Abstract
Objective
There are currently an increasing number of mobile health (mHealth) programs offered to patients with breast cancer undergoing chemotherapy, but their rate of adherence to app usage has remained low. This study aimed to examine the feasibility of an mHealth app-based program such as the adherence rate of app usage and determine the preliminary effects on self-efficacy, quality of life, symptom burden and healthcare utilization in these patients.
Methods
We conducted a randomized controlled pilot trial. Ninety-six participants were randomly allocated into either an intervention group or a control group (routine care plus a placebo app). The intervention group engaged in a 6-week self-regulation activity and received nurse-led social support via the app. The intention-to-treat principle was adopted. The generalized estimating equation was utilized to analyze the between-group, within-group and interaction effectiveness of this program.
Results
Totally 96 participants were enrolled from 16 May to 23 August 2022. The average rate of adherence to app usage increased from 4.8% at week 3 to 51.2% at week 6. There was a statistically significant reduction in the physiological efficacy scores of the intervention (p < .001) and control groups (p < .001) at week 6, compared with the baseline. At week 6, the intervention group reported a significantly lower symptom burden (p = .042) and significantly better physical well-being than the control group (p = .024).
Conclusions
It is feasible to perform an mHealth app-based self-management program for patients with breast cancer receiving chemotherapy. Nurses can utilize this program to facilitate patient self-management of symptoms during chemotherapy.
Registration
Clinicaltrials.gov, https://clinicaltrials.gov, (NCT05192525).
Keywords
Introduction
Breast cancer (BC) is the leading cause of cancer mortality and ranks first for incidence in females worldwide. 1 There were currently an estimated 124,002 deaths and 429,105 new cases for Chinese breast cancer patients in 2022. 2 Despite chemotherapy being a common systemic therapeutic strategy for improving the survival rate of BC patients, its side effects, such as alopecia, vomiting, diarrhea and febrile neutropenia, remain problematic.3,4 Nowadays, there are increasing number of BC patients receiving chemotherapy in outpatient settings. 5 BC patients are unable to receive timely medical support at home in the transitional period of outpatient chemotherapy. When they are not able to control symptoms at home, their first choice is to go to outpatient departments. During the COVID-19 pandemic, BC patients undergoing chemotherapy were at a higher risk of getting infected in crowded outpatient departments. 6 Especially for those dwelling in remote rural areas, traveling a long way to hospital may increase their susceptibility to viral infections. 7 Owing to the fear of infection and the closure of clinics, many patients have fewer chances to come into contact with healthcare professionals. This puts patients at risk of having their symptoms deteriorate and can lead to life-threatening situations. 7 Finding an innovative channel such as mobile health (mHealth) to support self-management among this group of patients is warranted.
mHealth is defined as “the utilization of mobile apps to promote health-related behaviors and deliver timely and tailored healthcare for improving health-related outcomes of an individual.” 8 As an important carrier of mHealth, mHealth applications have paved the way for delivering healthcare services, enabling patients who live in rural areas to consult healthcare providers and minimizing unscheduled visits and risk of infection of patients.9–12 The health authorities of developed and developing countries have been adopting mHealth apps to support self-management and symptom tracking for cancer patients. 11 During the COVID-19 pandemic, Chinese hospitals promoted the delivery of transitional care services via mHealth apps for cancer patients undergoing ambulant chemotherapy. 13 Thus far, there has been limited evidence about the effectiveness of mHealth app-based interventions for Chinese BC women undergoing chemotherapy.
According to the CONSORT-EHEALTH, the use (adherence) and non-use (non-adherence) of an intervention (application) are important issues for the effectiveness of mHealth app-based studies. 14 Psychological and physical outcomes seem affected by the adherence of mHealth app-based interventions (i.e. the adherence of app usage). 15 In one e-support program for patients with BC during the treatment of chemotherapy, patients’ app usage was positively related to self-efficacy, QoL and social support. 16 It is unclear regarding the mechanism of how the adherence of app usage influences the outcomes of mHealth app-based intervention. Previous evidence indicated that the mechanisms of adherence effect may be similar to the mechanisms of placebo effect. In this case, it suggested to examine the effectiveness of an mHealth app-based program under placebo and active condition. 15 Additionally, prior mHealth self-management programs reported that the rate of adherence to the app usage in BC patients had a decrease of 25.5%∼50% from the beginning to the completion of the course of chemotherapy. 17 Self-efficacy, which has been identified to be a crucial factor affecting the self-management behavior of BC patients, may have an impact on the patients’ adherence to app usage. 18 Baydoun et al. proposed that patients with higher self-efficacy levels have demonstrated better at changing their self-management behavior than those with lower self-efficacy levels. 19
To explore the mechanisms underlying the adherence effect of mHealth app-based intervention, we developed a novel nurse-led mHealth self-management program (mChemotherapy) under the guidance of Individual and Family Self-management Theory (IFSMT). 20 The IFSMT proposed three facilitators to adherence to self-management behaviors, which included (a) one macrosystem level where social facilitation was used to positively support patients in engaging a recommended behavior; (b) one mesosystem level where patients were empowered to carry out self-management behaviors and (c) one microsystem level where patients gained confidence after performing the intended behavior repeatedly. 21 In the macrosystem level, nurses played a crucial role in delivering social support and providing care such as symptom tracking, case management, surveillance, remote health education and medical referrals. In the mesosystem level, patients with BC were empowered to participate in self-regulation activities. Six components of self-regulation activities, namely goal setting, self-monitoring, reflective thinking, decision-making, planning and action and self-evaluation were integrated into a mobile application to foster self-monitoring behaviors and promote symptom self-management.21,22 In the microsystem level, patients recognized that they were able to comply with self-regulation activities and improve symptom burden, which in turn increase in their self-efficacy levels and adherence to the apps. The current study was designed to capture the phase of the first two cycles of chemotherapy to observe the behavioral strategies of patients with regard to how they respond to social support and establish self-regulation routines.
The current mChemotherapy program was designed to increase the self-regulation activities of the patients with BC undergoing chemotherapy, offer them with nurse-supervised social support with the aim of evaluating the adherence effect of an mHealth app-based intervention and improving their self-efficacy. The purposes of the pilot study were to examine the feasibility (i.e. adherence rate of app usage) and preliminary effects of this program.
Methods
Design
A randomized controlled pilot study was adopted with two parallel groups at the allocation ratio of 1:1. The registration of this study has been posted on ClinicalTrials.gov since 14 January 2022 (NCT05192525). The study protocol has been published elsewhere. 22 All recruited patients received the information explanation for the program and their written informed consents were obtained prior to the baseline data collection. Two follow-up visits were provided to participants at week 3 and week 6, respectively.
Study setting and sampling
The study was conducted in an outpatient chemotherapy ward of a medical hospital in Shanghai, China. The sample size was calculated by G*Power, with the effect size value of 0.38 reported from the same primary outcome in a previous similar mHealth-based study. 23 A sample of 220 (with 110 per group) was needed with α of 0.05, and power (1 − β) of 0.80. Since this is a pilot study, literature suggested to reduce the sample size of the main study by 65%. 24 Eventually, 94 subjects (47 per arm) were estimated for recruitment, assuming an attrition rate of 20%.
Inclusion and/or exclusion criteria
Participants were enrolled following inclusion criteria: (a) adult women (≥18 years); (b) being newly diagnosed with BC; (c) commencing chemotherapy and being scheduled at least four courses of chemotherapy prescription; (d) owning a smartphone; (e) having Wi-Fi at home and (f) being able to read, write and speak Chinese. Exclusion criteria were: (a) being pregnant; (b) being diagnosed with stage IV BC; (c) being scheduled targeted therapy or radiation treatment; (d) having mental disorders and (e) already participating in other mHealth trials.
Recruitment and randomization
The recruitment was carried out from 16 May to 23 August 2022. Four trained nurses enrolled the participants and screened them for eligibility in the hospital. The nurses who were responsible for recruitment screened the participants for eligibility and were not in charge of the process of generating random assignments to the groups. The participants were blinded to their group assignments before the participants signed the consent forms. Then, the eligible participants were randomly allocated to two groups by using the software “the Randomizer.” A researcher who did not engage in the recruitment and intervention carried out the randomization. The allocation results were exhibited only to the nurses who performed intervention in the mChemotherapy platform. To minimize contamination between groups, manuals specifically designed for the intervention/control groups were provided to the participants. The data were collected via online questionnaires. The questionnaires for the two groups bore the same titles and introductions in order to blind the participants in the control group to the random allocation. The nurses who were responsible for caring for the intervention group were not involved in providing the routine care for the control group.
Study interventions
Intervention group
Patients in the intervention group received a 6-week nurse-led mChemotherapy program starting from the self-regulation process of BC patients undergoing chemotherapy. 22 The mChemotherapy program consisted of one core intervention “mChemotherapy application,” one pre-chemotherapy visit and two scheduled follow-up visits. The mChemotherapy application was provided to patients in the intervention group at the commencement of chemotherapy.
A mChemotherapy application
The mChemotherapy application was developed based on algorithm and contained six modules: (a) self-monitoring, (b) knowledge base, (c) alerts, (d) consultations, (e) reminders and (f) my prescriptions (Figure 1). The algorithm structure of the mChemotherapy app encompasses patient-initiated behavior, system behavior and nurse-led behavior (Figure 2). The patient-initiated behavior, self-monitoring, was set to be correlated to three system behaviors (reminder, alerts and knowledge base). Of the three system behaviors, the alerts could trigger nurse-led behaviors. The nurse-led supportive behaviors, such as consultations and nursing prescriptions, could be provided to patients via the app.

Six modules of the mChemotherapy app for the intervention group.

The structure of the mChemotherapy app.
The intervention protocols to self-management of eight symptoms (i.e. fever/febrile neutropenia, nausea and vomiting, diarrhea, constipation, oral mucositis, insomnia and fatigue) were designed based on the clinical guidelines and the systematic reviews for cancer patients undergoing chemotherapy.4,25,26 A professional team, composed of three nursing experts, two advance practice nurses, one nursing teacher and two software engineers, constructed and reviewed the intervention protocols and the algorithm of the app.
(i) Self-monitoring: The “self-monitoring” module was used by participants of intervention group to monitor their symptoms by themselves during the 6 weeks. After the commencement of chemotherapy, the participants were required to report weekly on eight symptoms or whenever they felt unwell. In addition, the participants could report the results of a blood test after undergoing a blood routine examination in an outpatient department. After each report, the participants would be notified of the overall severity of their symptoms (mild, moderate or severe).
(ii) Knowledge base: When the reported symptoms were rated as mild, the self-management advice from the knowledge base was sent automatically to the patients. In the “Knowledge base” module, articles or videos on the self-management of eight chemotherapy-related symptoms were updated every 2 weeks. The patients could also search articles or videos in the knowledge base through keywords.
(iii) Alerts: The “Alerts” module has two functions: emergency hotline calls and medical referrals. When the reported symptoms were rated as severe, an alert message could be sent to nurses. A hotline call link was sent to the patient so that he or she could immediately contact the nurses. Nurses were required to initiate the emergency hotline calls and arrange a medical referral to the emergency department (ED) for the patients within 1 h, if necessary.
(iv) Consultation: The “Consultation” module was set for participants to inquire about their symptoms and for the nurses to perform a remote reassessment. When the reported symptoms were rated as moderate, the participants needed to complete a reassessment questionnaire via the consultation module. For those whose reassessed results were again rated as moderate, nurses delivered non-pharmacological self-management advice to patients within 24 h. For those whose reassessment results were rated as severe, nurses could immediately send patients a referral to an ED or outpatient department. Nurses could also provide individualized feedback to participants on their questions after the participants had completed the reassessment questionnaires.
(v) Reminder: The “Reminder” module was utilized to send automatic reminders for weekly self-monitoring. The participants could receive two automatic reminder messages for their weekly task of self-monitoring, at 7:00 AM and 7:00 PM, during the 6-week intervention period. For those participants who had not adhered to the self-monitoring protocols, nurses reminded them to perform the self-monitoring at the follow-up visits, if necessary.
(vi) My prescriptions: The “My prescription” module was adopted by participants to review the previous nursing prescriptions. Nurses delivered three sorts of nursing prescriptions during the 6 weeks. The first kind of nursing prescription, self-management advice (i.e. the engagement in exercise), was automatically sent to patients who had been assessed with mild symptoms. The second kind of nursing prescription, non-pharmacological nursing advice (i.e. the use of cryotherapy for mucositis), was delivered within 24 h to those who had been assessed with mild symptoms. The third sort of nursing prescription, such as a medical referral, was made by nurses to an ED within 1 h for those with severe symptoms.
In the pre-chemotherapy visit, an individualized goal-setting and a self-management planning were conducted by the nurse. The nurses provided education about the treatment regimen and the self-management of chemotherapy-induced side effects via the Official WeChat platform to the patients.
In the two scheduled visits (week 3, week 6), nurses reviewed the comprehensive symptom reports by clicking on the “History record” button on the mChemotherapy platform. For those participants who did not adhere to the weekly self-monitoring protocols, nurses asked them about the potential reasons for their nonadherence to self-monitoring and tried to provide encouragement to patients. The nurse and the patients would co-design a long-term self-management plan according to the patients’ specific and individualized needs.
Control group
Participants in the control group were treated with routine care plus a placebo app. The routine care included one pre-chemotherapy visit and two planned visits. The placebo app, which provided online consultation, reminders for chemotherapy and education corner, was given to each control group participant. The education corner provided information and knowledge on breast cancer and cancer treatments (i.e. introduction to breast cancer, causes of breast cancer and prevention of breast cancer). In the pre-chemotherapy visit, the nurses provided face-to-face education about the treatment regimen and the symptom self-management skills and knowledge to the patients. The two nurse-initiated visits were provided to control group via telephone calls at the 3rd week and the 6th week after the commencement of intervention. Patients consulted the nurses via telephone or the consultation module if they had any questions during the chemotherapy. Participants in the control group were not able to use the mChemotherapy app until they completed the post-data collection. The differences in protocols between the two groups were presented in Table 1.
Comparison of protocols between the two groups.

Fidelity of intervention
To ensure a complex intervention is implemented as intended, a framework of intervention fidelity strategies was adopted during the implementation phase of the study, which includes (a) study design, (b) training the providers, (c) delivery of treatment, (d) receipt of treatment and (e) enactment of treatment skills.27,28 The current study followed the five domains of fidelity strategies to ensure the rigor of the nurse-led mHealth self-management intervention protocol. The researcher evaluated the e-records of the mobile app on a weekly basis for patients’ adherence to the nursing prescriptions. At the end of the pilot study, the nurses completed a self-report using a performance checklist.
Study variables
Feasibility outcomes
Feasibility of participant recruitment and follow-up process. Five feasibility outcomes were measured, which included the time cost on subject enrollment, the eligibility rate, recruitment rate, retention rate and withdrawal rate.
Participant adherence of app usage. Participant adherence of using the app was tested by: (a) the adherence to the self-monitoring protocol and (b) data on the personal app usage. The rate of adherence to the self-monitoring protocol was assessed through the overall number of self-monitoring days divided by 6. For those participants who conducted self-monitoring once per week for 6 weeks, their adherence rate would be 100%. Data on the personal app usage for 6 weeks, such as log-in frequency was tracked via the app database. Log-in frequency was assessed as the number of times that a subject logged into the modules of the app during 6-week intervention.
Effectiveness outcomes
Primary outcome: self-efficacy. The 29-item strategies used by people to promote health (SUPPH) was utilized to measure the self-efficacy of the participants. The patients who reported higher scores indicated a higher level of self-efficacy. 29 The SUPPH was validated and adapted in Chinese cancer patients, having a high reliability (Cronbach's α=0.970). 30
Secondary outcome: quality of life. The Chinese version of Functional Assessment of Cancer Therapy-Breast (FACT-B) was used to assess the QoL of BC patients. Higher scores represented a higher level of QoL. FACT-B has been proven to be reliable and valid tool with internal consistency ranging from 0.82 to 0.85. 31
Secondary outcome: symptom burden. The 28-item Memorial Symptom Assessment Scale-Short Form (MSAS-SF) was used to determine the symptoms burden of patients during chemotherapy. The patient who reported higher scores experienced symptoms with more frequency, greater severity and higher distress. 32 The simplified Chinese version of MSAS-SF has been validated with good reliability of Cronbach α (0.782∼0.874). 33
Secondary outcome: healthcare utilization. Three types of chemotherapy-induced healthcare utilization were collected for analysis, involving patient-initiated emergency calls and unplanned visits to the outpatient and emergency departments. These records were obtained from the database of the app.
Data collection
There were a total of three-time points for data collection, namely, baseline (T0), week 3 (T1) and week 6 (T2). We compared self-efficacy and QoL at three-time points. Symptom burden was compared at two-time points (T1 and T2) and healthcare utilization was compared at T2. In addition, sociodemographic data was collected at baseline. At week 6, the feasibility outcomes were measured. All the questionnaires were collected through an online questionnaire website. For the adherence of app usage, the logged data of the subjects in both groups (the mChemotherapy group and the control group) were collected during the 6-week intervention via the app.
Data analysis
The intention-to-treat (ITT) was employed as the primary analysis for this pilot study. 34 For the ITT anaysis, all participants who are randomly assigned at baseline are included in the data analysis, even if they have dropped out of the study, did not adhere to the treatment, have not received a scheduled intervention or have been lost to the planned follow-ups. 34 The student t-test and chi-square tests were utilized to compare the continuous and categorical baseline characteristics of participants, respectively. Descriptive analyses were utilized to demonstrate the feasibility outcomes (i.e. the recruitment rate, dropout rate, questionnaire feasibility, patient adherence to app usage). Effectiveness outcomes, such as self-efficacy, QoL and symptom burden, were compared for the group, time and group × time effects by the generalized estimating equation (GEE) model. A Mann–Whitney U test was adopted to analyze the total value on frequency for patient healthcare utilization among two arms. Multiple imputations were adopted for the missing data, on the assumption that the data had been missing completely at random. 35 Missing continuous variables were imputed using a Markov chain Monte Carlo method and dichotomous or categorical variables were imputed using a fully conditional specification model. 36 Missing values were imputed within each group with adjustments for the baseline level of the imputed variable. 36 Despite the ITT method can preserve the benefits of randomization and yield an unbiased and accurate estimate, it may also lead to an underestimation of the results of an effective intervention. Therefore, sensitivity analyses were conducted by the comparisons of effectiveness outcome results under different analytic and statistical methods (i.e. ITT vs. per-protocol [PP] analysis, covariate-unadjusted vs. covariate-adjusted models, with vs. without multiple imputations). Since age has been identified as a crucial predictor of SUPPH scores and participants had statistically marginal between-group differences with regard to ages at baseline (intervention: 48.6 [12.2] years vs. control: 53.0 [10.9] years; 95%CI [−9.08 to 0.29], p = .065), it was used as a covariate in the adjusted GEE analyses. 37 Statistical analyses were carried out via IBM SPSS Statistics 26.0. The significance levels were assumed at 0.05 for two-tailed tests.
Ethical considerations
The study was granted ethical approval by the Human Subjects Ethics Sub-Committee (HSESC) of the Hong Kong Polytechnic University on 20 September 2021. Participants had the option to decline participation or withdraw at any point without consequence. Refusal or withdrawal could not impact their regular care. Participants received information on the study's purpose, procedures, risks, benefits, incentives, security and privacy. They received unique login credentials for the app. Personal data remained confidential, and all participants were de-identified. Only the research team had access to the data, which was securely stored. No data was saved on personal devices. The pilot study's findings were used in publication, with no personal information disclosed.
Results
Feasibility outcomes
Feasibility of participant recruitment and follow-up process
From 16 May to 23 August 2022, 96 patients (mean [SD] age, 58.9 [12.5] years) were enrolled. Of them, 64.6% (62/96) were at stage I of breast cancer and the majority (78.1%) received adjuvant chemotherapy (Table 2). The recruitment rate was 82.1% (96/117) and six out of 96 participants withdrew from the study intervention (6.3%). Figure 3 exhibited the Consolidated Standard of Reporting Trials (CONSORT) Flow Diagram.

A consolidated standard of reporting randomized trials (CONSORT) flow diagram.
Baseline characteristics of the participants.

Note: Statistically significant at p < .05.
Homogeneity of the baseline variables between the two groups
No statistically significant differences in the demographic and clinical data of the groups were found, with the p-value ranging from 0.065 to 0.936. No statistically significant between-group differences on self-efficacy and QoL were identified at baseline.
Patient adherence of using the app
The average adherence rate to app usage increased from 4.8% at week 3 to 51.2% at week 6. The total frequency of reported symptoms was 498. Of them, fatigue was the most frequently reported symptom (n = 138), followed by insomnia (n = 96) and constipation (n = 80) (Figure 4). The least frequently reported symptom was fever (n = 10). Regarding the login frequency of knowledge base, the self-management advice for fatigue was the most frequently read paper by patients, followed by the advice for infection prevention and self-management of constipation.
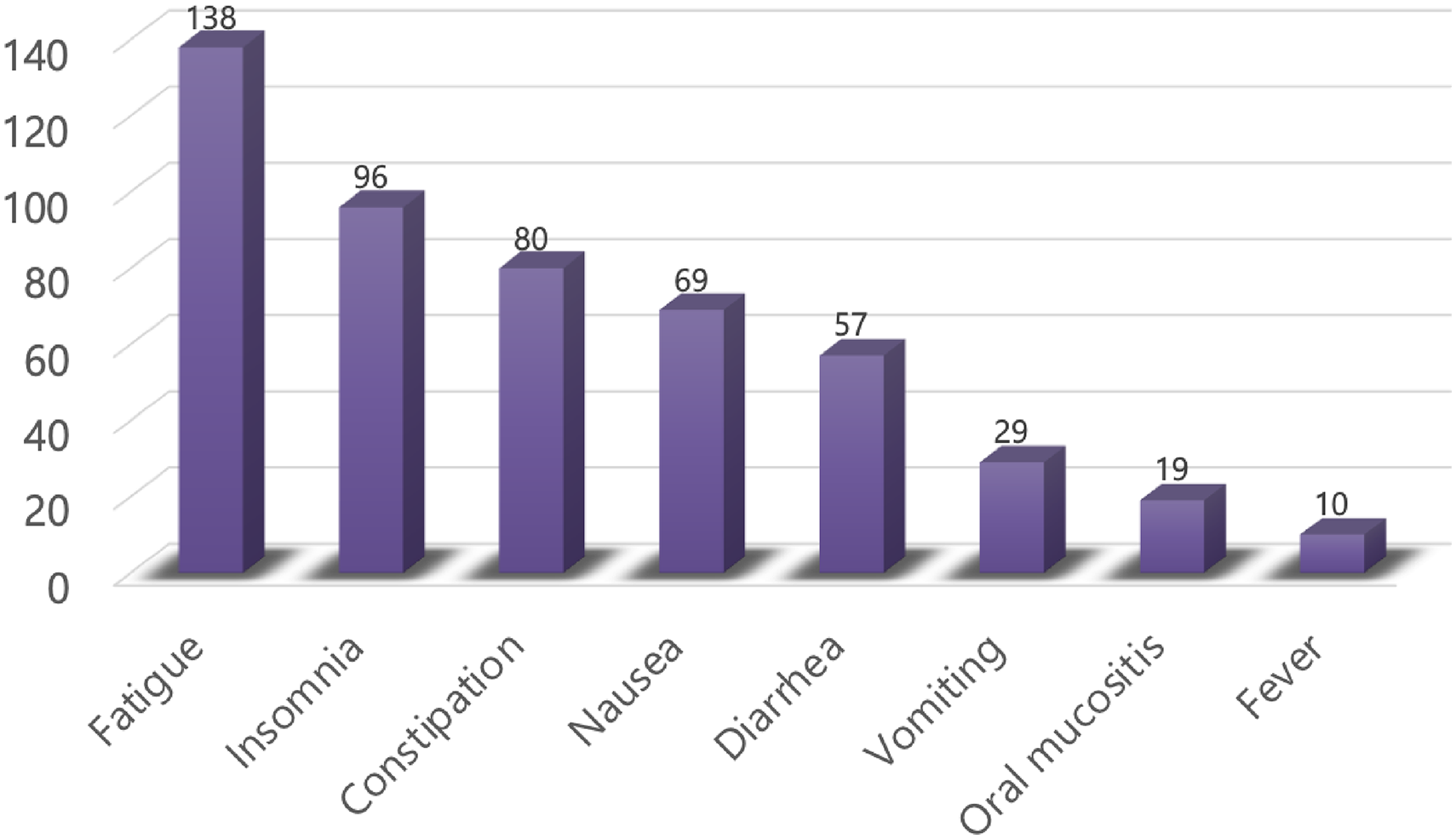
Distribution of symptoms reported via the app over 6 weeks (n = 498).
Fidelity of intervention
According to the results of the researcher's evaluation and the nurses’ self-reported checklist, all of the nurses had adhered to the intervention protocols.
Effectiveness outcomes
Self-efficacy
No statistically significant between-group and interaction effects of both total and sub-scale self-efficacy scores were obtained over the 6 weeks. The scores of physiological efficacy domain reached a statistically significant reduction in both two groups from baseline to week 3 (intervention, B = −60.16, 95%Cl [−64.93 to −55.38], p < .001; control, B = −61.25, 95%Cl [−66.64 to −55.86], p < .001). In the contrary, the performance efficacy scores exhibited a statistically significant increase from baseline to week 3 in both groups (Table 3).
The between-group, within-group and interaction effects of self-efficacy and QOL by ITT analysis.

Note. QoL: Quality of life. aP value for between-group effects. bP value for within-group effects. cP value for interaction effects. b1P value for time effects of intervention group. b2P value for time effects of control group. c1P value for time (T1)*group effects. c2P value for time (T2)*group effects. dHigher score indicating higher level of self-efficacy. eLower scores indicate better QoL.
Higher scores indicate better QoL.
Quality of life
The between-group effectiveness exhibited that the participants in the intervention group reported significantly better physical well-being domain scores than those in the control group (B = −2.39, 95%Cl [−4.41 to −0.37], p = .021). The intervention group reported significant higher functional well-being scores (B = 2.33, 95%Cl [−0.13 to 4.79], p = .047) and lower physical well-being scores (B = −3.10, 95%Cl [−5.80 to −0.40], p = .024) than the control group at week 6 (Table 3).
Symptom burden
The MSAS scores increased from week 3 to week 6 in the control group (B = 7.07, 95%Cl [2.96 to 11.18], p = .001). At week 6, the intervention group showed a statistically significantly lower MSAS scores (B = 8.03, 95%Cl [0.31 to 15.75], p = .042) than the control group (Table 4). The prevalence of six symptoms (lack of energy, nausea, feeling drowsy, vomiting, problems with sexual interest or activity, weight loss) exhibited statistically significant between-group differences at week 6. Figure 5 displays the distribution of symptom prevalence reported in MSAS at week 6.

Distribution of symptom prevalence reported in MSAS at week 6.
The between-group, within-group and interaction effects of symptom burden by ITT analysis.

Note. GDI: Global Distress Index; MSAS: Memorial Symptom Assessment Scale; PHYS: Physical Symptom dimension; PSYCH: Psychological Symptom dimension. aP value for between-group effects. bP value for within-group effects. cP value for interaction effects. b1P value for time effects of intervention group. b2P value for time effects of control group. c1P value for time (T1)*group effects. c2P value for time (T2)*group effects. dLower scores indicate better symptom burden.
Healthcare utilization
The total number of healthcare utilization for the control group (n = 405) was more than that of the intervention arm (n = 298). Significant less instances of WeChat consultation were found in the intervention group (n = 85), compared with the control group (n = 134) (p = .050).
Sensitivity analysis
The majority of self-efficacy results were the same when using the ITT or PP analytical methods. No obvious changes were noted in those statistics with and without controlling for the factor of age. The minor variations in the mean difference indicated that the confounding effects of potential factor (older age) were non-significant.
Discussion
Overall, this mHealth nurse-led self-management program displayed a high recruitment rate and a low withdrawal rate. The adherence rate to app usage had an increment from 4.8% in the middle of the program to 51.2% at the end of the program. Despite this program had no significant effectiveness on self-efficacy, patients in the mChemotherapy group reported significant lower symptom burden and higher QoL scores than those in the control group. Meanwhile, this program had significant benefits on healthcare utilization regarding the WeChat consultation.
The feasibility of this mHealth nurse-led self-management program
The recruitment for the current pilot RCT commenced on 16 May 2022, when the pandemic in Shanghai led to great changes in treatment for patients with cancer. Numerous clinics were closed, cancer treatments such as chemotherapy were suspended and cancer patients seldom visited to hospitals due to the fear of becoming infected and to the COVID-19-related social restrictions. Patients were also much less willing to take part in clinical studies after the onset of the pandemic in Shanghai in March 2022. However, our recruitment rate was higher than previous mHealth studies.38–40 Individuals are easier to sign up for an mHealth intervention as they feel it provides a channel in which they can receive online social support quickly and facilitate better management of their symptoms. 41
This program provided timely online social support by nurses to better manage patients’ symptoms, which contributed to a low withdrawal rate in the intervention group. Conversely, the patients in the control group had a higher dropouts than those in the intervention group. This could be explained by the results from the post-intervention group interviews, which have been submitted to a journal but unpublished yet. For example, the patients in the control group commented that they did not receive timely social support from nurses. It suggested that more strategies should be used to reduce the attrition rate such as weekly reminder to participation via telephone rather than messages. Moreover, several patients have commented that they were happy to accept the invitation of enrollment since it was not cumbersome to use the mHealth app. We conducted a usability testing before the recruitment and high perceived usability scores were obtained from the patient–testers, which indicated that the usability of this mChemotherapy intervention featured with a high quality. Furthermore, the results of group interviews showed that most patients agreed to engage in the study as they perceived this mHealth app provided them a reliable and valuable healthcare resource from a top hospital in China. A number of patients residing in rural locations underwent outpatient chemotherapy in Shanghai. Typically, they would return home in between their chemotherapy sessions. In rural areas, the availability of healthcare resources is not as extensive as it is in Shanghai. This program has the potential to offer remote care through mHealth to breast cancer patients residing in underdeveloped regions.
The app usage adherence rate exhibited a significant jump, rising from 4.8% to 51.2%. The improving adherence rate of using apps across the program was not in line with the previous mHealth studies that reported decreasing rates of 25.5%∼50% app usage from the beginning to the completion of the course of chemotherapy.42,43 The low adherence rate of 4.8% at week 3 may be attributed to the lack of motivation among breast cancer patients to engage in self-monitoring, particularly during the initial chemotherapy cycle's transitional periods when severe symptoms are experienced. The increase in adherence rate of app usage may be explained by two reasons. First, IFSMT proposed that patients who have high self-efficacy levels will be more intended to comply with self-management activities (i.e. the adherence behavior of using apps). Second, the low symptom burden and high awareness of symptom control in turn fostered patients to maintain the app usage in symptom self-management and consequently increase their adherence rate of app usage. 44
The effectiveness of this mHealth nurse-led self-management program
At week 6, the patients in the intervention group reported a higher performance domain score of self-efficacy compared with those in the control group, although there were statistically significant interaction effects for self-efficacy. The result was different from one previous study, which tested the effects of mHealth-based social support and reported that the e-support intervention had significant effects on BC patients’ self-efficacy. 16 Two reasons were identified to interpret the non-significant between-group effects of this program on self-efficacy. First, the patients in the control group received the social support via a placebo app, which could potentially mimic the effects of the current intervention program on self-efficacy. Second, the intervention effects could be influenced by the rate of adherence to app usage. The low rate of adherence to app usage for some participants might diminish the intervention’s effectiveness on self-efficacy.
Regarding the quality of life, the mChemotherapy group had statistically significantly better physical well-being than those in the control group. This finding was not in agreement with one RCT from Taiwan. 45 For the emotional and social domain of QoL, in the current study patients in the intervention group reported better emotional and social well-being than those in the control group, although there were no significant differences between the groups. This finding was in line with one recent RCT from Sweden and another cohort study from Slovenia.38,46 At the mesosystem level, BC patients were empowered to participate in iterative self-regulation activities. The self-regulation process increased the patients’ health awareness. They paid more attention to the self-management of physical symptoms. Symptom self-management behaviors affect the symptom burden, which may conversely influence health-related QoL. 19 Additionally, the clinical physical improvement can contribute to further improvement in functional well-being. 47 Thus, the patients in the intervention group reported slightly better functional well-being scores at week 6 than those in the control group.
This mChemotherapy program had encouraging results regarding symptom burden. The intervention group reported statistically significant lower scores for the total MSAS and PHYS domains than the controls at week 6. Our findings were similar with previous studies.12,48 Additionally, patients’ symptom prevalence was higher in the placebo app group than those in the mChemotherapy app group. The lower frequencies of visits to outpatient department were observed in the patients of mChemotherapy group compared with the controls. In addition, the less healthcare utilization such as the readmission rate may lead to the reduction of medical expenses.
Strengths and limitations
One strength of this pilot RCT is that several strategies of sensitivity analyses such as ITT and PP methods were adopted. The similar results from the sensitivity analyses pointed to the high reliability and robustness of the study. The preliminary effectiveness of this program could be supported by the results from the sensitivity analyses.
Our pilot study has several limitations. First, the attrition bias could not be excluded. A number of participants did not answer the questionnaires due to forgetfulness or symptom distress. To reduce the attrition bias, the missing data was checked and addressed by multiple imputations. Moreover, the univariate association analyses were used to compare the participants who were with and without missing values. These analyses for comparisons facilitate to discern the variables which could be included in the imputation model and provide proof for the plausibility of the missing at random assumption. 49 Second, the selection bias for participants should be taken into consideration, although no significant difference of the variables was found between the two groups at baseline. Most participants (75%) who were aged below 60 years were included in this study. The patients who were unable to use smartphones were commonly elderly patients and were excluded during the process of recruitment. Thus, the effectiveness of the intervention should be used with caution for patients at all ages. The lack of a longer study duration and follow-up period is also one limitation of this pilot study. The duration of the interventions in similar previous mHealth RCTs varied from 6 weeks to 160 days. 17 Although several positive results on QoL and symptom burden were identified, the long-term effectiveness and sustainability of the intervention have yet to be exhibited. A follow-up period of 12 weeks or longer should be used in a future full-scale study to evaluate the sustained effectiveness of this program.
Conclusions
It is feasible to conduct a 6-week nurse-led mHealth app-based self-management program for patients with BC undergoing chemotherapy. Self-efficacy is a facilitator to the adherence behavior of app usage in BC patients undergoing chemotherapy, despite there being no significant difference in group and interaction effects on self-efficacy. This program has preliminary effectiveness in improving BC patients’ symptom burden and physical well-being during chemotherapy. A future double-blinded full-scale RCT is needed to definitely determine the effectiveness of this program among BC patients during chemotherapy.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241231560 - Supplemental material for Feasibility of a mobile health app-based self-management program for Chinese patients with breast cancer receiving chemotherapy: A randomized controlled pilot study
Supplemental material, sj-docx-1-dhj-10.1177_20552076241231560 for Feasibility of a mobile health app-based self-management program for Chinese patients with breast cancer receiving chemotherapy: A randomized controlled pilot study by Nuo Shi, Arkers Kwan Ching Wong, Frances Kam Yuet Wong, Nan Zhang, Weiyi Zhu, Kunwei Shen, Xiaobin Lai, Yufei Jin, Chengjia Gu, Lijing Nie and Xiaojing Dong in DIGITAL HEALTH
Supplemental Material
sj-doc-2-dhj-10.1177_20552076241231560 - Supplemental material for Feasibility of a mobile health app-based self-management program for Chinese patients with breast cancer receiving chemotherapy: A randomized controlled pilot study
Supplemental material, sj-doc-2-dhj-10.1177_20552076241231560 for Feasibility of a mobile health app-based self-management program for Chinese patients with breast cancer receiving chemotherapy: A randomized controlled pilot study by Nuo Shi, Arkers Kwan Ching Wong, Frances Kam Yuet Wong, Nan Zhang, Weiyi Zhu, Kunwei Shen, Xiaobin Lai, Yufei Jin, Chengjia Gu, Lijing Nie and Xiaojing Dong in DIGITAL HEALTH
Footnotes
Acknowledgments
Nuo Shi thanked Qingyun Xue, Junxian Chen and Shuai Li who were responsible for developing the app.
Contributorship
NS, AKCW and FKYW contributed to conceptualization, methodology and software. NS contributed to data curation and writing—original draft preparation. XL and NZ contributed to software. NZ, YJ, CG, LN and XD contributed to investigation. WZ and KS contributed to project administration. AKCW and FKYW contributed to writing—reviewing and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The ethical approval for this study was granted by the Hong Kong Polytechnic University (HSEARS20210816002).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by thesis funding for the pursuit of the degree of Doctor of Health Science, Faculty of Health and Social Sciences, The Hong Kong Polytechnic University. NS was supported by the Scientific Research Funding Project of the Education Department of Liaoning Province (grant number JYTMS20230574, HSEARS20210816002).
Guarantor
NS and AKCW.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
