Abstract
Introduction
Markerless motion capture (MMC) technology has emerged as a clinical tool to assess the physical performance of patients. This study evaluates: (a) differences in upper limb joint angles between stroke survivors with different functional levels and their healthy counterparts in controlled indoor and uncontrolled outdoor environments; and (b) the relationship between the kinematic information obtained by the MMC system and the scores of manual motor assessments.
Methods
A customized MMC system using an iPad Pro captured the participants’ movements. Stroke survivors underwent three upper limb assessments and performed seven sets of upper limb tasks with their non-affected side, followed by their affected side. Healthy participants performed the same tasks with their dominant and non-dominant sides. The sensitivity and specificity of the machine models were calculated for classifying upper limb motor function levels using kinematic data from the MMC system.
Results
Fifty stroke survivors and 49 healthy adults were recruited. Significant differences were found between the affected and non-affected sides of stroke participants in most tasks. Significant positive correlations were found between the results of the manual motor assessments and most of the kinematic parameters. The results of the four selected machine learning models revealed ≥0.85 sensitivity in the stroke upper limb functional level classification.
Conclusion
The MMC system and machine learning algorithms provide accurate data for evaluating upper limb recovery in stroke survivors. Further research is needed to explore the use of the MMC system by stroke survivors at home during remote therapy.
Introduction
The motor recovery of stroke survivors often requires continuous monitoring by therapists to customize exercises for different recovery stages. 1 Conventional practices undertaken by therapists, such as regular functional assessments and the manual measurement of range of motion (ROM), require patients’ regular attendance in clinical settings. 2 The use of marker-based motion analysis systems and other kinematic evaluation techniques—such as extracting data like joint angles and movement counts from wearable sensors to quantify stroke rehabilitation progress—is often costly and time-consuming to set up and implement. 3 With advances in technology, markerless motion capture (MMC) technology has emerged as a clinical measurement tool that can be used to assess the physical performance of patients more naturally, as well as reducing time-consuming tasks in manual measurements conducted by therapists. 4 It also has the advantage over wearable sensors or complex optoelectronic laboratory setups for motion analysis while maintaining high kinematic accuracy.5–7 A previous study has been conducted to capture kinematic data regarding stroke survivors and healthy adults when performing tasks through a stroke-specific performance-based impairment index: the Fugl–Meyer Assessment (FMA). Movement data from stroke survivors and healthy adults can be successfully classified with a rate of above 90% using machine learning classification models. 8 Therefore, the MMC system has the potential to identify symptomatic movement patterns in stroke survivors through artificial-intelligence-assisted detection technology, in order to monitor patients’ motor performance and activities of daily living, especially during remote assessments as part of telerehabilitation. 9
While MMC systems facilitate the tracking of movement kinematics, it is difficult for therapists to interpret this kinematic data into a comprehensive understanding of patients’ actual motor functions. 10 Researchers have also proposed using mobile devices as MMC systems for remote rehabilitation assessments, which enhances accessibility by eliminating the need for additional hardware. 11 However, little research has been carried out to investigate the application of MMC systems in mobile devices for clinical motor assessments among stroke survivors.12,13 Since most of the MMC systems have been applied only to healthy subjects, the validity and efficacy of the MMC system in classifying the motor impairment of various severities in the clinical populations remains unclear. Moreover, there are limited studies to standardize the use of MMC for clinical interpretation and translate the kinematic data captured by the MMC to be used together with clinical assessment scales such as the FMA. These hence create a large research gap concerning the application of mobile MMC systems, particularly regarding their efficacy in providing accurate measurements with which to evaluate the motor performance of stroke survivors with varying levels of severity. Our pilot study demonstrated the good reliability of a customized MMC system on an iPad Pro for measuring active range of motion (AROM) and angular waveforms in healthy adults. 14 This system might help to measure the motor performance of stroke survivors who demonstrate movement impairment; this study therefore further evaluates the performance of the customized MMC system in detecting : (a) the differences in upper limb joint angles between stroke survivors with different motor functional levels and their healthy counterparts in both controlled indoor and uncontrolled outdoor environments; and (b) the relationship between the kinematic information obtained by the MMC system and the scores of manual motor assessments. This study also investigates the sensitivity and specificity of the classification of upper limb motor functional level using machine learning methods, based on the kinematic data from the MMC system on the mobile device.
Methods
Study design
This cross-sectional experimental study received ethical approval from the Human Subjects Research Ethics Committee of the Hong Kong Polytechnic University (HSEARS20230214010). All subjects provided informed written consent before participating.
Stroke survivors completed three upper limb assessments conducted by an occupational therapist: (a) the Functional Test for the Hemiplegic Upper Extremity (FTHUE)
Instrumentation
A customized MMC system developed using Xcode on the basis of the ARKit6 and RealityKit framework in an iPad Pro with a LiDAR scanner, which had undergone reliability testing for upper limb kinematic measurement,
14
was used to capture participants’ movements in this study. The detection of the human body and the joint positions were extracted and visualized through computer-vision algorithms using convolutional neural networks (CNNs). A total of 14 3D body-joint positions and the timestamp of the motion detection were captured by our motion-tracking platform. The capture frequency of the MMC system was set at 30 Hz. The normalized coordinates of the angles were relative to the center of the pelvis and defined as the origin of the ARKit's coordinate system. The adjacent 3D joint coordinates extractions were used to calculate the angles of interest (AOI). Angle 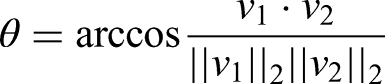
Sample size calculation
We assumed a two-tailed comparison with a type I error rate at 0.05, with 80% power. The stroke survivors were stratified according to the lower and higher functioning of their upper limb performance using the FTHUE. 15 As a conservative estimation with a discard rate of 15% due to bad data or outliers, according to our previous pilot study, 14 we presumed that 42 subjects in both healthy and stroke groups would be required for the final data analysis. Power analysis conducted with GPower 3.1.9.2 calculated the effect size as 0.74, which is between medium (0.5) and large (0.8). 18
Participants
Stroke survivors were recruited from community self-help groups and healthy counterparts through convenience sampling.
Inclusion and exclusion criteria for the participants
Inclusion criteria
To be eligible to take part in the study, participants were included if: (a) they were adults aged 18 years or above; (b) they had been diagnosed with a hemiplegic stroke; (c) they did not have a history of previous neurological or orthopedic diseases or congenital disorders of the upper or lower extremities or the spine; (d) they possessed adequate cognitive ability to understand instructions; and (e) they were able to engage in a 1-hour experimental session.
Exclusion criteria
Participants who met the following conditions were excluded: (a) medically unstable; (b) previous injuries or medical conditions over the upper limbs or spine affecting their upper limb functions (for healthy participants); (c) stroke survivors with a functional level of two or below, as measured using the FTHUE.
Motion capture tasks
In the motion capturing experiment, participants performed seven sets of upper limb tasks. The tasks included: (a) Task 3 in the FMA-UE, bringing the hand to the same side of the ear; (b) Task 4 in the FMA-UE, extending the arm to the opposite knee; (c) Task 6 in the FMA-UE, with shoulder flexion to 90° with the elbow at 0°; (d) Task 8 in the FMA-UE, with shoulder abduction 0° to 90° with the elbow fully extended and the forearm pronated; (e) Task 9 in the FMA-UE, with shoulder flexion beyond 90° with the elbow at 0° and the forearm in the mid position; (f) Task 3D in the FTHUE, holding a pouch; and (g) Task 24 in the FMA-UE, which is a finger-to-nose test. All of the tasks in this session were repeated five times. The stroke survivors were instructed to perform each task with their unaffected side first, followed by their affected side. Figure 1(a) to (g) illustrates the desired postures for these tasks.
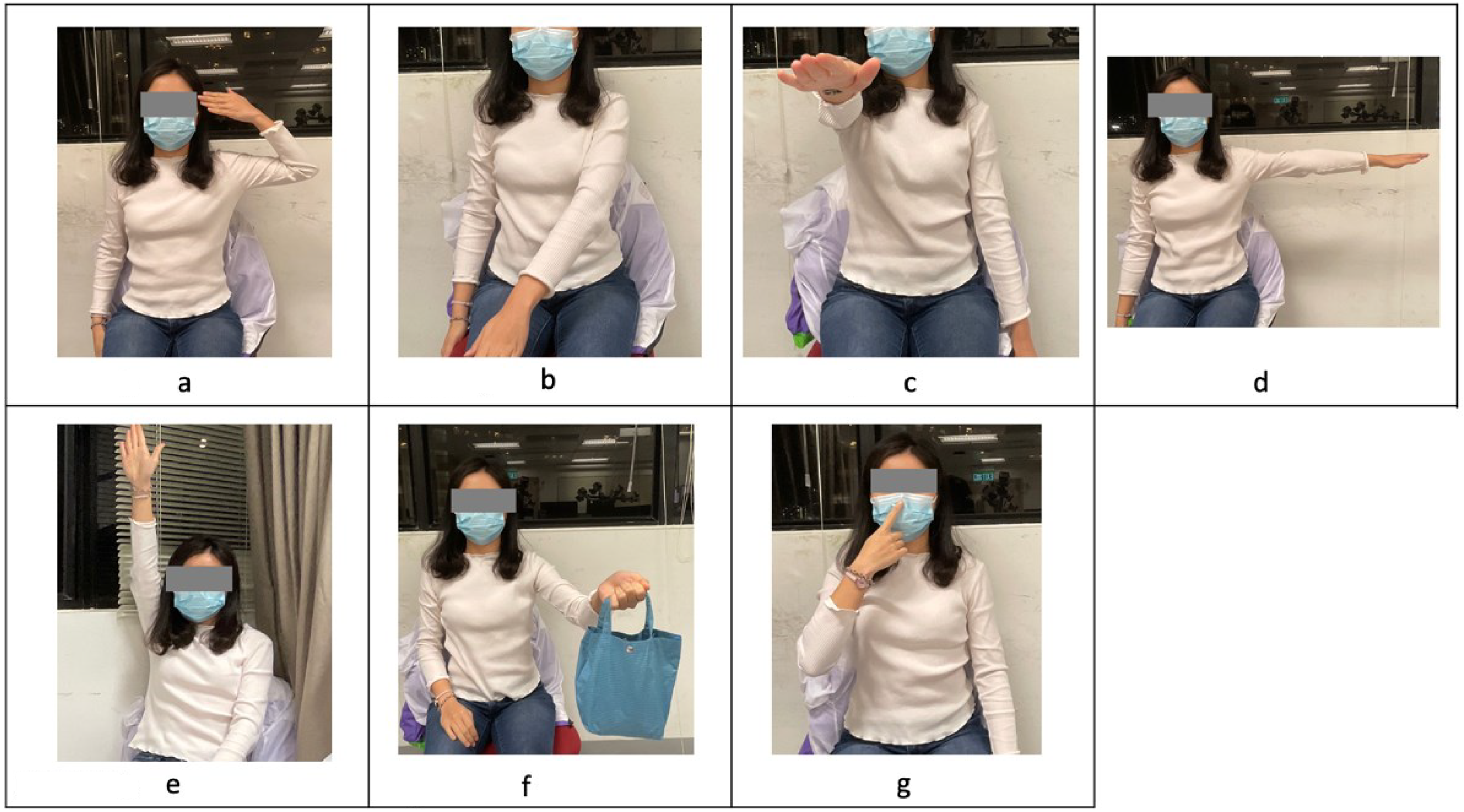
(a–g) Left to right, top to bottom, the desired postures for Task 1 to Task 7.
Experiment setup
Controlled indoor environment
The experiment was conducted at the university laboratory, where the floor was covered with vinyl to prevent it from being slippery. For the motion capturing session, participants stood in front of a plain wall in the same laboratory. One iPad Pro was placed two meters in front of the participant, and another two iPad Pros were placed at the lateral left and right sides of the participants, respectively. All iPads were connected to the same local network, and the motion capture system was launched on each device to establish a handshake signaling. At the start of recording, the master iPad sent a timestamped “start” command, triggering all devices to begin logging data with synchronized timestamps. Post-processing alignment of the data streams was performed using the shared timestamps to ensure temporal precision.
Uncontrolled outdoor environment
Three open areas at the university were chosen as uncontrolled outdoor environments. A 2.5 m × 2.5 m area was marked. Two 1 m × 1 m anti-slip mats were placed on both sides of the participants to prevent them from slipping. Three iPad Pros placed on a tripod stand were brought to the locations. The iPad Pro placement was the same as that in the laboratory environment.
Statistical analysis
Kinematic data, including completion time and joint angles in the target positions (ATP), were extracted from the MMC system, excluding the first trial. The normality of data distribution was assessed using the Shapiro–Wilk test to ensure the data meet the assumptions for parametric analysis. Averages from the second to fifth trials were analyzed using independent
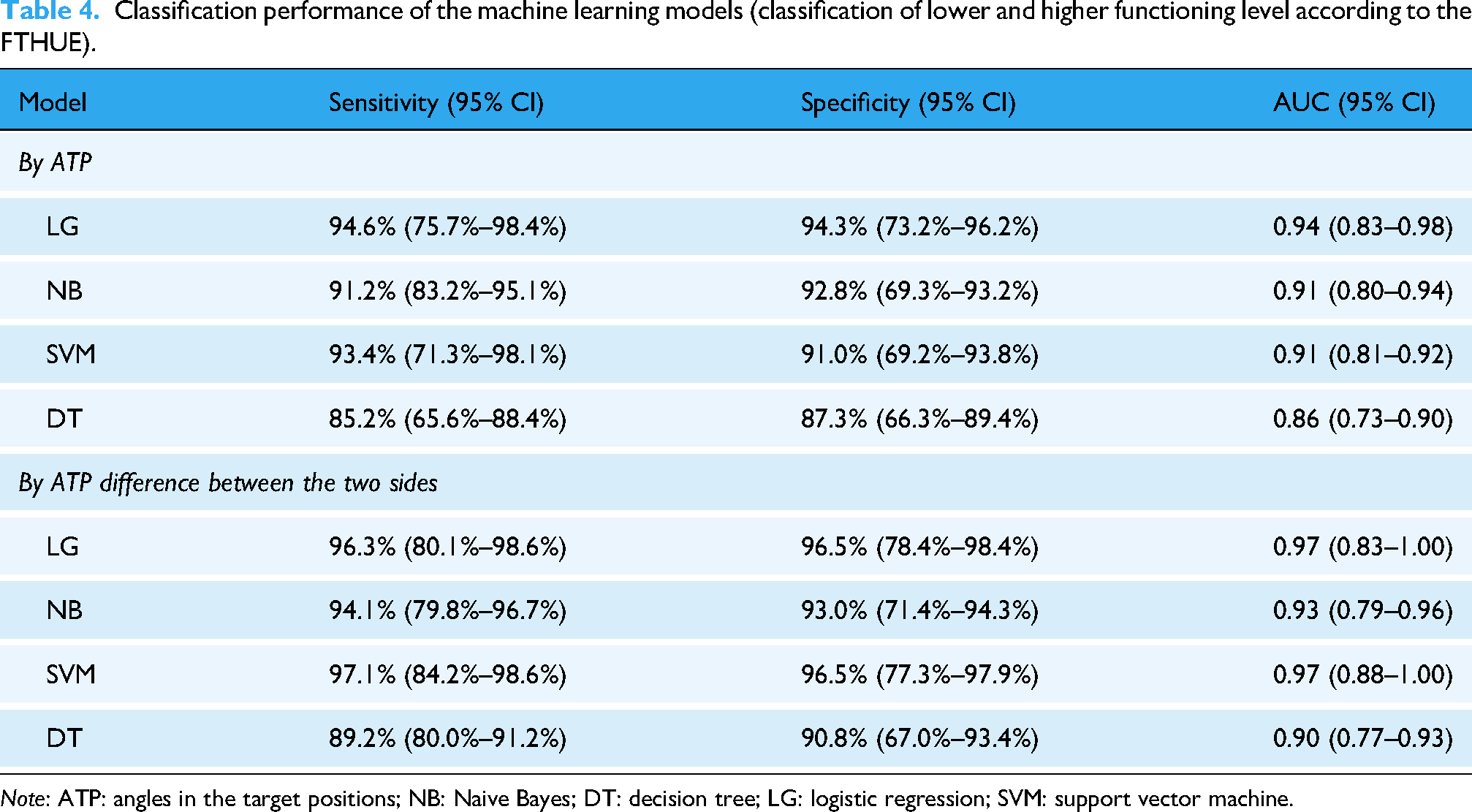
Four machine learning models, including logistic regression (LG), a Naive Bayes (NB) classifier, a support vector machine (SVM), and a decision tree (DT) model were used to investigate the trajectory in the predictions of the stroke participants’ upper limb assessment results, specifically the impairment levels according to the FMA-UE scores and the functional levels according to the FTHUE scores, based on kinematic data from the MMC system—the ATP and differences between the affected and unaffected sides. Regarding the FTHUE classification, participants with stroke were stratified into two groups: lower functioning (levels 3–4) and higher functioning (levels 5–7) group. 19 Regarding the FMA-UE classification, patients were categorized into three groups based on FMA-UE scores: higher functioning (FMA-UE > 52), moderate functioning (31 ≤ FMA-UE ≤ 52), and lower functioning (FMA-UE < 31). 20 The dataset was divided into training and test splits, using five-fold subject-wise stratified cross-validation. 21 These machine learning models were evaluated using standard classification and regression performance metrics. Regarding the FTHUE and FMA-UE groups prediction, the sensitivity, specificity, and AUC of each classification models were assessed.
All of the statistical tests were performed using IBM SPSS 26, while the CMC and RMSE values were generated by MATLAB R2020a. All machine learning models were run using the Scikit-learn package in Python.
Results
Fifty stroke survivors and 49 healthy adults were recruited. The mean age of the stroke survivors and the healthy adults was 58.9 years (SD: 11.7) and 60.2 years (SD: 8.5), respectively. Detailed demographic information regarding the participants is presented in Table 1.
Demographic information of the participants.
The shoulder and elbow joint angles at the targeted positions (ATP) for Tasks 1 to 6, are shown in Figure 2(a) and (b). Significant differences were found between the affected and non-affected sides of stroke survivors with lower functioning in all tasks, except for the shoulder angles in Tasks 1 and 2 (MD = −3.0 and −4.1). For stroke survivors with higher upper limb functioning, significant differences were observed in all tasks except for the shoulder angles in Tasks 3 and 5 (MD = −2.2 and −5.0). No significant differences were found between the two sides of healthy participants, except for shoulder and elbow angles in Task 3 (MD = 11.5 and 4.2) and elbow angles in Task 6 (MD = 14.8). Comparisons of the affected sides of higher and lower functioning stroke survivors showed significant differences in all tasks except for shoulder angles in Task 2 (

(a) Comparison of the ATP of the shoulder in Task 1 to Task 6. (b) Comparison of the ATP of the elbow in Task 1 to Task 6.
Differences in angular waveforms between lower and higher functioning stroke groups and healthy participants were calculated using CMC and RMSE, as shown in Table 2. Lower functioning stroke survivors had CMC values ranging from 0.23 to 0.67 across all tasks. In contrast, higher functioning stroke survivors had CMC values ranging from 0.39 (elbow in Task 1) to 0.86 (elbow in Task 4), while healthy participants ranged from 0.80 to 0.92 in Tasks 1 to 4.
CMC and RMSE values of the angular waveform comparison between the two sides.

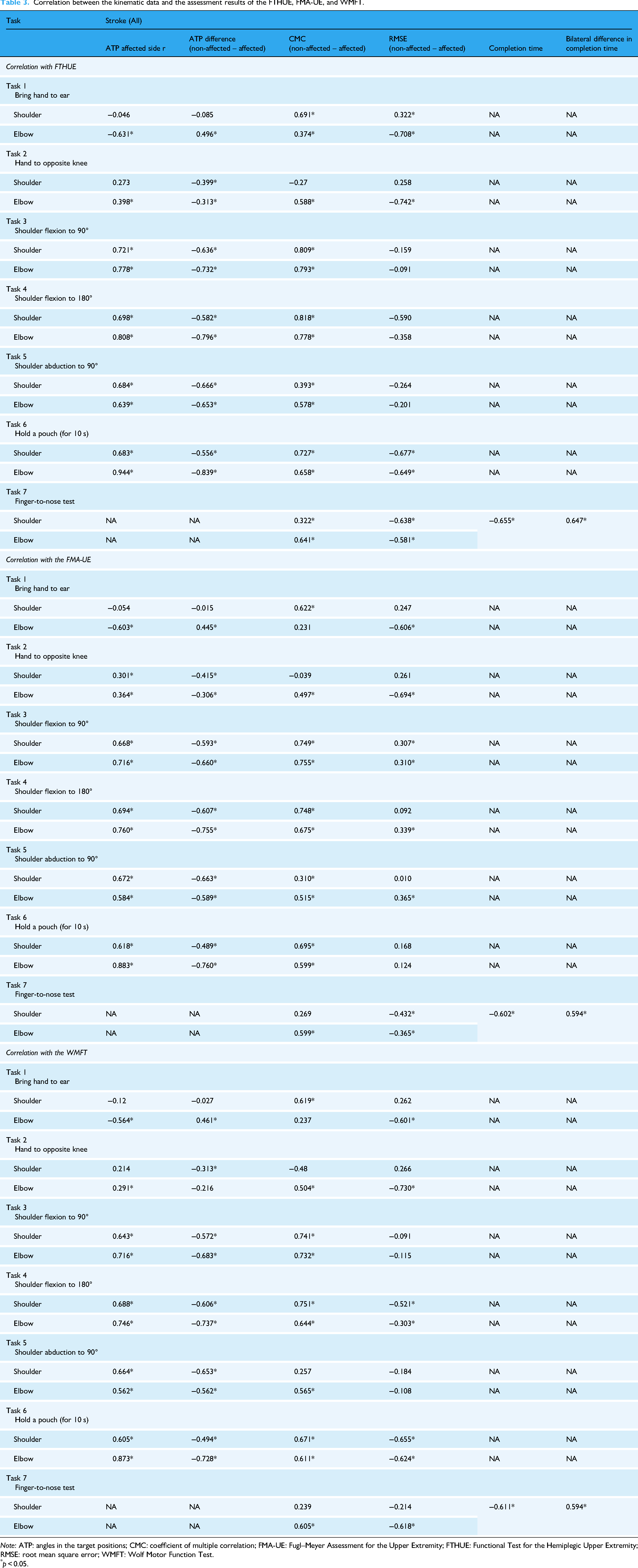
Correlations between the kinematic data and assessment results (FTHUE, FMA-UE, WMFT) are summarized in Table 3. Significant correlations were found for most kinematic parameters. The elbow ATP of the affected side in Task 6 (“hold a pouch”) had the strongest positive correlations with FTHUE, FMA-UE, and WMFT (
Correlation between the kinematic data and the assessment results of the FTHUE, FMA-UE, and WMFT.
Four machine learning models—LG, SVM, NB classifier, and DT—were trained to classify upper limb function according to FTHUE levels, based on kinematic data from the MMC system (Table 4). Models using the ATP of the affected side in Tasks 1 to 6 and completion time in Task 7 achieved sensitivities of ≥0.85, with the LG model showing the highest sensitivity and specificity (0.94). Models using ATP differences between affected and non-affected sides achieved sensitivities of 0.89 (DT model) to 0.97 (SVM model), with all models having an AUC ≥ 0.86. Feature importance analysis indicated that bilateral ATP differences in the shoulder and elbow during Tasks 3, 4, and 5 were key predictors of upper limb function. Results were consistent across cross-validation, with an average accuracy of 86.3% (SD = 2.7%). The SVM model classified participants into four functioning levels according to FMA-UE scores. Table 5 summarizes the SVM coefficients and optimal cutoff scores, with Tasks 3, 4, and 5 ranked highest. The cutoff scores showed over 90% sensitivity for the three stroke functioning groups based on the bilateral elbow angle difference in Task 5.
Classification performance of the machine learning models (classification of lower and higher functioning level according to the FTHUE).
Optimal cutoff score, coefficient, sensitivity, and specificity for each parameter in SVM classification (according to the FMA-UE).

The kinematic data captured by the MMC system in the outdoor environment contained a significant number of noise signals and missing data points, which hindered the formation of a complete angular waveform. Over half of the data had to be discarded due to noise signals. Due to the significant amount of outdoor data discarded, analysis of the outdoor data could not be performed. Figure 3(a) and (b) depicts the angular waveform extracted from a participant performing two tasks outdoors, demonstrating the noise signals and missing data points captured by the MMC system in the outdoor area. Figure 3(c) depicts the complete angular waveform of the same participants performing the task indoors.

(a) Angular waveform of one participant performing Task 3 in an outdoor area. (b) Angular waveform of one participant performing Task 4 in an outdoor area. (c) Angular waveform of one participant performing Task 4 in an indoor area.
Discussion
In this study, we found that the MMC system detected significant differences in joint angles at task completion between the affected and non-affected sides of stroke survivors in all selected tasks, except for shoulder angles in the “bring hand to the same side of the ear” task among those with lower upper limb function. The affected side showed significant limitations in shoulder and elbow ranges, likely due to limited control, spasticity, or muscle weakness post-stroke. 22 There were no significant differences between the dominant and non-dominant hands of healthy participants in most tasks. The CMC and RMSE values indicated greater differences between the affected and non-affected sides in stroke survivors with lower upper limb functioning compared to those with higher functioning. Our findings suggest that the MMC system in mobile devices is sensitive to capturing the kinematic differences between affected and non-affected sides, indicating its potential to provide kinematic information that distinguishes upper limb function levels in stroke survivors. The MMC system on a mobile device might hence serve as an alternative screening tool for motor ability in post-stroke patients, given that it can effectively detect symptomatic movements based on ROM differences and movement pattern variations.
All the selected classifiers achieved sensitivities above 0.84 based on joint angles from the affected side in lower and higher functioning classification, based on the FTHUE (FTHUE level five or above = higher functional level; FTHUE level four or below = lower functional level). 23 Our results indicate that combining the MMC system with machine learning can effectively distinguish between higher and lower levels of upper limb impairment in stroke patients. 24 Additionally, our AI models, trained on performance differences between affected and non-affected limbs, showed sensitivities above 0.89 and specificities of at least 0.90, indicating excellent performance. 25 Classifying based on these performance differences yielded higher sensitivity than considering only the affected side. One possible explanation for this result is that the non-affected hand typically reflects how an individual normally performs a motor task. Thus, comparing angular differences between the affected and non-affected sides may indicate how much the hemiparetic limb deviates from normal performance. A smaller difference suggests better recovery of motor function on the affected side and hence a higher functional level in classification.
In addition to FTHUE classification, FMA-UE scores were used to evaluate intervention effectiveness which supports the evidence-based practices for therapists. The SVM classifier identified optimal cutoff points for predicting FMA-UE performance, revealing that the lower functioning group had bilateral elbow angle differences exceeding 20°, while the higher functioning group had differences under 15° in most of the selected tasks. The SVM model highlighted that Tasks 3, 4, and 5, which involve specific elbow positions during shoulder movements, significantly contribute to functional classification. In contrast, tasks with minimal upper limb activity, such as the “hand to opposite knee” task, were less predictive of FMA-UE scores. Therefore, we recommend careful selection or combination of tasks for analyzing stroke survivors’ hemiplegic recovery, especially when comparing the non-affected side. The machine learning model suggests that combining the MMC system with classifiers can effectively distinguish upper limb function in stroke patients through a limited number of tasks, which can hence facilitate rapid screening of motor function in monitoring rehabilitation progress.
Although only seven tasks from the standardized upper limb assessment were selected, they are representative of the common functional tasks in standardized upper limb assessments, such as the FTHUE, FMA-UE, and WMFT. These seven tasks were chosen in this study due to their nature of being straightforward to administer, making them suitable for home settings where resources and supervision are limited. In addition, our tracking algorithm only included large joints in order to test its ability to identify and analyze participants’ gross motor abilities; mainly gross movements performed by the shoulder and elbow joint angles were investigated in this study. Future studies could consider tracking more complicated features, such as the contour of the hand and fingertips, so as to determine the ability of the MMC system to capture and analyze the movement of the wrist and the fine motor ability of stroke survivors.
Our findings indicate that kinematic data captured by the MMC system outdoors is affected by noise, background clutter, and light intensity. 26 The MMC often loses track of the target participant's joints when pedestrians pass by, misidentifying the pedestrian's limbs as the limbs of the target participant. A cluttered background can also confuse the system, which sometimes interprets tree branches as human limbs. Dim lighting results in sparse waveforms with missing data points as insufficient light hampers the capture of clear images, which affects tracking accuracy. We therefore recommend positioning the MMC in front of a plain background with adequate lighting and no moving objects to enhance data quality. Modifying the feature extraction function and including an initial calibration step may further improve joint identification and tracking accuracy.
The overall results of our study are consistent with previous recommendations by Bonnechère and colleagues 27 that an MMC system could be utilized to evaluate the upper limb motor performance of stroke survivors. Although it might not be appropriate for motion capturing in outdoor areas with a cluttered background, the kinematic data captured in a structured indoor environment provides a high level of sensitivity in regard to upper limb function classification. Although our study employed three iPad Pro devices to capture kinematic information from multiple angles, stroke survivors have the flexibility to utilize a single iPad Pro or their personal mobile device for motion capturing in a home setting. The use of three iPad Pro devices was solely intended to capture movements from diverse perspectives, while individuals can easily adjust the capturing angle independently when employing a single mobile device. Together with its portability, user-friendly setup, and low cost, an MMC system on a mobile device can facilitate remote monitoring of motor recovery during telerehabilitation. 28 Additionally, the precise data collected could enable therapists to conduct regular screenings of patients’ functional abilities at home, reducing the need for frequent clinic visits.
Even though current studies support the utilization of MMC technology in telerehabilitation, researchers must close the gap between research findings and the real-life implementation of MMC technology in order to promote its actual adoption in remote rehabilitation programs in the future. Future research should focus on designing a user-friendly interface that enables patients to operate the MMC system and transmit data to therapists effectively.
Limitations
This study employs data from 49 healthy adults and 50 stroke survivors. The sample size is considered small for training and testing machine learning models. Second, the ratio of stroke survivors with different functioning levels was not balanced. Future experiments examining the actual effect of different light levels on motion tracking quality in MMC systems are still warranted.
Conclusion
This study utilizes an MMC system on a mobile device to detect significant differences in the affected upper limbs of stroke survivors and healthy adults. The data provided by the MMC system reflects significant kinematic differences between the stroke survivors with lower upper limb functioning and those with higher functioning in all of the selected tasks. Strong correlations were found between upper limb motor assessment scores and kinematic performance. Combining the MMC system with a machine learning classification algorithm can provide precise data for evaluating upper limb recovery during telerehabilitation. Further research on the use of MMC systems by patients in home settings is warranted.
Footnotes
Abbreviations
Author note
Study was performed in the Hong Kong Polytechnic University, Hong Kong SAR
Acknowledgments
The authors thank the Research Impact Fund, Research Grants Council, University Grants Committee, Hong Kong SAR, for their support.
Ethical considerations and consent to participate
Experimental procedures were approved by the Human Subjects Research Ethics Committee of the Hong Kong Polytechnic University (Reference No.: HSEARS20230214010). The work was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). After the nature, purpose, risks and benefits of the study were explained, participants gave written informed consent to participate in the study.
Consent for publication
All authors have approved this manuscript for publication. It has not been previously published, nor is it pending publication elsewhere.
Author contributions
The authors confirm that all authors have contributed to the work presented in this manuscript and agree with the following contributorship statement:
Winnie W. T. Lam: Conceptualized the study, designed the methodology, writing of the manuscript, data collection. Contributed to data analysis and interpretation. Kenneth N. K. Fong: Supervised the project, conceptualized the study, designed the methodology. Drafting the manuscript and reviewed the final version. Reviewed and edited the manuscript for intellectual content. Chi-Wen Chien: Conducted the literature review, contributed to study design, and performed statistical analysis. Participated in revising the manuscript.
All authors have approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was partially funded by Research Impact Fund (Ref. no.: R5028-20F), Research Grants Council, University Grants Committee, Hong Kong SAR, to KNKF, and Research Centre for Assistive Technology, The Hong Kong Polytechnic University (Ref. no.: CE0E) to KNKF.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author [Kenneth NK Fong]. The data are not publicly available due to ethical restrictions, their containing information that could compromise the privacy of research participants.
Appendix 1. Details of the statistical analysis
Kinematic data, including completion time, angular waveforms, and joint angles at target positions, were extracted from the MMC system. The first trial in each task was a practice trial and was excluded from analysis. Averages from the second to fifth trials were used for statistical analysis. Comparisons of the joint angles in the target positions (ATP) were carried out using independent
