Abstract
Background
Reduced cardiorespiratory fitness levels are associated with increased short-term complications after surgery, and potentially exert long-lasting effects on the postoperative lives, work and educational pursuits of patients. Currently, research suggests that lifestyle interventions, such as preoperative physical exercise undertaken by patients themselves, may improve patients’ cardiopulmonary fitness and reduce post-operative complications. This study aims to investigate the effectiveness and feasibility of a remote medical supervision model for prehabilitation exercise in patients undergoing thoracoscopic lung tumour resection surgery.
Methods/Design
All enrolled patients will participate in a 4-week pre-operative exercise intervention to improve their cardiorespiratory fitness. During this period, patients will wear wearable devices and exercise at home based on exercise prescriptions. The exercise prescription comprises aerobic exercise (three times a week or more), muscle strengthening exercise (twice a week or more), and respiratory muscle exercise (once a day). The primary aim is to investigate whether baseline VO2max could be improved following a 4-week preoperative exercise program. Secondary objectives include changes in forced expiratory volume in 1 s and forced vital capacity, degree of acceptance of the technology, quality of life, handgrip strength, postoperative complication rate and length of hospital stay.
Discussion
This study aims to evaluate the influence of preoperative prehabilitation exercises in a telemedicine active supervision mode in patients undergoing thoracoscopic lung tumour resection. As such, results of this trial might have some impact on future implementations of group- and home-based prehabilitation exercises in lung cancers.
Trial registration
This study was approved by the Medical Ethics Committee of Tongji Hospital Affiliated to Tongji Medical College of Huazhong University of Science and Technology (approval number: TJ-IRB20220564) with registration at ClinicalTrials.gov (identifier: NCT05608759).
Keywords
Introduction
Background
Lung cancer is the most common and the leading cause of cancer death worldwide. 1 Although surgery offers a potential cure for certain lung cancers and is the gold standard treatment for early-stage lung cancer, 2 it is physiologically stressful and associated with significant postoperative morbidity and mortality.3–6 Furthermore, patients with reduced cardiorespiratory fitness (CRF) levels are associated with severe cardiovascular, respiratory comorbidities, increased short-term complications after surgery and potentially exert long-lasting effects on the postoperative lives, work and educational pursuits hence may be denied access to surgical resection therapy due to the high postoperative risks.7–10 Research has shown that exercise training has positive effects on CRF in patients with non-small cell lung cancer (NSCLC).11–13 Therefore, more and more doctors have introduced exercise training into the preoperative management of people with scheduled lung resection for NSCLC. Exercise training can improve CRF to overcome the physiological stress caused by surgery, decrease postoperative morbidity and mortality and improve long-term prognosis.11–13 Besides, the systemic inflammatory response that occurs following lung resection will also be reduced by the anti-inflammatory effects of exercise. 14
There are international recommendations that have been published to guide specific perioperative cardiopulmonary exercise testing (CPET) prior to the prescription of prehabilitation. The paradigm of pre-operative exercise as neoadjuvant therapy to reduce morbidity is increasingly promoted within general surgery and surgical oncology.4,15–17 Prehabilitation exercises generally involve breathing exercises and aerobic exercises at least five times a week. 18 Patients with lung cancer who participate in pre-operative exercise have better aerobic capacity, pulmonary function, and shorter hospital length of stay after surgery.18–20 Similarly, some studies in China have shown that the level of postoperative recovery can be improved by active preoperative exercise. However, compared to the CPET method used abroad to assess patients’ exercise status, the six-minute walk test or other subjective assessment methods are often used in China, with less objectivity.
While highly supervised, on-site exercise programs could be considered the gold standard for research and clinical care,17,19 they are labor-intensive for staff that administer them and requires a significant number of medical resources. Moreover, these programs are often inaccessible to patients who live in rural communities.
To address the stated challenges of the medical human resources and economic burden and to achieve effective active supervision under the family model of prehabilitation exercise in China, we propose the concept of ‘home exercise, wearable device, and remote active supervision’, so the exercise prescription can be adjusted in real-time, and the patient's compliance can be improved through active supervision. This study is a proof-of-concept, single-arm, pre-post design study. The aim is to evaluate the effectiveness of preoperative prehabilitation exercises in a telemedicine active supervision mode in patients undergoing thoracoscopic lung tumour resection, by considering the patient's CRF, postoperative complications and quality of life scores.
Objectives
The primary objective of this study is to investigate whether baseline VO2max could be improved following a 4-week preoperative exercise program that will take place at home. VO2max will be examined by CPET and a comparison of the results will be made between the baseline and the data after 4 weeks of prehabilitation exercise.
Secondary objectives will include an assessment of the adherence to exercise (as determined by the number of completed pre-operative exercise sessions divided by the number of prescribed sessions, according to the exercise log). Alternative measures of the functional reserve will be evaluated, in particular change in forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC) during CPET.
Other secondary objectives include degree of acceptance of the technology, the impact of preoperative exercise on validated quality of life (QoL) scores (EORTC QLQ-C30, EORTC QLQ-LC13), and the effect of prehabilitation on nutritional status as assessed using handgrip strength (HGS). Postoperative complications will be assessed using the Clavien-Dindo classification. The length of hospital stay will be analyzed up to 30 postoperative days.
Methods/design
This study, which aims to improve the cardiorespiratory fitness of the patients undergoing thoracoscopic lung tumour resection, was approved by the Medical Ethics Committee of Tongji Hospital Affiliated to Tongji Medical College of Huazhong University of Science and Technology (approval number: TJ-IRB20220564) with registration at ClinicalTrials.gov (identifier: NCT05608759) before the first participant enrollment. We followed the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guideline for the reporting of the study protocol. 21
Study design
This is a proof-of-concept, single-arm, pre-post design study, so blinding is not performed in this study. Participants will be recruited in Wuhan, China. Existing research suggests that delaying the timing of surgery may lead to tumour progression and prolonged prehabilitation exercises,22–24 increasing patient anxiety and poorer adherence, affecting the outcome of the patient's prehabilitation. Research from leading thoracic surgery journals has concluded that for patients with NSCLC, a treatment strategy with a target time of 28 days from the decision to operate until surgery is performed does not increase the risk of tumour ascending. 25 Therefore, all enrolled patients will participate in a 4-week pre-operative exercise intervention to improve their CRF. During this period, patients will wear wearable devices and exercise at home on the basis of exercise prescriptions. This preoperative protocol has been standardized following a certain framework. These usual prehabilitation sessions include warm-up exercises, aerobic exercises, muscle strengthening exercises (active resistance exercises), muscle stretching exercises, inspiratory muscle exercises (using breath training device, abdominal breathing, thoracic control training, pursed lip breathing, cough training). The study is planned to start in November 2022 and end in March 2024.
Participants
This study was conducted in accordance with SPIRIT 26 (Figure 1). It has been shown that there are no certain guidelines for the size of a prospective study in healthcare research, 27 hence we plan to recruit 30 patients who meet the inclusion criteria to participate in this study from November 2022 to December 2023. During the outpatient visit process, patients will be preliminarily screened by physicians according to the inclusion criteria. Subsequently, our research team will conduct further screening and enrolment of patients, such as obtaining informed consent and administering the baseline assessment.

Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) flow diagram.
Inclusion and exclusion criteria
Individuals interested in the study who meet the following basic eligibility criteria will be scheduled for the informed consent process and baseline survey. Individuals will be eligible if they: (a) have the capacity to give informed consent, (b) can tolerate surgery (i.e. segmentectomy, lobectomy) as indicated by standard clinical pre-op evaluation, including pulmonary function tests and CPET, (c) can use a smartphone application, (d) are suspected or diagnosed with stage I or II NSCLC, with or without pre-operative histologic confirmation, (e) can perform an exercise program for at least 4 weeks from the first visit to the preoperative clinic to elective surgery. Participants will be excluded if they: (a) have known contraindications for CPET, (b) have a major medical or psychiatric disorder that is expected to affect exercise, (c) are pregnant or lactating women, (d) have received neoadjuvant therapy. 28
Guidelines to the cessation of participation in the study include if the patients: (a) are converted to open thoracotomy during surgery, (b) themselves or their legal representative requests withdrawal, (c) are judged by researchers on the emergencies that will significantly affect the assessment of the patient's clinical condition during the research process.
Features of the telemedicine active supervision mode
The medical-grade wrist wearable supervisory device bound with APP used in this project can collect patients’ heart rate and oxygen saturation indexes non-invasively, accurately and does not interfere with patients’ home rehabilitation and rest. This wearable device automatically uploads data, reports and risk information to the cloud, and jointly monitors and analyzes key health information such as daily exercise, sleep quality, heart rate risk and hypoxia risk. It then establishes a full-cycle health data file for patients and records objective indexes for patients’ prescription implementation. Through the dynamic monitoring and management of the cloud platform, doctors can access patients’ rehabilitation data and evaluate whether the patient has completed the daily exercise program. In addition, patients can receive exercise prescriptions from doctors, fill in and upload exercise logs, record and view rehabilitation data and receive abnormal health warning information (Figure 2).

The feature of the study mode.
Device characteristics
The wearable device JIULE T3(Jiuletech, China)provided by the researchers to the enrolled patients is of medical grade and complies with China's Class II medical device standards, which can non-invasively and accurately collect participants’ heart rate, blood oxygen saturation and other indicators without affecting participants’ home rehabilitation and rest. The device will upload the collected data to the AI database in the cloud, where the AI will analyze and evaluate the data, and ultimately generate reports and risk information. The generated reports include daily exercise reports, health reports (including, heart rate, blood oxygen, sleep monitoring, heart rate anomaly statistics, blood oxygen anomaly statistics, etc.). Based on these reports, the AI will create a full-cycle health data file for the participant, recording all data during the exercise cycle. Doctors can access the participant's recovery data through the cloud-based platform to assess the participant's exercise condition. When the cloud-based platform detects abnormal data during the exercise process, it will contact the researchers and the participants to prevent adverse events (AEs) from occurring.
The AI app will capture and visualize the patient's physiological data in real time, and the app can also enable ‘exercise mode’ to be used in conjunction with a wearable device to show the patient specific information about the exercise prescription. After the patient has exercised, the software will analyze the patient's data and produce reports and conclusions to guide the patient's exercise (Figures 2 and 3).
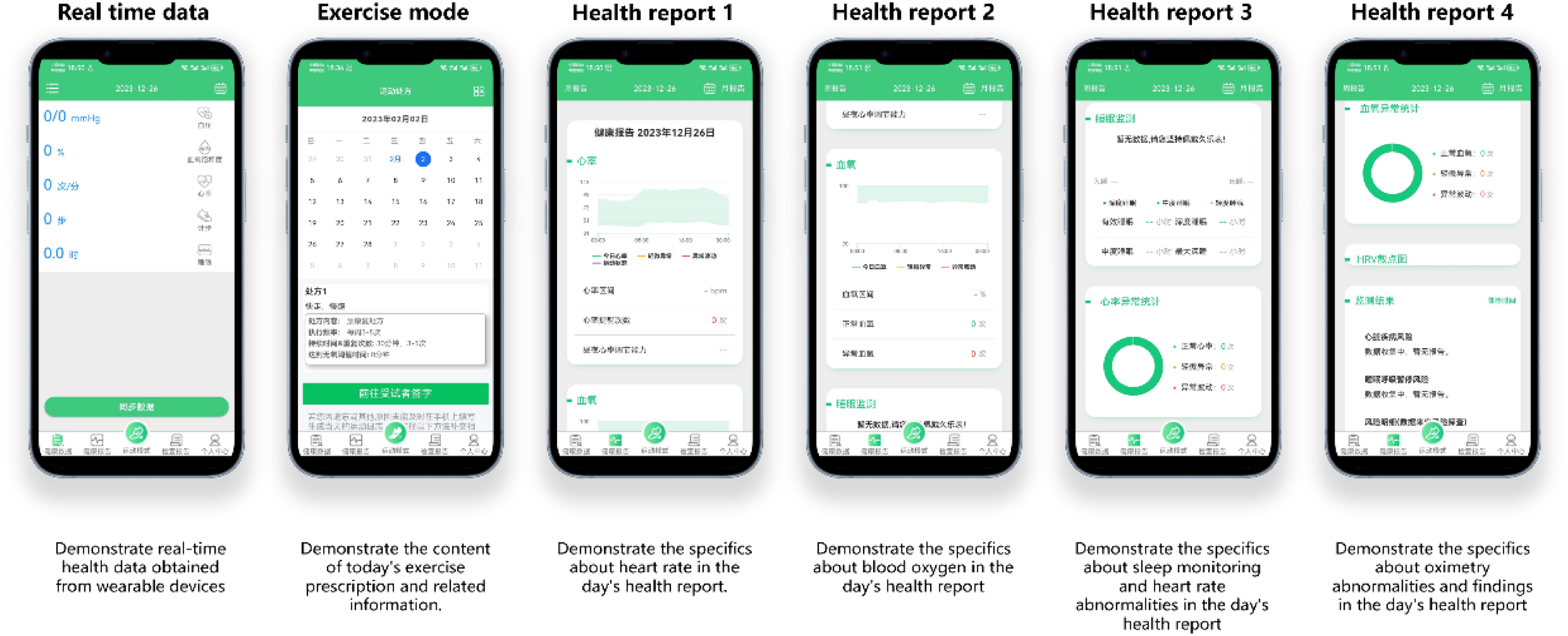
Based on wearable recorded patient physiological data, comprehensive analysis and assessment is performed and feedback is provided to the patient to provide personalized health advice.
Intervention
The cardiologist will perform CPET using BICYCLE ERGOMETERS Ergoselect 100 (ergoline GmbH, Germany) and MasterScreenTM CPX (Jaeger, Germany) in accordance with national guidelines, and based on the results, the participant will be given an exercise prescription.13,29–31 After the physician made the necessary registration or setting of the wearable supervisory device according to the exercise prescription, such as the initial threshold and warning threshold, the patient could take it home and execute the exercise prescription at home. During the home exercise period, the study investigators can access the patient's exercise status based on the real-time information collected (Tables 1 and 2, more detailed information on exercise prescription can be found in the Additional file 1), actively monitored the patient's exercise effect, and monitor whether the patient completed the daily program.
Exercise prescription.

Patient exercise log card.

The exercise prescription needs to be implemented for four weeks and consists of five parts, including warm-up exercise (Additional file 2), aerobic exercise, muscle strengthening exercise (Additional file 4), muscle stretching exercise (Additional file 3), and respiratory muscle exercises (Additional file 5). Warm-up exercises and muscle stretching exercises should be done before each exercise, aerobic exercise at least thrice per week, the muscle strengthening exercise at least twice per week, and respiratory muscle exercises once a day.
Participation in the study is entirely voluntary, and subjects have the unreserved right to withdraw from the study at any point without fear of discrimination, retaliation or prejudice to their medical treatment and rights. The investigator reserves the right to terminate a subject's participation in the study under circumstances such as the need for additional treatment, non-compliance with the study plan, occurrence of a study-related injury, or for any other justifiable reason.
Data collection during exercise
In this study, a log card will be used, which will be filled out by the patient at home according to the prehabilitation exercise done, if the patient withdraws in the middle of the study, the researcher will record the prominent reasons, and later analyze the relevant factors affecting the patient’s exercise compliance according to this data.
To collect basic vital signs data such as heart rate during rest and exercise, patients will be required to wear the wearable device at all times except during charging of the wearable device and washing. Patients will also be reminded to log in daily to fill in the exercise log card via the prehabilitation remote supervision APP, and the contents recorded are shown in Table 2.
Safety assessment
Although exercise training is a relatively safe treatment with a low risk of AE, 32 any AEs during the study period will be assessed, treated and documented in the case report form. Patients utilizing wearable supervisory devices may experience skin allergies, rashes and other related issues. Additionally, individuals exercising at home may encounter risks such as sports injuries, cardiovascular and cerebrovascular events. To minimize these risks, we have established a stringent risk assessment protocol for this project. Patients who meet the criteria for tolerance will undergo screening. Severe AEs occurred would be reported to the independent ethics committee. The ethics committee will conduct an annual follow-up review of this study.
Data monitoring
All records pertaining to patient identities will be maintained with the utmost confidentiality and, in accordance with applicable laws and regulations, will not be disclosed to the public. Access to patient identifying information will be limited solely to individuals directly involved in the trial, such as investigators and research nurses.
To further safeguard patient confidentiality, the case report form will not contain the subject's name. Instead, only the subject's assigned number and initials will be recorded on the form. In the event that the subject's name appears on any other document, such as a pathology report or imaging, it must be redacted on the document copy.
For studies utilizing computerized storage, strict compliance with local data protection laws is imperative. The confidentiality of subjects’ identities will also be preserved when publishing the study results. An investigator-maintained list will be used solely for the purpose of identifying subjects for record-keeping.
Study auditing
In accordance with local regulatory requirements, the designated inspector will establish regular contact with and conduct periodic visits to the investigator. These visits will entail the inspection of various trial records, including case report forms and other pertinent data, as deemed necessary.
Throughout the course of the study, the Supervisor assumes the responsibility of routinely reviewing case report forms. Their tasks encompass ensuring strict adherence to the study protocol and verifying the completeness, consistency and accuracy of the data entered. To facilitate this process, the Supervisor will be granted access to video recordings of procedures and other patient records to cross-verify the information documented in the case report forms.
The investigator or their designated representative commits to collaborating closely with the monitor to promptly address any issues or concerns identified during these monitoring visits.
Data management
All the data collected will be documented in case report form first by clinical research coordinators and checked by two trained clinical research associates before double data entry into REDCap by trained research assistant. This rule is set to ensure the data is accurate, authentic and traceable. The database will be protected by a password, and only the principal investigator will have access to the final dataset. A data monitoring committee that is independent of researcher and sponsor and has no conflict of interest with this study will be estimated. It will be responsible for the management of data quality.
Outcome measures
The primary outcome (change in VO2max) will be measured by an incremental symptom limited CPET test performed by an experienced consultant cardiologist, external to the trial study group. All patients will undergo CPET testing at baseline (before the start of prehabilitation) and after 4 weeks of prehabilitation exercise. Other CPET outcomes (FEV1 and FVC) will also be measured.
Degree of adherence and acceptance of technology will be assessed by monitoring patient attendance at exercise, patient-reported log, adherence to the supervised exercise program, as well as patient drop-out. Patients will be deemed compliant with the intervention if they complete >75% of scheduled prehabilitation sessions. Weekly steps recorded via a medical-grade wrist wearable device.
QoL will be assessed at specific time points: before the prehabilitation exercise (T1), after 4 weeks of prehabilitation exercise (T2), 7 days after surgery (T3) and 30 days after surgery (T4). The primary outcome will be the exercise capacity testing on a cycle ergometer (VO2 max). Validated questionnaires will include EORTC QLQ-C30, EORTC QLQ-LC13.
HGS will be measured at the same time points that questionnaires are taken. Measurements will be recorded by two individuals, one of whom will be external to the trial group. Surgery will be performed in a standard manner by three experienced lung cancer consultants. All patients will be transferred to postoperative intensive care and will follow a dedicated lung cancer Enhanced Recovery After Surgery pathway. Length of intensive care and hospital stay will be recorded, as well as postoperative complications which will be measured using the Clavien-Dindo classification. Table 3 demonstrates the study schedule (Table 3).
The study schedule.

Statistical methods
The baseline characteristics of the enrolled patients were statistically analyzed using descriptive analysis. Categorical variables were expressed as percentages (%); continuous variables were described as mean ± standard deviation or median and interquartile range. Histograms were used to describe the distribution of continuous variables. In this study, patients who did not complete 75% of the weekly exercise program were defined as low-adherence patients. Based on adherence, patients were classified as high-adherence patients and low-adherence patients, and a comparative analysis of these two groups was performed based on the study results. A linear mixed-effects model was used to analyze the intervention effect of prehabilitation exercise considering the results of the study in one patient with repeated measurements at different time points. Intervention modality, the time point of outcome assessment as categorical variables, and the interaction between intervention modality and time point were used as fixed effects models. SPSS was used for statistical outcome analysis, and the chi-square test or Mann–Whitney U-test was used depending on the type of data of the study results. The significance level (alpha) for all hypothesis tests was 0.05 bilaterally if not otherwise specified.
Discussion
The increasing incidence of lung cancer and its gradual rejuvenation pose a serious challenge to mankind. 1 Although the existing treatment programs are very mature, research on how to further improve patients’ postoperative rehabilitation is rare. 2 Improving patients’ cardiorespiratory fitness and overall physical fitness can help accelerate their postoperative recovery. Developing more efficient and acceptable protocols for supervised patient exercise is important for patients about to undergo lung surgery.
Improving patients’ postoperative cardiorespiratory fitness and long-term outcomes holds significant relevance within the context of China's current healthcare landscape. China, like many nations, faces a complex set of challenges in its healthcare system, including uneven distribution of medical resources and rising healthcare costs.33–35
The uneven distribution of medical resources across urban and rural areas in China is well-documented. Larger cities tend to possess more medical facilities and experts, leaving rural regions underserved.36,37 This disparity impacts postoperative care accessibility, as patients in rural areas may struggle to access timely medical interventions for complications. Furthermore, the escalating healthcare costs in China are a growing concern. As medical expenses continue to rise, patients and families face financial burdens.33,34 Research suggests that patients who engage in preoperative exercise programs, even in rural areas, are more likely to benefit from enhanced fitness and better surgical outcomes, reducing the need for emergency interventions and hospital readmissions.38,39
Engaging in prehabilitation requires patients to actively participate in their own healthcare journey. They become proactive in making lifestyle adjustments, such as adopting healthier eating habits, adhering to regular exercise routines, and managing stress. This heightened involvement in their well-being can lead to an increased awareness of the significance of preventive health measures and long-term health maintenance, and may lead to the establishment of long-lasting habits that contribute to overall well-being.
By improving patients’ overall health, prehabilitation can alleviate some of the financial stress associated with postoperative care, enhancing healthcare affordability for individuals and society as a whole which contributes to the sustainable progress of universal healthcare coverage in China. Additionally, it can also improve people's lifestyles, encouraging the adoption of healthier dietary habits, regular exercise routines and stress management practices, help improve the overall public health status in China.40–43
This study has several advantages. Firstly, the CPET is a widely recognized and well-established evaluation tool that has been deemed the ‘gold standard’ in assessing the cardiopulmonary capacity of patients prior to undergoing surgery. To the best of our knowledge, there is no prehabilitation project based on CPET and telemedicine active supervision model for the surgical treatment of patients with lung tumours in China. Secondly, this study is supervised by wearable devices and a rigorous, tailored exercise program will be established by a clinical exercise scientist for each patient based on their baseline functional capacity as assessed by CPET and the outcome will be objectively measured by an experienced consultant cardiologist. The exercise component of the prehabilitation program is supervised by a clinical exercise scientist who will construct a rigorous, tailored exercise program for each patient based on their baseline functional capacity as assessed by CPET.
This study also has certain weaknesses. As this study is performed in the central Chinese population hence limitations associated with geographical regions exist. Future randomized controlled trial will be conducted to validate the effect of prehabilitation in patients with lung tumours based on the results obtained.
Supplemental Material
sj-pdf-1-dhj-10.1177_20552076241258362 - Supplemental material for Your preoperative rehabilitation assistant: A study protocol for the impact of a telemedicine-supported preoperative home rehabilitation program on the prognosis of patients undergoing thoracoscopic surgery
Supplemental material, sj-pdf-1-dhj-10.1177_20552076241258362 for Your preoperative rehabilitation assistant: A study protocol for the impact of a telemedicine-supported preoperative home rehabilitation program on the prognosis of patients undergoing thoracoscopic surgery by Qihang Sun, Willis Wasonga Omindo, Wanjun Liu, Yan Huang, Ruijie Zhang, Yan Qian, Xianping Li, Ruixing Qiu, Shubin Zheng, Wei Ping and Ni Zhang in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076241258362 - Supplemental material for Your preoperative rehabilitation assistant: A study protocol for the impact of a telemedicine-supported preoperative home rehabilitation program on the prognosis of patients undergoing thoracoscopic surgery
Supplemental material, sj-docx-2-dhj-10.1177_20552076241258362 for Your preoperative rehabilitation assistant: A study protocol for the impact of a telemedicine-supported preoperative home rehabilitation program on the prognosis of patients undergoing thoracoscopic surgery by Qihang Sun, Willis Wasonga Omindo, Wanjun Liu, Yan Huang, Ruijie Zhang, Yan Qian, Xianping Li, Ruixing Qiu, Shubin Zheng, Wei Ping and Ni Zhang in DIGITAL HEALTH
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Author Contribution
QS and WWO contributed equally to this manuscript. NZ, BZ and WP initiated the conception and design of the study, while QS, WWO, WP, WL, YH, RZ, YQ, XL, RQ and SZ helped with the implementation of the study. QS, WWO and NZ prepared the first draft of the manuscript. All authors contributed to the study protocol and approved the submitted version.
Consent for publication
All data, findings and intellectual property rights arising from this study are the exclusive property of the sponsor, who may employ them in various capacities. Concurrently, the researcher is permitted to utilize study-derived information for their own scientific purposes, subject to prior consultation with and written consent from the Sponsor before publication. The Sponsor acknowledges the Investigator's right to publish the study's results upon its conclusion. However, the researcher must share a draft or abstract of the article with the Sponsor at least 30 days before submission for approval. The Sponsor commits to promptly review the article and will not unreasonably withhold approval. In the event of any disagreement between the sponsor and the researcher, a constructive dialogue shall be initiated to reach a mutually agreeable resolution concerning the content of the intended publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Tongji Hospital Affiliated to Tongji Medical College of Huazhong University of Science and Technology, China (approval number: TJ-IRB20220564) with registration at ClinicalTrials.gov (identifier: NCT05608759). All patients will be fully informed about the trial and given enough time to decide whether to participate in the study.
The informed consent forms will be obtained from all participants before taking part in the study. This informed consent form describes what is the objective of the research and what personal information will be collected. The study results will be disseminated through conferences and academic journals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received support from the Hubei Chen Xiaoping Science and Technology Development Foundation grant number 2022077.
Guarantor
Huiling Shao
Trial situation
The trial is currently in the recruitment phase. This protocol version is V2.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
