nutrient groups with green light/ red light visualisation to reflect the nutrient groups that should be avoided and provides insights into the patient's eating habits. The nutrition game is followed by at least three nutrition-based questions to increase awareness of healthy eating.
During TH-001 usage the patient also has access to learning modules which can be accessed at any time. The patient can navigate through the educational content, and the content is designed to provide information covering various aspects of prediabetes and diabetes, as well as information covering the benefits of increased physical activity and medical nutrition.
Study procedures
Participants first underwent a screening process to determine eligibility based on predefined inclusion and exclusion criteria. Those who met the criteria were enrolled and randomised in a 1:1 ratio into either the intervention or control group. The intervention group received TH-001 on top of standard care, and the control group received standard care alone.
In this study, standard care was defined as the routine clinical management for prediabetes provided at our centre. This included an initial consultation with an endocrinologist and individualised dietary and lifestyle counselling by a dietitian at the start of the study.
At the baseline visit, all participants underwent a clinical assessment, including anthropometric measurements (body weight, height, BMI, waist circumference, and waist-to-hip ratio) and laboratory evaluations (fasting BG, HbA1c, fasting plasma insulin, and 2-h post-load glucose from an OGTT). Throughout the study, engagement and adherence metrics were remotely monitored via app analytics.
All participants attended an in-person follow-up visit on Day 90, during which changes in metabolic and anthropometric parameters were evaluated as part of the endpoint assessments. AEs and ADEs were documented at the Day 90 visit. To ensure comprehensive safety monitoring, a robust adverse event protocol was implemented, incorporating systematic enquiry at each visit to proactively identify any potential issues. At baseline and the Day 90 follow-up visit, participants were systematically questioned using a standardized questionnaire to capture any AEs or ADEs, including their nature, onset, duration, severity, and resolution. All reported events were documented using the Medical Dictionary for Regulatory Activities (MedDRA) terminology to ensure consistency and facilitate accurate classification. The relationship of each event to the TH-001 intervention was assessed by an independent safety monitoring committee, which applied a standardized causality algorithm considering factors such as temporal association, biological plausibility, and dechallenge/rechallenge data where applicable. Events were classified as unrelated, possibly related, probably related, or definitely related to the intervention, with all assessments reviewed by the principal investigator. Additionally, participants had access to a dedicated technology support team to report any usability concerns or technical issues related to the digital intervention.
Statistical considerations
The sample size was estimated using the assumption that the mean HbA1c difference between groups would be 0.4% with a common standard deviation of 0.5, which is based on similar studies from the literature.16,20 With a type 1 risk of 5.0% (two-sided), a type 2 risk of 20.0%, and a dropout rate estimated at 20.0%, each group would need 31 subjects based on a 1:1 randomisation.
Data management and analysis were conducted using the Statistical Package for the Social Sciences (SPSS, IBM Corp.) version 27. Normal distribution and homogeneity of the results were evaluated using Shapiro‒Wilk and Levene's test, respectively.
For normally distributed data with homogeneous variances, chi-square tests were used for categorical variables to evaluate differences in demographic characteristics between the intervention (TH-001 plus standard care) and control (standard care alone) groups at baseline. To assess changes in continuous outcomes (HbA1c, fasting BG, 2-h post-load glucose, fasting plasma insulin, and anthropometric measures) from baseline to Day 90, a repeated-measures analysis of variance (ANOVA) was employed, with time (baseline, Day 90) as the within-subject factor and group (intervention, control) as the between-subject factor. This approach evaluated both within-group changes over time and between-group differences in change scores. When significant effects were detected, post hoc pairwise comparisons were conducted using the Tukey Honestly Significant Difference (HSD) test to identify specific differences. To account for multiple endpoints, the Bonferroni correction was applied to maintain the family-wise error rate at 5.0%. If normality or homogeneity assumptions were violated, nonparametric alternatives were used: the Mann‒Whitney U test for between-group comparisons at each time point and the Wilcoxon signed-rank test for within-group changes from baseline to Day 90. Statistical significance was defined as a p-value less than 0.05 for all analyses, adjusted as necessary for multiple comparisons.
To address missing data due to dropouts (15% in the intervention group and 20% in the control group), multiple imputation was used to estimate missing values for continuous outcomes at Day 90, assuming data were missing at random (MAR). Five imputed datasets were generated using predictive mean matching, incorporating baseline characteristics and available outcome data. Pooled estimates were analysed using repeated-measures ANOVA. To clarify, no intermediate weekly data were collected for primary or secondary endpoints, as assessments were conducted only at baseline and Day 90. To assess the robustness of findings to different missing data assumptions, sensitivity analyses included: a complete case analysis, including only participants with data at both baseline and Day 90; a worst-case scenario analysis, where missing Day 90 values in the intervention group were imputed with the control group's mean change plus one standard deviation, and vice versa; and a last observation carried forward (LOCF) approach for missing Day 90 data, using baseline values as a conservative estimate. Additionally, to evaluate the impact of outliers, a sensitivity analysis was conducted by excluding participants with outcome values (e.g. HbA1c, weight) beyond 1.5 times the interquartile range from the first or third quartile in each group, with results compared to the primary analysis to assess robustness. These sensitivity analyses confirmed the stability of the primary findings under varying assumptions.
Study organisation
This study is sponsored by Tesu Sağlık Teknolojileri A.Ş. The research was conducted at the Istinye University Health Application and Research Centre Hospital in Gaziosmanpaşa, Istanbul, Türkiye.
Results
Enrolment
Between February and September 2024, a total of 119 patients were evaluated for eligibility. Out of these, 51 patients did not meet the inclusion criteria, leaving 68 subjects randomly assigned to the intervention group (n = 33) and the control group (n = 35). During the 90-day treatment period, five subjects (15%) in the intervention group and seven subjects (20%) in the control group were lost to follow-up. Consequently, the final number of participants who completed the study was 28 in each group (Figure 1). Upon completion of the study, data from the participants who completed the trial were included in the final analysis.
Demographics and baseline characteristics
Demographics and baseline characteristics are summarised (Table 2). The demographic and baseline clinical characteristics of the participants were comparable between the control (n = 35) and intervention (n = 33) groups. The mean age was 49.0 ± 12.4 years in the control group and 48.3 ± 12.0 years in the intervention group (p = .814). The proportion of female participants was slightly higher in both groups (77.1% in the control group vs. 75.7% in the intervention group, p = .894). Anthropometric measurements, including weight, BMI, waist circumference, hip circumference, and waist-to-hip ratio, showed no statistically significant differences between the groups. The mean weight was 82.2 ± 12.1 kg in the control group and 88.2 ± 18.8 kg in the intervention group (p = .127), while BMI was 31.3 ± 4.1 kg/m² and 32.7 ± 7.3 kg/m², respectively (p = .323). Glycaemic control parameters, including HbA1c, fasting glucose, post-prandial glucose, and insulin levels, were also similar between groups. The mean HbA1c was 5.7 ± 0.3% in the control group and 5.8 ± 0.4% in the intervention group (p = .667). Fasting glucose levels were 104.5 ± 9.0 mg/dL and 107.1 ± 9.9 mg/dL, respectively, in the intervention and control group (p = .255). Post-prandial glucose levels were 137.0 ± 31.3 mg/dL in the control group and 140.0 ± 26.1 mg/dL in the intervention group (p = 0.717). Insulin levels also showed no significant difference (p = .325).
Demographics and baseline characteristics.
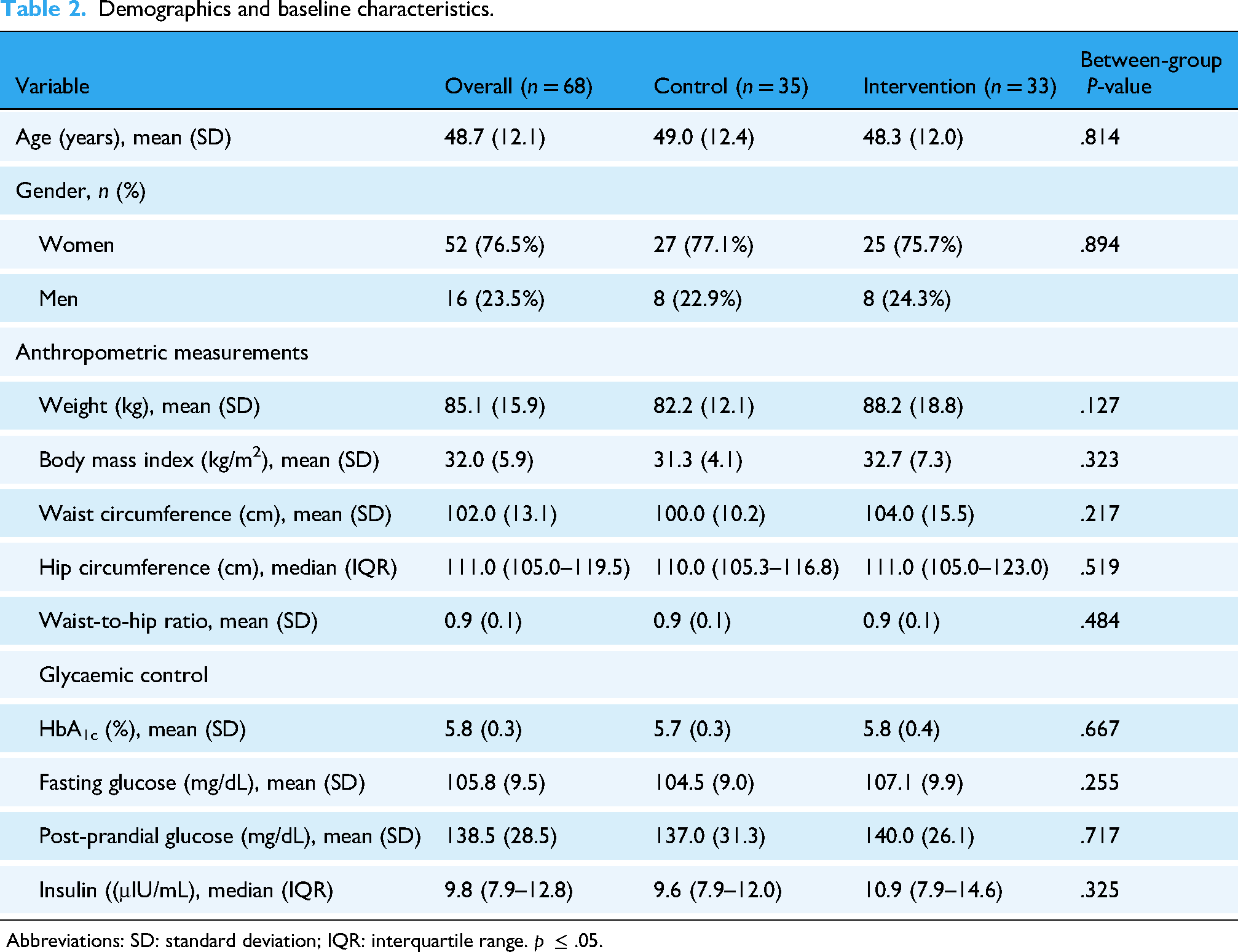
Abbreviations: SD: standard deviation; IQR: interquartile range. p ≤ .05.
The baseline characteristics of participants who dropped out (n = 12) versus those who completed the trial (n = 56) were compared to assess potential attrition bias in our trial (Table 3). No significant differences were observed between the two groups across key variables, including age (47.2 ± 11.8 vs. 49.0 ± 12.2 years, p = .632), gender distribution (75.0% vs. 76.8% female, p = .890), anthropometric measures such as weight (87.3 ± 16.4 vs. 84.6 ± 15.7 kg, p = .574) and BMI (33.1 ± 6.2 vs. 31.8 ± 5.8 kg/m², p = .491), and glycaemic control parameters like HbA1c (5.9 ± 0.4 vs. 5.8 ± 0.3%, p = .456) and fasting glucose (106.5 ± 10.1 vs. 105.6 ± 9.4 mg/dL, p = .753).
Baseline characteristics: dropouts vs. completers.

Abbreviations: SD: standard deviation; IQR: interquartile range. p ≤ 0.05.
Note: P-values are derived from chi-square tests for categorical variables (gender) and t-tests or Mann–Whitney U tests for continuous variables (age, anthropometric measurements, and glycaemic control), depending on normality and variance assumptions.
Outcomes
HbA1c levels showed a statistically significant reduction in the intervention group compared to the control group, with a mean difference of −0.22% (95% CI: −0.41 to −0.031, p = .023) (Figure 2(a)). Fasting glucose levels decreased in the intervention group (−3.50 mg/dL, SD: 10.22), while the control group showed an increase (+2.00 mg/dL, SD: 13.73). However, the between-group difference was not statistically significant (mean difference: −5.50 mg/dL, 95% CI: −11.99 to 0.99, p = .095). Postprandial glucose levels showed no significant difference between groups, with a mean difference of 1.23 mg/dL (95% CI: −22.36 to 24.81, p = .916). Insulin levels decreased more in the intervention group (−2.10 µU/mL, SD: 5.91) compared to the control group (0.43 µU/mL, SD: 4.27). The between-group difference was −2.53 µU/mL (95% CI: −5.33 to 0.27, p = .075), which was not statistically significant.
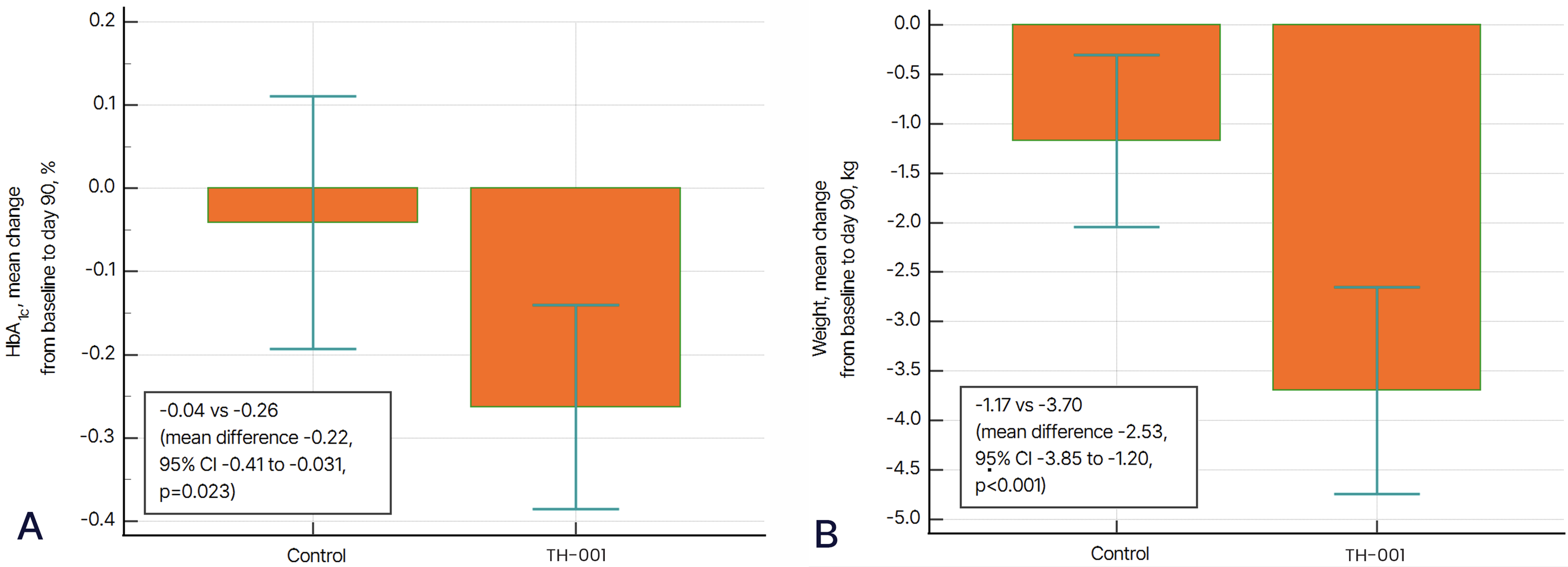
(a) Mean change in HbA1c levels from baseline to day 90 in the control and TH-001 groups. The TH-001 group showed a significantly greater reduction in HbA1c compared to the control group, with a mean difference of −0.22% (95% CI: −0.41 to −0.031, p = .023), indicating improved glycaemic control. (b) Mean change in body weight from baseline to day 90. The TH-001 group experienced significantly greater weight reduction (−3.70 kg) compared to the control group (−1.17 kg), with a between-group mean difference of −2.53 kg (95% CI: −3.85 to −1.20, p < .001). Error bars represent standard deviations. Abbreviations: HbA1c: hemoglobin A1c; CI: confidence interval. p ≤ .05.
Weight reduction was significantly greater in the intervention group (−3.70 kg, SD: 2.69) compared to the control group (−1.17 kg, SD: 2.24), with a statistically significant between-group difference of −2.53 kg (95% CI: −3.85 to −1.20, p < .001) (Figure 2(b)). Body mass index (BMI) also showed a significant reduction in the intervention group (−1.35 kg/m², SD: 0.98) compared to the control group (−0.46 kg/m², SD: 0.87), with a between-group difference of −0.88 kg/m² (95% CI: −1.38 to −0.39, p < .001). Other anthropometric measures, including waist circumference, hip circumference, and waist-to-hip ratio, showed reductions in the intervention group; however, these differences were not statistically significant (Table 4).
Mean changes in primary and secondary outcomes from baseline to day 90 and mean differences between control and intervention groups.
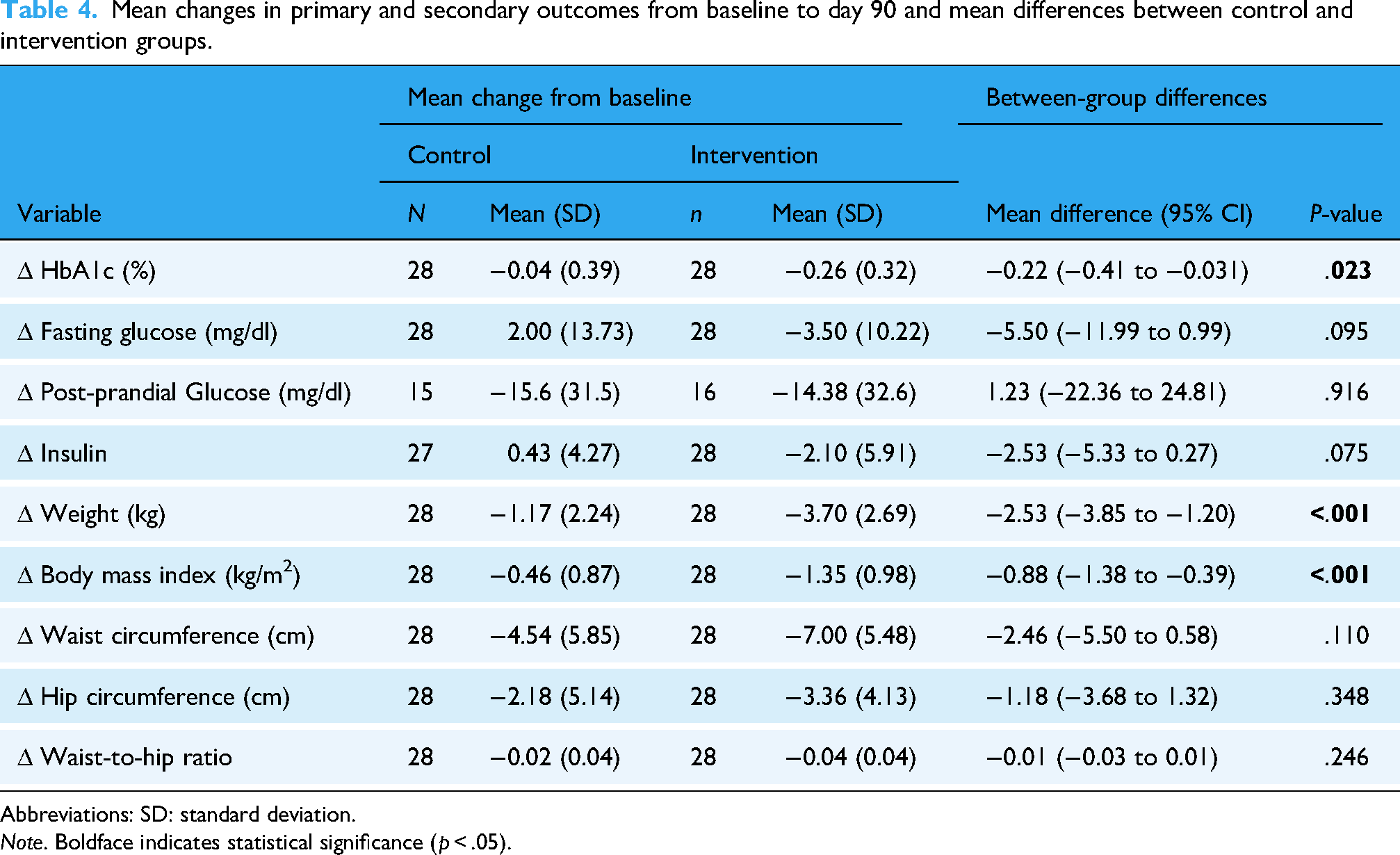
Abbreviations: SD: standard deviation.
Note. Boldface indicates statistical significance (p < .05).
The analysis revealed a strong correlation between the number of days using the TH-001 app and the reduction in HbA1c levels (r = .95, p < .001). As shown in Figure 3, Participants with higher app usage experienced greater reductions in HbA1c.

Scatter plot illustrating the relationship between app usage days and changes in HbA1c levels among TH-001 users. The regression model y = 0.121−0.00852 x shows a strong inverse correlation (r = .95, p < .001), indicating that each additional day of app engagement was associated with a mean HbA1c reduction of 0.00852%. Abbreviations: HbA1c: hemoglobin A1c; App, TH-001. p ≤.05.
TH-001 was well tolerated, with no reported AEs or adverse device effects during the study.
User engagement metrics
While engagement metrics were not predefined in the study protocol, they were assessed based on commonly used measures in similar digital health trials and digital DPP (these terms are also summarised in the Appendix). The primary and secondary endpoints were pre-specified, while engagement metrics represent additional exploratory analyses that provide context for understanding intervention utilization. App uptake, defined as the proportion of participants who logged into the application and completed at least one assigned daily task, was recorded at 84.8% for TH-001. Engagement duration, measured as the number of days from study enrolment to the last day within the study period that a participant from the uptake group logged into the app, averaged 62.4 days, with a median of 69.0 days. Additionally, participants successfully completed at least one assigned task on 42.2% of study days, as reflected in the goal completion rate (Table 5). The adherence score, which quantifies adherence relative to the intended use by calculating the ratio of actual use to intended use, was 49.8%. Retention, determined by attendance at least 60% of assigned sessions, was 48.4%. Lastly, completion, defined as attendance at the final intervention session or the recording of a final health measure, plus at least 60% attendance, was 32.3% (Table 6).
Engagement metrics for TH-001.

App uptake: The proportion of users randomised to the intervention who logged in to the app and successfully completed their assigned tasks at least once. App usage days: The total number of days users from the uptake group logged into the app, regardless of task completion. Engagement duration days: The number of days from the date of study enrolment to the last day within the study period that a user from the uptake group logged into the app. Goal completion: The proportion of days during the study period that a user in the uptake group successfully completed at least one assigned task.
Comparison of engagement metrics and values for TH-001 and reference studies, including adherence score, feasibility, uptake, retention, and completion.

Abbreviations: DPP: diabetes prevention programme; HbA1c: hemoglobin A1c.
aAverage of nutrition, physical activity, and weight loss apps.
App uptake, defined as the proportion of participants who logged into the application and completed at least one assigned daily task, was notably higher for TH-001 at 93.9%, compared to a benchmark of 56.0%. 21 Engagement duration is defined as the number of days from the date of study enrolment to the last day within the study period that a user from the uptake group logged into the app. The engagement duration averaged 62.4 days, with a median of 69.0 days. Additionally, participants successfully completed at least one assigned task on 42.2% of study days, as reflected in the goal completion rate (Table 5). The adherence score, measuring the adherence relative to the intended use derived as the quantified ratio of intended use to actual use, was calculated at 49.8%, closely aligning with a reference value of 50.9%. 22 Feasibility, defined as the proportion of participants who successfully completed the study protocol, was recorded at 84.8%, similar to the reference value of 85.4%. 23 Retention, determined by attendance at least 60% of assigned sessions, was 48.4%, exceeding the reference value of 34.0%. 21 Lastly, completion, which accounted for attendance at the final intervention session or successful recording of a concluding health measure, reached 32.3%, surpassing a reference value of 22.0% 21 (Table 6).
Discussion
TH-001 demonstrated significant improvements in glycaemic control among adults with prediabetes, reducing HbA1c by 0.22% at 90 days (p = .023) compared to the control group. A dose-response relationship was observed, with greater exposure to the intervention associated with a more pronounced HbA1c reduction (p < .0001). Participants in the intervention group also experienced significant reductions in body weight (p < .001) and BMI (p < .001), reinforcing the potential of this intervention in addressing key risk factors for diabetes progression. The efficacy of digital interventions in reducing HbA1c has been demonstrated in multiple randomised controlled trials. Block et al. reported a significant HbA1c reduction of 0.26% (p =0.002) using a digital intervention incorporating tailored websites, a mobile app, and interactive emails based on the principles of the Centers for Disease Control and Prevention's DPP. 18 Similarly, Katula et al. observed a shift from prediabetes to normal glycaemic levels (−0.23% [95% CI −0.26, −0.20], p = .001) through an internet-based intervention delivered via mobile phones, computers, and smartwatches. 19 A meta-analysis of 33 studies involving 14,398 participants further supports the effectiveness of digital health interventions in improving glycaemic measures, particularly HbA1c (p < .01). 24 These interventions have also been shown to facilitate weight loss, which is crucial for improving insulin sensitivity and lowering HbA1c in individuals with prediabetes. 25 Given that a 5–7% reduction in body weight significantly reduces diabetes risk, digital health solutions present a scalable and cost-effective approach to managing prediabetes. 26
TH-001 demonstrated outcomes consistent with those reported in traditional DPPs. The UK NHS in-person DPP reported HbA1c reductions of 0.12% to 0.19% over 9–12 months, with greater reductions observed in participants attending more than 60% of sessions. 27 Similarly, the NHS Digital DPP pilot achieved an average HbA1c reduction of 0.15% over 12 months, while the original U.S. DPP trial reported HbA1c reductions (∼0.4% at three years), alongside a 58% reduction in diabetes incidence.9,28
Despite their efficacy, traditional DPPs face challenges such as logistical barriers, inconsistent long-term engagement, and reliance on in-person attendance, leading to high attrition rates. These limitations reduce their scalability, particularly among underserved populations.29,30 TH-001 addresses these challenges through a fully digital delivery model incorporating gamification, personalised notifications, and cognitive-behavioural strategies to enhance adherence. By leveraging mobile technology, the programme ensures continuous access to tailored content, eliminating the need for in-person sessions and facilitating sustained behaviour change. Beyond glycaemic outcomes, TH-001 demonstrated a clinically significant reduction in body weight and BMI. Participants lost an average of 3.70 kg (4% of total body weight, p < .001) and experienced a BMI reduction of 1.35 kg/m² (p < .001) over 90 days. In comparison, real-world DPP implementations in the U.S. report an average weight loss of 4.2%, with 35% of participants achieving at least 5% weight loss. 29 The UK NHS DPP reported an average weight loss of 2.3 kg (2.7% of body weight) over 12 months, increasing to 3.3 kg (3.9%) among highly adherent participants, while the NHS Digital DPP achieved a mean weight loss of 3.1 kg over 12 months.27,28 These findings suggest that TH-001 delivers comparable weight loss outcomes within a shorter timeframe, though long-term evaluations are needed to assess the sustainability of these effects. User engagement with TH-001 was notably high, with an app uptake rate of 84.8%, a retention rate of 48.4% at 90 days, and a completion rate of 32.3%. In comparison, the NHS DPP reported an uptake rate of 56%, a retention rate of 34%, and a completion rate of 22%. 21 The U.S. National DPP reported a median program duration of 134 days out of 365, with participants attending a median of 14 out of 22 sessions. 29 TH-001 demonstrated a median engagement of 69 days out of 90, with a goal completion rate of 42.2%, indicating that participants actively engaged with the intervention on nearly half of the study days. These findings highlight the feasibility of this intervention in supporting sustained participation, overcoming common barriers associated with in-person programmes, and facilitating adherence to structured lifestyle modifications.31,32 A strong dose-response relationship was observed, with greater app usage correlating with greater HbA1c reduction (r = .95, p < .001), reinforcing the importance of sustained engagement. This is in line with previous findings on digital health interventions, which have emphasized the importance of user adherence in achieving clinical benefits. 22
TH-001's positive outcomes stem from its cognitive behavioural approach, delivered through a 90-day digital therapeutic program with six modules. It promotes lifestyle changes via behavioural guidance, daily tasks, gamification, virtual scoreboards, education, and self-monitoring tools. The program encourages 150 min of weekly moderate-intensity activity and uses a food-card swipe interface with “traffic light” visualization to teach healthy eating. Educational content on prediabetes, diabetes, activity, and nutrition supports sustained behaviour change. Real-time physician monitoring enhances tailored support and adherence. This scalable, evidence-based digital approach overcomes the limitations of resource-intensive traditional programs.27,29,30
High engagement duration, averaging 62.4 days with a median of 69.0 days within the 90-day study period, and a goal completion rate of 42.2%, strongly indicates the intervention's feasibility and acceptability. The powerful dose-response relationship between app usage and HbA1c reduction (r = .95, p < .001) indicates a strong association between engagement with TH-001 and improved glycaemic control, suggesting that sustained interaction is critical for achieving clinical benefits. This study suggests DTx require both clinical efficacy and engaging design to promote consistent use.33,34 The strong correlation between app usage and HbA1c reduction, however, may not solely indicate a dose-response relationship. Confounding factors, such as baseline motivation, socioeconomic status, or concurrent lifestyle changes (e.g. better diet or increased exercise), could contribute. Motivated users may engage more with the app while adopting healthier behaviours, enhancing HbA1c reductions. Future research should measure these confounders to clarify the causal link between app usage and clinical outcomes.
TH-001 demonstrated a strong safety profile, with no AEs or device effects reported during the 90-day study, supporting its potential for widespread adoption and patient adherence in prediabetes management. Additionally, TH-001 achieved a 4% weight loss, a clinically significant outcome that may help prevent or delay type 2 diabetes and related complications, including cardiovascular disease, 35 kidney disease, 36 blindness, 37 stroke, and dementia.5,6
TH-001’s clinically significant weight loss and HbA1c reduction in prediabetic individuals support its integration into primary care as an adjunct or alternative to traditional diabetes-prevention programs, especially for those with limited access. A dose-response relationship between app engagement and outcomes allows clinicians to monitor adherence and tailor interventions. Its effectiveness across diverse, self-directed users, scalability, accessibility, and safety profile make it a potential first-line option that may help delay the need for pharmacological intervention. The 90-day results suggest cost-effectiveness and reduced healthcare utilisation, supporting favourable reimbursement policies. Successful adoption requires standardised protocols, clear patient-selection criteria, comprehensive education, and investment in digital infrastructure, training, and oversight.
Limitations of the study
This trial has several limitations. First, the study was open-label: participants and investigators were aware of group assignments (it was not possible to blind the use of a smartphone app). To minimize performance and reporting bias, the study relied on objective, laboratory-based metabolic endpoints (HbA1c, fasting glucose, etc.), which are not subject to participant or assessor interpretation
While TH-001 demonstrated a rapid impact on glycaemic control and weight reduction within the first 90 days, its long-term effectiveness remains to be determined. Evidence from the NHS and CDC DPPs suggests that even modest weight loss (3–5%) can significantly reduce diabetes incidence if maintained over time.9,27,29 Future research should extend the follow-up of digital interventions like TH-001 to at least 12 months or more to determine whether the short-term metabolic gains are sustainable and whether they indeed lead to a reduction in the development of type 2 diabetes. Relatedly, the absence of intermediate assessments (e.g. at 30 or 60 days) limits insights into the trajectory of changes, such as whether improvements in HbA1c or weight loss occurred progressively or plateaued early, which could inform optimization of the intervention's modular structure.
Fasting glucose and insulin levels showed an improving trend, though the changes did not reach statistical significance. This may be attributed to the relatively short study duration, as previous research suggests that metabolic adaptations, particularly those affecting glucose regulation and insulin sensitivity, often require longer intervention periods to yield significant effects. 38 The study's modest sample size (n = 68 randomized, with 56 completers) may have further limited statistical power to detect smaller but clinically meaningful differences in these secondary outcomes, especially given the observed dropout rates (15% in the intervention group and 20% in the control group), despite the use of multiple imputation to handle missing data.
A correlation between engagement and HbA1c reduction was observed; however, highly motivated individuals may be more likely to engage with the intervention while also adopting other healthy behaviours, such as dietary changes and increased physical activity, which independently contribute to HbA1c reduction. This self-selection bias limits the ability to establish a direct causal relationship between app use and clinical outcomes. Future studies employing stratified randomised controlled trial designs could help disentangle these effects. Additionally, engagement metrics were analysed exploratorily rather than as predefined endpoints, potentially introducing post-hoc bias in their interpretation.
Additionally, the study was conducted in a single country, which may limit generalisability due to variations in cultural norms, healthcare access, dietary habits, and digital literacy across different populations. Addressing these limitations in subsequent investigations will contribute to refining digital therapeutic interventions for broader clinical applicability.
The generalisability of our findings is also limited by the study's context. This was a single-site study conducted in Istanbul, Turkey, and the participant population was relatively homogenous in terms of ethnicity and cultural background. Results may differ in other countries or healthcare settings where cultural norms, healthcare access, dietary habits, and digital literacy vary. Furthermore, our trial cohort was predominantly female (∼76% of participants). This gender imbalance reflects the observation that women are more likely to enroll in lifestyle change programs than men (in the U.S. National DPP, roughly 121,000 females vs 39,000 males enrolled by 2017, an approximately 3:1). 39
Conclusion
The findings of this randomised controlled trial demonstrate that the digital therapeutic TH-001 shows promising short-term efficacy in HbA1c reduction that requires longer-term evaluation to assess sustained benefits and diabetes prevention outcomes. Participants using TH-001 experienced a statistically significant reduction in HbA1c and achieved meaningful weight loss and BMI reduction compared to the control arm. These improvements were observed without any AEs, indicating the safety of the intervention. User engagement metrics indicated a high level of interaction and adherence, suggesting TH-001's potential to overcome traditional barriers to DPPs. A notable dose-response relationship between app usage and HbA1c reduction underscores the importance of sustained engagement for achieving clinical benefits. Further research with extended follow-up and broader population samples is necessary to establish the sustained effectiveness and generalizability of TH-001 as a scalable intervention for prediabetes management.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251376260 - Supplemental material for A randomised clinical trial to evaluate a digital therapeutic (TH-001) for improving glycaemic control in adults with prediabetes
Supplemental material, sj-docx-1-dhj-10.1177_20552076251376260 for A randomised clinical trial to evaluate a digital therapeutic (TH-001) for improving glycaemic control in adults with prediabetes by Ramazan Cakmak, İlker Tosun, Hasan Avcu, Gurkan Caner Birer, Ozer Akgul, Kamal S. Saini, Jonathan Carnell and Orkide Kutlu in DIGITAL HEALTH
Footnotes
ORCID iDs
Authorship contributions
R.C.: conceptualisation (lead); methodology (equal); investigation (lead); supervision (lead); writing–original draft (lead); writing–review and editing (equal); and project administration (equal).
İ.T.: methodology (equal); investigation (supporting); writing–original draft (equal); and writing–review and editing (equal).
H.A.: methodology (equal); visualisation (lead); and writing–review and editing (equal).
G.C.B.: methodology (supporting); and writing–review and editing (equal).
O.A.: formal analysis (lead); data curation (lead); and writing–review and editing (supporting).
K.S.S.: writing–review and editing (equal).
J.C.: writing–review and editing (equal).
O.K.: writing–review and editing (equal); formal analysis (equal); data curation (equal).
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Funding
The study is funded by Tesu Health. Tesu Health is the sole sponsor of the study. No external grants were utilized for this research. The digital therapeutic software application used in this study was provided by Tesu Health. Tesu Health reviewed the manuscript before submission, but the co-authors maintained the authority of final approval of the manuscript to submit for publication.
Declaration of conflicting interests
İ.T., H.A. and G.C.B. are employees and shareholders of the study sponsor. K.S.S. reports consulting fees from the European Commission, and stock and/or other ownership interests in Labcorp Inc., Fortrea Inc., and Quantum Health Analytics (UK) Ltd., outside the submitted work. The other authors do not report any conflicts of interest.
To manage potential conflicts of interest, data analysis was conducted by independent co-authors (O.A. and O.K.) not affiliated with the study sponsor, and the study was overseen by the Istinye University Clinical Research Ethics Committee, which had no financial ties to Tesu Health. The sponsor, Tesu Health, had no role in data collection, data analysis, data interpretation beyond providing the digital therapeutic software and funding. All authors, including those with financial interests, reviewed and approved the final manuscript, ensuring compliance with the Digital Health Journal's policy on authorship and transparency.
Data availability
The data that support the findings of this study are available from the corresponding author, İ.T., upon reasonable request.
Prior presentation
Data from this study were presented at the 18th International Conference on Advanced Technologies & Treatments for Diabetes, 19–22 March 2025, Amsterdam, The Netherlands and at the 61st National Diabetes Metabolism and Nutrition Diseases Congress organized by the Turkish Diabetes Foundation and the Turkish Diabetes Association, 21–25 May 2025, Cyprus.
Supplemental material
Supplemental material for this article is available online.
Appendix: Glossary of digital engagement metrics
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
