Abstract
Purpose
To investigate the feasibility of implementing a remote patient monitoring system using an electronic patient-reported outcomes (ePROs) platform in a tertiary cancer center in the Republic of Ireland.
Methods
Patients receiving oral chemotherapy and oncology clinicians were invited to participate in the study. Patients were asked to submit weekly symptom questionnaires through an ePRO mobile phone application (app)—ONCOpatient®. Clinical staff were invited to use the ONCOpatient® clinician interface. After 8 weeks all participants submitted evaluation questionnaires.
Results
Thirteen patients and five staff were enrolled in the study. The majority of patients were female (85%) with a median age of 48 years (range 22–73). Most (92%) were enrolled over telephone requiring on average 16 minutes. Compliance with the weekly assessments was 91%. Alerts were triggered by 40% of patients who then required phone calls to aid with symptom management. At the end of study, 87% of patients reported they would use the app frequently, 75% reported that the platform met their expectations, and 25% that it exceeded their expectations. Similarly, 100% of staff reported they would use the app frequently, 60% reported that it met their expectations, and 40% that it exceeded their expectations.
Conclusions
Our pilot study showed that it is feasible to implement ePRO platforms in the Irish clinical setting. Small sample bias was recognized as a limitation, and we plan to confirm our findings on a larger cohort of patients. In the next phase we will integrate wearables including remote blood pressure monitoring.
Introduction
Symptom management through optimal supportive care is a cornerstone of quality in cancer care. 1 Clinicians are unaware of up to half of their patients’ symptoms and systematic symptom monitoring can close this gap.2–5 Monitoring using electronic patient-reported outcomes (ePROs) improves patient–provider communication, allows systematic review of symptoms and enables a timely and appropriate response by the care team.2,6,7 Consequently patient satisfaction, quality of life (QoL) and overall survival3,8 all improve, while hospitalization rates fall.4,9
Currently in Irish hospitals, patients communicate their symptoms during scheduled interval visits to the chemotherapy day-wards and outpatient clinics. The vast majority rely on personal recollection which jeopardizes reliability. European Society of Medical Oncology (ESMO) guidelines recommend implementation of ePROs in routine clinical practice. 1 Furthermore, randomized studies have shown that the use of ePROs is feasible without increasing clinical burden. 10 To date, electronic symptom monitoring has never been implemented in clinical oncology in the Republic of Ireland. This leads to delays in symptom control interventions and potentially increases the need for emergency department visits.
Our primary objective was to test the feasibility of using a smart phone-based ePRO app ONCOpatient® in the Cork University Hospital (CUH), a tertiary cancer center. Secondary objectives included assessment of the workload required for patient profile setup and further optimization of the end-user experience using participant feedback.
Methods
Recruitment and enrollment
Suitable patients were identified by the clinical staff during outpatient clinic visits. They were approached in person and recruited by convenience sampling. Inclusion criteria were age ≥18 years, capable of informed consent, life expectancy ≥3 months, metastatic solid tumor malignancy, on oral anti-cancer treatment and access to a smart phone device. Patients were set up with a unique digital profile containing identifying information and details pertaining to their disease and treatment. Staff were recruited during regular clinical hours and granted access to the ONCOpatient® platform through a secure ONCOassist® account. All participants signed an informed consent.
Study design
This non-randomized, prospective, single arm study was conducted between January and April of 2022. Study received approval by the University College Cork Clinical Research Ethics Committee (CREC) and a Service Provider Data Processing agreement was signed between the senior author and the ONCOassist®. Feasibility was defined as ≥70% compliance with the weekly symptom assessments.10,11
ONCOpatient® development and overview
ONCOassist® provided English language-based digital solutions compatible with Android and iOS platforms. Patient and clinician interfaces were developed in collaboration with the CUH Medical Oncology team and concordant with the ESMO guidelines on ePROs.1,12–14 National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (NCI-PRO-CTCAE™) 15 was used to generate the adaptive questionnaires. 16 The patient facing interface allowed symptom assessment (Figure 1) medication management and communication with the care team. Alerts were generated (Figure 2) and acted upon by the clinical team within 24 hours if a patient reported severe symptoms (≥grade 3) or a deterioration of any symptom by ≥2points. 8 ePRO reports were assessed weekly to ensure continuous functioning of the platform and whenever an adverse change in symptom score triggered an alert. Medication reminders were sent to patients via push notifications and compliance was self-recorded. The ePRO clinician facing interface (Figure 3) provided an organized overview of the patients’ diagnosis, symptom tracking, treatment and compliance.

Patient facing interface—symptom assessment feature.

Alerts triggered by patients’ responses are transmitted via email or sms as per end-user preference. Patients reach appropriate team members directly via pre-programmed phone numbers.

ONCOpatient® clinician facing interface providing information on patients’ treatment, symptoms and compliance.
Data collection and processing
Responses to weekly assessments of 12 common cancer-related symptoms were captured for 8 weeks. 3 Patients reported on pain, fatigue, nausea, vomiting, constipation, diarrhea, shortness of breath, cough, numbness, painful urination and hot flashes. They recorded symptoms on a scale of 0 to 4 using the validated NCI-PRO-CTCAE™ assessment scale. These specific symptoms were chosen as baseline as their use in ePRO assessments has been shown to improve clinical outcomes for cancer patients in a randomized clinical trial. 8 Patients were allowed to modify the questionnaires if they wanted to add a new symptom or remove an irrelevant one. At the end of the study all participants submitted anonymous questionnaires supplemented by free text to reflect their experience with the platform.
Encrypted data was safely stored on the ONCOpatient® Amazon Web Services (AWS) cloud infrastructure residing in the Republic of Ireland and in compliance with both Health Insurance Portability and Accountability Act (HIPAA) and European Union General Data Protection Regulation (GDPR) requirements.
Statistical analysis
Statistical analysis was performed using Excel software. Categorical variables were reported as frequencies (
Results
Study population
In total 18 participants entered the study, 13 patients and 5 clinical staff members. All staff and patients who were approached during recruitment, agreed to participate. Most patients were female (
Baseline patient characteristics.

CDK4/6: cyclin-dependent kinases 4 and 6; BRAF: proto-oncogene BRaf inhibitor; MEK: mitogen-activated protein kinase inhibitor. EGFR: epidermal growth factor receptor; TKI: tyrosine kinase inhibitor.
Feasibility
Average compliance with the weekly symptom assessments was 91%, range (38%–100%), and the self-reported compliance with oral medication was 63% (range 9%–97%) (Table 2).
Assessments of feasibility, treatment compliance and workload.

Added workload
Twelve patients (92%) were set up by telephone and the average time it took to complete the process was 16 minutes (range 9–25 minutes). In-person setup was completed for 1 patient and the time it took was 10 minutes. Alerts were triggered by 5 patients, and they received advice over telephone from their clinical team. No patient triggered more than one alert. Symptoms which triggered alerts were pain, nausea, cough, numbness/tingling and hot flushes.
End of study feedback
At the end of the 8-week study period 8 of 11 patients and 5 of 5 clinical staff members completed the web-based survey (Figure 4, Appendix A). The majority (87%) of patients reported that they would use the app frequently, 75% felt that the app met their subjective expectations and 25% that it exceeded their expectations. Three clinicians (60%) reported that the app met their subjective expectations and 40% that it exceeded their expectations. All participants found the app easy to use and reported that they would use it frequently. Standard 12 symptom questionnaires were modified by 5 (40%) of patients and presence of this feature was confirmed as favorable in the free text feedback. Free text suggestions were heterogenous and mostly pertained to the suggestions on how to improve the app graphics as well as medication management features of the app.
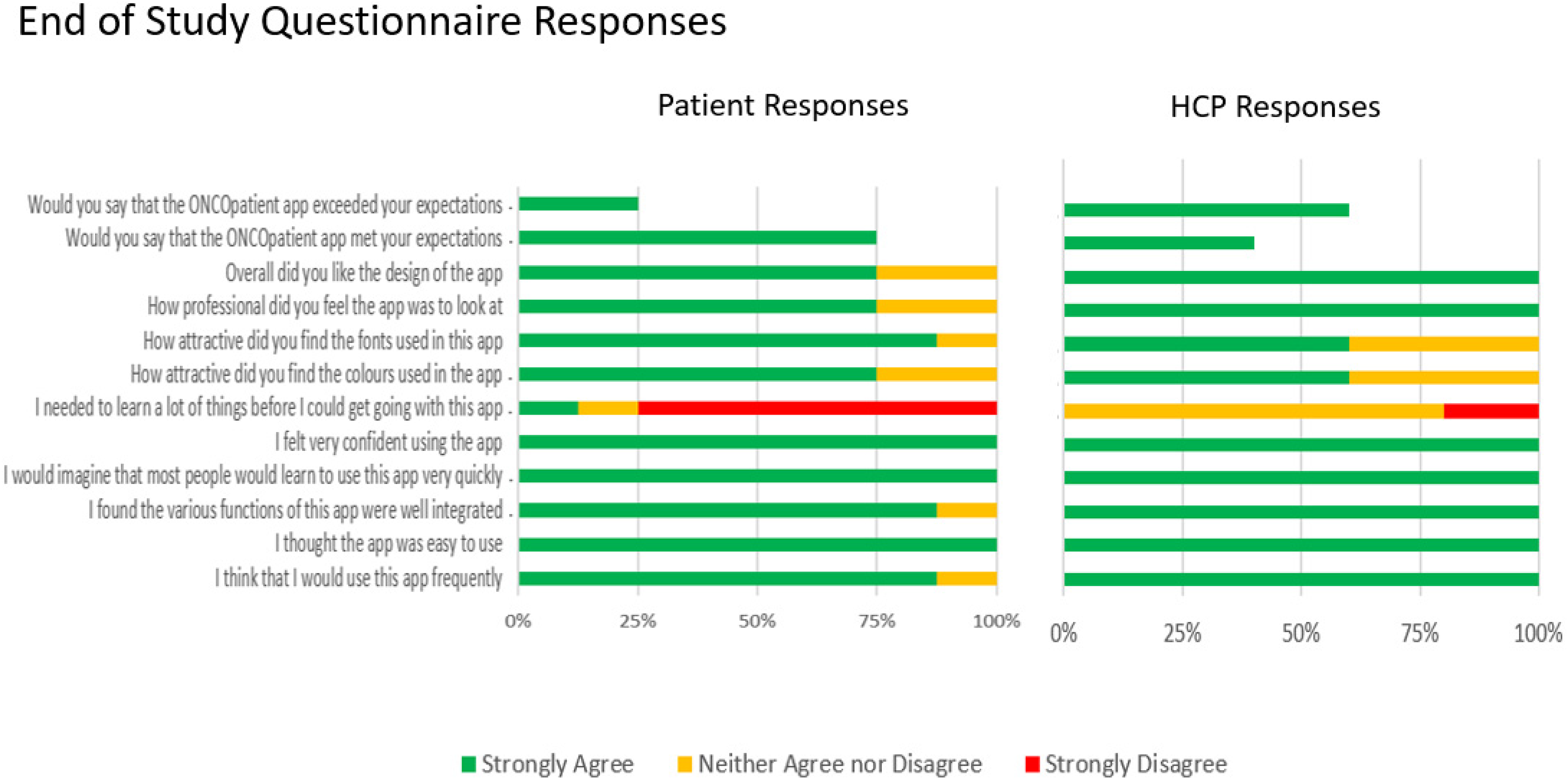
End of study responses (see Appendix A).
Discussion
To our knowledge, this is the first study in the Republic of Ireland to assess the feasibility of implementing an ePRO platform in the setting of clinical oncology. Limitations of the study were the small number of participants and focus was on the oral chemotherapy patients only.
Most patients were women with breast cancer on CDK inhibitors. It was felt that this would not affect the goals of our study as previous publications indicated no significant differences in terms of compliance regardless of sex. 6 However, we will ensure better gender balance in future studies to avoid the interpretation bias.
In terms of added workload we only studied the time required for the initial patient profile setup and additional aspects of the clinical workload will be addressed in the future studies
Our pilot study was not designed to assess the impact of the interventions on the symptom burden and hospitalization rates. However multiple prior studies on PRO implementation already showed favorable impacts on these factors3,17–19
Most patients with low (<50%) medication compliance were in the CDK4/6 inhibitor group where the treatment schedule depended on both pre-cycle absolute neutrophil counts as well as the standard treatment schedule of 3 weeks on and 1 week off. Possible explanations for the low recorded compliance could be issues with initial app set up and treatment delays due to low neutrophil counts. This was recognized as one of the limitations of the ONCOpatient® app. Further iterations of the platform have been implemented to ensure more accurate compliance measurement. We are planning to add a feature to help differentiate whether patients missed doses due to compliance versus safety issues.
Next steps will involve enhancing ONCOpatient® to perform digital phenotyping by integrating data captured
Conclusion
Our pilot study showed that it is feasible to implement an ePRO platform in an Irish tertiary cancer center with high levels of patient and clinician satisfaction. It will be important to continue developing and testing the platform in a larger cohort of patients and in a wider variety of clinical settings.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231185428 - Supplemental material for A pilot project investigating the use of ONCOpatient®—An electronic patient-reported outcomes app for oncology patients
Supplemental material, sj-docx-1-dhj-10.1177_20552076231185428 for A pilot project investigating the use of ONCOpatient®—An electronic patient-reported outcomes app for oncology patients by Bojan Macanovic, David O’Reilly, Harry Harvey, Danial Hadi and Maeve Cloherty, Pauline O’Dea, Derek G. Power, Dearbhaile C. Collins, Roisin M. Connolly, Richard M. Bambury, Seamus O’Reilly in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to thank Mr Kevin Bambury and Mr Eoin O’Carroll of Portable Medical Technology Ltd for their efforts and perseverance in developing the ONCOpatient® ePRO platform and providing technical support during the course of the pilot project. They would also like to acknowledge and thank all of the patients and clinical staff involved in the study.
Contributorship
BM researched literature, conceived the study and developed the study protocol with SOR and RB. BM and SOR contributed to gaining ethical approval and data analysis. DOR helped with protocol development and final protocol submission. BM, HH, DH, MC and POD helped with patient recruitment, symptom monitoring and data acquisition. BM wrote the first draft of the manuscript. All authors reviewed the initial draft, provided critical feedback and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was reviewed and received the approval from the University College Cork Clinical Research Ethics Committee (CREC). CREC Review Reference Number: ECM 4 (x) 09/03/2021 & EC 3 (y) 11/05/2021.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Portable Medical Technology Ltd who provided software development for the ONCOpatient® ePRO platform used in this pilot study. Portable Medical Technology Ltd paid for the cost of the final manuscript submission. Portable Medical Technology Ltd did not have any role in the design of the study or the analysis and interpretation of the data.
Guarantor
Professor Seamus O’Reilly.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
