Abstract
Purpose
This study aims to identify independent risk factors for mixed urinary incontinence (MUI) in parous women using a multicenter epidemiological study and to establish and validate a predictive nomogram.
Methods
A large-scale survey was conducted from June 2022 to September 2023, including parous women aged over 20 selected through stratified random sampling. Data encompassed sociodemographic and obstetric histories, comorbidities, and standardized questionnaires. The primary goal was to identify high-risk factors for MUI, while the secondary was to develop a nomogram. Risk factors were determined using univariable and multivariable analyses. The nomogram's performance was assessed via concordance index (C-index) and calibration plots through internal and external validation.
Results
A total of 7709 women participated, with an MUI prevalence of 6.8%. Independent risk factors included higher body mass index, urban residence, postmenopausal status, multiple vaginal deliveries, history of pelvic surgery and macrosomia, family history of pelvic floor dysfunction, hypertension, and constipation. The area under the curve for the nomogram model was 0.717 in the training set, 0.714 for internal validation, and 0.725 for external validation. The calibration plots showed a good agreement between the predicted and observed outcomes.
Conclusion
This study identifies key risk factors for MUI in parous women and introduces a validated nomogram with high but not perfect predictive accuracy. The model enables early identification and management of MUI, though further refinement could enhance accuracy.
Introduction
The International Continence Society (ICS) defines mixed urinary incontinence (MUI) as involuntary leakage associated with urgency, as well as physical efforts such as exertion, sneezing, and coughing. 1 Previous large-scale epidemiological studies conducted across several European countries, including France, the United Kingdom, Spain, and Germany, have reported that the prevalence of MUI in women ranges from 25% to 40%. 2 In contrast, two large-scale epidemiological surveys conducted in China revealed a lower prevalence of 6.5% to 8.0% among women.3,4 Additionally, compared to the other two primary types of urinary incontinence (UI)—urge urinary incontinence (UUI) and stress urinary incontinence (SUI)—MUI exerts a more pronounced negative impact on women's quality of life (QoL). 5 This heightened impact is partly due to the fact that women with MUI are more likely to experience larger volumes and more frequent episodes of leakage compared to those with UUI or SUI alone.6,7
MUI is characterized by features of both UUI and SUI, suggesting that its underlying pathophysiology may involve two distinct disease processes. 8 This complexity makes the etiology of MUI more intricate and elusive compared to UUI or SUI alone, posing challenges for early diagnosis and targeted interventions. As a result, healthcare providers often focus on educating and promoting awareness of high-risk factors for MUI when addressing female patients during primary prevention efforts. Previous studies have identified several well-established risk factors for MUI, including age, obesity, postmenopausal status, and a history of multiple vaginal deliveries.4,9–11 However, most large-scale studies on female MUI were conducted over a decade, or even two decades ago. Given the increasing life expectancy, the aging population, and the rising prevalence of obesity, it is crucial to update our understanding of the risk factors associated with MUI.
As mentioned earlier, merely identifying high-risk factors for MUI is insufficient for accurately predicting which women are likely to develop MUI in the future. Traditional risk factor assessment provides general population-level insights but lacks the precision needed for individualized risk stratification. This limitation hampers the precise identification of high-risk populations, making it difficult to establish effective early diagnosis and intervention strategies. Predictive models, by integrating multiple risk factors into a statistical framework, offer a more accurate and personalized estimation of MUI risk. Over the past decade, numerous studies have demonstrated that combining women's sociodemographic characteristics, obstetric history, and certain auxiliary examination parameters can effectively predict the risk of developing SUI using nomogram models.12–15 These studies have reported area under the receiver operating characteristic curve (AUC) values ranging from 0.757 to 0.853, indicating good predictive performance. However, to the best of our knowledge, no studies have been published to date on the application of such models for predicting MUI. 16
Therefore, the aim of our study is to utilize data from a large-scale, multicenter epidemiological survey on female UI to provide an updated analysis of the high-risk factors for MUI. Additionally, we seek to develop a nomogram to predict the occurrence of MUI and to evaluate its predictive performance through both internal and external validation.
Material and methods
Data collection
This study is the second phase of the Female Pelvic Floor Health Management Center Project in Fujian Province, which we are conducting to understand the epidemiology and treatment of female pelvic floor dysfunction (PFD) in Fujian Province. The first phase of the project aimed to establish a provincial female UI epidemiology database using data from a large-scale, multicenter cross-sectional female UI epidemiological survey conducted between June 2022 and September 2023 in various regions of Fujian Province, China, which was the source of data for this study. This epidemiological survey was simultaneously carried out in four regions: Sanming, Nanping, Quanzhou, and Ningde, with Sanming's Shaxian district serving as the primary center and the other regions functioning as sub-centers. The study targeted at women over the age of 20, with participants selected through multistage stratified random sampling. This study adhered strictly to the principles outlined in the Declaration of Helsinki and received approval from the Ethical Review Committee of local institution. All participants provided written informed consent prior to their inclusion in the survey.
We aimed to include 10,000 women in our study at the primary center of Shaxian, representing 10% of the local female population. We employed a multistage stratified cluster random sampling method. In the first stage, based on the urban-rural population distribution, we selected 4000 women from urban areas and 6000 women from rural areas. In the second stage, Shaxian includes two urban streets and 10 rural towns; we selected 2000 participants from the urban streets and 600 from each town. In the final stage, communities under the urban streets and villages under the towns were randomly selected following the principle of simple random sampling. All eligible women from the selected communities and villages were required to participate in the survey until the target sample size was reached. The detailed sampling process has been thoroughly described in previously published studies.3,17 In the sub-centers of Nanping, Quanzhou, and Ningde, one urban community and one rural village were selected, respectively. Data from the female population in these areas were collected through face-to-face interviews and standardized questionnaires. All selected women were invited to local health service stations where general practitioners (GPs) in their respective areas collected data through face-to-face interviews. These GPs collected data using a standardized questionnaire to gather relevant information from the respondents. The questionnaire, which was carefully designed in advance, included sections on sociodemographic information, obstetric history, and certain medical histories and comorbidities. Recognizing the critical role of data collectors and tools in epidemiological research, we provided standardized training to these GPs before the formal survey began. This training was aimed at ensuring the accurate and consistent collection of data. Additionally, a pilot study involving 300 participants was conducted to assess the feasibility and accuracy of the survey methodology.
To determine whether respondents had MUI, we used a validated Chinese version of the International Consultation on Incontinence Questionnaire-Short Form (ICIQ-SF). 18 The ICIQ-SF is highly recommended by ICS for diagnosing MUI, receiving a high-grade A recommendation. The questionnaire comprises four sections: the first three assess the frequency and amount of urinary leakage and its impact on QoL, while the final section identifies UI subtypes based on symptoms associated with leakage: (1) never—no urine leakage; (2) leakage before reaching the toilet; (3) leakage during coughing or sneezing; (4) leakage during sleep; (5) leakage during physical activity or exercise; (6) leakage after urination, once dressed; (7) leakage without an apparent cause; (8) continuous leakage. Respondents who selected option (1) were classified as not having UI. Those who chose options (3) and/or (5) were categorized as having SUI, while those selecting one or more of options (2), (4), and (6) were classified as having UUI. If a respondent met the criteria for both SUI and UUI, or selected options (7) and/or (8), they were identified as having MUI.
Model development and validation
The training dataset for the model was derived from
The model's predictive performance was assessed using measures of discrimination and calibration. Discrimination was assessed using the concordance index (C-index), derived from the area under the receiver operating characteristic curve (AUC). A higher AUC value, approaching 1, signifies stronger predictive accuracy for the outcome, whereas an AUC of 0.5 suggests that the model's predictions are equivalent to random guessing. Calibration curves were plotted to visualize the predicted and observed risks of individuals, where a perfectly calibrated model follows a 45° line. Internal validation of the model was performed using fivefold cross-validation, while external validation was conducted using a dataset of subsites conducted in different regions from the training set.
Statistical analysis
All analyses were conducted using R software (version 4.3.0) and its associated packages. Continuous variables were first assessed for normality using the Kolmogorov–Smirnov test. Those that followed a normal distribution were summarized as mean ± standard deviation, with differences compared using Student's t-test. Variables not normally distributed were presented as medians with ranges and compared using the Mann–Whitney U test. Categorical variables were reported as frequencies and percentages, with differences analyzed using the chi-square test or Fisher's exact test as appropriate. Statistical significance was set at a p-value of <0.05.
Results
A total of 10,000 respondents were randomly selected at the major center. Of these, 2291 were excluded due to refusal to participate, lack of information on childbirth and comorbidities, or having no history of childbirth. Consequently, 7709 parous women were included in the final analysis, as outlined in Figure 1. Among this cohort, the prevalence of MUI was 6.8% (528/7709). The baseline characteristics of the training set and external validation set are presented in Table 1.

Flowchart of data obtaining and model development and validation.
Baseline characteristics of participants.

US dollars: According to the exchange rate on 20 November 2022.
Abbreviations: BMI: body mass index; SD: standard deviation; MUI: mixed urinary incontinence; PFD: pelvic floor dysfunction; NA: not applicable.
Continuous variables are given as mean ± standard deviation; categorical variables are presented as number (percentage). P-value was calculated with a Pearson Chi-square test, Fisher exact, Student's t test.
Univariate and multivariate analysis of factors associated with MUI
Univariate analysis identified 17 significant risk factors (p < 0.05) for further consideration (see Table 1 for full details on all analyzed variables). These factors include age, body mass index (BMI), region, educational level, income level, postmenopausal status, mode of delivery (vaginal or cesarean), age at first delivery, birth weight, history of macrosomic delivery, history of pelvic surgery, family history of PFD, hypertension, diabetes mellitus, chronic coughing, and constipation. Some variables, such as smoking, were analyzed but did not show statistical significance (Table 1).
Subsequent multivariate analysis revealed several independent risk factors for MUI in this population. These factors are higher BMI (odds ratio [OR] = 1.101, 95% confidence interval [CI] = 1.074–1.128, p < 0.001), urban residence (OR = 1.760, 95% CI = 1.439–2.155, p < 0.001), postmenopausal status (OR = 2.065, 95% CI = 1.521–2.803, p < 0.001), multiple vaginal deliveries (OR = 1.320, 95% CI = 1.178–1.480, p < 0.001), history of macrosomia (OR = 5.365, 95% CI = 3.619–7.954, p < 0.001), history of pelvic surgery (OR = 2.625, 95% CI = 1.647–4.183, p < 0.001), family history of PFD (OR = 2.653, 95% CI = 1.916–3.673, p < 0.001), hypertension (OR = 1.822, 95% CI = 1.353–2.453, p < 0.001), and constipation (OR = 3.941, 95% CI = 2.251–6.900, p < 0.001). Detailed results are presented in Table 2.
Multivariate logistic regression analyses of factors influencing MUI.
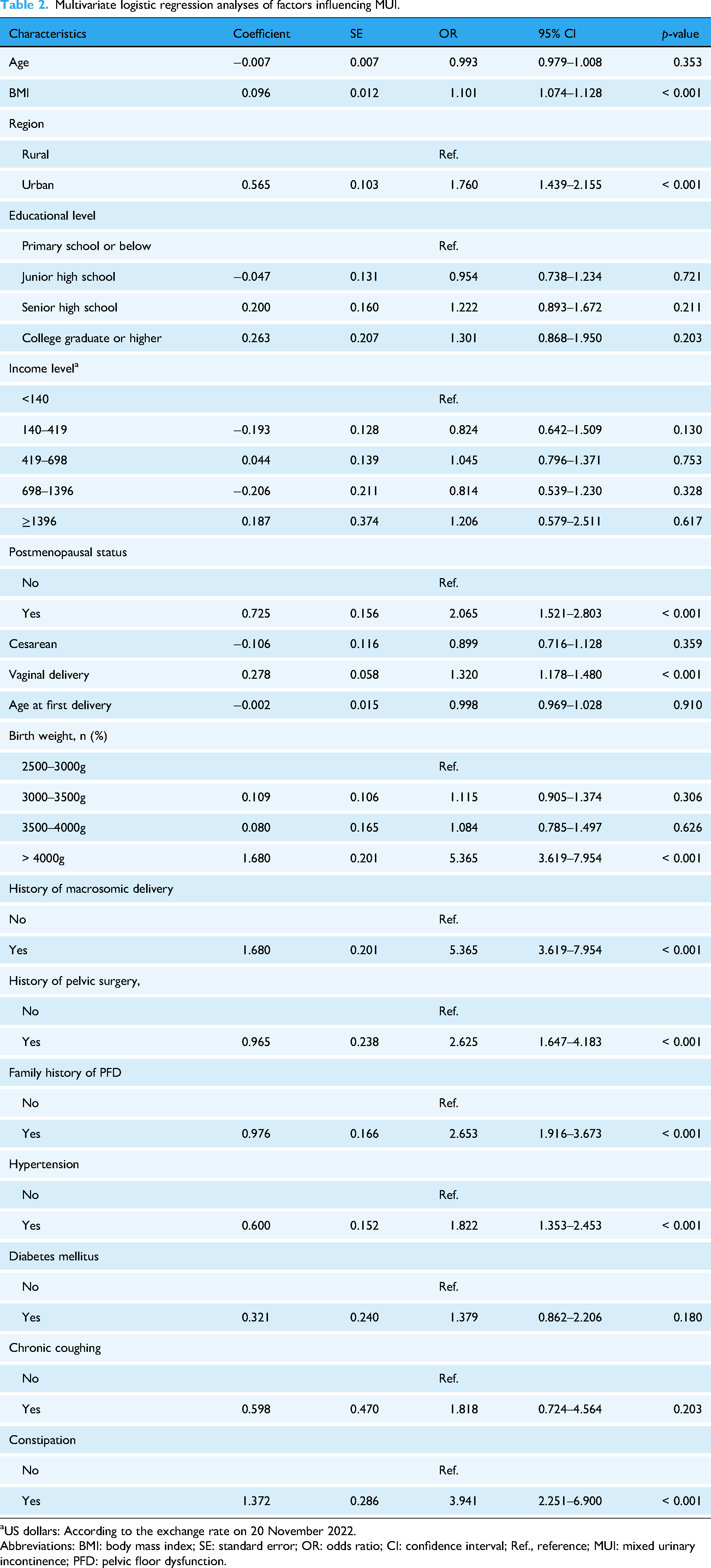
US dollars: According to the exchange rate on 20 November 2022.
Abbreviations: BMI: body mass index; SE: standard error; OR: odds ratio; CI: confidence interval; Ref., reference; MUI: mixed urinary incontinence; PFD: pelvic floor dysfunction.
Interaction analysis was conducted among the independent risk factors identified in the multivariable regression model. A statistically significant synergistic effect was observed between vaginal delivery and history of macrosomia (OR = 1.64, 95% CI: 1.15–2.33, p = 0.0061),suggesting that women with both risk factors were more likely to develop MUI than women with only one of these risk factors. No significant interactions were observed among the other independent risk factors.
Establishment and validation of nomogram model
Based on the above analysis, a risk prediction model for MUI was established using nine independent risk factors: BMI, region, postmenopausal status, vaginal delivery, history of macrosomic delivery, history of pelvic surgery, family history of PFD, hypertension, and constipation. This model was visualized as a nomogram, as shown in Figure 2.

The nomogram for predict the risk of mixed urinary incontinence (MUI). Each variable is assigned a specific score based on its value along the variable axis. The individual scores are then totaled to provide an overall score, which corresponds to the estimated risk of MUI prevalence.
Internal validation of the model was performed using fivefold cross-validation. Additionally, external validation was carried out with a dataset of 1006 parous women collected from three other centers in different regions. The ROC curves for the model's training set, internal validation, and external validation are presented in Figure 3A, 3B, and 3C, respectively. The AUC values for the training set, internal validation, and external validation were 0.717 (95% CI 0.694–0.740), 0.714 (95% CI 0.691–0.736), and 0.725 (95% CI 0.664–0.786), indicating good discrimination of the model. The calibration curves for the training set, internal validation, and external validation are shown in Figure 4A, 4B, and 4C, demonstrating good agreement between the predicted and observed values.

The receiver operating characteristic curve of predictive model in the training set (A), the internal validation (B) and the external validation (C). The AUC of each model and its 95% confidence interval are shown in the labeling in the lower right corner.

The calibration curve of predictive model in the training set (A), the internal validation (B) and the external validation (C).
Discussion
Based on a large-scale, multicenter epidemiological database, this study provides an up-to-date analysis of the characteristics of MUI in parous women. The identified independent risk factors include higher BMI, urban residence, postmenopausal status, multiple vaginal deliveries, history of macrosomia and pelvic surgery, family history of PFD, hypertension, and constipation. Furthermore, the nomogram model developed from these findings demonstrated strong predictive performance in both internal and external validations, underscoring its potential as a valuable tool for the early identification and precise intervention of MUI.
Consistent with previous studies,4,9–11 our research identified a strong association between higher BMI, postmenopausal status, multiple vaginal deliveries, history of macrosomia, history of pelvic surgery, family history of PFD, constipation, and hypertension with the occurrence of MUI. While these findings are not entirely novel, the underlying mechanisms contributing to these associations have been extensively explored in the literature. These mechanisms include changes in female hormone levels, deterioration of bladder and urethral function, congenital or acquired degenerative changes in pelvic floor muscles and fascia, and acute or chronic injuries to pelvic support structures. The pathophysiological processes through which these risk factors contribute to MUI have been well documented in previous studies,19,20 and our research further confirms that these factors remain the primary triggers for MUI. Furthermore, our interaction analysis demonstrated a significant synergistic effect between vaginal delivery and history of macrosomia (p = 0.0061), emphasizing the importance of considering combined obstetric risk factors when assessing an individual's susceptibility to MUI.
In addition to these well-established risk factors, our study identifies urban residence as a significant risk factor for MUI, contrasting with earlier studies that primarily focused on physiological and obstetric factors. Similar findings were reported by Ojengbede et al., 21 who found that women in rural areas were approximately three times less likely to report MUI than women in urban areas (OR 0.31, 95% CI 0.12–0.75). A plausible explanation for this could be that urban residents are more willing to discuss symptoms related to urinary leakage, rather than adhering to the stigma that incontinence is shameful and keeping such symptoms secret. Despite our survey being conducted by GPs familiar with the respondents, it is still challenging to completely eliminate the impact of social stigma on the reporting of incontinence symptoms. 22 Moreover, this finding may reflect the growing influence of social and environmental factors on MUI, particularly in the context of urbanization. Rapid urbanization and its associated lifestyle changes—such as reduced physical activity, high-calorie diets, increased stress, depression, and anxiety—have all been shown to contribute to a higher prevalence of MUI.23–25
Notably, our multivariable analysis revealed that age was not an independent risk factor for MUI, which differs from many previous reports. This discrepancy could be attributed to the characteristics of our study population. The mean age of participants in our study was 46.3 ± 10.5 years, whereas previous studies have reported a marked increase in MUI prevalence after the age of 60. 26 Additionally, as our study focused on parous women, the effects of age may have been overshadowed by childbirth-related factors, resulting in the non-significant impact of advancing age on MUI in our cohort.
Cesarean delivery has been widely hypothesized to reduce the risk of SUI by preventing direct injury to pelvic floor muscles and supportive structures. 27 Cesarean delivery, however, was not significantly associated with MUI in our study. The etiology of MUI is multifactorial, involving both mechanical and neurological factors, and mode of delivery may not be a deciding factor. Variations in populations examined, follow-up duration, and adjustment for confounders may also influence these results. In addition, many women who underwent cesarean delivery may still have experienced labor-associated pelvic floor stress, such as prolonged second-stage labor or a failed trial of vaginal delivery, which could weaken potential protective effect. A recent study by Stroeder et al. reported that both cesarean and vaginal delivery were associated with significant increases in bladder neck mobility (BNM) at 3 months postpartum, and that increased BNM was associated with the occurrence of UI in women. 28
With the continuous advancement of artificial intelligence and big data technologies in recent years, an increasing number of diseases now rely on database-driven risk prediction models, transforming them into effective risk screening tools. 29 , 30 These tools provide more precise guidance for primary and secondary disease prevention. Wang et al. reported a nomogram model for predicting postpartum UI, based on demographic and obstetric data from 1186 postpartum women. 15 The final model included predictors such as birth mode, UI before and during pregnancy, place of residence, feeding pattern, parity, and age at first birth. The model demonstrated good predictive performance, with an AUC of 0.759 (95% CI 0.70–0.82) in the validation cohort and a well-calibrated fit. In another study with a smaller sample size, a nomogram was developed by integrating patient characteristics, obstetric data, and pelvic floor ultrasound parameters. The predictive factors included age, parity, vaginal delivery, bladder neck descent, and the angle of the internal urethral orifice funnel. This model showed enhanced predictive performance, with an AUC of 0.807 (95% CI 0.723–0.891) in the validation set. 31 The successful development of these predictive models facilitates the creation of more straightforward and efficient screening tools for MUI, enabling healthcare providers to offer women personalized assessments of their future risk of developing MUI, rather than merely providing generalized information about high-risk factors. This also lays a solid foundation for the formulation of individualized early intervention strategies for MUI.
Strengths and limitations
One of the major strengths of our study is that it represents the first attempt to apply a MUI-specific database and nomogram methodology to develop a predictive model for MUI. The model underwent both internal and external validation, yielding promising results that enhance the reliability of its predictive capabilities. Additionally, we have provided a relatively straightforward and visually intuitive risk prediction tool, which is crucial for facilitating the broader application of the model in clinical practice.
However, this study has several limitations that must be acknowledged. First, the study's reliance on data from a large-scale epidemiological survey precluded the possibility of conducting specialized pelvic examinations. Consequently, the diagnostic outcomes were based solely on questionnaire responses. Although we used a highly validated and evidence-based questionnaire, recall bias among respondents could potentially impact the results. Moreover, the inability to capture critical factors such as cystocele and uterine prolapse—conditions that may significantly influence the development of MUI—could affect the model's predictive accuracy. Second, due to the constraints inherent in the large-scale epidemiological data source, we were unable to include pelvic floor ultrasound examination parameters, which have been identified in previous studies as potentially important predictors of UI. The absence of these parameters may limit the model's comprehensiveness and predictive power. Despite external validation, broader multicenter validation is still needed to confirm the model's generalizability and promote adaptive modifications to improve predictive performance and real-world applicability. Finally, while the primary aim of developing the predictive model was to provide a reliable risk identification tool to aid in the early detection and intervention of high-risk populations for MUI, the design of this study did not allow for the evaluation of the actual effectiveness of early interventions based on the risk assessment results. Thus, future studies may should focus on validating the practical utility of this model in reducing MUI prevalence through targeted early interventions.
We believe that applying the preliminary findings of this study to a smaller, more targeted cohort—by incorporating physical examination results and pelvic floor ultrasound parameters—will likely enhance the predictive power of our current model. Such improvements could provide healthcare professionals with a more accurate and user-friendly tool for screening the risk of MUI in parous women. Furthermore, these advancements would lay a solid foundation for future prospective cohort studies aimed at early interventions based on risk prediction results, ultimately contributing to more effective prevention strategies for MUI.
Conclusion
This study, based on a large-scale, multicenter epidemiological database, confirmed multiple independent risk factors for MUI, including higher BMI, urban residence, postmenopausal status, multiple vaginal deliveries, history of macrosomia, pelvic surgery, family history of PFD, hypertension, and constipation. The identification of these factors underscores their critical role in the development of MUI. Additionally, this study is the first to apply the nomogram method to establish a risk prediction model for MUI. The model demonstrated high predictive performance and reliability through internal and external validation. The visual risk prediction tool developed in this study offers a straightforward and intuitive approach, enabling healthcare providers to improve early screening and risk assessment for MUI. Furthermore, this tool lays the groundwork for future studies aimed at individualized early interventions based on predictive outcomes. Follow-up research should be aimed at prospective cohort studies to cross-validate and refine the model in clinical practice. Additionally, the integration of objective clinical parameters, such as pelvic floor ultrasound findings or hormonal biomarkers, may enhance the predictive power of the model. Lastly, the practicality and clinical utility of this instrument in decision-making in the clinic needs to be assessed to maximize its application. As it gets further validated and refined, this model can significantly improve precision prevention and control measures for MUI. Based on individualized risk profiles, early intervention strategies—such as weight management and pelvic floor exercises—may be implemented to mitigate risk. Additionally, targeted monitoring of high-risk individuals could facilitate the early detection of MUI and potentially prevent its progression.
Footnotes
Acknowledgements
We would like to express our sincere gratitude for the full support of the many GPs and respondents who participated in the study. In the process of revising and proofreading the manuscript, we used ChatGPT4.0 to improve sentence fluency and ensure grammatical accuracy. We would like to express our sincere gratitude to the development team of ChatGPT for their valuable assistance.
Guarantor
XJ, as the guarantor of this study, ensures that the data in this study are accurate and free from academic misconduct.
Ethical considerations
Ethical approval was granted by the Ethical Review Committee of Fujian Maternity and Child Health Hospital (2023KYLLR01045).
Informed Consent
All participants provided informed consent prior to participating in the study and agreed to the publication of the findings.
Author contributions/CRediT
QW did conceptualization, data curation, formal analysis, investigation, methodology, and writing—original draft. SM did data analysis, visualization, and writing—review and editing. HL did data curation, formal analysis, and investigation. XL did data curation, formal analysis, and investigation. CL did formal analysis, investigation, and writing—review and editing. XJ did formal analysis, investigation, and writing—review and editing. All authors approved the final article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Joint Funds for the innovation of science and Technology,Fujian province(Grant number:2023Y9367).
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data of this study are available from the corresponding author upon reasonable request.
