Abstract
Objective
This review aims to systematically map and categorize the current state of wearable applications among oncology patients and to identify determinants impeding clinical implementation.
Methods
A Medline, Embase and clinicaltrials.gov search identified journal articles, conference abstracts, letters, reports, dissertations and registered studies on the use of wearables in patients with malignancies published up to 10 November 2021.
Results
Of 2509 records identified, 112 met the eligibility criteria. Of these, 9.8% (11/112) were RCTs and 47.3% (53/112) of publications were observational. Wearables were investigated pre-treatment (2.7%; 3/112), during treatment (34.8%; 39/112), post-treatment (17.9%; 20/112), in survivors (27.7%; 31/112) and in non-specified or multiple treatment phases (17.0%; 19/112). Medical-grade wearables were applied in 22.3% (25/112) of publications. Primary objectives ranged from technical feasibility (8.0%; 9/112), user feasibility (42.9%; 48/112) and correlational analysis (40.2%; 45/112) to outcome change analysis (8.9%; 10/112). Outcome change was mostly investigated regarding physical activity improvement (80.0%; 8/10). Most publications (42.9%; 48/112) and registered studies (39.3%; 24/61) featured multiple cancer types, with breast cancer as the most prevalent specific type (22.3% in publications, 16.4% in registered studies).
Conclusions
Most studies among oncology patients using wearables are focused on assessing the user feasibility of consumer-grade wearables, whereas rates of RCTs assessing clinical efficacy are low. Substantial improvements in clinically relevant endpoints by the use of wearables, such as morbidity and mortality are yet to be demonstrated.
Introduction
An aging population with increasing cancer prevalence poses significant challenges to healthcare systems worldwide. 1 Personalized pre-treatment assessment becomes paramount due to diverse treatment options, including CAR-T cell therapy. During cancer treatment, patients may encounter complications like infections, frailty, cardiac disorders and treatment-specific side effects, for example, cytokine release syndrome.2–4 Decreased physical activity (PA), fatigue syndrome and mental health disorders are commonly observed during follow-up of cancer patients.5,6 Early detection of health deterioration could reduce rates of hospitalization or emergency visits, as shown in other medical fields.7–9 Moreover, delayed diagnosis and treatment of complications are associated with higher morbidity and mortality. 10 As outpatient treatment becomes more prevalent, disease monitoring largely depends on patient self-management and regular clinic visits, posing challenges to patients and healthcare professionals (HCP) and underscoring the need for effective monitoring techniques. 10
Wearable devices equipped with non-invasive sensors offer continuous health data monitoring under daily life conditions. These “wearables” utilize well-established sensor technologies, such as accelerometry, temperature probes and photoplethysmography, enabling subsequent or real-time data acquisition. Generating and processing vast amounts of “big data” is challenging yet offers vast opportunities in healthcare. It enables the creation of a “digital twin”—a virtual patient representation that helps anticipate clinical events and guide personalized treatment decisions by monitoring changes in real-time parameter. 11
Wearables have been successfully applied to early detect COVID-19, 12 heart failure, 13 Alzheimer’s disease 14 and seizures in patients with epilepsy, 15 as well as health deterioration or adverse events (AE) of certain therapies. 16 Additionally, wearables can objectify patient-reported outcomes (PRO), otherwise prone to subjectivity and highly depend on patient adherence, such as quality of life (QoL) 17 and pain. 18 Furthermore, wearables can serve as interventional tools to enhance PA,19–23 thereby improving overall well-being.20,23
Despite their potential across diverse applications, wearables are rarely implemented clinically in oncology. This scoping review aims to explore the current state of wearable application among oncology patients, focusing on determinants impeding clinical implementation by evaluating and extracting information from published clinical studies.
Methods
For this scoping review, the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-ScR) were used. 24 The final protocol was registered with the Open Science Framework on 16 November 2021 (https://osf.io/bgmya). 25
We included publications focusing on wearable application in adult patients with a confirmed oncologic disease. The term “wearable” is defined as a mobile hardware device with integrated software that collects clinical data via non-invasive sensors by measuring different parameters. Medline, Embase and clinicaltrials.gov were searched up to 10 November 2021 for full-text journal articles, letters, reports, dissertations, conference abstracts and trial registrations in any language (with keywords in English) with no limitation in the date of publication. The search strategies employed are described in Supplemental Tables 1 and 2. Keywords for the search on clinicaltrials.gov were “wearable” and “oncology.”
Three authors (KC, MV and MJ) independently assessed the eligibility of records through title and abstract screening, followed by full-text screening. Disagreements were settled by a fourth author (JB) through consensus. Data extraction was performed by the first author (KC) and revised by a second author (MJ or MV), using a standardized form (Supplemental Table 3).
For quality assessment, full-text records’ adherence to research guidelines was evaluated using aspects of STROBE, CONSORT and TREND, which we merged into a shorter checklist (Supplemental Table 4). A three-point grading system was used to categorize guideline adherence as “full,” “partly” or “none.” Due to a heterogeneous nomenclature of study designs, publications were categorized according to the common nomenclature of study designs. 26 Study objectives were categorized by stage of maturity, modified after the World Health Organization “Monitoring and Evaluating Digital Health Interventions” manual to reflect the stages a wearable passes through from prototype to clinical implementation. 26 Collected parameters were categorized to warrant a coherent data analysis. A full list of categories with specifications is presented in Supplemental Table 5. In the case of study characteristics with more than one feature option (e.g. malignancy type, wearable type) reported shares might not add up to 100%. Registered planned studies from clinicaltrials.gov are presented as future perspectives.
Results
The literature search yielded a total of 2509 publications, with 112 publications meeting eligibility criteria after title, abstract and full-text screening (Figure 1). Main characteristics of included publications are listed in Table 1.

PRISMA flow diagram of the screening and selection of publications.
Main characteristics of included publications; n = 112.

An increasing number of published studies can be observed from the earliest eligible publication in 2007 until 2021 (26 publications) with a short-lasting decrease in 2020 (14 studies) (Figure 2). Most studies were published by first authors located in the USA (55.4%; 62/112), followed by Europe (29.5%; 33/112), Asia (6.3%; 7/112), Canada and Australia (both 4.5%; 5/112) (Supplemental Figure 4).
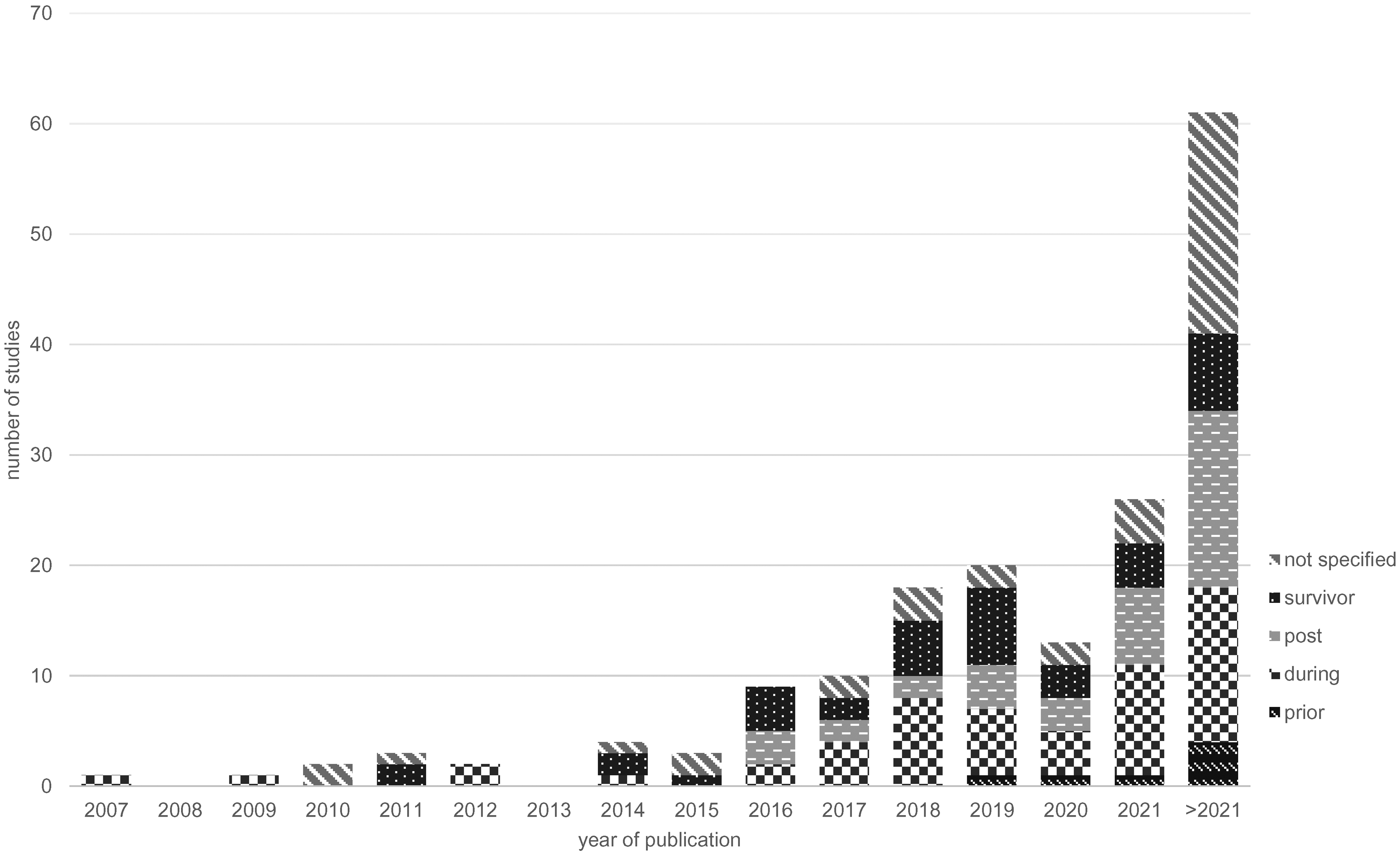
Number of published studies (year 2007–2021) and registered studies on clinicaltrials.gov (year >2021) per treatment phase per year of publication.
The search of clinicaltrials.gov resulted in 117 registrations, of which 61 were eligible. 63.9% (39/61) of those were registered by researchers located in the USA (Supplemental Figure 5).
Study characteristics
Study characteristics are summarized in Table 2. Around 10% of publications were RCTs and nearly half of publications applied a prospective longitudinal design (47.3%; 53/112) followed by 19.6% (22/112) cross-sectional studies.
Main characteristics in publications and registered studies.

Only 8.9% (10/112) of publications conducted outcome change analyses (Figure 3), applying a weighted mean duration of 80.2 days (47.8–112.7).

Primary study objectives of published studies according to the stage of maturity, modified after “WHO monitoring and evaluating digital health interventions.” The sizes of the boxes do not correspond to the number of studies of the respective category.
Interestingly, compared to this, the share of registered RCTs is four times higher. The share of registered outcome change analysis is three times higher compared to publications, applying a higher planned average duration of 129.1 days.
Device characteristics
Device characteristics are shown in Table 2. 22.3% (25/112) of publications applied medical-grade devices. Within the subset of EU-based publications, 27.8% (5/18) incorporated medical-grade devices, while in USA-based publications, medical-grade devices were used in 17.7% (11/62) of publications.
The most commonly used sensors were accelerometers and actigraphs (85.7%; 96/112), followed by HR sensors (16.1%; 18/112), temperature sensors (3.6%; 4/112) and ECG (1.8%; 2/112). In 76.8% (86/112) of publications, one sensor was applied, 21.4% (24/112) utilized two to four sensors and 1.8% (2/112) did not specify applied sensors.
Registered studies do not show a clear trend to applying multiple sensors: 65.6% (40/61) of registered studies are planning to employ one sensor, followed by two sensors (18.0%; 11/61), five sensors (1.6%; 1/61) or not specified sensor count (9.9%; 9/91).
Participants’ characteristics
Participants’ characteristics are reported in Table 2. Included populations had a weighted mean age of 50.1 (48.2–51.9) years and a mean female participant ratio of 51.5%, or 45.2%, excluding studies assessing a sex-specific malignancy. Most publications (42.9%) and registered studies (39.3%) featured multiple cancer types, with breast cancer as the prevalent specific type (22.3% in publications, 16.4% in registered studies).
Primary objectives
Figure 3 depicts the primary study objectives of publications according to the maturity stage of the assessed wearable.
Technical feasibility
8.0% (9/112) of publications assessed the accuracy, reliability, or validation of wearables, for example, minimum number of nights needed to obtain accurate results of objective sleep 29 or association of wearable and pedometer measurements. 27
User feasibility
Most publications (42.9%; 48/112) examined user feasibility, for example, by assessing wearable usage, trial completion, data transmission or adverse events. This heterogeneity of feasibility definition is described in detail by other reviewers. 28 A single study described wearable applications as “potentially feasible,” 29 and all remaining studies concluded a high level of feasibility.
The share of feasibility assessment as the primary objective will be exceeded by registered studies (50.1%; 31/61).
Correlational analysis
Correlational analyses of wearable data to clinical outcomes and to PRO were conducted by 40.2% (45/112) of publications. Wearable data were demonstrated to correlate to hospitalization rate, 30 AE, 31 overall survival, 32 performance, 33 risk, 34 symptoms35,36 and QoL. 37 However, consistent correlations of wearable data to subjectively reported PA or sleep were not established due to inaccurate sleep tracking and misinterpretation of inactivity as sleep by wearables.38–40 Similarly, according to the authors, a correlation between activity and hospitalization duration could not be demonstrated consistently due to variations in the study population. 41
Fewer registered studies plan to conduct correlational analyses (19.7%; 12/61), resembling clinical endpoints of published studies. One novel approach is a correlational analysis of wearable data and pain.
Outcome change analysis
A small share of publications analyzed outcome change (8.9%; 10/112), predominantly aiming to improve PA as the primary endpoint (80%; 8/10). Two publications measured weight loss as a primary endpoint (20%; 2/10).42,43 A beneficial effect of wearables was mainly apparent among patients characterized as posttreatment and cancer survivors. Two publications report a lack of positive outcomes after wearable-supported interventions, due to a lack of motivation 44 and underpowered study conditions, 45 according to the authors.
Registered trials plan to conduct more outcome change analyses (27.9%; 17/61) with a broader variability of clinical endpoints, which will be reported in the respective treatment phases of the following paragraph.
Treatment phases
The distribution of treatment phases in which the studies were conducted is presented in Table 2. Study endpoints within respective treatment phases and respective stages of maturity are presented in Figure 4.

Study endpoints within the continuum of oncology care, categorized according to study objectives’ stage of maturity (n = 112). The number in brackets represents the number of studies in the respective category. Technical feasibility is not displayed.
Pretreatment phase
Of the three publications that assessed patients pre-treatment, two studies were conducted as prospective longitudinal studies and one study was an RCT. 46 Wearables were reported to be successfully applied for surgical risk assessment 34 and as a prehabilitation tool before surgery. 46
Registered studies show a relative increase in studies utilizing wearables in the pre-treatment phase. Of the three studies that plan to apply wearables for prehabilitation, two will conduct outcome change analysis (NCT03902834, NCT03689634) and one will assess feasibility (NCT03162718).
During treatment phase
Of the 39 publications that assessed patients during treatment, one was designed as RCT, aiming to increase PA 47 and 26 publications (66.7%) conducted correlational analyses.
Of the 20 registered studies, during treatment, wearables will mainly be applied for feasibility assessment. An exercise intervention for balance improvement by wearable application represents a novel investigational approach in oncology (NCT02773329).
Posttreatment phase
Of the 20 publications which assessed patients post-treatment, one was designed as RCT. 45 Wearables were reported to be successfully applied to increase PA,42,48,49 predict hospitalization50,51 and toxicities 52 and objectify symptoms, 53 among others. One study implementing an intervention to increase PA did not show a significant benefit due to underpowered study conditions, according to the authors. 45
Of the 16 registered studies, wearables post-treatment are planned to be applied to conduct exercise interventions to increase PA mostly, either the latter being the primary endpoint (NCT05046756, NCT05031052, NCT04907591, NCT04783168) or investigating subsequent effects of PA increase, for example, reduced readmissions (NCT04314778), decreased insulin resistance (NCT04649671), improved bone mineral density (NCT04144127) and changes in mental health (NCT03874754). Further registered studies aim to decrease ultraviolet (UV) exposure (NCT03344796, NCT03518229).
Survivor phase
Of the 31 publications including cancer survivors, 21 conducted feasibility assessments. Seven studies were designed as RCTs, assessing feasibility and outcome change of exercise interventions. Wearables were reported to be successfully applied to increase PA 54 and to reduce body weight. 43
Of the nine registered studies, wearable-based interventions aim to increase PA (NCT03612596) and reduce UV exposure (NCT03927742). Feasibility assessment of a breathing intervention represents a novel approach to wearable application in oncology (NCT03773380).
No specific treatment phase
Of the 19 published studies that did not specify a treatment phase or analyzed cohorts from multiple treatment phases, most studies included cohorts with multiple malignancy types (57.9%; 11/19).
One study of 12 registered studies plans to decrease cardiotoxicity risk by implementing a preventive, artificial intelligence-based intervention in a clinical setting (NCT04885088).
Quality assessment
The full evaluation is given in Supplemental Table 4. The majority of publications eligible for quality assessment did not specify their study design in the title or abstract by using commonly recognized terms (56.3%; 36/64). Eligibility criteria, sources and methods of selection of participants and methods of follow-up were not defined by 31.3% (20/64) of studies.
Study size determination was described in only 12.5% (8/64) of studies. Most studies did not address missing data handling 81.3% (52/64). Number of participants at each stage of the study was not reported by 31.3% (20/64) of studies. Generalizability was not discussed by 70.3% (45/64) of studies. Reasons for limited generalizability were selection bias to patient selection, for example, due to required technical 55 /smartphone access56,57 or required smartphone literacy 58 and small sample size.59,60
Discussion
For this scoping review, 112 research papers, published between 2007 and 2021, were identified that examined wearable applications in oncology, regarding their technical and user feasibility, correlation of wearable data to PRO and to clinical outcomes and their capability to change participants’ lifestyles. This study indicates a rising use of wearables addressing variable endpoints in oncology research across the continuum of oncology care.
The surge in studies on wearables in oncology is driven by technological advancements, a focus on patient empowerment and personalized medicine, the potential for cost-effectiveness and early detection and intervention, interdisciplinary collaboration, demand for increasing outpatient treatment and surveillance and growing regulatory interest. This exponential growth of publications on wearable applications has been documented not only in oncology28,61–63 but also in other medical fields,64,65 for example, chronic diseases. 66 In oncology, prior reviews assessed wearable devices, but limited their research to specific data items, for example, activity data, 62 analyzing wearable usage in a certain oncologic treatment phase, 28 or investigating particular objectives, for example, adherence and clinical outcomes.28,67
The average age of study participants was ∼50 years, with this younger demographic particularly receptive to new technologies. A restricted access to wearable usage due to lack of familiarity with smart devices and technical requirements such as reliable internet connection or the necessity of owning a compatible private smartphone limit generalizability of included studies.55–57 Further selection biases discussed by authors of included studies arise due to higher motivation 68 of study participants, higher educational level 69 and a healthier lifestyle 70 compared to the general cancer populations. In order to antagonize digital health disparities, these aspects should be considered while interpreting the results of the respective trials. For real-world implementation, these aspects should be addressed by wearable manufacturers, HCPs and research centers.
Further, the evaluation of the efficacy of wearables on clinical outcomes, a crucial step before clinical implementation, was only conducted sparsely, primarily showing success in survivor cohorts and in posttreatment cohorts. Despite a slight trend toward assessment during and posttreatment, critical treatment phases require further investigation, addressing clinical endpoints beyond change in PA.
More homogenous participant cohorts, especially concerning a specific cancer type, could add value to research results since physical conditions of patients with different cancer types and treatment modalities often vary. Evidence of wearable application in specific cancer cohorts might facilitate clinical implementation of wearables, since patients are often bound to cancer-specific centers or wards and thus could be elected and approached easily.
The clinical impact on oncology outcomes currently mainly seems favorable to PA improvement, in line with Jo et al., who investigated clinical outcomes of wearable application in patients with chronic diseases. 66 Successful long-term PA improvement and subsequent clinical changes remain to be shown. Only two studies confirmed a significant PA increase after 3 months compared to a control group with no intervention and no wearable.46,68 Of those, one was a single-center study with 22 participants, which achieved a significant improvement in the 6 Minutes-Walk-Test. However, a significant change in daily active minutes could not be achieved. 46 Lynch et al. describe a significant PA increase and sedentary time reduction in breast cancer survivors after three months. 68 Further studies indicated PA increase or weight loss up to 3 months, but without the comparison to a control group, 42 or with no significant difference to included control groups.42,43,48,49,69 Only two studies present outcome change results after 6 months: Lemanska et al. report a PA decrease after initial PA increase after 3 months (with no control group) and Kong et al. found increased PA levels with no significant difference to a control group without wearable.46,69
In line with an increase in PA, two of the above-mentioned publications observed significant weight loss after 3 months,42,43 whereas QoL remained unaltered. 42 Future studies should address additional clinical endpoints induced by altered PA. Limited change can be observed considering the number of applied sensors in published studies and planned studies. Wearable research in oncology populations is predominantly based on one sensor—both in published and registered studies. Despite these trends, longer study duration and inclusion of well-defined patient populations with respective control groups are desirable. The high share of feasibility analyses of publications and planned studies should proceed to more advanced study objectives. Innovative technology with multi-sensor approaches could enhance wearables’ potential in predicting diseases and optimizing treatment selection through the concept of a “digital twin.”
A high share of studies published by US-based authors was observed in line with other reviews.62,70 Strict regulatory laws concerning medical certification in the EU were presumed as a cause. 70 This assumption can only be agreed with regarding prospective medical certification since this review found a high share of studies that applied consumer-grade devices without medical certification—both in the EU and the USA. More strict data privacy and security regulations in the EU complicate research in this sensitive field, both from researchers’ and from participants’ points of view, who might be skeptical concerning the adaption of new technologies and especially concerning the processing of sensitive personal health data. 71 One-third of the adult US population is estimated to use wearables 72 and more than 80% of US wearable users report willingness to share wearable information with HCPs. 73 In contrast, only 19% of the EU population between 16 and 74 years reported to use wearables in 2020. 74 Although this information cannot be directly compared, wearable usage is likely more common in the USA than in Europe.
“No evidence, no implementation—no implementation, no evidence”—this paradox stated by Guo et al. encapsulates the challenges of digital health, relating to a lack of real-world evidence of digital health solutions. 75 Evidence-based evaluation of wearables is crucial for patients, HCPs, investors, regulatory bodies, wearable developers and benefactors. Observational study designs with small sample sizes were prevalent in this scoping review, mainly focusing on feasibility and correlational analyses during cancer treatment and in cancer survivors. Despite low flexibility and long periods from planning to publication and difficulties of blinding, RCTs remain the gold standard for efficacy assessment. Only a small proportion of publications were RCTs, while a higher share of registered studies plans to conduct RCTs, focusing on during and post-treatment evaluations.
Innovative study designs are needed to address the challenges encountered in digital health research, for example, dynamics of digitization, software updates and software bugs. “Pragmatic RCT,” adaptive designs and virtual trials are possible approaches to complement conventional RCT. 76 Alternative study designs such as Continuous Evaluation of Evolving Behavioral Intervention Technologies (CEEBIT), Multiphase Optimization Strategy (MOST), Sequential Multiple Assignment Randomized Trials (SMART), Factorial/Fractional RCT and Micro-Randomized Trials (MRT) are seen as helpful tools for the development of digital health interventions.76–78 Various frameworks and guidelines for the evaluation of digital health interventions are reviewed by Kernebeck et al., including, for example, a CONSORT-/STROBE expansion for eHealth interventions. 79
Moreover, our review indicates a need for further efforts to use medical-grade wearables for accurate data collection. The results call for investigations of wearables’ short-term and long-term effectiveness in oncology populations, including survival rates, malignancy recurrence rates and hospitalization rates. Wearables’ capability to potentially predict symptoms and identify patients’ deterioration early may facilitate patient care with malignant diseases and result in improved clinical outcomes. Besides health outcome changes, positive effects of wearable application on healthcare processes should be demonstrated, for example, in terms of economic or financial evaluation and workload reduction of HCPs.
The strengths and limitations of the present study merit consideration. First, the application of a wearable and measurement of parameters, which can be consciously controlled by the patient (e.g. PA), could be seen as an intervention per se, according to the Hawthorne effect. 80 In this review, studies intending to change collected parameters (e.g. through a step goal) were considered interventional studies and studies which applied wearables to, for example, foresee clinical events, were considered observational studies. Second, the categorization of parameters was conducted carefully, with an attempt to achieve a meaningful and objective evaluation. However, any categorization can lead to a certain bias in the results—especially when analyzing and classifying heterogeneous studies. Furthermore, the categories are partly overlapping and are not as clear in practice as they are in theory, which might lead to restricted reproducibility. Third, technical devices new to the market, such as wearables, are prone to fast-moving development of hardware and software. Since trials often present momentary assessment of devices, technical changes, especially in early published studies, cannot warrant continued validity. This also applies to this scoping review. Fourth, the interpretation of clinicaltrials.gov is limited by the fact that details of the study, such as trial performance, completion and publication, are not reliable. Aimed study designs and methods could be adapted due to altered circumstances and downward adjustment of planned study quality seems more likely than vice versa.
Conclusions
Although less than 10% of clinical evaluations of wearables in oncology patients were done by RCTs so far, wearables appear to show promise for improving clinical care of patients with malignant diseases. Challenges in evidence creation, study design and regulatory aspects need to be addressed for their effective implementation. Long-term outcome change of wearables should be analyzed to demonstrate their effectiveness. Wearable application should be assessed in critical treatment phases, analyzing endpoints beyond PA change. Application of medical-grade wearables is desirable, optimally able to process big data sets or even enabling the adaption of algorithms based on the “digital twin” concept.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-docx-1-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-2-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-2-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-3-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-3-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-4-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-4-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-5-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-5-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-6-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-6-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-xlsx-7-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-xlsx-7-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Supplemental Material
sj-pptx-8-dhj-10.1177_20552076241233998 - Supplemental material for Application of wearables for remote monitoring of oncology patients: A scoping review
Supplemental material, sj-pptx-8-dhj-10.1177_20552076241233998 for Application of wearables for remote monitoring of oncology patients: A scoping review by Katharina Cloß, Marlo Verket, Dirk Müller-Wieland, Nikolaus Marx, Katharina Schuett, Edgar Jost, Martina Crysandt, Fabian Beier, Tim H Brümmendorf, Guido Kobbe, Julia Brandts and Malte Jacobsen in DIGITAL HEALTH
Footnotes
Acknowledgments
None.
Contributorship
KC contributed to conceptualization, methodology, data curation, formal analysis and writing–original draft. MV contributed to conceptualization, methodology, data curation and review and editing. DM-W contributed to supervision, conceptualization and review and editing. NM contributed to review and editing. KS contributed to review and editing. EJ contributed to review and editing. MC contributed to review and editing. FB contributed to review and editing. TB contributed to review and editing. GK contributed to review and editing. JB contributed to project administration, conceptualization, methodology, data curation, visualization and review and editing. MJ contributed to project administration, conceptualization, methodology, data curation, visualization and review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from the internal grant program of the University Hospital Aachen, RWTH Aachen University, Germany. Dr Marx is supported by the Deutsche Forschungsgemeinschaft (German Research Foundation; TRR 219; Project-ID 322900939 [M03, M05]).
Guarantor
MJ, JB.
Informed consent
This manuscript presents a literature review of publicly available information. Since no individual data were used for the analysis presented in this manuscript, the requirement for consent is not applicable.
Registration
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
