Abstract
Objective
The interactions between fasting insulin levels, high blood pressure and nonalcoholic fatty liver disease (NAFLD) are still unclear. We examined the causal mechanisms between these three cardiometabolic traits using Mendelian randomization (MR) approach by utilizing genetic instruments.
Methods
Three different genome-wide association studies resources of European ancestry were utilized for the present study. Two-sample MRs were used to assess causal effects between fasting insulin levels, high blood pressure and NAFLD. Multivariate MR was used to calculate the mediating effect. The inverse variance-weighted method was used as the main analysis method.
Results
Our study confirmed a causal effect of fasting insulin levels (IVW-OR = 9.54, P = 0.001) and high blood pressure (IVW-OR = 3.926, P = 0.005) on NAFLD risk. And fasting insulin level was positively casually associated with high blood pressure risk (IVW-OR = 1.170, P < 0.001). However, the impact of high blood pressure on fasting insulin levels was still uncertain because of the presence of horizontal pleiotropy. Reverse MR showed NAFLD had a positive correlation with fasting insulin levels (IVW-OR = 1.010, P < 0.001) and a negative causal effect on high blood pressure risk (IVW-OR = 0.997, P = 0.037). Combined the multivariate MR result revealed high blood pressure partially mediated the contribution of fasting insulin level to NAFLD risk (proportion mediated: 9.091%).
Conclusions
Our study suggests there is a bidirectional causal relationship between fasting insulin levels and NAFLD. High blood pressure seems to play a mediating role in the development of NAFLD caused by changes in fasting insulin levels. However, it is uncertain whether high blood pressure is a mediator between NAFLD and the risk of fasting insulin level.
Keywords
Introduction
Nonalcoholic fatty liver disease (NAFLD) is a disease in which excessive free fatty acids enter the liver and exceed the liver’s ability to handle them leading to continuous lipid deposition and inducing liver steatosis. A meta-analysis published in 2022 suggested that the total global prevalence of NAFLD was approximately 32.4% and was continuing to increase at an alarming rate. 1 Approximately one in five patients with NAFLD can develop nonalcoholic steatohepatitis (NASH), and NASH can progress to cirrhosis, liver fibrosis and even liver cancer, and is a major cause of liver-related morbidity and mortality.1,2 Nevertheless, the current focus on NAFLD is still insufficient. A study that collected data from 102 countries around the world revealed that no country had developed a national or sub-national strategy aimed at NAFLD. 3
The correlation between NAFLD and type 2 diabetes mellitus (T2DM) is well established. Over the years, researchers have done numerous studies on the interaction between NAFLD and diabetes. Studies showed that the prevalence of NAFLD was five times higher in people with diabetes than in people without diabetes, and about two-thirds of patients with T2DM have NAFLD. 4 Another meta-analysis demonstrated that the risk of T2DM in NAFLD patients was more than twice that of non-NAFLD patients. 5 NAFLD and T2DM are often accompanied by insulin resistance (IR), and thus IR is thought to play an essential role in the progression of both diseases. In IR, effector organs require more insulin to produce normal physiological effects, which can cause an increase in fasting insulin level. Therefore, elevated fasting insulin level may promote the development of NAFLD. A meta-analysis revealed that elevated fasting insulin level was independently associated with an increased risk of high blood pressure in the general population. 6 Another research showed that high blood pressure was considered as a risk factor for NAFLD. 7 The above results indicated that high blood pressure was closely related to the fasting insulin level and NAFLD, but it is unclear whether it plays a mediating role in both.
Mendelian randomization (MR) analysis is a method that uses genetic variants as instrumental variables (IVs) to represent specific exposures in order to infer a causal relationship between exposure and outcome. It can overcome the major limitation of unmeasured confounding in observational study evidence. 8 Currently, most studies have concentrated on the relationship between fasting insulin level and NAFLD in diabetic patients. In the present study, the potential causal relationship between fasting insulin levels in people without diabetes and NAFLD was determined by a bidirectional two-sample MR. We also conducted multivariate MR analysis to test the mediating role of high blood pressure.
Methods
Study design
The present MR study was performed in two steps that are described in Figure 1(a). In the first step, we performed a bidirectional two-sample MR using genome-wide association studies (GWASs) data to assess the causal effects of fasting insulin levels 9 on NAFLD risk and NAFLD on risk of increased fasting insulin levels, respectively. In the second step, two-sample MR was applied to evaluate whether high blood pressure was a potential mediator in the causal relationship between fasting insulin levels and NAFLD, and combining the result of multivariate MR to calculate the mediating effect.
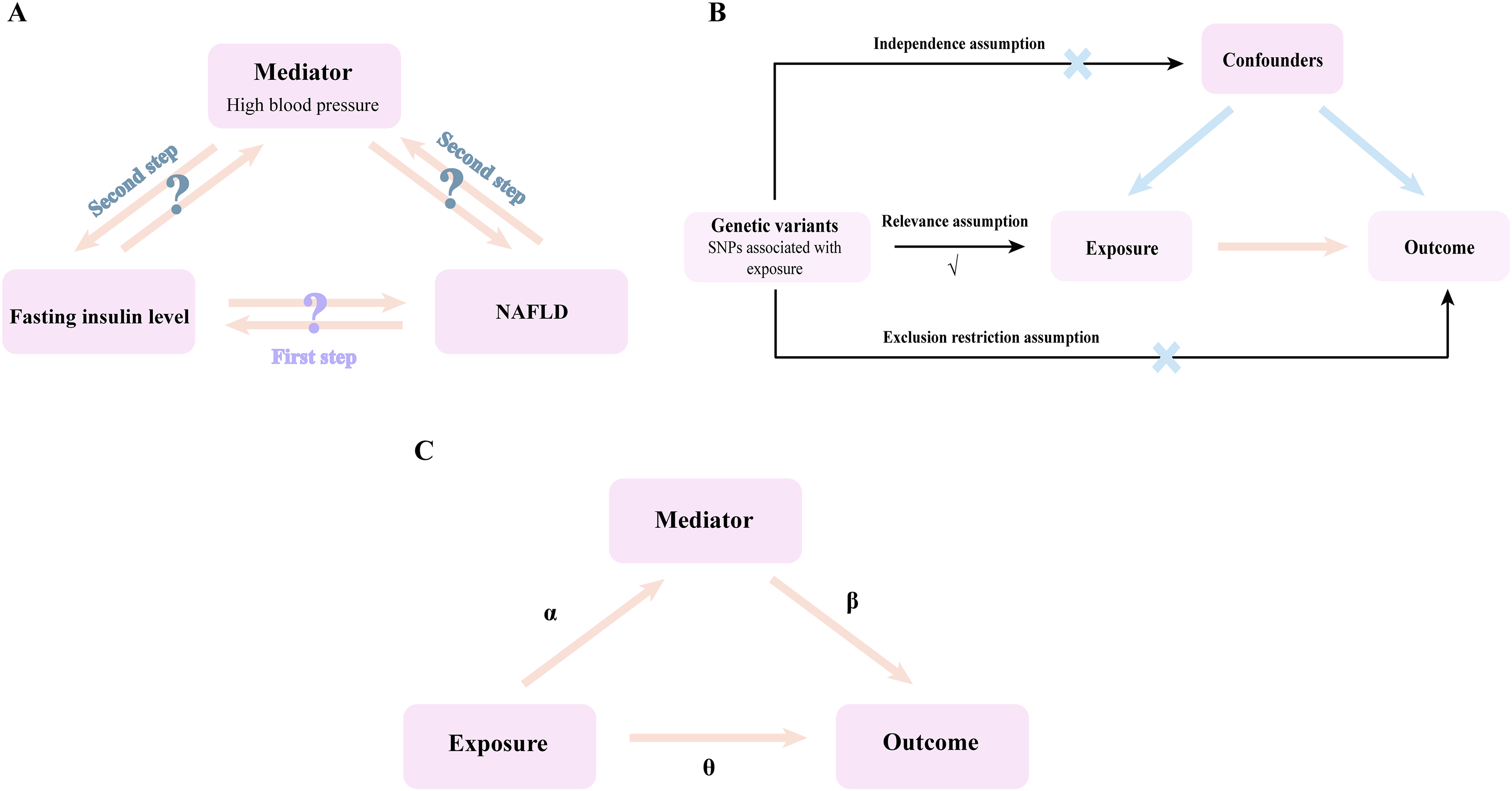
Overview of the design of this study. (a) Study design; (b) three core assumptions that MR needs to fulfill; (c) calculation method for Mediation effect and mediation proportion. NAFLD, nonalcoholic fatty liver disease.
Data sources
Different publicly available de-identified GWAS datasets were utilized for the present study that are described in Table 1. Data for fasting insulin levels was derived from a meta-analysis based on multiple cohort studies enrolling 151,013 European ancestry individuals. These individuals were from Croatia, Denmark, Finland, France, Germany, Greece, Iceland, Italy, Netherlands, Republic of Ireland, Spain, Sweden, Switzerland, UK, US, and Australia, and the data had been adjusted based on body mass index. 9 Summary data on high blood pressure included 124,227 cases and 337,653 controls, which were published in 2018. All individuals included in the study were of European ancestry. FinnGen is the first research project to bring together almost all Finnish biological libraries. The biological bank was established by universities, hospital areas, and other research institutions. NAFLD data collection began in August 2017 and ended in February 2020, including 218,792 Finns. Ethical approval and notified consent were available for all original studies cited in this study.
GWAS data sources.
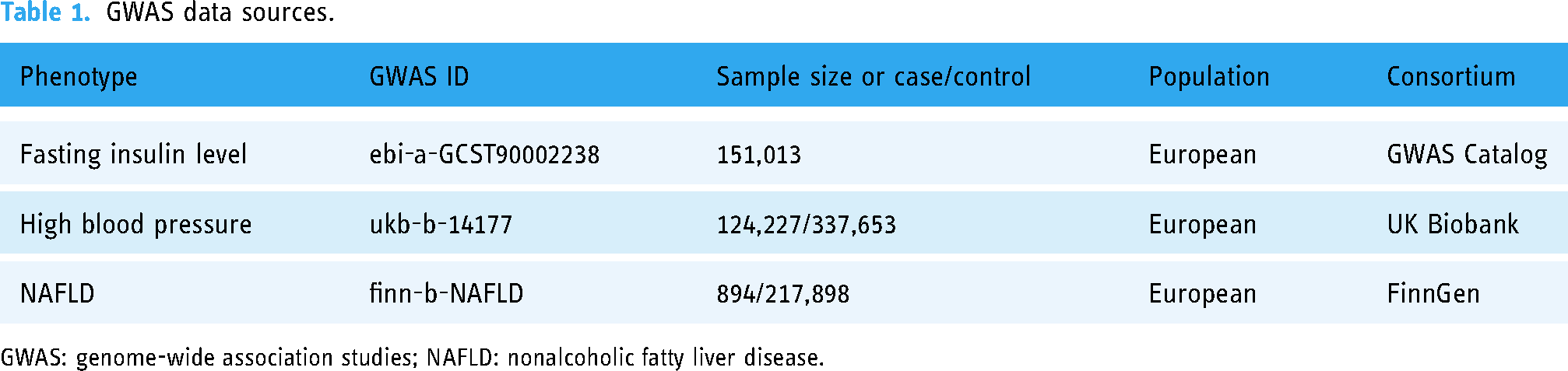
GWAS: genome-wide association studies; NAFLD: nonalcoholic fatty liver disease.
Genetic instrument selection
MR study should satisfy three core assumptions, as shown in Figure 1(b). First, single nucleotide polymorphisms (SNPs) are strongly associated with exposure (relevance assumption). Second, SNPs are not associated with confounders affecting exposure and outcome (independence assumption). Third, SNPs can only have an effect on outcome through exposure (exclusion restriction assumption).
For the relevance assumption, we selected SNPs with genome-wide significance from the corresponding GWASs by the R package “TwoSampleMR” (P < 5× 10−8 for fasting insulin level and high blood pressure). To select independent genetic variants in the 10,000 kb window, SNPs were further aggregated to a linkage disequilibrium threshold of r2 < 0.001. Then, the information of SNPs in the outcome NAFLD was extracted separately, and the data of fasting insulin levels, high blood pressure and the outcome NAFLD were combined separately, and the palindromic SNPs with intermediate allele frequencies were removed. F-statistics can assess the strength of the IVs selected in the MR analysis and test whether MR studies may be influenced by weak instrumental bias. F > 10 considered IVs to be strongly correlated with exposure. 10 F = β2exposure/SE2exposure. 11 SNPs with F < 10 will be excluded. For the exclusion restriction assumption, outlier test was conducted by the R package “MRPRESSO,” which selected SNPs that caused bias and generated data for MR analysis (Supplemental Table 1). Afterward, to reduce the risk of bias due to potential violations of independence assumption, we considered obesity as a potential confounder of the relationship between fasting insulin levels, 12 high blood pressure 13 and NAFLD, 14 as previous MR studies have shown it to be a common causal risk factor for all three. We excluded obesity-associated SNPs identified in the GWAS Catalog (https://www.ebi.ac.uk/gwas/) from the MR analysis, and all data were derived from European pedigrees, as described in Supplemental File 1.
When NAFLD was the exposure, the conditions for the SNPs selected by NAFLD were P < 5× 10−7, distance >10,000 kb and linkage disequilibrium r2 < 0.01. The results of the data obtained according to the above method of excluding SNPs were presented in Supplemental Table 1.
Statistical analyses
To further refine the exclusion restriction assumption, we conducted a series of statistical analyses. The random-effects inverse variance-weighted (IVW) method was used as the main analysis method in this study. Four sensitivity analysis methods, MR–Egger, weighted median, simple mode and weighted mode, were supplemented to assess the robustness of IVW estimates under different assumptions. 15 If the IVW result showed P < 0.05 and the results of other methods were in the same positive or negative direction as the IVW results, they were considered statistically significant in this study. 16 In addition, we used Cochran's Q test to assess the heterogeneity of the causal effect between exposure and outcome. P < 0.05 was considered the presence of heterogeneity. Directional pleiotropy was determined from the intercept term in the MR–Egger regression model. MR–Egger intercept analysis tested whether the intercept was 0 when the SNP effect was 0. A P < 0.05 indicated that the intercept was not 0 and that there was directional pleiotropy in the sense that the outcome itself existed when there was no exposure yet, at which point exposure could not be shown to be a risk factor for the outcome. The funnel plot was also used to detect directional pleiotropy, and if the SNPs were distributed in a funnel shape, it indicates no causal estimates from weaker variants tend to be skewed in one direction (Supplemental Figure 2). 17 The presence of horizontal pleiotropy was determined based on the results of MR–PRESSO global test, if P < 0.05 then it was considered that there was horizontal pleiotropy. 18 Leave-one-out analysis was conducted by eliminating SNPs one by one and then performing MR analysis to test whether the overall results of MR–Egger and IVW methods were driven by a single SNP (Supplemental File 2).
In order to calculate the mediating effect, we first used two-sample MRs to estimate the causal effects of genetically determined exposure on mediator (α) and exposure on outcome (total effect θ). Then, multivariate MR was used to assess the causal effect of mediator on outcome risk with adjustment for exposure (β). Indirect effect = αβ. The proportion of total effect of exposure on outcome mediated by mediator = indirect effect/total effect (Figure 1(c)). For multivariate MR, data were read and analyzed by R packages “TwoSampleMR.” The multivariate inverse variance weighted (MV-IVW) method was used as the dominant analysis method.
Results
A bidirectional two-sample MR proved the causal effect between fasting insulin levels and NAFLD
As displayed in Table 2, 38 SNPs were examined at fasting insulin level as genetic instruments when NAFLD was used as the outcome (Supplemental Table 1). MR–PRESSO did not detect the outlier SNP and there was no SNP related to obesity. IVW result showed that the fasting insulin level was associated with an increased risk of NAFLD (OR = 9.536, 95% CI: 2.662–34.162, P < 0.001). In addition, the robustness of the IVW result was supported by the weighted median in the sensitivity analysis (P = 0.024), but MR–Egger, simple mode and weighted mode lacked significant causally associated (P > 0.05) (Figure 2(a) and Supplemental Figure 1(a)). Cochran's Q statistic found no evidence of heterogeneity (IVW Q = 48.012, P = 0.106) and MR–Egger intercept test (P = 0.947) and the funnel plot showed no directional pleiotropy). MR–PRESSO global test revealed no horizontal pleiotropy (P = 0.106). Leave-one-out analysis indicated that causal estimates were not driven by a specific SNP (Supplemental File 2). The F statistic for each selected SNP was >10, and the average F statistic = 51.802 (ranging from 22.438 to 173.130) (Supplemental Table 1). The above results suggested that the weak instrumental bias in MR analysis was low and the selected SNPs were all strongly associated with fasting insulin level.

Two-sample MR analysis results on the interaction between fasting insulin level, high blood pressure and NAFLD. (a) Fasting insulin level on NAFLD; (b) NAFLD on fasting insulin level; (c) fasting insulin level on high blood pressure; (d) high blood pressure on fasting insulin level; (e) high blood pressure on NAFLD; (f) NAFLD on high blood pressure. MR, Mendelian randomization; NAFLD, nonalcoholic fatty liver disease.
Heterogeneity and pleiotropy assessment.

IVW: inverse variance weighted; NAFLD: nonalcoholic fatty liver disease; SNPs: single nucleotide polymorphisms.
Six SNPs were examined in NAFLD (Supplemental Table 1). No SNP was related to obesity and no outlier SNP was detected by MR–PRESSO. IVW result indicated that NAFLD was positively associated with increased risk of fasting insulin level (OR = 1.010, 95% CI: 1.005–0.016, P < 0.001). Moreover, the same results were observed in the weighted median and weighted mode methods (P < 0.05). However, MR–Egger and simple mode methods showed a lack of significant causally associated (P > 0.05) (Figure 2(b) and Supplemental Figure 1(b)). No evidence of heterogeneity (IVW Q = 2.983, P = 0.703) and directional pleiotropy (P = 0.895) was found (Table 2). The result of MR–PRESSO global test indicated no horizontal pleiotropy (P = 0.646). Leave-one-out analysis showed that none of the SNPs motivated the results of this step (Supplemental File 2). The F statistic of each selected SNP was >10, and the average F statistic = 42.006 (ranging from 25.359 to 102.495) (Supplemental Table 1), indicating that SNPs are strongly associated with NAFLD. All of the above results were compelling evidence for a causal effect between NAFLD occurrence and increased risk of fasting insulin level.
The above results demonstrated that the fasting insulin level and NAFLD were positively causally related to each other.
Two-sample MRs demonstrated high blood pressure was a potential mediator in the causal effect between fasting insulin levels and NAFLD
In order to verify whether the high blood pressure was a potential mediator between fasting insulin levels and NAFLD, we first determined the causal effect between fasting insulin levels and high blood pressure.
First, we analyzed fasting insulin level as exposure and high blood pressure as outcome using two-sample MR, and a total of 37 SNPs were examined. Since MR–PRESSO detected 15 outlier SNPs, we finally included 22 SNPs (Supplemental Table 1). There were no obesity-related SNPs in them. The IVW result confirmed that fasting insulin levels were causally associated with the risk of high blood pressure (OR = 1.170, 95% CI: 1.131–1.210, P < 0.001). In addition, the MR–Egger, weighted median, simple mode and weighted mode in sensitivity analysis observed the same results as IVW (P < 0.05) (Figure 2(c) and Supplemental Figure 1(c)). Cochran's Q statistic discovered evidence of heterogeneity (IVW Q = 33.090, P = 0.045), but MR–Egger intercept test (P = 0.958) and MR–PRESSO global test (P = 0.116) suggested that there was no pleiotropy (Table 2). Leave-one-out analysis indicated that causal estimation was not driven by a specific SNP (Supplemental File 2). The F statistic for each selected SNP was >10, and the average F statistic = 52.896 (ranging from 26.214 to 173.130) (Supplemental Table 1).
Subsequently, we used high blood pressure as exposure and fasting insulin level as outcome for analysis, and examined a total of 222 SNPs. Since MR–PRESSO detected 15 outlier SNPs and no obesity-related SNP was detected, we finally included 211 SNPs (Supplemental Table 1). IVW result confirmed that there was no causal effect between high blood pressure and fasting insulin levels (OR = 1.004, 95% CI: 0.950–1.062, P = 0.876). Also, four other sensitivity analyses supported the robustness of the IVW result (P > 0.05) (Figure 2(d) and Supplemental Figure 1(d)). Although MR–Egger intercept test showed no pleiotropy (P = 0.968), Cochran's Q statistic identified evidence of heterogeneity and MR–PRESSO indicated the presence of horizontal pleiotropy (P < 0.05). Leave-one-out analysis implied that none of the SNPs drove the results of this step (Supplemental File 2). The average F statistic = 62.488 (Supplemental Table 1), suggesting that there was no weak instrumental bias in this analysis. Although the above results indicated that high blood pressure did not affect the risk of fast insulin levels, due to the presence of horizontal pleiotropy could significantly increase the false positive rate of MR studies, the above results were not reliable and require verification with larger sample sizes in the future.
Third, we analyzed high blood pressure as exposure and NAFLD as outcome using two-sample MR. After excluding 1 outlier SNPs and 0 obesity related SNPs, 218 SNPs were examined in high blood pressure (Supplemental Table 1). The IVW result suggested that there was a positive causal effect between high blood pressure and NAFLD risk (OR = 3.926, 95% CI: 1.513–10.187, P = 0.005). Furthermore, the same result was observed in the MR–Egger method of sensitivity analysis (P = 0.029), the weighted median, simple mode and weighted mode lacked significant causally associated (P > 0.05) (Figure 2(e) and Supplemental Figure 1(e)). Cochran's Q statistic found no evidence of heterogeneity (IVW Q = 247.454, P = 0.076), MR–Egger intercept test indicated no directional pleiotropy (P = 0.207) and MR–PRESSO global test showed no horizontal pleiotropy (P = 0.085). Leave-one-out analysis indicated that causal estimates were not driven by a specific SNP (Supplemental File 2). The F statistic of each selected SNP was >10, and the average F statistic = 61.657 (ranging from 29.822 to 459.958) (Supplemental Table 1). Fourth, we used NAFLD as exposure and high blood pressure as outcome for analysis. Since no outlier SNP and obesity-related SNP was detected, we finally included five SNPs (Supplemental Table 1). IVW result confirmed that there was a negative causal effect between NAFLD and high blood pressure risk (OR = 0.997, 95% CI: 0.995–1.000, P = 0.037). Four sensitivity analyses methods lacked significant causally associated (P > 0.05) (Figure 2(f) and Supplemental Figure 1(f)). As shown in Table 2, Cochran's Q statistic identified no evidence of heterogeneity (P = 0.324), MR–Egger intercept test showed no directional pleiotropy (P = 0.989), and MR–PRESSO indicated the absence of horizontal pleiotropy (P = 0.507). Leave-one-out analysis implied that none of the SNPs drove the results of this step (Supplemental File 2). The average F statistic = 62.488 (Supplemental Table 1), suggesting that there was no weak instrumental bias in this analysis.
As shown in Figure 3, the above results proved that the fasting insulin level was positively causally associated with the risk of high blood pressure. At the same time, high blood pressure was positively causally associated with the risk of NAFLD. However, although there was a causal effect between NAFLD and high blood pressure risk, it was uncertain whether there was a causal effect between high blood pressure and the risk of fasting insulin levels due to the presence of horizontal pleiotropy. Therefore, the current results only proved that high blood pressure was a mediator in the causal effect between fasting insulin levels on NAFLD risk.

The causal effect diagram.
Multivariate MR estimated for the mediation effect of high blood pressure in the association between fasting insulin levels and NAFLD
In multivariate MR analysis, a total of 228 SNPs were examined out (Supplemental Table 1). The IVW result was shown in Table 3, After correction for high blood pressure, the causal effect of fasting insulin level on outcome NAFLD remained significant (P = 0.002). Besides, after correcting for fasting insulin level, the same result was obtained, that was, the causal effect of high blood pressure on outcome NAFLD remained significant (P = 0.011).
Multivariable MR analysis of the association between fasting insulin level /high blood pressure and NAFLD.

MV-IVW: multivariate inverse variance weighted; MR: Mendelian randomization; NAFLD: nonalcoholic fatty liver disease.
As shown in Figure 1(c), when fasting insulin level as exposure, NAFLD as outcome, and high blood pressure as the mediator, α = 0.157, β = 1.303, the indirect effect = αβ = 0.205, the total effect θ = 2.255. And high blood pressure mediated 9.091% of the total effect of fasting insulin levels on NAFLD risk. However, since our inverse MR showed a negative causal effect between NAFLD as exposure and high blood pressure as outcome and a positive causal effect between NAFLD as exposure and fasting insulin level as outcome, these suggested that we underestimated the mediating effect of high blood pressure.
Discussion
This MR study assessed the causal relationship between fasting insulin level and NAFLD in non-diabetic patients and identified the mediating role of high blood pressure in the pathway between fasting insulin level and NAFLD.
IR is associated with the pathogenesis of NAFLD and the progression of disease from steatosis to NASH. 4 Fasting insulin level is widely recognized as an indicator of IR. 19 In IR, this feedback mechanism by which insulin reduces hepatic glucose production by inhibiting glycogenolysis and limiting postprandial glucose elevation is impaired, resulting in a continued increase in hepatic glucose production. And glucose toxicity is associated with elevated glucose levels, which can further lead to IR, creating a vicious cycle. 20 In addition, Mansouri et al. 21 pointed out that high levels of insulin during IR led to sustained postprandial hormone-sensitive lipase activity, which increased lipolysis in adipocytes, leading to an increase in circulating free fatty acids (FFAs) and facilitating its uptake by the liver. Excess FFAs entered the liver and induced hepatic lipid deposition causing NAFLD. The above results explained the positive causal effect between fasting insulin level and NAFLD risk. However, FFAs accumulated in the liver can induce changes in insulin signaling pathways by activating serine kinases, which aggravated IR and created a vicious cycle. 22 In addition, adiponectin played an important role in the treatment of NAFLD and its related metabolic diseases. When adiponectin levels decreased during NAFLD, FFAs synthesis increased and FFAs oxidation decreased, leading to liver lipid accumulation and decreased insulin sensitivity, thereby inducing an increase in fasting insulin level. 23 The above may be the reason why NAFLD promoted higher risk of fasting insulin level. NAFLD, as part of the metabolic syndrome, has an intricate relationship with multiple organs of the body, and further studies are still needed to explain the interaction between NAFLD and fasting insulin level in the future.
A meta-analysis noted that elevated fasting insulin level was independently associated with an increased risk of high blood pressure in the general population, 6 which was consistent with the results of our current MR. This may be due to reduced insulin sensitivity and loss of the vasodilatory effect of insulin in the IR state, resulting in vasoconstriction and thus high blood pressure. 24 In addition, IR conditions, activation of the sympathetic nervous system, renal sodium retention, and the pro-growth effect of vascular smooth muscle cells can induce high blood pressure. 25
In the reverse MR with high blood pressure as the exposure and fasting insulin level as the outcome, we were unable to determine the causal association between high blood pressure and the risk of altered fasting insulin level because of horizontal pleiotropy. It has been investigated that elevated fasting insulin level was mainly seen in patients with first-degree high blood pressure, while the same did not occur in secondary high blood pressure. 26 There is further evidence that in high-risk patients, IR precedes high blood pressure. 27 In the future, larger sample size data is still needed to test the causal association between high blood pressure and the risk of changes in fasting insulin levels.
Our study proved a positive causal effect between high blood pressure and NAFLD risk, and the results of some clinical researches also supported our results. Observational studies have identified that approximately 50% of middle-aged and elderly patients with high blood pressure have NAFLD, 28 and that high blood pressure can promote the development of NAFLD. 29 A clinical study of children also confirmed that the prevalence of NAFLD was significantly higher in children with high blood pressure than in children without high blood pressure. 30 Although there is no conclusive evidence on how high blood pressure affects NAFLD, it may be associated with vascular endothelial dysfunction.31,32
It is worth noting that our MR suggested that NAFLD reduced the risk of high blood pressure, which contradicted clinical research results. Clinical studies have confirmed that the prevalence of arterial hypertension among patients with NAFLD ranges from 40 to 70%, and NAFLD as an independent risk factor for hypertension, was strongly associated with an increased risk of prehypertension and hypertension.33,34 In addition, our study showed that high blood pressure partially mediated the effect of fasting insulin on the development of NAFLD (proportion mediated: 9.091%). And since our study showed a positive causal relationship between NAFLD and the risk of fasting insulin level alteration and a negative causal relationship with the risk of high blood pressure, this suggested that we underestimated the mediating role of high blood pressure in the effect of fasting insulin level on the risk of NAFLD. The above results suggested that we should also pay active attention to blood pressure changes when we found changes in fasting insulin level, and reasonable and effective control of blood pressure may help to prevent the occurrence of NAFLD. In the future, we need to further evaluate the mediating role of high blood pressure in the causal effect of fasting insulin level and NAFLD with a larger sample size.The current MR study still has some limitations. First, the present study only selected data from European lineages as the study population, which lacked the representativeness of all regions worldwide. Second, all data in this study were obtained from the GWAS public database, and there may be sample overlap. However, all F statistics were >10 in this study, indicating that sample overlap had no significant effect on the conclusions we reached. Third, our study only chose one GWAS study data for fasting insulin level, high blood pressure and NAFLD, respectively, and not conducted replication analysis. Fourth, fasting insulin level and NAFLD were found to be causally associated in both the directions that need to be examined further.
In addition, NAFLD is closely related to glycemic changes. Fasting insulin level is an important indicator of glycemic changes, and there are still many other indicators reflecting glycemic characteristics that have not yet been explored in terms of their causal relationship with NAFLD, which is the direction of our future research.
Conclusions
The present study indicated a bidirectional causal relationship between fasting insulin levels and NAFLD. Fasting insulin levels can positively contribute to NAFLD risk by promoting high blood pressure. The mediating role of high blood pressure between fasting insulin levels and NAFLD suggests the potential clinical and public health implications of controlling high blood pressure in NAFLD prevention and intervention. However, it is uncertain whether high blood pressure is a mediator between NAFLD and the risk of fasting insulin level. This requires selecting GWAS with a larger sample size for MR analysis in the future.
Supplemental Material
sj-rar-1-dhj-10.1177_20552076231216682 - Supplemental material for High blood pressure mediated the effect of fasting insulin level on nonalcoholic fatty liver disease risk: A Mendelian randomization study
Supplemental material, sj-rar-1-dhj-10.1177_20552076231216682 for High blood pressure mediated the effect of fasting insulin level on nonalcoholic fatty liver disease risk: A Mendelian randomization study by Ziwen Wang, Mengpei Zhu, Yumei Huang, Jiali Cao and Zhifan Xiong in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors gratefully acknowledge all the researchers who shared research data for free and websites that collect data.
Contributorship
ZW performed data analysis and wrote the first draft. MZ, YH and JC prepared figures and tables. ZX designed the project and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No ethical review was required for this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Scientific Research Fund of Liyuan Hospital, Tongji Medical College, Huazhong University of Science and Technology [grant numbers 2023LYYYCXTD0002] and the National Key Research and Development Program of China [grant numbers 2018YFC2002000].
Guarantor
ZX.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
