Abstract
Background
Ensuring accurate and continuous monitoring of patients’ physiological parameters is paramount for evaluating their health status and guiding clinical decision-making. Technological advancements have the potential to significantly improve patient care and outcomes by offering a seamless continuum of healthcare experiences. Biobeat Technologies Ltd has pioneered a non−invasive wearable approach to acquiring advanced hemodynamic parameters, employing devices such as the BB−613WP wrist monitor and the BB−613P chest patch. Biobeat devices have been applicable across many clinical settings, as substantiated by a growing body of research. This systematic review endeavours to comprehensively consolidate the evidence regarding using Biobeat monitors across various clinical scenarios.
Methods
From 2016 to 2024, a thorough literature search was conducted across multiple databases. The inclusion criteria for selected studies comprised adult patients aged 18 years or older in any healthcare setting, employing Biobeat monitoring devices (wrist monitors and/or chest patches), reporting at least one outcome or finding, and presenting fully published original research studies, including randomized controlled trials and prospective or retrospective cohort studies. The quality and risk of bias assessment for the studies was performed using the Newcastle−Ottawa scale and COSMIN scoring system.
Results
Among 27 studies identified, 15 met the inclusion criteria, involving 4248 patients. These included 14 prospective observational studies and one retrospective cohort study; no randomized control trials were identified. Notably, eight studies were conducted in ambulatory settings, with 1 study focusing on patients undergoing labor and delivery. Additionally, three studies were carried out in general inpatient wards, 1 in a medical ICU and another in a cardiac surgery ICU (CSICU). Furthermore, 1 study presented results from 3 separate investigations— 2 in ambulatory settings and 1 in the CSICU. Across all studies, Biobeat devices were consistently utilized, with each study reporting positive outcomes associated with their use.
Conclusion
This systematic review demonstrates that Biobeat's non-invasive wearable devices have been effectively utilized across various clinical settings, consistently contributing to positive patient outcomes. The versatility and reliability of these devices highlight their potential to enhance patient care and support clinical decision-making, warranting further research to explore their broader applications.
Keywords
Introduction
Accurate and continuous monitoring of patients’ physiological parameters is critical in assessing their health and informing clinical decision-making processes. 1 This is significant during medical treatments, perioperative care, and remote monitoring programs. Various methods exist for obtaining hemodynamic data, with intensive care units (ICU) often relying on invasive techniques. Noninvasive cuff-based devices offer advantages in ambulatory settings.1–4 Technological advancements can enhance patient care and outcomes by providing a continuum of healthcare experiences.
A notable development in wearable, non-invasive photoplethysmography (PPG) technology introduces an alternative approach to obtaining advanced hemodynamic parameters.1–15 Biobeat Technologies Ltd, based in Petach Tikva, Israel, has developed a wearable, non-invasive monitoring technology with devices such as the BB−613WP wrist monitor and the BB−613P chest patch. Biobeat, established in 2014 and cleared by the FDA with CE mark certification, offers wearable and disposable remote patient monitoring (RPM) devices. These include long-term wrist and short-term chest monitors capable of continuously and accurately measuring various health parameters, including heart rate (HR), blood oxygen saturation (SpO2), blood pressure (BP), cardiac output (CO), and respiratory rate (RR). Biobeat provides timely alerts on patient health conditions by utilizing an automated, real-time Early Warning Score (EWS) system and advanced artificial intelligence (AI)−based algorithms in a secure Health Insurance Portability and Accountability Act of 1996 (HIPAA) and General Data Protection Regulation (GDPR)−compliant, cloud−based patient management platform. 16
Since its founding in 2014, Biobeat devices have been employed in numerous clinical settings, as evidenced by a growing body of research.1–15 These studies have investigated the agreement, accuracy, and reliability of Biobeat devices in diverse contexts, ranging from assessing cardiovascular changes in Coronavirus 2019 (COVID−19) patients and post-COVID-19 vaccination to applications in ICU settings, hemodynamic monitoring during labour and delivery, and monitoring congestive heart failure patients.1–15 Notably, studies such as Nachman et al. (2021, 2022)3,4 have demonstrated high agreement and reliability of the Biobeat cuffless device compared to standard sphygmomanometers. In contrast, others have highlighted its accuracy in blood pressure measurements among post−cardiac surgery patients. An observational study conducted within a randomized controlled trial (RCT) suggested that remote monitoring with Biobeat could aid in early pre-symptomatic flu detection. 9 Although the outcomes are consistently positive, variations in methodologies, sample sizes, data limitations, and clinical settings across different studies underscore the need for a systematic review to comprehensively summarize the available evidence on using the Biobeat monitoring device in diverse clinical scenarios. This review seeks to provide a comprehensive summary of the literature regarding the application of the Biobeat monitor across diverse clinical settings.
Methods
The International Prospective Register of Systematic Reviews (PROSPERO; CRD42024503346) registered the systematic review protocol. This study adhered to the Preferred Reporting Items for Systematic Review and Meta−Analyses (PRISMA) guidelines. 17 According to standard protocol, the institutional review board did not need to approve this systematic review, as it did not involve research on human participants. The review focused on using the Biobeat monitor in various clinical settings (Appendix Supplementary File 1).
Literature search
An expert librarian conducted a systematic and structured literature search (Appendix Supplementary File 2). All searches were conducted from 2014 (the founding year of Biobeat) to the present, using the following databases: MEDLINE/PubMed, Scopus, Web of Science, and Google Scholar. These databases were searched according to the thesaurus of the NCBI MeSH browser for the following terms and combinations of keywords in full text: ‘Biobeat,’ ‘outcome,’ ‘Healthcare,’ ‘remote monitoring,’ ‘complications,’ and ‘risk’. Results were restricted to English-language publications. Conference abstracts were excluded from the source where possible. Bibliographies of identified publications and published reviews were hand-searched for potentially relevant articles. The Biobeat clinical publication web page was also reviewed for relevant articles. 18
Selection criteria
Titles and abstracts, where available, were independently reviewed for relevancy by two reviewers (I.Z. and M.N.). The final selection of articles was based on the independent full−text reviews conducted by the same two reviewers. In cases of disagreement, both reviewers and co-authors discussed the materials until a consensus was reached. The inclusion criteria for selected studies were as follows: 1) adult patients 18 years or older in any healthcare setting; 2) use of the Biobeat monitoring device, either wrist monitor and/or chest patch; 3) at least one outcome or finding reported; and 4) fully published original research studies, either RCTs, prospective or retrospective cohort studies. Studies that did not meet the inclusion criteria were excluded. Case reports, review articles, conference abstracts and editorials were also excluded.
Data extraction
The following data were extracted from the selected studies: author, year of publication, country, study design, cohort size, demographic data (age, gender, BMI), clinical settings used, aim, primary outcomes related to physiological measurements obtained by Biobeat, and study findings. Where studies reported data in median and range, data were converted to mean and standard deviation using a conversion tool. 19 Two authors (I.Z. and M.N.) performed data extraction independently utilizing a data collection form. An independent reviewer (Y.S.) confirmed all data entries and checked for completeness and accuracy at least twice. Any disagreements were addressed by consulting another author (NW) and ultimately reached a consensus with the entire team.
Quality of study assessment
The quality and risk bias of the included studies were independently assessed by two reviewers (I.Z. and M.N.) using the Newcastle-Ottawa Scale (NOS) for cohort studies and COSMIN (Consensus-Based Standards for the Selection of Health Status Measurement Instruments).20,21 The NOS checklist appraises studies through three arms: participant selection, comparability, and outcomes. For participant selection, four criteria were assessed: (1) representativeness of the exposed cohort; (2) selection of the non-exposed cohort; (3) ascertainment of exposure; and (4) demonstration that the outcome of interest was not present at the start of the study. For comparability, two criteria were assessed: (1) study controls for confounders and (2) the study controls for at least three additional risk factors. For outcome, three criteria were assessed: (1) assessment of outcome; (2) follow-up duration long enough for an outcome to occur; and (3) adequacy of follow-up of cohorts. One star (*) was awarded for each criterion fulfilled according to the scale. The COSMIN evaluation included five study design and methodology components (the percentage of missing data, missing data management, adequate sample size, acceptable criterion comparison, and design or methodological flaws) and one analysis component (acceptable accuracy analyses). Each dimension is graded as excellent, good, fair, or poor based on the COSMIN validation subscale for scoring criteria. 22
Statistical analysis
Descriptive statistical analysis was performed for all included studies. Mean ± standard deviation (SD) or median (range / interquartile range [IQR]) were reported where applicable for continuous outcomes. Numbers and percentages or proportions were reported for categorical outcomes. Data on sample size, age (years), gender (male or female percentage), and BMI (kg/m2) were collected for the individual studies, and pooled estimates were calculated across the studies using the GraphPad Prism software. Outcome data were pooled when appropriate. Otherwise, the outcomes of the individual studies were summarised in a tabular column.
Results
Search results
The literature search yielded 30 results (Figure 1). No duplicates were found. After title and abstract screening, 25 articles were eligible for full−text review. Of these articles, 15 studies met the inclusion criteria.1–15 Each of these studies employed a Biobeat monitor in various healthcare settings.

PRISMA flow diagram of the study selection process.
Fourteen out of fifteen studies were prospective observational,1–11,13–15 and one was a retrospective observational. 12 No randomized control trials were identified in our literature search. All studies were conducted in Israel. Nachman is the first author in four studies,2–4,13 whereas Eisenkraft leads as the first author in three.7,8,12 The remaining authors each lead one study.1,5,6,9–11,14,15 These studies included 4248 patients whose physiological parameters were measured using Biobeat exclusively or concurrently with other currently accepted methods (Table 1). The average age of the participants was 45.0 ± 21.1 years, with a 95% confidence interval of [38.2, 58.9], and 56.3% were male gender. The average BMI was 25.2 ± 5.0 kg/m2, with a 95% CI [25.0, 28.9].
Summary of the demographic data.

PC: Prospective Cohort; RC: Retrospective Cohort.
Study outcomes
The studies varied in location, patient populations, and comparisons (Table 2). They were conducted in ambulatory to in-hospital settings, including labour and delivery, general inpatient wards, and the ICU. A wide range of physiologic data was collected across the studies.
Biobeat monitor utilization in various healthcare settings: a systematic review in the tabular column.

Ambulatory settings
Of the eight studies conducted in ambulatory settings, Biobeat monitors were utilized for various applications.2–5,9,10,13,14 One study measured physiological parameters every 60 s in patients with heart failure undergoing diuresis with furosemide in an outpatient setting. 5 The Biobeat wrist monitor captured readings for CO, stroke volume (SV), cardiac index (CI), HR, systemic vascular resistance (SVR), systolic blood pressure (SBP), diastolic blood pressure (DBP), pulse pressure (PP), and mean arterial pressure (MAP). 5 Notable trends were observed between pre- and post-diuresis timings. Significant differences in changes from baseline measurements between males and females for CO (p = 0.006), SV (p = 0.011), CI (p = 0.017), HR (p = 0.003), and SVR (p = 0.011) were reported. 5 Additionally, changes from baseline at the end of diuresis were reported based on k−means clustering, with three total clusters. 5 Cluster 3 consisted of males and females who showed no significant parameter changes at the end of diuresis. 5 Cluster 1 consisted mainly of males, who, compared to Cluster 3 males, showed higher baseline PP and CI (p = 0.056 and p = 0.051, respectively). 5 In this cluster, significant decreases in SBP, DBP, PP, MAP, SV, CI, and CO were observed, as well as a significant increase in SVR (p < 0.05). 5 Cluster 2 comprised mainly females, who, compared to Cluster 3 females, were younger and had higher baseline PP and lower CO (p = 0.004, p = 0.037, and p = 0.056, respectively). 5 Significant increases in HR, DBP, SV, CO, CI, CO, and a decrease in SVR at the end of diuresis were reported in this cluster (p < 0.05). 5
Two studies used Biobeat monitors to track physiological changes post−vaccination for COVID−19 and influenza.9,10 Biobeat captured physiological data in each study, and significant trends were observed. Gepner et al. (2022) reported peak measurements within 48 h of receiving the BNT162b2 mRNA COVID−19 vaccine. They found an increase in HR (9.85%), SBP (3.91%), and DBP (3.78%) compared to measurements taken the day before receiving the vaccine. 10 In patients who self-reported being asymptomatic without side effects, significant changes in physiologic parameters were observed only on the first-night post-vaccination.
In contrast, physiologic changes were observed in patients who reported side effects on both the first and second-night post-vaccination. 10 No significant differences were observed between men and women. 10 Goldstein et al. (2021) challenged subjects intranasally with the H3N2 human influenza virus 2 months after receiving either a novel flu vaccine or a placebo injection. 9 Continuous monitoring with Biobeat began 24−48 h before virus inoculation and continued for 9 days afterward. 9 Nasal swabs were taken twice daily to assess viral load. 9 In flu-positive subjects, an increase in HR, SBP, MAP, and SV were observed compared to baseline and a decrease in SVR at day 3 post-inoculation (p < 0.05). 9 In the flu-negative subjects, a reduction in SV was observed (p < 0.05). 9
Three studies utilized Biobeat wrist monitors to assess BP in ambulatory settings.2–4 Comparable findings were reported between BP measurements obtained by Biobeat and other methods in each study. One study compared readings from Biobeat wrist monitors to standard manometry. 2 For agreement between devices, a 100% sensitivity was detected in identifying hypertension, with a specificity of 92.4%. 2 A Bland-Altman analysis of healthy subjects showed a bias of −0.08 [− 7.06, 6.90] and 0.002 [− 6.88, 6.87] for SBP and DBP, respectively. 2 In subjects with hypertension, the bias was 0.49 [− 7.13, 6.14] for SBP and − 0.63 [− 7.11, 5.86] for DBP. 2 Another study compared readings from a Biobeat wrist monitor to an ambulatory blood pressure monitoring (ABPM) device. 4 Of the readings obtained, 87.6% of DBP readings and 81.5% of SBP readings were within ±5 mmHg of those obtained by the ABPM device, and 95.3% (DBP) and 94.2% (SBP) of measurements were within ±10 mmHg of those measured by ABPM. 4 The R2 values for DBP and SBP between Biobeat and ABPM were greater than 0.95 (p < 0.0001). 4 Bland−Altman analysis of mean 24-h values were −1.1mmHg for both DBP and SBP, with 95% limits of agreements [−4.1, 1.8] and [−4.2, 1.9] mmHg, respectively. 4 Daytime measurements of DBP and SBP showed a −1.9 mmHg bias for both, with limits of agreement (LOAs) [−6.3, 2.5] and [−7.0, 3.3], respectively. 4 Nighttime measurements showed a bias of 0.7 mmHg for DBP and 0.4 mmHg for SBP (DBP: −1.5, 2.9, SBP: −1.7, 2.6mmHg). 4 A third study compared Biobeat wrist monitors to a cuff−based blood pressure device, analyzing the influences of sex, BMI, and skin colour on the measurements obtained. 3 The overall Bland-Altman analysis showed that Biobeat had a bias of −0.02 ± 3.7 [−7.2, 7.3mmHg], and r = 0.985 for SBP, and −0.3 ± 4.2mmHg [−8.6, 8.0], r = 0.931 for DBP. 3 In male subjects, Biobeat had a bias of 0.2 ± 3.5 [−6.6, 7.1], r = 0.987 for SBP, and 0.2 ± 3.8 [−7.7, 7.3], r = 0.949 for DBP. 3 In females, the bias for SBP was 0.1 ± 3.8 [−7.7, 7.4], r = 0.982, and the bias for DBP was −0.4 ± 3.9 [−9.3, 8.5], r = 0.914. 3 Bland−Altman analyses were also conducted on BMI subgroups. In subjects with normal weight, biases were −0.08 ± 3.8 [−7.5, 7.3], r = 0.981, and −0.5 ± 4.0 [−8.3, 7.3], r = 0.931, for SBP and DBP, respectively. In the overweight group, the respective biases for SBP and DBP were 0.1 ± 4.1 [−8.2, 8.0], r = 0.978, and −0.4 ± 3.6 [−7.4, 6.6], r = 0.935. 3 Finally, in the obese group, Biobeat showed a bias of −0.7 ± 3.5 [−7.6, 6.3], r = 0.991 for SBP, and −0.7 ± 3.3 [−7.3, 5.8], r = 0.965 for DBP. 3 Data for skin tone was also analyzed according to the Fitzpatrick scale. 3 In subjects assigned types 1−3, Biobeat had a bias of −0.1 ± 3.9 [−7.4, 7.4], r = 0.982 for SBP and −0.4 ± 3.8 [−7.9, 7.1], r = 0.936 for DBP. 3 For subjects assigned types 4–6, Biobeat showed a bias of −0.1 ± 3.8 [−7.5, 7.2], r = 0.983 for SBP, and −0.6 ± 3.9 [−8.3, 7.1], r = 0.938 for DBP. 3
In another study, the correlation between RR measured using handheld capnography and Biobeat chest patch was r = 0.991, with a bias of <0.1 breath per minute [LOA −2.3, 2.3]. 8
Labour and delivery setting
Biobeat was used to monitor physiological parameters during labour and delivery in 1 study. 6 The device continuously recorded physiological data throughout the labour and delivery process. Significant changes in CO and SVR were reported in measurements taken 10 min before and after the administration of epidural anesthesia (p < 0.05). 6 When comparing parameters taken 30 min after delivery to those taken 30 min before and to peak values during delivery, significant changes were observed in CO, HR, SV, BP, and SVR (p < 0.05). 6 Significant physiological changes in HR, SVR, and CO were also captured 10 min before and after placental expulsion (p < 0.05). 6
General inpatient settings
Three studies used Biobeat in general inpatient wards.7,12,15 One study analyzed 5-day trends in several physiological parameters in patients with COVID-19. 12 Within the first 24 h, significant temperature, RR, and SVR increases were observed, along with significant decreases in SpO2, DBP, CO and CI (p < 0.01). 12 Additionally, within the first 48 h, a sustained reduction in SpO2 in both males and females was observed, accompanied by significant RR increases in males, obese patients, and all age sub-groups except the young (p < 0.001). 12 HR was also noted to be decreased in men and increased in women within this period (p < 0.01). 12 Other significant trends in SBP, DBP, CO, and CI were observed throughout the 5-day monitoring period (p = 0.002, p < 0.001, p < 0.01, p < 0.001, respectively). 12 Moreover, a study comparing RR measured by capnography versus Biobeat chest monitor in COVID−19 patients showed a high correlation (r = 0.888) and bias of <0.1 breath per minute [LOA −3.9, 3.9]. 8
Another study used readings from Biobeat monitors to validate a new tool called the Multiparameter Real-Time Warning Score (MPRT-WS) against the National Early Warning Score (NEWS). 7 The distribution of risk values according to NEWS versus MPRT-WS was comparable (p = 0.863). 7 Of the patients who deteriorated (n = 39), only 6 received a “High” NEWS score, while 30 patients received a “High/Urgent” MPRT-WS score. 7
Itelman et al. (2022) compared various early warning scores (NEWS, ABCNO, and “wish list” criteria) that Biobeat detected when medical staff identified actual clinical deterioration in high-risk patients on telemetry wards. 15 Of the 24 patients who experienced actual deterioration, the Biobeat monitor captured physiological data that would have triggered a NEWS early warning in 16 patients, on average 29 h before medical staff detected the actual deterioration. 15 Likewise, on average, ABCNO criteria were met in 18 patients at 38 h before actual deterioration was detected. 15 Finally, early warning based on “Biobeat detected wish list” criteria in all patients who experienced deterioration 40 h before being detected by medical staff. 15
Intensive care settings
Two studies assessed the use of Biobeat in intensive care settings.1,11 Dvir et al. (2022) compared CO measurements obtained from Biobeat chest patches to those obtained from thermodilution in ICU patients. 1 They found a high correlation in mean CO measurements between Biobeat and thermodilution (r = 0.906). 1 Moreover, the bias for Biobeat PPG was 0.3 L/m [−1.6, 2.2]. 1
Another study by Kachel et al. (2021) compared measurements obtained simultaneously by the Biobeat wrist monitor and chest patch with those obtained from an arterial line (AL) for HR, SBP, DBP, and MAP in postoperative cardiac surgery patients. 11 Bland−Altman analysis for values obtained by each method demonstrated a small bias of <1mmHg in SBP, DBP, and MAP for both the wrist monitor and chest patch compared to AL with narrow LOAs (wristwatch: SBP: −10,10 mmHg, DBP: −6,6 mmHg, MAP: −5,5 mmHg; chest−patch monitor: SBP: −9,9 mmHg, DBP: −7,7 mmHg, MAP: −6,6 mmHg). 11 Similarly, HR values also showed a small bias of <1 beat per minute (bpm) with LOA [−3,3] for both devices. 11 Measurements for SBP, DBP, MAP, and HR were all strongly correlated with those obtained by AL (r > 0.93, p < 0.001 for all). 11 Additionally, another study compared RR values obtained by the Biobeat chest patch versus Maquet Servo−I ventilators in CSICU patients and found a high correlation (r = 0.884), and a bias of <0.1 breath per minute [LOA −1.6, 1.7] in the Biobeat chest patch. 8
Study quality assessment
Table 3 and Appendix Supplementary File 3 provide the quality assessment using the Newcastle−Ottawa Scale (NOS) scoring system for observational studies. The comprehensive NOS scoring system thoroughly evaluated the three essential domains: selection, comparability, and outcome. In the selection domain, studies were assessed for the representativeness of the exposed cohort. The study population consisted of adults exposed to the Biobeat monitor in various healthcare settings. Regarding selecting the non-exposed cohort, most studies included an active intervention to compare with the Biobeat monitor. All these studies identified the use of Biobeat monitor as an intervention, and the outcome of interest was not present at the start of the study. In the comparability domain, the studies provided data on important variables like age, sex, BMI, medical disease, skin colour, etc. This ensures a fair comparison between Biobeat technology and traditional care. Finally, the studies performed the appropriate outcome assessment with statistical methods in the outcome domain, and the follow-up period was adequate and complete to capture the outcome in most of the studies. The NOS helps evaluate methodological quality and ensures reliable evidence. This assessment reduces bias and enhances the validity of Biobeat monitors in various healthcare settings. All studies were of moderate to high quality.1–12,15
Quality assessment using the Newcastle−Ottawa scale scoring system.

Total Score: 8−9: High-quality study; 7−5: Moderate quality study: < 5: Low-quality study; Some of these studies may not need the control for additional factors as they are comparative observational trials.
Table 4 and Appendix Supplementary File 4 provide the COSMIN risk of bias assessment for studies that validated Biobeat wearable devices. Three studies were ranked poor due to the small sample size (patients <30).1,4,11 One study was ranked good in the participant's sample size domain (patients <100), 14 and another study was ranked good in the measurement's sample size domain (readings <100). 1
Risk of bias assessment for studies that validated biobeat wearable device readings for continuously monitored patients in various healthcare settings.
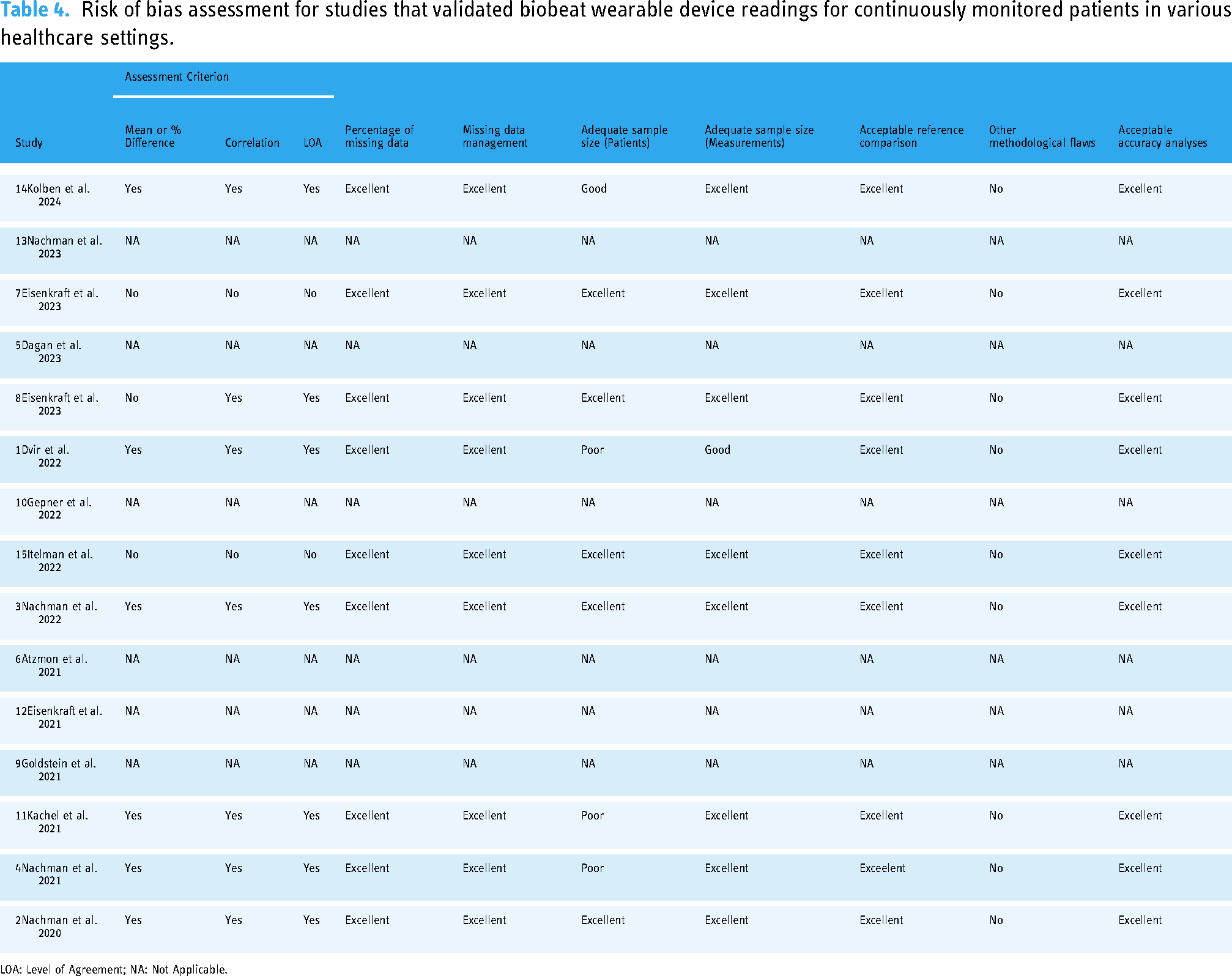
LOA: Level of Agreement; NA: Not Applicable.
Discussion
This systematic review is the first to examine the use of Biobeat monitors across various clinical scenarios. Our review suggests that Biobeat monitors are applicable in ambulatory settings, critical care, labour and delivery, and inpatient medicine units. They offer the benefits of continuous, noninvasive, wireless and real-time monitoring of several hemodynamic parameters and insights into disease diagnosis and progression, warning scores, and treatment responses within these settings.
Given the high correlation between blood pressure measurements obtained by Biobeat and standard manometry, Biobeat monitors may provide a routine method to conduct 24−hour ABPM to diagnose hypertension. Currently, the gold standard for diagnosing hypertension involves cumbersome ABPM devices, which can limit patient compliance. 23 Biobeat monitors address this problem as they can be applied to record blood pressure throughout the day while patients engage in activities of daily living. Importantly, these blood pressure measurements obtained by Biobeat are not influenced by sex, BMI, or skin colour. 3 This serves as a promising means of reliably measuring blood pressures across diverse populations, improving equity and addressing concerns of accuracy variations from person to person.
Further studies may be needed to validate the same for other physiologic parameters, as wearable devices can be susceptible to variations in readings among different patient demographics. Altogether, these findings may help address a leading risk factor for cardiovascular morbidity by providing continuous and accurate measurements across diverse populations. 3 Continuous monitoring using Biobeat may also provide insights into treatment response and disease onset before the manifestation of physical symptoms. Changes in advanced physiological parameters such as SVR, SV, CO, and CI may serve as early indicators of influenza infection or elevated inflammation following the administration of novel vaccines.9,10 In addition, they admitted patients with COVID−19 who were monitored using Biobeat demonstrated noticeable changes in physiological parameters over 5 days. 12 Thus, Biobeat monitors may have a role in assisting with identifying diseases and monitoring their progression.
Biobeat monitors may also help in the early identification of clinical deterioration based on several EWS systems. Machine learning enhances early warning systems (EWS) by identifying at-risk patients and alerting care teams for timely intervention. EWS uses data from wearable sensors to predict patient decline, often providing 30-h advance notice for proactive care that can save lives. EWS aims to promote timely and appropriate management of deteriorating patients admitted to the hospital. 15 Biobeat monitors identified high EWS scores in three models – NEWS, ABCNO, and “wish list” criteria – several hours before patients clinically deteriorated. 15 However, while the sensitivity of these findings was high, specificity remained low. 15 Another study developed a novel multi-parameter real-time warning score system derived from commonly measured parameters by Biobeat monitors. The scores from the novel MPRT-WS system obtained by Biobeat's measurements correlate with NEWS scores for the same patients. 7 Typically, EWS is calculated infrequently. 7 However, Biobeat offers the ability to calculate EWS using any model on a frequent and continuous basis, allowing for both earlier identification of the risk of clinical deterioration and real-time identification of ongoing clinical deterioration. Notably, alarm fatigue poses a challenge to healthcare staff, so further refinement of EWS criteria to increase their specificity even with Biobeat monitors is warranted. 7
Further, physiological data captured by Biobeat may contribute to increasing understanding of diseases and treatments. Dagan et al. (2023) utilized Biobeat to identify patterns in advanced hemodynamic parameters in patients with heart failure undergoing diuresis. 5 Their clustering of patients based on hemodynamic responses obtained by Biobeat monitors may inform future care directions, such as adjusting individual treatments, preventing deterioration, and maintaining the quality of life. 5 Likewise, Biobeat was used to monitor maternal hemodynamics in response to epidural anesthesia during labour and delivery. 6 Using Biobeat to monitor maternal hemodynamics in this process continuously yielded insights into the magnitude and timing of physiological adaptations, again broadening the knowledge of the procedure and individual patient responses.6
A major advantage of the Biobeat monitor is its ability to capture advanced hemodynamic information non-invasively. Compared to invasive monitoring methods such as thermodilution, AL monitoring, and ventilator readings, Biobeat monitors provide comparable readings with minimal bias.1,8,11 Through Biobeat monitors, advanced parameters such as CO, often considered challenging to obtain by noninvasive methods, can be measured accurately and safely. 1 Furthermore, noninvasive monitoring spares patients from the complications of invasive techniques, such as infection, local vascular damage, morbidity, and mortality. 24
Biobeat devices provide sophisticated remote monitoring features, tracking thirteen vital signs continuously, wirelessly, and in real time through photoplethysmography (PPG). Biobeat distinguishes itself from other wearable monitoring technologies, such as Masimo and consumer devices like the Apple Watch or Fitbit, through its clinical-grade accuracy and capability to integrate into hospital and remote patient monitoring systems. However, comparative studies directly evaluating Biobeat against other technologies are limited. Research indicates that devices such as Masimo are well-respected for their precision and are commonly employed in clinical environments, particularly for monitoring oxygen saturation and heart rate. 25 In contrast, consumer-grade wearables emphasize usability and fitness tracking but typically fall short of offering the extensive monitoring or medical-grade validation that Biobeat provides.
Various authors have utilized the Biobeat in distinct clinical environments, resulting in diverse outcomes across studies, which prevents data pooling and meta-analysis. We recognize that each unique healthcare setting calls for its own monitoring needs and contexts. For instance, ICUs utilize Biobeat to continuously monitor clinically unstable patients and inform care decisions based on dynamic changes to patients’ physiologic parameters. ICU clinicians may also prefer a more comprehensive continuous evaluation of patient hemodynamic measures, including CO, MAP, and RR.
In contrast, other studies conducted in ambulatory settings focused primarily on multiple measures of a single parameter, like BP, to diagnose hypertension. While it is valid for unique healthcare settings to have different applications for Biobeat, this presents a stark drawback to analyzing the existing data. Specifically, this limited our ability to perform meta-analyses or further statistical analyses on the primary outcomes.
Notably, our literature search in this systematic review found no RCTs; all included studies were observational. The lack of high-quality trials introduces substantial limitations to the robustness of our conclusions. Although the observational studies in this review indicated consistent results, findings from more rigorously designed studies, like multicentric RCTs, would more effectively validate our findings. Including randomized controlled trials (RCTs), of which none were present, would have more effectively addressed issues such as confounding variables and selection bias, thereby facilitating a more accurate assessment of the intervention.
Further, several studies included small sample sizes, with fewer than 100 patients undergoing hemodynamic monitoring using Biobeat monitors. These studies posed challenges related to the reliability of the findings. While positive findings were reported, the effects could have been overestimated, imprecise, and less generalizable. As such, findings from these studies with smaller sample sizes contributed less meaningfully to our conclusions.
Further, studies in other regions where healthcare infrastructure, patient populations, and clinical practice cultures may differ could enhance the generalizability of Biobeat utilization. All studies included in this review were conducted in Israel, despite Biobeat being marketed in North America and Europe. Conducting trials in these regions in the future could expand insight into the strengths and weaknesses of integrating Biobeat devices in healthcare settings universally.
All of the included studies on this topic present generally positive findings. Given the novelty of Biobeat's devices and the literature studies, there may be a potential publication bias. However, our search strategy was robust, and grey literature was hand-searched for other relevant articles to minimize systematic causes of publication bias. Consequently, although affirmative findings may indicate the presence of publication bias within the existing literature, our search strategy was methodically designed to encompass all studies pertinent to the utilization of Biobeat in healthcare environments, irrespective of the outcomes.
Future research should address these limitations by implementing more robust methodologies, including well-designed multicentric RCTs with larger sample sizes. These studies could provide more precise insights into the efficacy of Biobeat monitors across perioperative clinical scenarios and patient populations.
Moreover, incorporating additional outcome measures and exploring potential confounding factors would enhance the validity and reliability of research findings. By doing so, researchers can better understand the true impact of Biobeat monitors on postoperative patient outcomes and healthcare delivery. Furthermore, comparative studies comparing Biobeat monitors with existing monitoring technologies or standard care approaches would be valuable in assessing their added benefits and cost-effectiveness.
Conclusion
This systematic review demonstrates that Biobeat's non-invasive wearable devices, including the BB-613WP wrist monitor and the BB-613P chest patch, have been effectively utilized across various clinical settings, contributing to positive patient outcomes. The studies included in this review encompass a wide range of environments, from ambulatory care to intensive care units, indicating the versatility and reliability of Biobeat devices. The consistent reporting of favourable outcomes highlights the potential of these advanced hemodynamic monitoring tools to enhance patient care, support clinical decision-making, and ultimately improve health outcomes. Further research is warranted to expand upon these findings and explore the long-term benefits and broader applications of Biobeat technology in diverse patient populations.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251324012 - Supplemental material for Biobeat monitor utilization in various healthcare settings: A systematic review
Supplemental material, sj-docx-1-dhj-10.1177_20552076251324012 for Biobeat monitor utilization in various healthcare settings: A systematic review by Yifan Zhang, Jill Querney, Yamini Subramani, Kendra Naismith, Priyanka Singh, Lee-Anne Fochesato, Nida Fatima, Natasha Wood, Richard Malthaner and Mahesh Nagappa in DIGITAL HEALTH
Footnotes
Acknowledgement
We thank Brad Dishan, BSc, MLIS, Clinical Librarian at the Health Sciences Library, London Health Sciences Centre, for his assistance with the literature search.
Attestation
All authors approved the final manuscript.
Author contributions
All authors contributed to the study's design, literature search, data collection, data analysis, and manuscript writing.
Consent statement
Not applicable as this is a systematic review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Research ethics board approval
This does not apply to this study. According to standard protocol, the institutional review board did not need to approve this systematic review, as it did not involve research on human participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
