Abstract
Background
Alcohol use disorder (AUD) is one of the most common substance use disorders. People with AUD are in great need of highly accessible and comprehensive management, involving medicine, exercise, and psychotherapy. However, due to limited resources, providing comprehensive treatment for every patient is challenging. Virtual agent-assisted intelligent rehabilitation treatment can improve the accessibility of comprehensive management.
Objectives
This randomized controlled trial aims to test whether the virtual agent-assisted intelligent rehabilitation treatment (Echo-app-v2.0) plus treatment as usual (TAU) has greater efficacy than only TAU in AUD.
Methods
One hundred participants with AUD will be recruited and randomly assigned to either TAU or virtual agent-assisted intelligent rehabilitation treatment plus TAU based on the 1:1 ratio. The virtual agent-assisted intelligent rehabilitation treatment is delivered by the Echo app v2.0 developed by our research team. Participants will be assessed at baseline, week 4 of treatment, one month, and three months after the end of treatment. The primary outcome is the participants’ craving for alcohol, measured by Visual Analogue Scale. Other outcomes include the use of alcohol, motivation for treatment, coping style, depression, anxiety, impulsivity, stress, and sleep quality.
Discussion
The virtual agent, with vivid human image, high privacy, and the ability to interact with users, has the potential to play an important role in the delivery of digital psychotherapy. The development of Echo app v2.0 has the possibility to raise the availability of comprehensive treatment for people with AUD.
Keywords
Background
Alcohol use disorder (AUD) is defined as a problematic pattern of alcohol use despite adverse social, occupational or health consequences. 1 AUD is characterized by high prevalence, social functioning impairment and association with other severe comorbidities. 2 People with AUD also show impairments in their ability to perceive social information, process emotions and establish interpersonal relations.3,4 Common cooccurring psychiatric disorders of AUD include depressive disorders, personality disorders, and anxiety disorders. 5
Besides, AUD is associated with physical comorbidities, including higher rates of obesity, hypertension, low physical activity, and higher risk of all-cause and cancer-specific mortality.
Luckily, extensive research has shown that evidence-based treatment such as behavioral and psychological treatments or medication can help people with AUD achieve and maintain recovery.6,7 Physical examination and subsequent screening are also essential for participants with AUD. In clinical management practice, past research has revealed that comprehensive treatment plans that combine the medication plus behavioral treatment is considered most effective8,9 Involving patients in one or more psychological and pharmacological treatments is more important than advocating a specific treatment.
Recommended psychological interventions for AUDs include motivation interviewing (MI), cognitive behavioral therapy (CBT), problem-solving skills, and mindfulness.10,11 MI can improve clients’ treatment compliance and motivation. A common difficulty for people with AUD is lack of motivation to change or sense of frustration at the difficulty of changing. Therefore, to enhance their internal motivation and awareness of the possibility of change is an important goal in MI. 12 CBT includes a range of social learning theories and stress-coping skills. From the perspective of CBT, alcohol dependence is a learned behavior that can be modified by learning cognitive and behavioral techniques. Cognitive behavioral therapists first increase motivation to quit or reduce alcohol, help clients identify highly risk situations for drinking alcohol, understand the consequences of overusing alcohol, discuss the idea of controlling dysfunctional drinking to prevent relapse, and improve social skills and stress management. 13 Mindfulness is easy to learn and use. It can help clients with AUD develop self-awareness and attitudes of acceptance so that they can detect and adjust negative emotions in time in the process of alcohol abstinence. 14
Despite the effectiveness of treatment, there still exists a gap that many people with AUD do not actively seek treatment. 15 The reasons behind their attitudes towards treatment are varied, including high price, stigma of mental health services and lack of professionals.
Technology-based interventions are considered emerging ways for people with AUD to manage and continue care by themselves. 16 In the past decade, a growing body of research has recognized the effectiveness of interventions by technology on AUD, which provides more treatment options. Based on psycho-social interventions and medication management, a novel computer-delivered behavioral platform was developed in 2016 for people with AUD to reduce therapists’ bias and the cost of implementing clinical trials. 17 Our research team from Shanghai Mental Center also developed a community-based addiction rehabilitation electronic system, which showed high levels of continued participants and social workers’ acceptability. 18
However, current studies are mainly based on unidimensional indicators such as scale evaluation, cognitive tasks or physiological indicators to propose the pathological characteristics of people with substance use disorders and interventions are relatively simple, less accurate, and less targeted. Several systematic reviews of mobile interventions on AUD showed that unidimensional interventions are not sufficient to support participants’ recovery needs.19–21 Comprehensive management is the most effective in AUD recovery.8But even after considering components of comprehensive interventions and specified implementation strategies, what factors play an important role in the recovery of participants in the comprehensive management are still unknown.
In addition, as the traditional diagnosis, evaluation and psychological interventions are highly dependent on empathy and interaction between clients and professionals, the current digital interventions are hard to simulate the therapist’s image with a high degree of imitation, which directly affects the clients’ experience and therapeutic effect. The construction of highly simulated virtual images of psychotherapists is crucial for the application of digital interventions. Virtual agent, with the vivid image of human and the ability to interact with users, has the potential to assist in delivering psychotherapy.
Our research team have developed a tablet-based app with the function of a virtual digital psychotherapist called Echo-app, which has gained positive preliminary results in people with methamphetamine use disorders. The participants showed a significant increase in motivation for abstaining from drugs and satisfaction with the app. 22 The participants with methamphetamine use disorders also identified the facilitators in their experience of using the Echo app, including the detailed and thought-provoking treatment content, vivid image of the virtual therapist, the flexibility of time, the novelty of form, and emotional approachability (unpublished).
As we have only conducted a single-arm study, a more robust research design such as randomized controlled trial (RCT) needs to be considered to verify the long-term treatment effect in the future. Thus, based on the Echo app, of which the efficacy had been estimated around people with methamphetamine use disorders, we have developed a “psychological-cognitive-physiological” multidimensional assessment and rehabilitation management system (Echo app v2.0). The system consists of multidimensional assessment including psychological scales, cognitive tasks, and physiological indicators and virtual agent-assisted intelligent rehabilitation treatment. Based on evidence-based psychological interventions for AUD, including MI, CBT, and mindfulness the system provides targeted rehabilitation treatment for AUD. This system also adopts virtual images and audio of human psychotherapists to create vivid interactions with clients.
Accordingly, we aimed to test the efficacy of virtual agent-assisted intelligent rehabilitation treatment in participants with AUD with RCT. Specifically, the objective of this trial is to determine whether the virtual agent-assisted intelligent rehabilitation treatment plus treatment as usual (TAU) has greater efficacy than mere TAU in treating AUD, measured by their craving for alcohol.
Methods
Design and implementation of the Echo-app v2.0 system
Echo-app v2.0 is a “psychological-cognitive-physiological” multidimensional assessment and rehabilitation management system. This system consists of the user side, administer side, and server side. The administrator side is used to manage patient accounts, access and download assessment and intervention data. The server is connected to the administrator side and the user side to obtain and integrate data in both sides. The user side is composed of assessment module and intervention module. The assessment module, connected to the administrator and server, is used to assess the severity of substance use disorders, psychological emotional state, and physiological indicators. The intervention module, which is connected assessment module, administrator and server sides, can interact with participants through virtual digital image of a therapist(Figure 1).

Schematic diagram of the architecture of Echo app v2.0.
The assessment module includes psychological scales, cognitive tasks, and physiological indicators. Psychological scales are used for screening the participants’ mental and emotional state. Cognitive tasks include dot-probe task, balloon analogue risk task (BART), stop signal task (SST), and delay discounting task (DDT).
The dot-probe task aims to measure the attentional bias of participants. The computer presents a pair of images to the participant each time, a neutral stimulus, and alcohol-related object. The probe point then appears randomly in the location of one of the images. The participants respond to the position where the probe point appears. In the task, the relative positions of the two images and the probe point and the cue image appear randomly, to balance the practice effects and fatigue effects. The task assumes that participants respond faster to the dot when the salience of an alcohol-related object captures the attention of the participants. 23
BART is a laboratory-based measure used to test the risk taking of the participants, in correlation with the self-reported occurrence of addictive behaviors. 24 In this task, a simulated balloon will be displayed on the screen and participants need to blow up the balloon gradually. Each time the balloon is blown up, the corresponding points of the balloon increase. If participants stop before the bursting of the balloon, the corresponding points will be obtained. If the balloon reaches a certain level and the balloon bursts, the points of the balloon will be zero. The participants are required to get as many points as possible.
SST aims to test the inhibitory control of participants. 25 Participants need to make the corresponding choice or classification response to the target stimulus (Go condition). Once the stop signal appears on the target stimulus, the participants need to stop the response (Stop condition). The task uses the staircase model to set the stop signal delay time. The stop signal delay time of each trial is changed by 50 ms (increase or decrease) according to the time of the previous trial, so as to determine the stop signal delay time of the participants. The main evaluation is the stop signal reaction time, which refers to the mean reaction time in the Go trials minus the stop signal delay time.
DDT assesses the participants’ level of control and decision-making through their changes in choice for time and reward. The main evaluation is the discount parameter K-value. A larger K-value indicates that participants tend to receive immediate and small rewards, with higher impulsivity and poor self-control. Past research has indicated that people with substance use disorder prefer immediate and smaller rewards than larger and delayed ones in DDT. 26
Physiological indicator, namely EEG, is acquired simultaneously when participants complete cognitive tasks. We use a high-density 64-channel sintered Ag/AgCl electrode cap with BrainProducts BrainAmp ERP System (BrainCap with Asia Cut, Germany) to collect EEG data, with sampling rate of 1,000 HZ and electrode impedance kept below 5 kΩ. Two vertical electrooculography electrodes are placed above and below the left eye to detect eye movement and blinking. At the same time, a reference electrode is placed at the tip of the nose, and the ground electrode is located at the FCz. This system automatically uses the assessment module to obtain the subjective and objective evaluation results of participants’ addiction severity, psychological status, and cognitive level. The results of the assessment, including severity of addiction, psychological and emotional status, cognitive function and physiological status, and individualized treatment recommendations will be fed back to the users in a visual way (example of visualized feedback seen in attachment file 1).The feedback of treatment recommendations include eight dimensions: impulsivity, sleep, stress, motivation for treatment, coping style (including positive coping and negative coping), anxiety, depression, and compulsion, listed in three levels of recommendations.
Based on treatment recommendations, participants will receive individualized and targeted treatment. For the top four recommended dimensions listed in first-level recommendation, the participants will receive the corresponding sessions related to the dimensions twice. The corresponding sessions to other dimensions in the first-level recommendations will be given priorly but only once. Participants will only receive the sessions related to the second-level recommendation once. Sessions related to dimensions in third-level recommendations are optional and not mandatory. The details of the sessions are given in the intervention section.
The treatment of the virtual addiction rehabilitation therapist system is achieved through a semistructured human–computer interaction. The treatment system combines the full-stack end-to-end AI performance animation solution technology, intelligent three-dimensional modeling, binding technology, speech synthesis technology, and intelligent interaction technology to form a virtual psychotherapist image (demo video seen in attachment file 2). Compared with the traditional text-based self-help dialogue agent, this system creates a significant sense of interaction.
The forms of interaction include inquiries, statements, recommendations summary, and knowledge assessment. Inquiry is an interactive way that the virtual therapist advances the treatment process, guide the treatment topic, and gathers information by asking participants questions. After participants answering the questions, targeted and summative statements and recommendations summary are fed back to the participants. To ensure the therapeutic effect, knowledge assessment is to test the participants’ mastery of key points of psychotherapy through multiple-choice or true–false questions. The logic diagram of Echo app v2.0 is shown in Figure 2.

Logic diagram of the virtual agent-assisted intelligent rehabilitation treatment system.
Given the problem of cognitive impairment in people with AUD, we are also developing cognitive interventions in parallel. In this study, cognitive impairment will be assessed before and after intervention, and the treatment content of cognitive recovery will be developed synchronously for Echo app v3.0.
Study design and participants
A parallel, randomized controlled, two-arm and single-blind trial was conducted. Eligible participants need to (a) between the ages of 18 and 55; (b) meet Diagnostic and Statistical Manual of Mental Disorders (DSM)-5 criteria for AUDs; (c) withdraw alcohol for less than three months; (d) ability to use computers; and (e) sign informed consent. We exclude participants over 55 years old because we want to control the participants’ ability to use computers and their cognitive functioning well enough to receive interventions delivered by digital devices.
Participants are excluded if they (a) have a comorbidity of other neuropsychiatric diseases or mental disorders; (b) have diseases that affect cognitive function, such as history of head trauma, cerebrovascular disease, epilepsy, etc. (c) have a family history of mental illness; or (d) not familiar with computer operation, unable to complete assessment and treatment. An overview of inclusion and exclusion criteria is in Table 1.
Inclusion and exclusion criteria.

Setting and recruitment
Potential participants are recruited from people who receive addiction treatment in Fuyang No. 3 People's Hospital and Shanghai Mental Health Center. After signing informed consent, the participants who are willing to participate will receive prescreening assessments to determine whether they meet the eligibility criteria. The prescreening assessments include DSM-5 criteria for substance use disorders and general information related to the inclusion and exclusion criteria. All the assessments are conducted by qualified psychiatrists in each hospital.
Eligible participants will proceed into the next step (participants’ timeline shown in Figure 3).

Participants’ timeline.
Sample size and power calculation
We adopt the superiority by margin tests to calculate sample size. 27 Based on previous literature and preliminary data from participants with methamphetamine use disorders, we take the effective response rate of craving reduction as the primary outcome, and the effective response is defined as the cue-induced craving reduction greater than or equal to 50%. At the significance level of 0.05, with a superiority margin of 0.1, 44 subjects for the treatment group and 44 subjects for the control group are needed to achieve 90% power when the response rate for control is 0.5 and for treatment is 0.7. Considering the dropout rate of 10%, the number of participants enrolled in each group is 50. This study aims to enroll 100 participants, with 50 in each group.
Randomization and blinding
In total 100 participants are randomly assigned to each of the treatment conditions based on the 1:1 ratio. We consider virtual agent-assisted intelligent rehabilitation treatment plus TAU group as the testing group, and the TAU group as the control group. Especially, each treatment group has 50 participants. In this study, the random number table method is used to randomly assign a participant to either the testing group or the control group.
Intervention
Both groups will receive TAU, including medication, exercise, and psychological education. Virtual agent-assisted intelligent rehabilitation treatment will be delivered to participants in the testing group through the Echo app v2.0.
Treatment as usual
TAU for participants with AUD includes medication, exercise, and psychological education. Participants in both treatment groups will be prescribed medications. Medications are prescribed based on clinician recommendation if needed. Participants with alcohol withdrawal symptoms will be prescribed benzodiazepines. 28 Benzodiazepines exert their stabilizing effects through their potentiating effect on the transmission of the inhibitory neurotransmitter gamma-aminobutyric acid. After the withdrawal symptoms are controlled, the benzodiazepines are generally stopped gradually within 7–10 days. Any discontinuation and adverse reactions will be reported.
Participants attend 30 min exercise intervention (15 min Baduanjin and 15 min aerobic exercise) every day for 4 weeks. Past evidence suggests that exercise has significant improvements in depression and physical fitness of AUD participants. 29 Baduanjin is a Chinese traditional activity for preventing illness and staying healthy. 30 Participants also receive psychological education to improve their compliance to treatment. 31
Virtual agent-assisted intelligent rehabilitation treatment
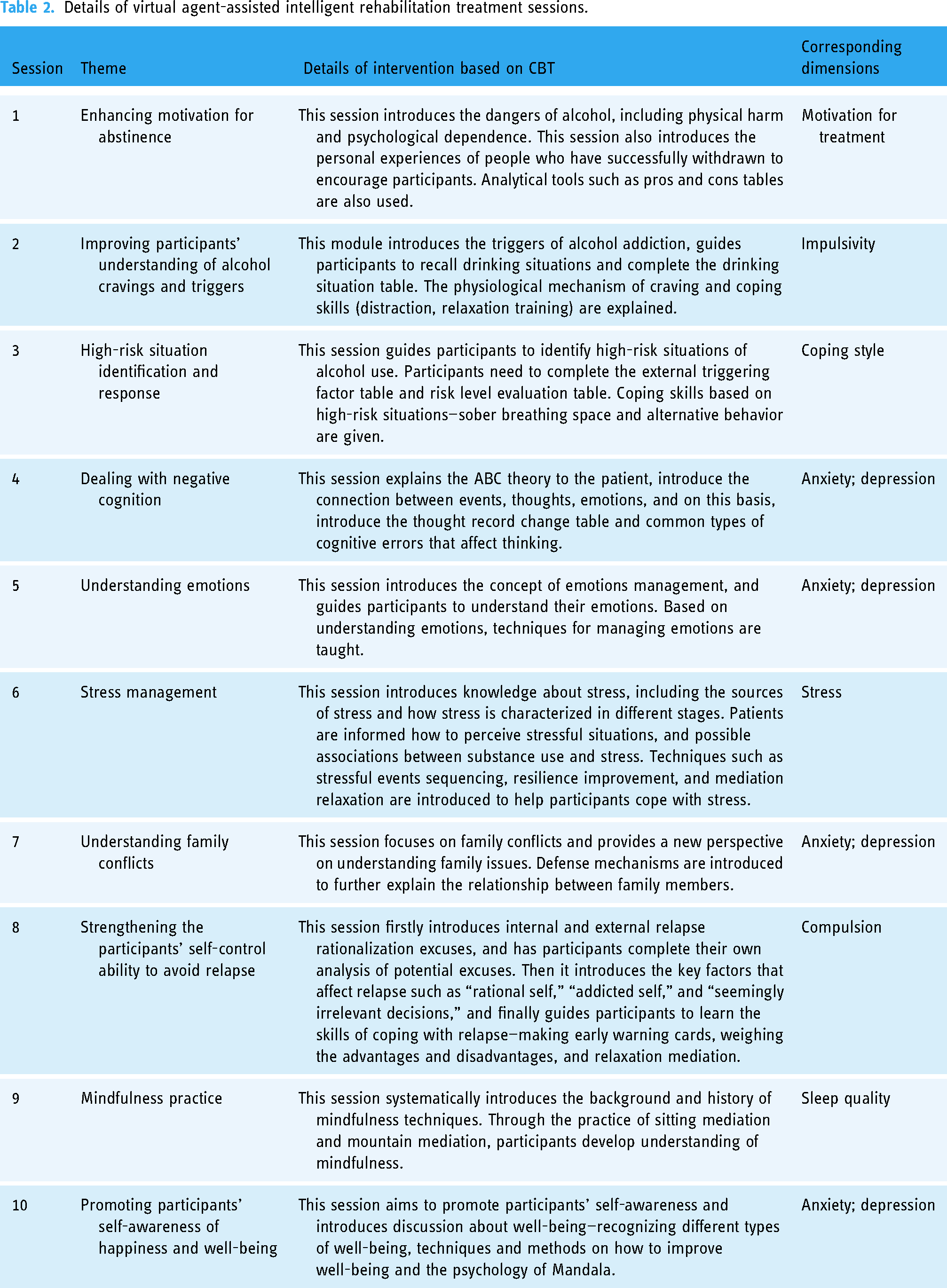
The virtual agent-assisted intelligent rehabilitation treatment consists of 10 sessions. The participants receive 2–3 sessions weekly for 4 weeks based on their treatment plan. The sessions include a variety of CBT tools, audio guidance on mindfulness for relapse prevention and related knowledge (for details see Table 2).
Details of virtual agent-assisted intelligent rehabilitation treatment sessions.
Measurement
At enrollment, participants are assessed for alcohol use and general demographic information. Our measurements for alcohol use is DSM-5 criteria for substance use disorders. At the treatment stage, both groups of participants will be then assessed at baseline, week 4, one month, and three months after the end of treatment. The primary outcome is participants’ craving for alcohol, measured by Penn Alcohol Craving Scale (PACS). The response is defined by at least a 50% score reduction in PACS at week 4 of treatment. Other measurements include the use of alcohol, motivation for treatment measured by Stages of Change Readiness and Treatment Eagerness Scale (SOCRATES), impulsiveness measured by the Barratt Impulsiveness Scale (Barratt), anxiety measured by Generalized Anxiety Disorder-7 (GAD-7), depression measured by Patient Health Questionnaire-9 (PHQ-9), stress measured by the Perceived Stress Scale (PSS), sleep quality measured by Pittsburgh Sleep Quality Index (PSQI) and coping style measured by Simplified Coping Style Questionnaire (SCSQ). PACS is the participants’ self-report about the frequency, intensity, duration of cravings, ability to resist drinking and an overall score of alcohol craving. 32 SOCRATES is a 19-item scale designed to assess participants’ readiness for change in alcohol use, including recognition, ambivalence, and taking steps subscales. 33 Barratt scale consists of 30 questions to assess whether and to what extent a person has impulse control disorders or pathological impulses. 34 GAD-7 is a seven-item self-report questionnaire to assess the level of participants’ anxiety. 35 PHQ-9 is a self-administered instrument for assessing depression severity with nine items. 36 PSS is a widely used assessment scale for overall stress perception. 37 PSQI is a self-rated instrument to screen participants’ sleep quality and disturbance over a month. 38 SCSQ consists of 20 items to assess the positive and negative coping styles. 39 The Chinese versions of the above scales have good reliability and validity.40–46 After receiving all of the treatment, participants are also invited to focus group interviews on their views of virtual agent-assisted intelligent rehabilitation treatment. The researchers will ask open-ended questions and encourage participants to fully express their ideas. The interview process lasts 1–2 h and will be recorded. The interview outline includes: (a) typical and special use scenarios of virtual agent-assisted intelligent rehabilitation treatment; (b) problems and solutions during virtual agent-assisted intelligent rehabilitation treatment; (c) evaluation of virtual agent-assisted intelligent rehabilitation treatment functions and (d) suggestions for improvement of virtual agent-assisted intelligent rehabilitation treatment (Table 3).
Assessment time schedule.

a. Week 4: after 10 treatment sessions.
b. Week 8: a month after quitting treatment.
c. Week 16: three months after quitting treatment.
Discontinuation
During the treatment and follow-up evaluation, all adverse events are recorded in the adverse events record form. Participants are informed of their right to withdraw and encouraged to report their experiences of adverse events. During the whole process of treatment and follow-up, any adverse event will be monitored and documented. Participants will be asked to withdraw from the program if (1) any serious adverse event happens ; (2) participants show low compliance with the treatment; (3) participants require to withdraw from the program; or (4) there are uncontrolled concurrent physical or psychological diseases.
Data collection
For the testing group, data will be automatically saved in the system, which will be downloaded in a protected computer. This computer is only accessible to the investigators of this study. For the control group, participants will be asked to complete the paper version of the assessment. All the data will be recorded in a specified excel document by an independent researcher. SPSS 22.0 will be used for quantitative data analysis. NVIVO 12 will be used for analyzing qualitative data from focus group interviews.
Data management and analysis
The aim of this trial is to explore whether the virtual agent-assisted intelligent rehabilitation treatment plus TAU has greater efficacy than TAU in the treatment of AUDs. We will adopt 5% significance level and calculate 95% confidence interval. The PACS score reduction from baseline to week 4 and one month after treatment is analyzed with generalized linear mixed models with fixed effects of intervention condition, time, and interaction between treatment allocation and time. Other variables including age, education level, and PACS score at baseline are taken into covariates. Restricted maximum likelihood estimation will be used for parameter estimation. We will address the missing results through a multiple imputation model. A descriptive analysis will be performed to determine whether the randomization procedure resulted in no difference between the two groups. If necessary, adjustments for multiple comparisons will be made. SPSS 22.0 is used for data analysis.
Since we conducted the superiority study, we adopted intention-to-treat (ITT) to use full analysis set for data analysis, to avoid exaggerated estimates of efficacy. In addition to including all participants with valid results in the analysis of results at the endpoint, participants assigned to randomization who for some reason discontinue the trial after a period of time or are transferred to another treatment should also be included. The missing values were made up by carrying over the most recent observation to the endpoint so that the number of participants in each group at the endpoint was the same as at the beginning of the trial. This analytical method ensured the principle of randomization.
The focus group interview will be recorded and transcribed into scripts verbatim. The scripts will be analyzed with thematic analysis based on grounded theory.
Ethics
This protocol has been approved by the Ethics Committee of Shanghai Mental Health Center (REC number: 2020–92C2). The participants will be fully informed of the purpose, procedures assessments, potential risks, and benefits of research and their consent will be obtained before their participation. We will also explain the participants’ rights to withdraw at any time during the research.
Discussion
To our knowledge, this will be the first research to examine the efficacy of virtual agent-assisted intelligent rehabilitation treatment based on a “psychological-cognitive-physiological” multidimensional assessment and rehabilitation management system. This system adopts psychological scales, cognitive tasks, and physiological indicators to provide participants with targeted and specific virtual agent-assisted intelligent rehabilitation treatment.
Past research has proven the effectiveness of multiple digital interventions, including web-based therapeutic education system, messaging, virtual reality and biofeedback. 47 Despite the vast promise, the development of digital interventions also brings the problems of high cost and workloads, causing mental health professionals’ resistance to digital interventions. In addition, a great number of digital interventions have not been validated due to high drop-out rates, no follow-up assessments and brief interventions. 48 By contrast, our study employs individualized and targeted treatment with various forms of interactions between users and the system. We also plan to implement qualitative and quantitive follow-up assessments to fully understand the participants’ experience.
In China, alcohol consumption is a public health problem. Currently, medicine as the primary treatment lacks good therapeutic effects due to poor patient compliance and drug-related adverse reactions. 31 Nonmedicinal therapies, such as psychotherapy, are both effective and acceptable. In clinical practice, participants are in great need of highly accessible, targeted and comprehensive management. However, the absence of professionals and services makes comprehensive and individualized treatment unattainable.
The results of the trial will further indicate whether the combination of TAU and virtual agent-assisted intelligent rehabilitation treatment is more effective than TAU alone. If the virtual agent-assisted intelligent rehabilitation treatment is proven effective, it will raise the possibility of the population-level use.
This study has several limitations. First, we use craving for alcohol as primary outcome because it can effectively reduce the workload of psychiatrists and improve the accuracy of evaluation. Craving has been suggested highly correlated with the disease progression of alcohol dependence. However, self-reports of the participants may not authentically reflect their craving for alcohol. Second, this study used a digital intervention to treat addiction. This implies that better access to Internet resources, computer skills and user engagement are required to implement this treatment, which has limited its application. 49 Third, participants may drop out of the study for reasons such as a lack of empathic support that they receive during traditional psychotherapy though we will fully illustrate the research to participants and address their potential concerns. 50 Thus, we adopt ITT principle to prevent the bias caused by missing data.
Conclusion
In conclusion, the aim of the present research is to examine the effectiveness of virtual agent-assisted intelligent rehabilitation treatment in reducing craving for alcohol. The results of this study will indicate the possibility of applying virtual agent-based interventions to the treatment of AUD. Provision of digital interventions will further enhance the accessibility of psychotherapy and alleviate the insufficiency of human psychotherapists.
Supplemental Material
sj-doc-1-dhj-10.1177_20552076231219432 - Supplemental material for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial
Supplemental material, sj-doc-1-dhj-10.1177_20552076231219432 for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial by Shuo Li, Dapeng Zhang, Jingyang Liu, Hang Su, Lei Guo, Qianying Wu, Jiang Du, Haifeng Jiang, Na Zhong, Chuanning Huang, Tianzhen Chen and Min Zhao in DIGITAL HEALTH
Supplemental Material
sj-png-2-dhj-10.1177_20552076231219432 - Supplemental material for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial
Supplemental material, sj-png-2-dhj-10.1177_20552076231219432 for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial by Shuo Li, Dapeng Zhang, Jingyang Liu, Hang Su, Lei Guo, Qianying Wu, Jiang Du, Haifeng Jiang, Na Zhong, Chuanning Huang, Tianzhen Chen and Min Zhao in DIGITAL HEALTH
Supplemental Material
sj-png-3-dhj-10.1177_20552076231219432 - Supplemental material for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial
Supplemental material, sj-png-3-dhj-10.1177_20552076231219432 for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial by Shuo Li, Dapeng Zhang, Jingyang Liu, Hang Su, Lei Guo, Qianying Wu, Jiang Du, Haifeng Jiang, Na Zhong, Chuanning Huang, Tianzhen Chen and Min Zhao in DIGITAL HEALTH
Supplemental Material
sj-png-4-dhj-10.1177_20552076231219432 - Supplemental material for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial
Supplemental material, sj-png-4-dhj-10.1177_20552076231219432 for The preliminary efficacy of virtual agent-assisted intelligent rehabilitation treatment (Echo app v2.0) in patients with alcohol use disorders: Study protocol for a randomized controlled trial by Shuo Li, Dapeng Zhang, Jingyang Liu, Hang Su, Lei Guo, Qianying Wu, Jiang Du, Haifeng Jiang, Na Zhong, Chuanning Huang, Tianzhen Chen and Min Zhao in DIGITAL HEALTH
Supplemental Material
Footnotes
Acknowledgements
We also thank the support from the Innovative Research Team of High-level Local Universities in Shanghai.
Authors’ contributions
MZ and TZC devised the study concept and is overseeing the conduct of the study at all sites. SL drafted the original draft. SL DPZ,JYL,HS, LG, QYW, HFJ, JD, NZ, and CNH participated in methodology, review, and editing.
Authors’ Notes
Shuo Li and Dapeng Zhang contributed equally.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The ethics committee of Shanghai Mental health center approved this study (REC number: 2020-92C2).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82130041, 82201650), Shanghai Shenkang Hospital Development Center (SHDC2020CR3045B), Shanghai Key Laboratory of Psychotic Disorders (13DZ2260500), Shanghai Rising-star Cultivation Program (22YF1439200), Capability Promotion Project for Research-oriented Doctor at SMHC (2021-YJXYS-01), Shanghai Municipal Science and Technology Major Project (2018SHZDZX05), Shanghai Engineering Research Center of Intelligent Addiction Treatment and Rehabilitation (19DZ2255200), Shanghai Clinical Research Center for Mental Health Q6 (19MC1911100), and Shanghai Science and Technology Program (21DZ2201000).
Guarantor
MZ.
Consent for publication
All authors have reviewed and approved this manuscript as being submitted.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
