Abstract
Objective
The incidence of intensive care unit acquired weakness (ICU-AW) has shown an increasing trend with still a lack of effective treatment options. The early assessment of the risk of developing ICU-AW can provide patients with targeted interventions. This study aimed to determine the independent risk factors of ICU-AW in patients receiving mechanical ventilation (MV) and develop a nomogram and verify its predictive efficacy.
Methods
This observational study included patients receiving MV therapy in the ICU of our hospital between January 2020 and January 2023. They were divided into the ICU-AW and non-ICU-AW groups. The training cohort (n = 264) and the validation cohort (n = 143) were constructed. Multivariate logistic regression analyses were used to select the risk factors, and a nomogram model was established. Calibration, receiver operating characteristic (ROC), and decision curves were used to evaluate the effectiveness of the model.
Results
The MV duration (OR = 1.24, 95%CI[1.11, 1.38]), APACHE II score (OR = 1.34, 95%CI[1.20, 1.50]), SOFA score (OR = 1.36, 95%CI[1.21, 1.53]), age (OR = 1.05, 95%CI[1.00, 1.10]), nerve blockers (OR = 3.26, 95%CI[1.34, 7.92]), and diabetes mellitus (OR = 3.12, 95%CI[1.10, 8.87]) were independent risk factors for ICU-AW. The nomogram had good predictive efficacy for both the training (area under the curve (AUC) = 0.950, 95%CI [0.93, 0.97]) and validation cohorts (AUC = 0.823, 95%CI [0.75, 0.89]).
Conclusion
The MV duration, APACHE II, SOFA, age, use of nerve blockers, and diabetes mellitus are independent risk factors for ICU-AW. The nomogram model based on them had good predictive efficacy and may be clinically useful.
Introduction
Intensive care unit acquired weakness (ICU-AW) is a clinical syndrome that occurs during critical illness, but it is not caused by critical illness and is mainly characterized by new-onset symmetrical weakness of the limbs. 1 Clinical symptoms of ICU-AW include difficult extrication, paralysis or quadriplegia, diminished reflexes, and muscle atrophy.2,3 During hospitalization, ICU-AW had a relatively high incidence. However, after discharge, it was still a problem that cannot be ignored. 4 The incidence of ICU-AW has shown an increasing trend, which may be related to the prolonged survival of patients with critical illness due to the advancement of medical technology and aging of the society.5,6 ICU-AW can increase the risk of short- and long-term mortality and cause severe physical dysfunction, including quadriplegia or paraplegia, which reduces the quality of life of patients.7,8 ICU-AW can also prolong the duration of hospitalization of patients, aggravate the economic burden of patients and their families, and consume medical and social resources. 9
Due to its severe impact on patients, ICU-AW has been a research hotspot domestically and internationally, attracting marked attention from scholars. The pathogenesis of ICU-AW is complex and involves functional and structural alterations of muscles and nerves, which have not yet been fully clarified.10,11 There is still a lack of effective treatment options for ICU-AW among healthcare professionals.12,13 Early control of risk factors may be the only preventive measure to reduce its incidence.13,14 Therefore, it is of marked clinical importance to identify the risk factors for ICU-AW at an early stage. The purpose of this study was to identify the independent risk factors for ICU-AW in patients receiving mechanical ventilation (MV), develop a nomogram model based on the independent risk factors, validate the predictive efficacy of the nomogram model, and provide a reference for the evaluation of the risk of ICU-AW and clinical decision-making.
Methods
Object of study
This observational study included patients with critical illness who were treated with MV in the ICU of our hospital between January 2020 to January 2023. The inclusion criteria were as follows: (1) age of ≥18 years; (2) patients admitted to the ICU for the first time to receive treatment, length of stay in ICU ≥7 days; (3) patients receiving MV for more than 48 h; and (4) enough to follow orders and make voluntary movements after treatment in ICU. Patients who met all the above criteria were enrolled in this study. The exclusion criteria were as follows: (1) patients with craniocerebral trauma and spinal cord injury; (2) patients with central and peripheral nervous system lesions; (3) patients with cerebrovascular diseases; (4) patients with diseases affecting the function of the spinal cord, such as lumbar intervertebral disc herniation and spinal stenosis; (5) patients with of those with a history of psychiatric disorders; (6) patients with coexisting malignant tumors; and (7) participants of other clinical research. Patients who met any of the above exclusion criteria were excluded. This study was approved by the Ethics Committee of the local hospital, and the ethics approval number is KY 2022-116-01. All patients or legally authorized representatives of patients agreed to participate in this study and signed informed consent.
Calculation of sample sizes
Based on the results of previous literature and pre-analysis, this study predicted that no more than eight independent variables would be included in the multifactorial logistic regression analysis. According to the method of estimating the sample size for multifactorial logistic regression analysis, the number of positive cases (for ICU-AW) would be at least 10 times the number of independent variables (80 cases). The predicted incidence of ICU-AW was 45–55%, and a total sample size of at least 178 cases was estimated. Finally, 264 patients were included in the training cohort. The ratio of the number of patients in the validation cohort to that in the training cohort was 3:7, and the validation cohort included at least 80 patients. Finally, 143 patients were included in the validation cohort in this study.
Diagnostic criteria of ICU-AW
Referring to the Official Clinical Practice Guidelines of the American Thoracic Society, 15 patients were evaluated using the British Medical Research Council (MRC) scale. From 24 h after admission to ICU, the patient's consciousness was assessed daily, and a total of five instructions were preset: “Open eyes, look at me, open mouth, extend tongue, and nod my head when I count to 5.” If the patient could cooperate with 3 or more, it was considered as awake cooperation. If the patient was conscious and can cooperate with muscle strength measurement, the day was recorded as T1, and the muscle strength measurement was performed on the patient with MRC, and the MRC score was recorded. According to MRC score, those with MRC score <48 were classified as ICU-AW group, and those with MRC score ≥48 were classified as non-ICU-AW group. The evaluation was done by three attending physicians, who need to reach a consensus.
Observation indexes
When the patients were admitted to the ICU, we collected general information about the patients, including age, gender, body mass index, history of diabetes mellitus, and history of hypertension. From admission to ICU to T1 day, we also collected their treatment information, including the duration of MV; whether they received enteral nutrition; whether they were activity limited or not; and whether they were administered a sedative, analgesic, nerve blocker, catecholamine, cortisol, and continuous renal replacement therapy. From admission to ICU to T1 day, the information was collected about their diseases included whether they had sepsis, electrolyte disorders, or multivariate organ dysfunction syndrome. When the patients were admitted to the ICU, Acute physiology and chronic health evaluation II (APACHE II) and Sequential Organ Failure Assessment (SOFA) scores of the patients were assessed.
Statistical analysis
This study used R (V3.5.1) software and SPSS 26.0 software for statistical analysis. Measurement data with homogeneous variance are expressed as mean ± standard deviation (x ± s), and t-test or one-way ANOVA was used to compare them. Measurement data with unequal variance are expressed as median and quartiles (M [Q1, Q3]), and the Mann–Whitney or Kruskal–Wallis test was used to compare them. Counting data are expressed in sum rate (N [%]), and the chi-squared test was used to compare them. Multivariate logistic regression analyses were used to screen the independent risk factors and develop a nomogram. Multiple logistic regression analysis was performed on the variables with inter-group differences, employing a stepwise regression approach to select independent risk factors and establish the final model. Each variable was tested as an exposure factor, and the rest were control variables. The predictive efficacy of the variables for disease occurrence was evaluated using the ROC curve, and larger areas under the curve (AUCs) were associated with better prediction efficacy. The calibration curve and the Hosmer–Lemeshow goodness-of-fit were used to determine if the descriptive curve had shifted. The predictive efficacy of the model was assessed using decision curve analysis (DCA). Two-sided p-values of <0.05 denoted statistical significance.
Results
Differences in clinical variables of the ICU-AW and non-ICU-AW groups
In this study, 264 patients with MV in the ICU were included in the training cohort. In the training cohort, 116 patients (43.93%) had ICU-AW, while 148 (56.06%) did not. Refer to Table 1, the age of patients in the ICU-AW group was higher than that in the non-ICU-AW group (p < 0.001). Diabetes mellitus (p < 0.001) and sepsis (p = 0.040) were more prevalent in the ICU-AW group than in the non-ICU-AW group. The APACHE II and SOFA scores were higher for the ICU-AW group than for the non-ICU-AW group (p < 0.001). The duration of MV for the patients in the ICU-AW group was longer than that for those in the non-ICU-AW group (p < 0.001). The proportion of patients with activity limitation (p = 0.034) was higher for the ICU-AW group than for the non-ICU-AW group. The proportion of patients who received sedatives (p = 0.019), neuroblockers (p < 0.001), and cortisol (p = 0.026) was higher for the ICU-AW group than for the non-ICU-AW group. The above details are provided in Table 1.
Comparison of clinical variables of the ICU-AW and non-ICU-AW groups.
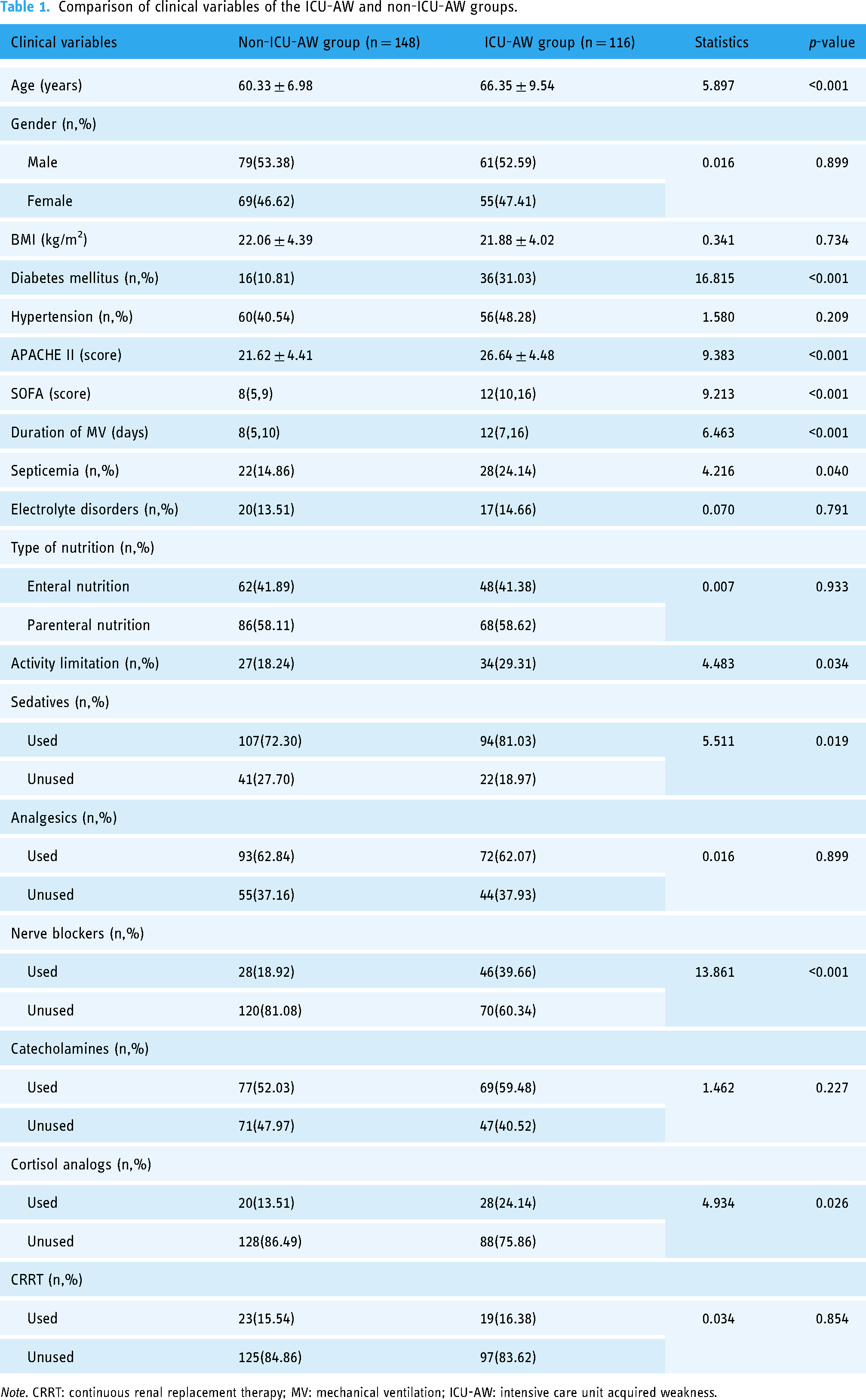
Note. CRRT: continuous renal replacement therapy; MV: mechanical ventilation; ICU-AW: intensive care unit acquired weakness.
Multivariate logistic regression analysis to determine the independent risk factors of ICU-AW
Multivariate logistic regression analysis showed that the duration of MV, APACHE II score, SOFA score, age, use of nerve blockers, and history of diabetes mellitus were independent risk factors for ICU-AW (OR > 1, p < 0.05), as shown in Table 2.
Screening of independent risk factors of ICU-AW using multivariate logistic regression analysis.
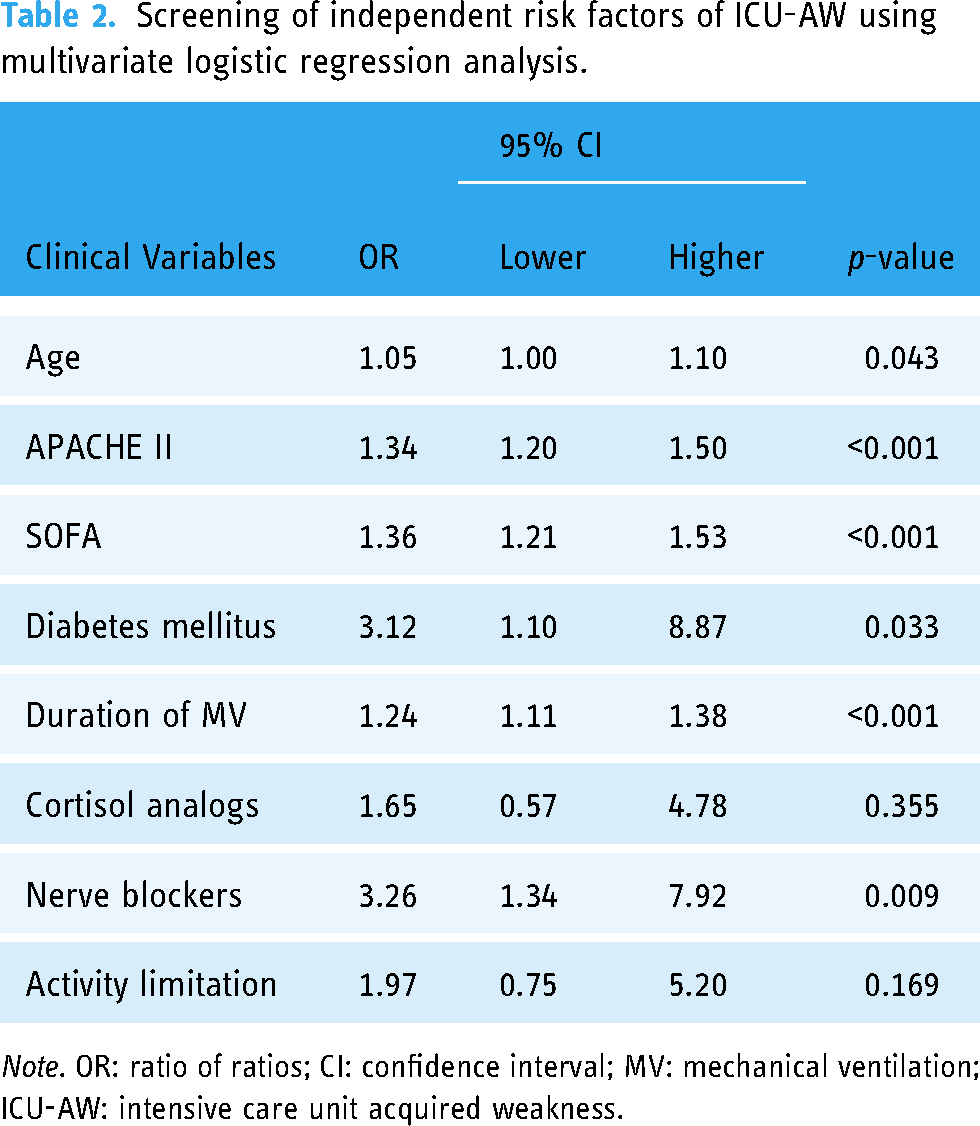
Note. OR: ratio of ratios; CI: confidence interval; MV: mechanical ventilation; ICU-AW: intensive care unit acquired weakness.
Nomogram model development and evaluation
Based on the above independent risk factors, a nomogram was developed to predict the risk of ICU-AW in patients in the ICU (n = 264), as shown in Figure 1. In this study, the score determined by the nomogram model was used as the risk score. The calibration curve of the nomogram model showed a mean absolute error of 0.018 and a mean squared error of 0.001 for the bias-corrected and ideal curves and no statistically significant deviation between the bias-corrected and ideal curves (Hosmer–Lemeshow goodness-of-fit test, p > 0.05), as shown in Figure 2. The ROC curve showed that the risk score had an AUC of 0.950 (95%CI [0.93, 0.97]), which was higher than that of the other independent risk factors, as shown in Figure 3. The AUC of age, APACHE II score, the MV duration, SOFA score, nerve blockers, and diabetes mellitus was 0.739(95%CI[0.68, 0.80]), 0.792(95%CI[0.74, 0.85]), 0.730(95%CI[0.67, 0.79]), 0.829(95%CI[0.78, 0.88]), 0.706(95%CI[0.65, 0.76]), 0.660(95%CI[0.60, 0.72]), respectively. The DCA curve showed that predicting the incidence of ICU-AW using the nomogram model, compared with the other independent risk factors, intervening in all patients, and not intervening in all patients, resulted in increased net patient benefit, as shown in Figure 4. The above results indicate that the nomogram model had good predictive efficacy.

Nomogram model for predicting the risk of ICU-AW. The red dots represent the risk factor scores and total scores for a random patient in the training cohort. ICU-AW: intensive care unit acquired weakness.

Nomogram model calibration curve. CI: confidence interval.
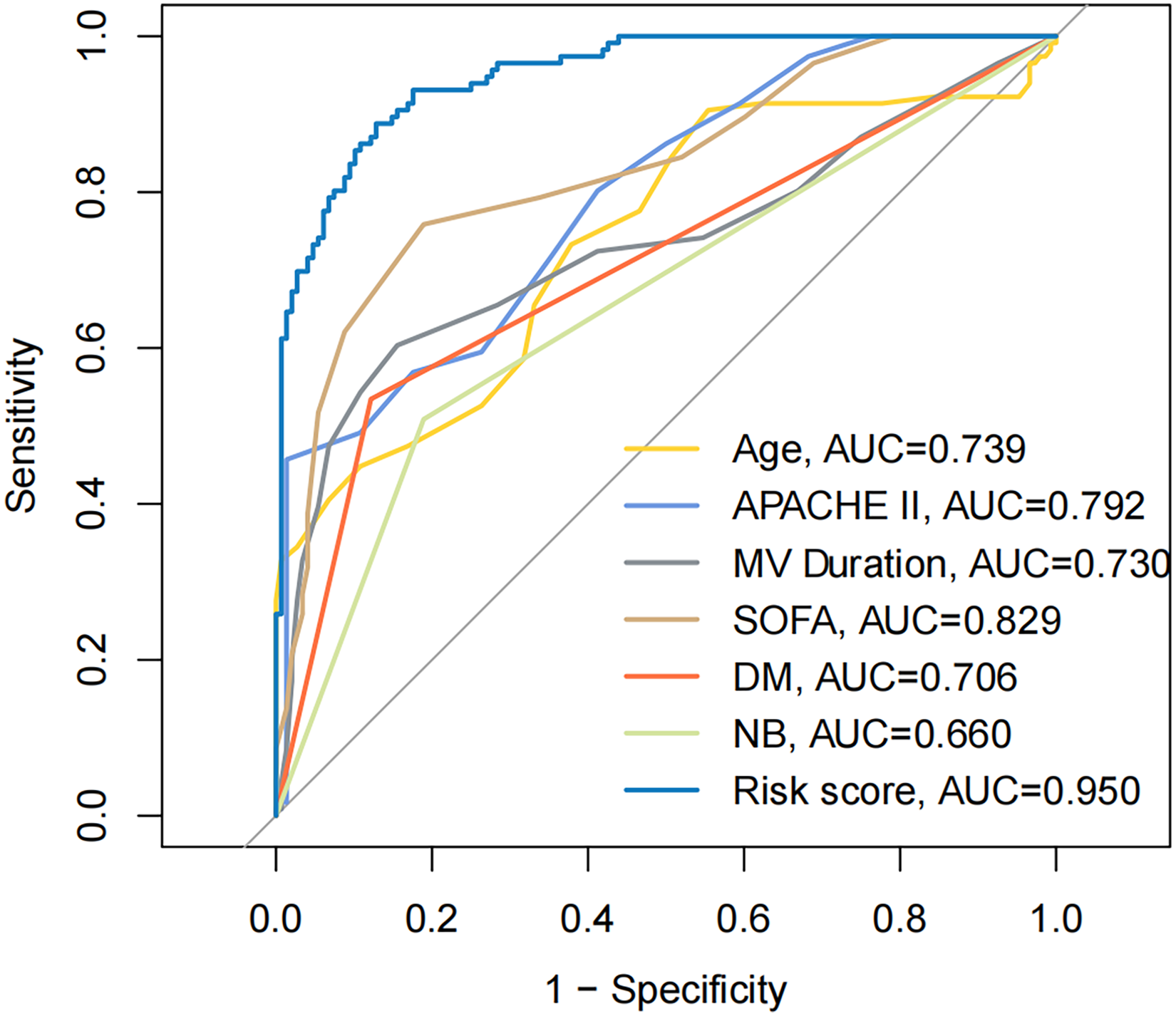
Receiver operator characteristic curves for the risk score and independent risk factors. AUC: area under the curve; MV: mechanical ventilation; DM: diabetes mellitus; NB: nerve blockers.

Decision tree curves for the risk score and independent risk factors. All represented intervention for all patients. None represented no intervention for all patients. MV: mechanical ventilation; DM: diabetes mellitus; NB: nerve blockers.
Validation of the effectiveness of the nomogram model
We used a validation cohort (n = 143) to externally validate the nomogram model. In the validation cohort, 58 (40.56%) had ICU-AW, and 85 (59.44%) did not. The calibration curve for the validation cohort showed a mean absolute error of 0.033 and a mean squared error of 0.001 for the bias-corrected curve and the ideal curve with no statistically significant offset (Hosmer–Lemeshow goodness-of-fit test, p > 0.05). The C-index was 0.823, as shown in Figure 5. The validation cohort ROC curve showed that the AUC of the risk score was 0.823 (95%CI [0.75, 0.89]), which was higher than other independent risk factors, as shown in Figure 6. The AUC of age, APACHE II score, the MV duration, SOFA score, nerve blockers, and diabetes mellitus was 0.601(95%CI[0.51, 0.70]), 0.643(95%CI[0.55, 0.74]), 0.651(95%CI[0.56, 0.75]), 0.676(95%CI[0.59, 0.77]), 0.665(95%CI[0.59, 0.74]), 0.612(95%CI[0.53, 0.69]), respectively. The validation cohort DCA curve showed that predicting the incidence of ICU-AW using the nomogram model increased the net patient benefit at the same threshold probability relative to the other independent risk factors, intervention for all patients, and no intervention for all patients, as shown in Figure 7. The above results indicate that the nomogram model had good predictive efficacy on the validation cohort.

Calibration curves for the nomogram model for the validation cohort. CI: confidence intervals.

Receiver operator characteristic curves of the risk score and independent risk factors for the validation cohort. AUC: area under the curve; MV: mechanical ventilation; DM: diabetes mellitus; NB: nerve blockers.

Decision tree curves for risk scores and independent risk factors for the validation cohort. All represented intervention for all patients. None represented no intervention for all patients. MV: mechanical ventilation; DM: diabetes mellitus; NB: nerve blockers.
Discussion
In this prospective cohort study, the independent risk factors for ICU-AW were screened. They included the duration of MV, APACHE II score, SOFA score, age, nerve blocker use, and history of diabetes. The reported independent risk factors for the development of ICU-AW were not consistent across studies but were generally compatible, which may be related to differences in the clinical variables included and subject selection bias. The risk factors for ICU-AW included age, weight, comorbidities, disease severity, organ failure, exposure to medications that negatively affect muscle fibers and neurons, inactivity, and other intensive care-related factors.6,16–19 Yang et al. 4 confirmed by meta-analysis suggesting that female sex, duration of MV in days, age, length of ICU stay, infectious diseases, renal replacement therapy, use of aminoglycosides, SOFA score, and hyperglycemia were independent risk factors for ICU-AW. An observational cohort study 20 showed that the APACHE II score and duration of MV were significantly correlated with the development of ICU-AW in patients who were critically ill. Shabana and others 21 concluded that the duration of MV was correlated with the occurrence of ICU-AW but could not be independent of free triiodothyronine. Liver and kidney function indicators and serum inflammatory factor indicators were not included as clinical variables in this study. On the one hand, most of the serologic indicators only suggest that the organism is in a certain pathological state and do not reflect the severity of the disease. On the other hand, serologic indicators fluctuate markedly over time and are affected by therapeutic measures, making it impossible to objectively evaluate the status of patients. Transforming continuous variables into dichotomous variables may result in the loss of data information. Therefore, continuous variables, including patient age, duration of MV, and scale scores, were retained in this study.
ICU-AW increases the risk of ICU mortality, in-hospital mortality, and long-term mortality; decreases the physical functioning and quality of life of patients; and consumes a large amount of healthcare and societal resources. The pathophysiological mechanisms of ICU-AW, including complex structural/functional alterations within muscle fibers and neurons, are incompletely understood, and clinically effective therapeutic strategies for ICU-AW are still lacking. Intervention based on identified risk factors can reduce the incidence of diseases. The kidney injury care bundle can significantly decrease the occurrence of acute kidney injury in myocardial infarction patients. 22 Early mobilization can reduce the incidence of ICU-acquired weakness. 23 Avoiding hyperglycemia and minimizing sedation are effective preventive measures for reducing the occurrence of ICU-acquired weakness. 24 Early prediction of the disease risk and development of preventive measures remain the best options for intervention. We emphasized that MV may be associated with a poor long-term prognosis in ICU patients, resulting in a range of severe organ damage. For example, MV was independently associated with acute kidney injury and the occurrence of severe acute kidney injury. 25 The incidence of ICU-AW was much higher in patients with MV than in patients without MV.26,27 Patients with longer MV may face a worse prognosis. 28 Therefore, this study developed a nomogram model to predict the development of ICU-AW among patients with MV in ICU based on the screened independent risk factors. The nomogram model was validated in this study using calibration, ROC, and decision curves. The validation results showed that the nomogram model had good predictive efficacy and was superior to a single independent risk factor. In the external cohort, the nomogram model showed good predictive efficacy. The nomogram model may help healthcare professionals to comprehensively and accurately assess the risk of ICU-AW in patients with MV in ICU and provide targeted interventions as early as possible to reduce the incidence of the disease, improve quality of life, and prolong survival. In older patients with a history of diabetes mellitus and high APACHE II and SOFA scores, it is necessary to use nerve blockers more cautiously, strictly control the blood glucose of patients, and rationally control the duration of MV.
The samples of the training and validation cohorts included in this study were small and obtained from a single center. This may have introduced selection bias, and studies involving larger samples and multiple centers are recommended. In addition, we included MV time as a variable in the model. It should be emphasized that the MV time is a continuous variable, and its time end point is extubation. However, in the application of predictive models, the occurrence of an end point in time is not required. The risk of developing ICU-AW increased with or without extubation for longer periods of MV. Although MV time, as an incomplete variable, will affect the evaluation efficiency of the model to some extent, which is the shortcoming of this study, MV time is still a variable that cannot be ignored.
Conclusion
In summary, the duration of MV, APACHE II score, SOFA score, age, nerve blockers, and history of diabetes mellitus were independent risk factors for ICU-AW. The nomogram model based on the above independent risk factors had good predictive efficacy and may be clinically useful.
Footnotes
Acknowledgements
The authors would like to thank all participants that took part in the study.
Contributorship
JW and CL researched literature and conceived the study. JW was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. JW wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was obtained from the Research Ethics Committee of the Jiangsu Taizhou People's Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
JW.
