Abstract
Background
The heterogeneous etiology of mild cognitive impairment (MCI) presents significant challenges in monitoring its progression and impeding its advancement toward dementia. Digital multidomain lifestyle interventions have shown promise as potential solutions for their ability to treat MCI.
Objective
This study is the first phase in a series of evaluations aimed at assessing various components of Silvia-Rx, which was originally designed as a digital multidomain lifestyle intervention. Specifically, this study focused on a 60-session core cognitive training program to evaluate its feasibility and efficacy in addressing cognitive decline in individuals with MCI.
Methods
Individuals aged 60 to 80 years diagnosed with MCI were enrolled to participate in a 60-session tablet-based cognitive regimen of Silvia-Rx. Feasibility was assessed through adherence and retention rates, while the Alzheimer's Disease Assessment Scale-Cognitive (ADAS-cog) scale was utilized to measure cognitive function as the primary indicator of efficacy.
Results
The mean age of the participants was 71.11 years, and 13 (68.42%) were women. Only one withdrawal occurred, resulting in a 95% retention rate (19 participants) post-intervention. The completion rate was excellent at 100%, indicating that the 60-session core cognitive program in Silvia-Rx was well tolerated by older participants with MCI. Regarding efficacy, there was a statistically significant improvement in cognitive function among MCI participants after the intervention, as evidenced by changes in total ADAS-cog scores.
Conclusions
Results demonstrated excellent adherence throughout the program and significant cognitive improvements after the intervention. This pilot study indicates that Silvia-Rx's digital cognitive program is feasible for people diagnosed with MCI, suggests potential for improving cognitive function, though further research with large sample size is needed to confirm these results.
Keywords
Introduction
Background
The aging population has been continuously increasing all over the world. In 2022, almost 10% of the global population was over 65 years of age, and it is anticipated to reach 16% by the year 2050. This highlights the increasing worldwide challenge posed by degenerative diseases, particularly dementia. Mild cognitive impairment (MCI) is characterized by a level of cognitive deterioration that exceeds expectations based on an individual's age and educational background yet does not substantially hinder daily functioning. 1 The prevalence of MCI among older adults is a growing concern since it is recognized as a transitional stage from normal cognitive aging to the development of dementia. 1 Worldwide, approximately 14.9% of individuals over 60 years old with MCI transition to dementia within the next two years, and one-third of those with MCI are diagnosed with dementia within five years. 2
At present, no particular drug treatments exist for managing MCI. 3 Given the heterogeneous etiology in the clinical spectrum of MCI, monitoring its progression and impeding its advancement toward dementia present significant challenges. 4 Therefore, an increasing number of non-pharmacological interventions have emerged as promising strategies for managing MCI to reduce the risk of transferring to the onset of dementia.5–7 With the increasing prominence of non-pharmacological interventions, studies have investigated single-domain interventions such as exercise, diet, and cognitive training. However, their effectiveness has often been limited, prompting the proposal of integrated multidomain interventions to achieve even greater health benefits. 7 The evolution of these interventions from single-domain to comprehensive multidomain lifestyle approaches has increased their preventive and therapeutic efficacy by tackling various risk factors. 8
Despite the acknowledged significance of multidomain interventions, the delivery of two or more interventions through face-to-face approaches has substantial limitations regarding time, space, and financial resources. To overcome these challenges, digital interventions have emerged as a promising alternative. However, current digital intervention studies for cognitive impairments are highly heterogeneous and often limited to specific domains, particularly exercise. 5 In previous studies, there was diversity not only in terms of types of interventions, modes of delivery, materials, procedures, location, duration of interventions, length, frequency, and intensity of sessions, but also in defining mild cognitive impairment using simple cognitive questionnaires without clinical evaluation. Furthermore, there is a lack of exploration on the key issues of successful digital interventions, such as applicability, accessibility, acceptability, and sustainability. These points indicate that there is insufficient development and research of multidomain digital interventions for individuals with mild cognitive impairment. They also highlight the need for accurate diagnosis and approaches based on underlying causes.
The existence of several modifiable risk and protective factors across the lifespan underscores the promise of multidomain lifestyle interventions, which encompass cognitive training, physical exercise regimens, and other lifestyle modifications, in amplifying their effectiveness in mitigating risk factors.9,10 Among these interventions, cognitive training has been posited as a means to fortify cognitive functions through the execution of repetitive, varied cognitive tasks grounded in the concept of neuroplasticity. 11 In this vein, cognitive training is also considered to work as ‘software’ by enhancing the operation and adaptability of neural circuits. 12
Cognitive reserve, closely related to brain plasticity, refers to a theoretical concept that compensates for age-related decline and pathological damage. 13 It has been introduced to explain the variance among individuals in their resilience to brain aging and pathological alterations, including those seen in Alzheimer's disease. Reserve serves as a buffer between pathology and clinical outcome, thereby explaining their observed discrepancies. 12 The protective role of cognitive reserve against the risk of dementia has been extensively documented.8,14,15 A recent study indicated that daily engagement with digital devices could develop an individual's cognitive reserve and further safeguard against cognitive decline in older adults. 16 Therefore, non-pharmacological digital multidomain intervention, particularly cognitive training, could offer the potential for personalized and scalable solutions to support brain health. 17 In this context, technology-driven evaluations afford a superior level of precision in both cognitive measurement and scoring processes, as well as automated feedback and results. 18 Digital solutions facilitate clinical services via smartphones or alternative portable devices, effectively minimizing time, frequency, and geographical obstacles to cognitive assessment. 19 Consequently, this approach enables efficient and continuous monitoring to identify early, subtle signs of cognitive impairment.
In this line, the digital multidomain lifestyle intervention platform, Silvia-Rx, was designed and developed. The primary aim of this single-arm pilot study, which mainly focused on its core cognitive training in Silvia-Rx, was to evaluate the feasibility and efficacy of the core cognitive program in older adults diagnosed with MCI. To assess feasibility, we examined adherence and retention to this program across the 60 sessions. We hypothesized that older adults with MCI would consistently engage with the core cognitive program in Silvia-Rx and maintain participation throughout the sessions. Regarding efficacy, we measured the global cognitive function of all participants pre- and post-intervention. It was hypothesized that there would be a maintenance or improvement in the overall cognitive performance of the participants following the completion of the 60-session program.
The secondary goal of the study was to assess a range of health-related outcomes through self-reported questionnaires, which included measures of depression, anxiety, stress, sleep quality, and motivation to prevent dementia. The aim was to examine whether these outcomes would show any changes following the intervention compared to their baseline levels. We anticipated that there would be an improvement in these health-related outcomes after completing the 60-session core cognitive program.
Methods
Participants
A total of 20 individuals diagnosed with MCI were recruited through notices at the regional center conducting dementia screening as part of the projects of the Korea National Institute of Dementia, and through the geriatric clinic at Wonju Severance Christian Hospital, which is affiliated with this center. All participants were community-dwelling older adults, diagnosed according to criteria proposed by MCI criteria of the International Working Group 20 based on a similar concept with mild neurocognitive disorder of the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5). 21 Diagnoses were confirmed by psychiatrists based on careful history-taking about symptoms, neurological tests, radiological examination, and genetic testing. Structured assessments of cognitive abilities were conducted, gathering information from both the patient and their caregiver and subsequently evaluating Clinical Dementia Rating (CDR) 22 and CDR-Sum of box (SOB) 23 scores. Objective cognitive impairment was ascertained 1.5 standard deviation (SD) or more below the norm using the neuropsychological assessment battery of the Korean version of the Consortium to Establish a Registry for Alzheimer's Disease assessment packet (CERAD-K) 24 by a trained research nurse or neuropsychologist. The neuropsychological assessment battery of CERAD-K comprised tests for memory (word list memory, word list recall, and word list recognition), language (fluency and naming), constructional ability (constructional praxis and constructional memory), and executive function (trail-making test and Stroop test).
Potential participants were informed about the research, and those who expressed interest in participating were contacted by the research team to go through the informed consent process. Participants were eligible for inclusion if they were: 1) aged 60 to 80 years old; 2) reported subjective cognitive complaints by themselves and/or informant; 3) ascertained objective impairment by more than one of the neuropsychological tests; 4) maintained or only slight impairments in their functional capacities and do not manifest indications of dementia; and 5) Korean speaking.
The exclusion criteria were 1) a history of severe psychiatric and neurological disorders such as bipolar disorder, schizophrenia, stroke, Parkinson's disease, or epilepsy; 2) a documented history of alcohol or substance abuse; 3) severe sensory and motor impairments that impact their ability to utilize a tablet device; 4) receipt of any concurrent cognitive and memory training program at the time of their initial screening; and 5) unable to manually manipulate the mobile device.
Before the initial screening, all patients were provided with comprehensive information regarding the goals and procedures of the study. All patients with MCI who participated in this study had the ability to read and understand the study procedures and consent form. Therefore, those who willingly consented to partake in the study formally acknowledged their agreement by signing the provided written consent forms without the involvement of their legally authorized representatives. This study was carried out in accordance with the Declaration of Helsinki. 25
Study design and intervention
This study was a pre-post pilot study to examine the feasibility and preliminary efficacy of the core cognitive program in Silvia-Rx. The application was used to collect all study data and monitor the completion of program-related tasks. All participants performed all study procedures remotely via the tablet device that we provided.
All patients could access the Silvia app (Silvia Health Inc., South Korea) to join a 60-session program of Silvia-Rx based on scientific evidence to improve brain functions and reduce dementia risk.
10
The multidomain lifestyle intervention was designed for 60-session core and supplementary programs. All participants were recommended to engage in the core program for one session per day, at least five days a week, with each session lasting 20 to 30 minutes. The core cognitive program consisted of 2 modules as follows (Figure 1):
Cognitive training: Cognitive training is an obligatory training regimen. Gamified cognitive training targeting a broad set of cognitive domains focused on memory, visuospatial, language, executive function, and attention was provided. Upon completion of each task, performance metrics are displayed, along with comparative data indicating the extent of cognitive improvement relative to previous results. Examples of the programs include detecting patterns, memorizing lists, and rotational shape activities. User interaction with these tasks is carried out through touchscreen responses. Episodic memory: Episodic memory training is also designated as an obligatory training regimen. It is structured to present a narrative about a single plant every 2–3 days for 60 sessions. Participants are subsequently required to recall these narratives to answer quiz questions about the respective plant. Each narrative includes the plant's name and its historical or etymological origins. Upon successfully answering the quiz questions, participants are awarded plant cards as a reward.
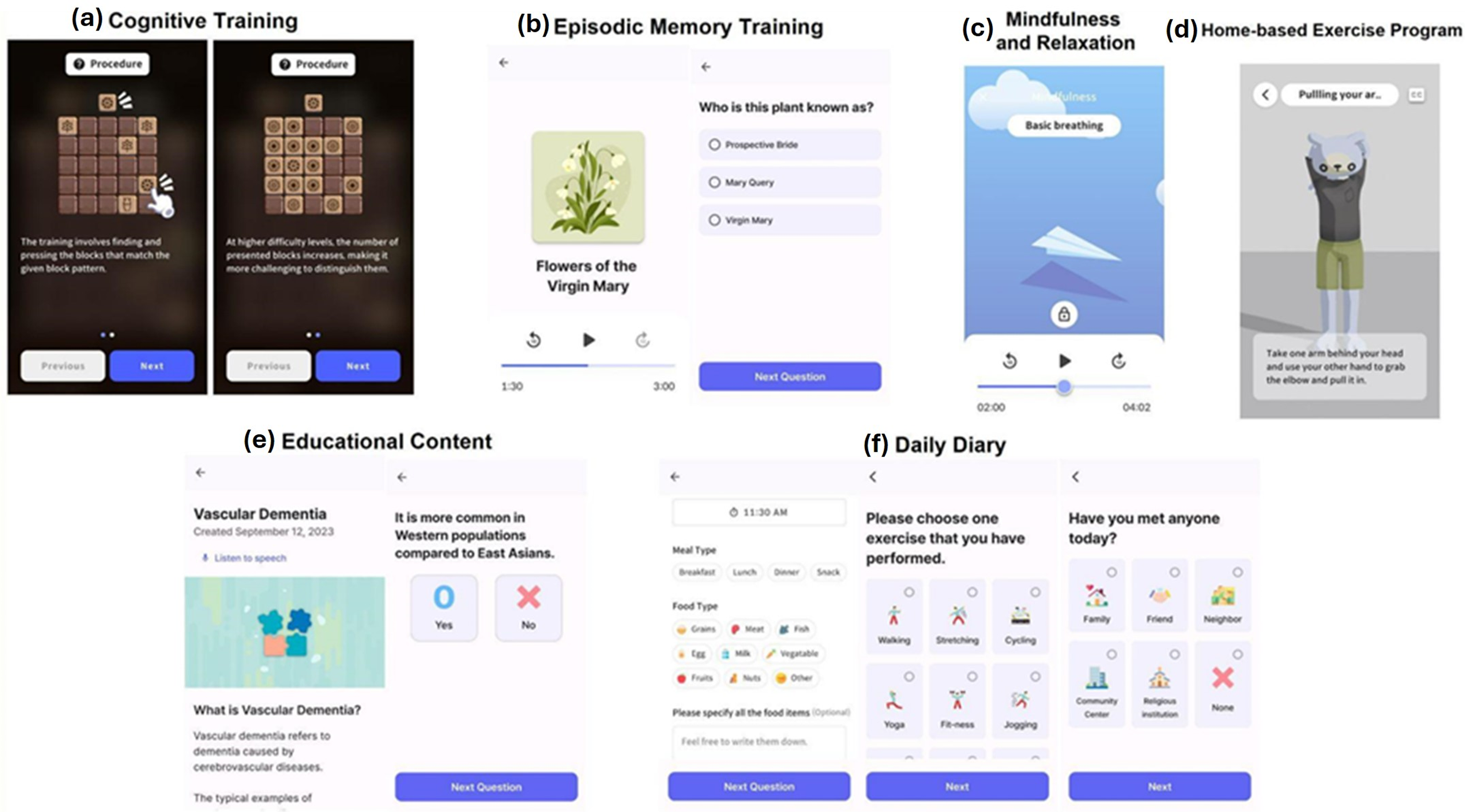
Representative images of Silvia-Rx. This multidomain lifestyle intervention was designed as a 60-session program, consisting of both core and supplementary modules. The core cognitive program comprised two modules: (a) cognitive training and (b) episodic memory training. The supplementary program included four modules: (c) mindfulness and relaxation, (d) a home-based exercise program, (e) educational content, and (f) a daily diary.
The supplementary program comprised 4 different modules as follows (Figure 1):
The video-assisted physical exercise program: This module consisted of two main pieces of training, warm-up and cool-down, and a physical module. The warm-up & cool-down regimens comprised videos instructing users on how to perform the activities. The physical module regimens consisted of videos that guided users through exercises targeting specific body parts such as the arms, legs, and shoulders. Mindfulness and relaxation: These activities included focused breathing, sensory awareness, tension relief, and guided imagery. Narration for these activities was provided in a calm and soothing voice. Users engaged with these activities by listening to them on a daily rotation and practicing the meditation techniques. Educational content: This module offered a curriculum of 60 topics encompassing a range of health-related topics, including the definition of dementia, its various causes, risk factors, and explanations about brain synapses and cognitive functions. Users were presented with health education articles available in audio and text formats for comprehensive understanding. After each article, users were required to complete a quiz related to the content, serving as a measure to assess comprehension of the material. Daily diary: A daily diary is a self-reported log where users can document various aspects of their daily lives. The parameters for documentation include the types of food consumed, the duration and kind of physical exercise undertaken, the number of glasses of water consumed daily, any changes in body weight, adherence to medication schedules, and the types of social contexts.
All participants were provided an access code to use the Silvia-Rx mobile app. During the introductory training session, trained instructors taught each participant how to use the app. All information transmitted between the tablet app and the server was encrypted.
Outcome measures
All outcome measures were administered at baseline (Visit 1) and after completing the 60-session core cognitive program (Visit 2). At Visit 1, demographic data, including age, gender, education level, chronic disease status (e.g., diabetes, hyperlipidemia), and living arrangements, were collected.
The primary feasibility outcomes were adherence to the protocol and the retention rate. Overall adherence to the protocol was calculated by averaging the completed session counts for the core cognitive module. Engagement in the supplementary program was not included in this feasibility outcome measure. The retention rate was calculated as the percentage of participants attending Visit 2 after completing all 60 sessions. Additionally, the primary efficacy outcome was the change from baseline (Visit 1) to post-intervention (Visit 2) in the Alzheimer's Disease Assessment Scale-Cognitive (ADAS-cog). The ADAS-cog assesses changes in global cognition by including 11 domain-specific items: four sub-test items (word recall, orientation, remembering test instructions, and word recognition) assessing the memory domain, five sub-test items (commands, naming, comprehension of spoken language, word-finding difficulty, and spoken language ability) assessing the language domain, and two sub-test items (constructional praxis, and ideational praxis) assessing the praxis domain. 26 A higher score represents poorer cognitive performance, and a positive change from baseline indicates worsening cognitive impairment. Secondary outcomes, including the Korean version of the prospective and retrospective memory questionnaire (K-PRMQ), 27 the Center for Epidemiological Studies-Depression (CES-D) questionnaire, 28 the State-Trait Anxiety Intervention-X-1 (STAI-X-1), 29 the Perceived Stress Scale (PSS), 30 and the Pittsburgh Sleep Quality Index (PSQI), 31 and the Motivation to Change Lifestyle and Health Behaviours for Dementia Risk Reduction (MCLHBDRR) scale 32 were measured at Visits 1 and 2.
Statistical analysis
Descriptive analyses were conducted to summarize the characteristics of the participants. A Wilcoxon signed-rank test comparing pre- and post-measures (primary efficacy and secondary outcome measures) was performed to examine significant improvement after the intervention. For all analyses,
Results
Participants
The demographic characteristics of the study participants are shown in Table 1. A total of 20 MCI patients were enrolled and consented to join the Silvia-Rx intervention. Only one patient withdrew from the study because of time constraints. The mean age of the participants was 71.11 years (SD 5.68), and 13 (68.42%) were women. Table 2 delineates the results from comprehensive cognitive assessments and provides etiological classifications for the MCI diagnoses among the 19 subjects: 7 were attributed to Alzheimer's disease, 6 to Lewy bodies, 3 to vascular disease, and 3 to an unspecified neurocognitive disorder.
Baseline characteristics and demographics of subjects with mild cognitive impairment (MCI).

MMSE-KC: Mini Mental State Examination in the Korean version of the Consortium to Establish a Registry for Alzheimer's Disease (CERAD-K) neuropsychological assessment battery. CDR-SOB: Clinical Dementia Rating-Sum of Box. Note: CDR-SOB, which is commonly used in clinical trials and practice for rating severity, in predementia or very mild dementia stages highly predicts progression to dementia.
The cognitive characteristics and related diagnosis in each participant.

+: cognitive impairment of the domain was ascertained 1.5 standard deviation or more below the norm of Koreans adjusted by the age, gender, and education. M: male, F: female, Edu: education (years), CDR: Clinical Dementia Rating Scale; SOB: CDR-Sum of boxes; MMSE-KC: Mini Mental State Examination in the Korean version of the Consortium to Establish a Registry for Alzheimer's Disease (CERAD-K) neuropsychological assessment battery; VF: Verbal Fluency; BNT: Modified Korean version of the Boston Naming Test; WLM: Word List Memory; CP: Constructional Praxis; WLR: Word List Recall; WLRc: Word List Recognition; CR: Constructional Recall; TM: Trail Making test; ST: Stroop test; MCI: mild cognitive impairment.
Feasibility outcomes
Excellent retention was observed, with 19 (95%) patients assessed at the post-evaluation point (Visit 2). Adherence to the protocol was substantial, with 100% of participants completing all 60-session core cognitive modules. None of the participants exhibited adverse reactions during the intervention.
Efficacy outcomes
The results of the analyses of the efficacy variables are shown in Table 3. Compared to baseline, the global cognition measured by the ADAS-cog was significantly improved after completing the 60-session core cognitive modules (

The ADAS-cog total scores. Compared to baseline (Pre), the global cognition measured by ADAS-cog was significantly improved after completing the 60-session core cognitive program (Post) (
Pre and post of the total ADAS-cog and sub-test scores.

ADAS-cog: the Alzheimer's Disease Assessment Scale-Cognitive.
() : range.
*Significance at
**Significance at
Pre and post of secondary outcome measures.

K-PRMQ: Korean version of the prospective and retrospective memory questionnaire; CES-D: Center for Epidemiological Studies-Depression questionnaire; STAI-X-1: State-Trait Anxiety Intervention-X-1; PSS: Perceived Stress Scale; PSQI: Pittsburgh Sleep Quality Index. MCLHB-DRR: Motivation to Change Lifestyle and Health Behaviours for Dementia Risk Reduction
Discussion
Principal findings
As part of the development of a digital multidomain lifestyle intervention, we explored the feasibility and preliminary efficacy of a 60-session core cognitive program in Silvia-Rx for older people with MCI. Our results indicated that older adults with MCI very well tolerated the 60-session core cognitive regimen. Furthermore, their overall cognitive function significantly improved after the intervention compared to baseline. Notably, the cognitive improvements were specifically observed in the praxis domain among subjects with MCI. No significant secondary health-related outcome measures were observed. Furthermore, given that the participants in the study had MCI etiology from several causes, including Alzheimer's disease, Lewy body’s disease, and vascular disease, these initial findings suggest that the core cognitive program of Silvia-Rx could have potential benefits for MCI patients with a diverse range of underlying conditions.
Our feasibility findings indicate excellent retention rates of 95% and completion rates of 100%, demonstrating that participants with MCI very well tolerated the 60-session core cognitive module. Moreover, the absence of any adverse effects related to the intervention further highlights the positive aspect of this intervention. It has been noted in prior studies that while nearly all computer-enhanced technologies are compatible with mouse input, utilizing a mouse demands a more significant cognitive load compared to touchscreens or other interfaces. 33 Given that mouse control may present challenges for older adults and people with dementia, tablet-based cognitive training could foster increased participation among individuals with MCI. Thus, our findings affirm the viability of implementing a self-administered cognitive regimen for older people with MCI.
In our study, there was a statistically significant improvement in cognitive functions among participants, as indicated by the total scores on the ADAS-cog. Sixteen out of 19 MCI patients exhibited sustained or improved global cognitive function after the 60-session core cognitive training, indicating a successful training effect on most participants. The ADAS-cog serves as a widely recognized tool for assessing cognitive impairment severity, ranging from mild to severe Alzheimer's disease. 34 This efficacy outcome highlights the positive effects of the core cognitive program in Silvia-Rx on the cognitive abilities of older people with MCI. The intervention's core cognitive modules, which addressed five distinct cognitive areas and episodic memory, likely acted as a potent stimulus for enhancing cognitive resilience against aging or pathological effects. Engaging in varied cognitive exercises repeatedly could strengthen the cognitive reserve in older adults with MCI, leading to improved cognitive function and potentially decelerating the rate of cognitive deterioration. Such findings emphasize the value of core cognitive training in promoting cognitive health among people with MCI.
Notably, among the cognitive sub-tests of the ADAS-cog measurement, significant enhancement was observed in the praxis domain following the completion of the 60-session core module. The ADAS-cog includes sub-tests assessing cognitive functions in the areas of memory, language, and praxis. 34 The inability to perform such praxis movements, known as apraxia, is important in assessing cognitive deficits and can aid in distinguishing between normal elderly individuals, those with MCI, and Alzheimer's disease patients.35,36 This aligns with previous findings and suggests that cognitive training may specifically bolster psychomotor activities that require motor skills and coordination, which are essential to praxis.37,38 However, the lack of significant improvement in memory and language could be attributed to the diverse etiologies present in our sample. Since impaired cognitive function varies across different domains according to etiology, it is plausible that the diverse etiologies of the participants influenced the results. All subjects were initially assessed for baseline cognitive function using the CERAD-K. However, only two out of 19 MCI patients exhibited pure memory decline with reduced recognition during the baseline CERAD-K assessment. Therefore, since most participants did not show significant impairments in memory and language cognitive functions at baseline, it is possible that no significant differences were observed post-intervention due to a ceiling effect. To confirm this hypothesis, further research with a larger sample with diverse etiologies is necessary. On the other hand, it is necessary to explore whether it would be more beneficial to enhance the memory improvement components in the cognitive training regimen or to focus on ‘praxis’ to potentially shorten the overall training duration.
The three subjects in this study did not show cognitive improvement, contrary to the expected outcome. Reasons for these differing responses include individual variability, disease progression stage, adherence to the intervention, baseline cognitive function, and other health conditions. Unique genetic, biological, and environmental factors, as well as differences in MCI stage and progression, adherence to protocols, initial cognitive levels, and comorbidities, may have influenced their responses. These factors highlight the complexity of treating cognitive impairment and the need for personalized approaches, indicating further research is necessary. 5
The health-related secondary outcomes such as stress, sleep quality, and mood, exhibited no substantial changes after the intervention. Since the intervention primarily focused on cognitive training, improvements in cognitive function were likely more immediately noticeable, whereas other secondary measures may not have shown sufficient effects within the same timeframe. On the other hand, the difference from previous studies that reported improvements in cognitive functions being associated with enhancements in mood, quality of life, and activities of daily living after the interventions,39,40 may suggest that the core cognitive training in Silvia-Rx serves as an effective cognitive enhancer independently of improvements in emotional well-being. Alternatively, this could be because participants whose cognitive function was impaired due to sleep disturbance or depression were excluded from the study based on expert clinical evaluation.
Despite the necessity of diagnosing and studying MCI based on precise clinical evaluation to ensure the reliability of the efficacy and usability results of digital therapeutics and to apply them to various groups in the real world, 41 existing studies often diagnose MCI based on simple survey assessments or exclude non-amnestic MCI, focusing instead on amnestic MCI, which is more likely to be associated with Alzheimer's disease. The current study can contribute to the existing literature by demonstrating the importance of accurate clinical evaluations in MCI research. One of the study's strengths lies in its inclusion of a diverse group of participants who were precisely diagnosed concerning the etiology of MCI. Additionally, individuals with conditions such as depression or sleep disorders, which could significantly impact cognitive function, were excluded based on clinical evaluations.
Limitations
A few limitations warrant mention. Firstly, the primary limitation of this pilot study is the small sample size. Despite providing preliminary data regarding the feasibility and efficacy of this core cognitive program, the sample size was still insufficient for drawing a broad conclusion. Hence, further studies with larger sample sizes are needed. Secondly, the study was conducted using a single-arm, pre-and-post design. Consequently, future studies should employ a more robust research design, such as randomized controlled trials, including comparisons of cognitive outcome changes between intervention and control groups, to validate and expand upon our findings. Third, we mainly focused on the core cognitive modules to investigate the feasibility and efficacy of our Silvia-Rx intervention program. In line with previous studies of multidomain lifestyle interventions, our supplementary modules, including physical exercise, mindfulness, and educational content, could also enhance brain function and overall health in older adults with MCI.42–44 Future research should extend its scope to include these supplementary modules to assess their collective impact on brain functioning and general health, and the relationship with the number of sessions, as well as to determine the optimal number of sessions. Lastly, while this study focused on the 60–80 age group, further research is needed to determine whether the program is effective and applicable to a broader adult population, including younger adults. For this training to be widely applicable across various age groups, it would be beneficial to establish age-specific criteria or thresholds to determine suitability and necessary modifications. This approach would facilitate a more targeted and efficient application of the intervention.
Conclusions
As part of the development process for a digital multidomain lifestyle intervention, this pilot study aimed to validate the feasibility and effectiveness of core cognitive training in older individuals diagnosed with MCI. Despite certain limitations, such as the small sample size, our preliminary findings indicated that the 60-session intervention was highly feasible for this population. Furthermore, the intervention was observed to enhance cognitive capabilities over time, indicating its potential to contribute to the augmentation of cognitive reserve. Therefore, core cognitive training may be a promising strategy for augmenting cognitive performance in older adults with MCI. Future research should aim to replicate these findings with a larger sample from various age groups and incorporate supplementary modules, such as physical exercise and mindfulness practices, to comprehensively determine the intervention's effects on cognitive abilities and overall health outcomes.
Supplemental Material
sj-zip-1-dhj-10.1177_20552076241284810 - Supplemental material for Development of a digital multidomain lifestyle intervention for mild cognitive impairment: A pilot study on the feasibility and efficacy of cognitive training
Supplemental material, sj-zip-1-dhj-10.1177_20552076241284810 for Development of a digital multidomain lifestyle intervention for mild cognitive impairment: A pilot study on the feasibility and efficacy of cognitive training by Hyun Jung Han, Myung Jin Ko, Ahyoung Park, Jaemin Cheun, Yeonjoo Nam and Tae Hui Kim in DIGITAL HEALTH
Footnotes
Acknowledgments
We would like to express our sincere gratitude to our research participants for their invaluable dedication to the clinical study. And we wish to thank Ms. Myeonghee Hong and Jiyeon Yu at Yonsei Geriatric Mental Health Research Center at the Wonju Severance Christian Hospital, who assisted in recruitment support.
Contributorship
Study conception and design: MJK, AP, and THK; Silvia-Rx development, design, server maintenance, and IT support: JC; data collection and intervention administration: HJH and THK; analysis and interpretation of results: HJH, MJK, YN, and THK; draft manuscript preparation: HJH and YN. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
H. J. Han, A. Park, and J. Cheun are employees of Silvia Health Inc. and M. J. Ko is the CEO of Silvia Health Inc.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Declaration of Helsinki. All participants provided written informed consent as approved by the Institutional Review Board of the Wonju Severance Christian Hospital (approval number: CR323074).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Korea Planning & Evaluation Institute of Industrial Technology (KeIT) grant funded by the Korea government (MOTIE, Ministry of Trade, Industry and Energy) [Project Name: Development and validation of a digital phenotype-based platform for the treatment and prevention of MCI using a multidisciplinary approach to decrease dementia risk / Project Number: 20024263].
Guarantor
THK.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
