Abstract
Digital biomarkers are quantifiable and objective indicators of a person's physiological function, behavioral state or treatment response, that can be captured using connected sensor technologies such as wearable devices and mobile apps. We envision that continuous and 24-h monitoring of the underlying physiological and behavioral processes through digital biomarkers can enhance early diagnostics, disease management, and self-care of cardiometabolic diseases. Cardiometabolic diseases, which include a combination of cardiovascular and metabolic disorders, represent an emerging global health threat. The prevention potential of cardiometabolic diseases is around 80%, indicating a promising role for interventions in the lifestyle and/or the environmental context. Disruption of sleep and circadian rhythms are increasingly recognized as risk factors for cardiometabolic disease. Digital biomarkers can be used to measure around the clock, that is, day and night, to quantify not only sleep patterns but also diurnal fluctuations of certain biomarkers and processes. In this way, digital biomarkers can support the delivery of optimal timed medical care. Night-time cardiometabolic patterns, such as blood pressure dipping, are predictive of cardiometabolic health outcomes. In addition, the sleep period provides an opportunity for digital cardiometabolic health monitoring with relatively low influence of artifacts, such as physical activity and eating. Digital biomarkers that utilize sleep as a window of health can be used during daily life to enable early diagnosis of cardiometabolic diseases, facilitate remote patient monitoring, and support self-management in people with cardiometabolic diseases. This review describes the influence of sleep and circadian rhythms on cardiometabolic disease and highlights the state-of-the-art sleep and circadian digital biomarkers which could be of benefit in the prevention of cardiometabolic disease.
Keywords
Introduction
Extension of the human lifespan with improved quality of life necessitates a healthcare transition from disease-focused to health-focused care with an emphasis on prevention, early diagnosis, and self-management. Novel connected sensor technology is a key enabler in this transition, especially for chronic lifestyle-related diseases such as cardiometabolic diseases (CMDs). CMDs include a combination of cardiovascular and metabolic disorders including hypertension and diabetes, which make up a large extent of the healthcare burden. Lifestyle factors, including physical activity and diet, are a large contributor to the incidence of CMDs. Notably, an estimated 80% of premature deaths from heart disease and stroke can be avoided with lifestyle changes. 1 In addition to established lifestyle factors, emerging research suggests that sleep disorders as well as circadian misalignment, contribute to the incidence of CMDs. 2
Good sleep quality is characterized by a sleep duration of around 7 to 9 h, regular sleeping patterns, and regular sleep architecture with a stable cyclical pattern without too many interruptions. 3 Many individuals do not meet an adequate quantity and/or quality of sleep.4,5 In the Netherlands, the prevalence of sleep disorders is 28.0 cases per 1000 residents in 2021. 6 Sleep duration and cardiometabolic risk are correlated, both for too short as well as for too long sleep duration. 7 The causal relations between sleep and CMDs are not fully elucidated and are complicated by various physiological, behavioral, and socio-economic aspects.
The sleep-wake cycle is the most well-known circadian rhythm. Almost all physiological processes in the body, including cardiometabolic processes, follow a circadian (∼24 h; from “circa”= approximately and “diem” = day) rhythm, which is generated by the central clock, located in the suprachiasmatic nucleus (SCN) of the hypothalamus. Rhythmic processes in peripheral organs are synchronized by the central clock, but also by external factors like the feeding-fasting cycle. An intact circadian system guarantees the necessary circadian variation in energy metabolism. For instance, the SCN, together with peripheral clocks, contributes to the circadian variation in glucose uptake and insulin release. 8 In the healthiest situation, central and peripheral clocks from the act are in synchrony. However, under circumstances that are inappropriately timed relative to the internal time, such as altered sleep-wake patterns, meal schedules, or lighting regimes, circadian misalignment can occur.9,10 Misalignment can lead to a desynchronization between the endogenous central clock and peripheral clocks. 11 Asynchrony between different tissue clocks may play a role in the etiology of CMDs resulting from a mismatch of carbohydrate and lipid metabolism between involved organs. 12
Insight into the relationship between sleep, circadian processes, and CMDs allows for new opportunities in the prevention and management of CMDs. We envision that continuous and 24-h monitoring of the underlying physiological and behavioral processes through digital biomarkers can enhance early diagnostics, disease management, and self-care of CMDs. Digital biomarkers are quantifiable and objective indicators of a person's physiological function, behavioral state or treatment response, that can be captured using connected sensor technologies such as wearable devices and mobile apps. 13 Known examples of medical digital biomarker applications include the detection of atrial fibrillation through a smartwatch, 14 diabetes management through continuous glucose monitoring, 15 and sleep staging using a wrist-worn wearable.16,17 Digital measurement is imperative in the healthcare transition facilitating high-quality care in an at-home setting at lower costs compared to standard of care. 18
Digital biomarkers have the advantage of measuring around the clock, that is, day and night, to quantify diurnal fluctuations of certain biomarkers and processes. In addition, nocturnal measurements, in particular during the sleeping period, provide a window of opportunity for digital measurement of aspects of cardiometabolic health, as the extent of motion and activity is lower during this period, leading to fewer motion-related artifacts and more accurate digital biomarkers that reflect a person's physiological function. Probably the largest benefit eventually comes from the ability of digital biomarkers to support real-time self-management and the evaluation of behavior and/or treatment effects related to a person's health. This together, allows for early interception of disease development and progression and even for health promotion.
This review first describes the role of sleep and circadian rhythm in cardiometabolic health. Then, the state-of-the-art in digital biomarkers of sleep, circadian rhythm, and CMD is reviewed and a perspective is provided on how these biomarkers can enhance the prevention and management of CMDs.
Sleep and cardiometabolic disease
Good sleep quality is essential for optimal physical and mental health. Sleep is regulated by the circadian activity of neural networks that drive the sleep-wake cycle. A healthy sleep architecture during a night consists of 4 to 5 sleep cycles, each of approximately 90 min. One sleep cycle comprises three non-rapid eye movement (NREM) stages and one rapid eye movement (REM) stage.
Sleep quantity as well as sleep quality are important for practically all physiological systems in the body, ranging from mental condition to immune state to cardiometabolic health.19,20 The relation between sleep and cardiometabolic health has recently been well-described by Baranwal et al. 4 Poor sleep quality is associated with hypertension, increased risk for arrhythmia, and increased risk for cardiovascular events (Figure 1). This is mediated through several pathophysiological mechanisms such as endothelial dysfunction, metabolic dysregulation, systemic inflammation, and increased autonomic arousal.4,21 Impaired sleep also increases the risk of diabetes and other metabolic disorders, through increased insulin resistance, unhealthy eating patterns, increased body weight, and accumulation of visceral fat. 4 Finally, sleep disruption is associated with increased levels of inflammatory cytokines that are risk factors for CMDs. 22 The most well-described and prominent relationships exist between obstructive sleep apnea (OSA) and CMDs. OSA was shown to cause systemic hypertension and possibly myocardial infection, cardiac arrhythmias including atrial fibrillation 23 and ventricular tachyarrhythmias, 24 congestive heart failure, stroke, and death.25,26 The effect of OSA on hypertension is mediated through hypoxemic stress and activation of the sympathetic nervous system. Treatment of OSA by, for example, continuous positive airway pressure (CPAP) also benefits cardiometabolic health. CPAP reduces blood pressure in patients with OSA and the risk of major adverse cardiac or cerebrovascular events in patients with established cardiovascular disease (CVD) and OSA.27,28 However, antihypertensive drugs do not influence OSA, indicating that the causal direction is from sleep disorder to hypertension. 21 Beyond hypertension, the etiology of OSA leading to heart failure is explained by greater sympathetic outflow, a hypoxia-induced increase of ventricular afterload, and by the augmented risk of myocardial infarction due to hypoxia and metabolic mismatch.21,29,30

Pathophysiological mechanisms induced by impaired sleep and involved in the development of cardiometabolic diseases. The orange line indicates the pathophysiological processes involved in the development of type 2 diabetes as a risk factor for CVD. Blue lines indicate the additional pathophysiological mechanisms involved in the development of CVD.
As sleep disruption is associated with CMDs, the question arises whether improving sleep can prevent, cure, or stop the progression of CMDs. There are indications that improving sleep quality or duration might reduce the risk of CMDs. The extension of sleep duration can lead to a reduction in blood pressure. 31 Indeed, appropriate sleep duration and sleep hygiene are recommended in the guidelines of the American Heart Association 2021 to support cardiovascular prevention. 32
Circadian rhythm and cardiometabolic disease
In addition to disturbed sleep quality as a risk factor for CMD, disturbances in circadian processes related to cardiovascular and metabolic health can contribute to the incidence of CMDs. Shift workers are at higher risk for developing CMD such as hypertension, ischemic stroke, coronary heart disease, obesity, and T2D, presumably due to disturbed physiological rhythmicity as a result of working and eating out of synchrony with the endogenous biological clock.3334–35 In the contemporary 24/7 society, many people suffer from social jetlag, which results in chronic sleep loss that is associated with an increased CMD risk.36,37
The occurrence of most types of cardiovascular disease follows a circadian pattern with an elevated risk of adverse cardiovascular events such as myocardial infarction, stroke, and ventricular arrhythmias during the morning hours. 38 Contributing factors to this morning peak in cardiovascular events are diurnal patterns in blood pressure, heart rate, and platelet aggregability. 39
There is a circadian rhythm in resting heart rate, likely coordinated by the SCN and the local cardiac clock, with an important bridging role for the autonomic nervous system. Heart rate is higher during the waking hours to facilitate the increased levels of physical activity and is lower during the night/resting hours and seems to be independent of the nocturnal fall in blood pressure.40,41 The activity of the autonomic nervous system also follows a diurnal pattern. 42 Autonomic nervous system activity is measured as heart rate variability (HRV), which reflects the variation of the cardiac beat-to-beat intervals. 43 Reduced HRV, in particular when its circadian oscillation is lost, is associated with an increased risk of cardiac arrhythmia, with sudden death in coronary artery disease, 44 and with an increased risk of having a first cardiovascular event in people without known CVD. 45
Furthermore, blood pressure shows a 24-h rhythm, with low values during the night, an increase in the early morning a stable period during the day (Figure 2). 46 A disrupted pattern in diurnal blood pressure levels is associated with a higher overall cardiovascular risk.47,48 In patients with hypertension, the absence of a nocturnal decrease in blood pressure, called non-dipping, is associated with increased target-organ damage, a higher risk for left ventricular hypertrophy leading to increased incidence of heart failure, and a wide variety of poor cardiovascular outcomes. 49 Major causal factors for non-dipping include OSA and a disrupted circadian rhythm, for example, in the case of night shift work. 50 Indeed, nocturnal dipping is a representative example of how continuous monitoring can provide valuable insight into someone's cardiometabolic risk profile.
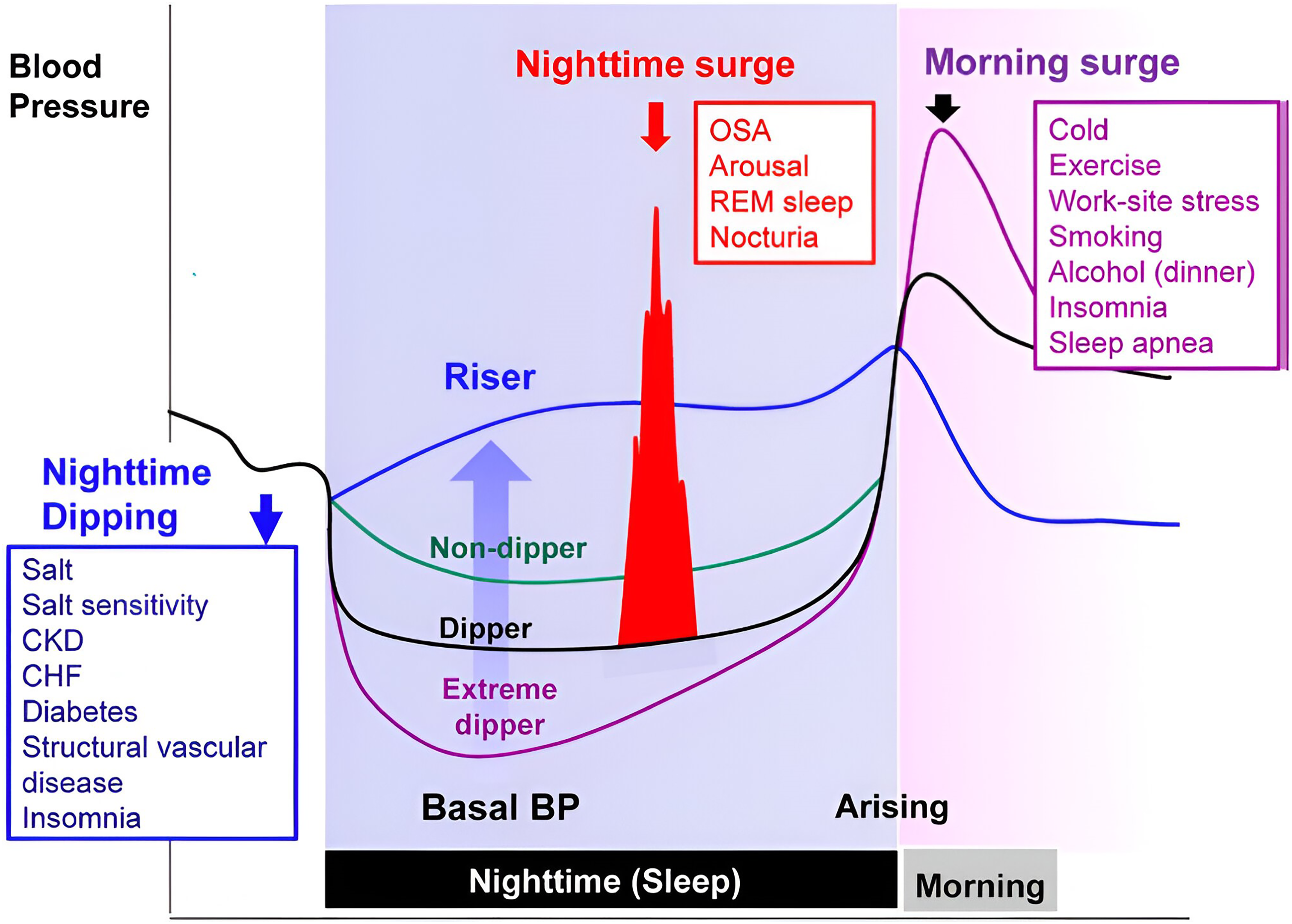
Nocturnal blood pressure dipping as a representative example of how continuous monitoring of sleep and diurnal physiological processes can provide insight to support the management and prevention of cardiometabolic disease. Taken with permission from a previous study. 50
Finally, blood glucose concentration fluctuates over the 24-h cycle. This rhythm is generated by the central brain clock through circadian control of glucose transporters, glucagon receptors, gluconeogenesis, and glucose tolerance and is independent of feeding conditions.12,51 In people with (pre)diabetes and obesity, abnormalities in the diurnal variation of glucose levels, glucose production, and glucose tolerance have been reported, together with disturbed rhythms in energy substrate oxidation, plasma free fatty acid levels, and key clock genes in muscle and fat tissue.525354–55
Continuous monitoring of sleep, circadian rhythm and cardiometabolic processes
Summarizing, both (disturbed) sleep physiology and circadian processes are reciprocally linked to CMD. Inadequate sleep, represented by short or long sleep duration, suboptimal NREM stages and abrupt changes during REM sleep all elevate the risk of hypertension and other cardiovascular diseases. This is mediated through various autonomic and circadian coordinated mechanisms like sympathetic activation, dysmetabolism, endothelial dysfunction, inflammation, and blood coagulation abnormalities. Additionally, circadian misalignment, common in shift work and social jetlag, disrupts synchrony between central and peripheral body clocks. This is associated with diurnal misalignment in blood pressure, heart rate, and glucose levels thereby raising the risk of CMD and cardiovascular events.
Several reviews have described the potential of wearable technologies to monitor these physiological patterns in relation to CMD.56,57 However, most of the recent research has focused on physical activity, heart rate monitoring, and arrhythmias. The integrated continuous monitoring of sleep, circadian rhythm, and nighttime cardiometabolic processes in a daily life environment to predict cardiometabolic outcomes has received little attention. It is therefore important that digital biomarkers that determine sleep quality and duration, circadian rhythm, and nighttime cardiometabolic physiology become available, utilizing the sleep period as a window of cardiometabolic health. The next paragraph will provide an overview of the state-of-the-art in digital biomarkers of sleep, circadian rhythm, and nocturnal digital biomarkers of CMD including heart rate, blood pressure, and glucose.
Digital biomarkers for the measurement of sleep, circadian rhythm, and cardiometabolic disease
Digital health technologies, in particular devices that utilize wearable sensor technology (wearables), can contain multiple sensor modalities, including accelerometry, photoplethysmography (PPG), and bio-impedance. They allow the monitoring of biometric and vital signs such as physical activity, heart rate, temperature, and oxygen saturation. In fact, the combination of accelerometry and PPG has significantly improved the possibilities for continuous sleep monitoring in daily life.16,17 Additionally, digital biomarkers can be used to measure around the clock, that is, day and night, they are well suited to capture diurnal fluctuations of certain biological processes and may allow for extension of the currently limited number of circadian rhythm biomarkers.
An additional advantage of digital biomarkers is the ability of physiological monitoring during nocturnal sleep. For several physiological signs, including blood pressure, heart rate, and blood glucose, it has been shown that nocturnal measurements are more sensitive for predicting health outcomes than daytime measurements. 58 Indeed, when individuals are awake physically and intellectually active, and experiencing emotional triggers underlying physiological mechanisms, such as cardiac activity, autonomic balance, and glycemic regulation are modified. Here, we describe how digital biomarkers of sleep- and circadian rhythm-related physiology can support the monitoring and prediction of cardiometabolic disease (Figure 3, Table 1).

Conceptual visualization of digital biomarkers that collect information about sleep architecture and duration, circadian rhythms, and nocturnal physiology to predict cardiometabolic outcomes, such as diabetes, coronary artery disease, and heart failure.
Representative examples of digital biomarkers for the measurement of sleep, circadian rhythm, and nocturnal physiology predicting cardiometabolic disease outcomes.
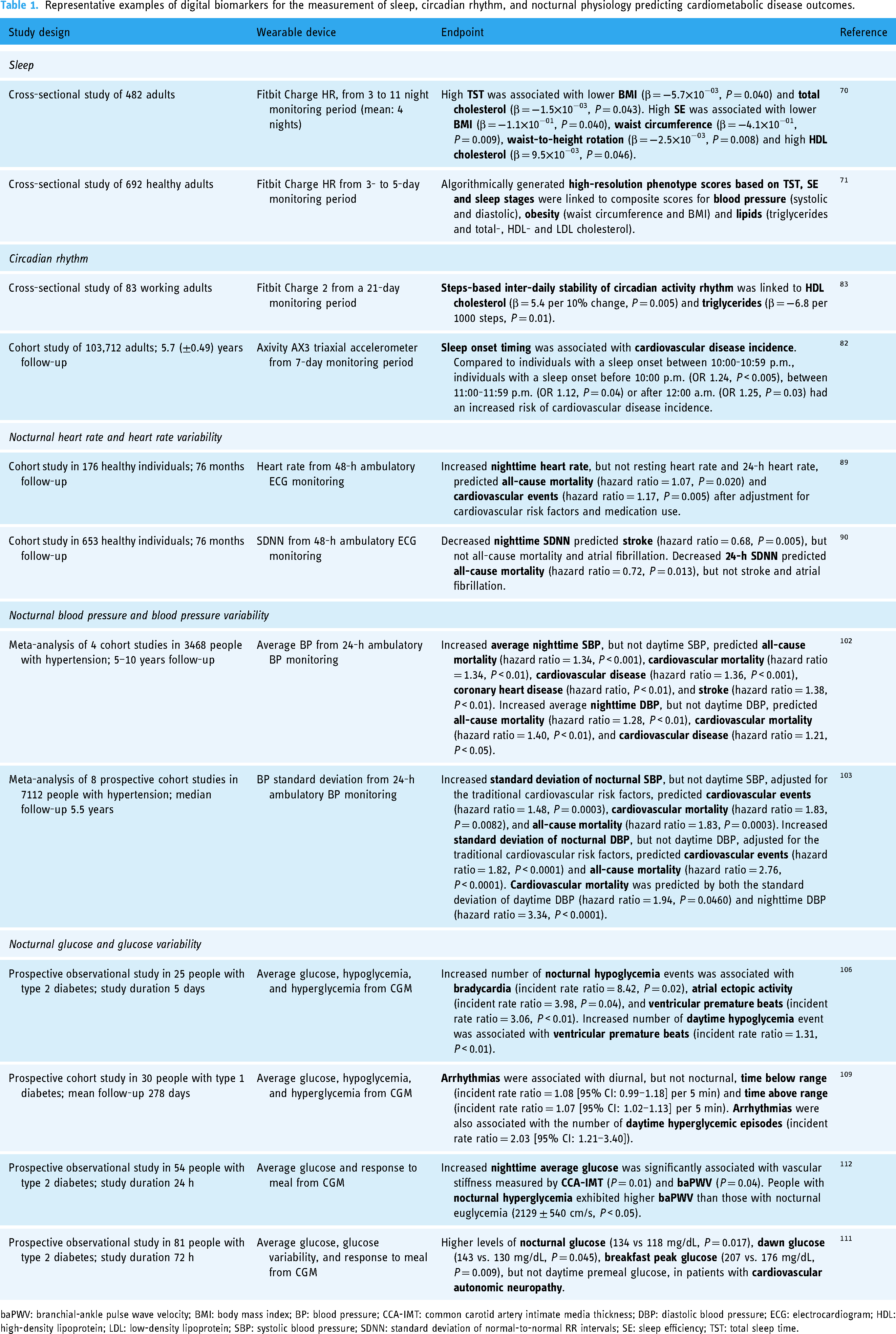
baPWV: branchial-ankle pulse wave velocity; BMI: body mass index; BP: blood pressure; CCA-IMT: common carotid artery intimate media thickness; DBP: diastolic blood pressure; ECG: electrocardiogram; HDL: high-density lipoprotein; LDL: low-density lipoprotein; SBP: systolic blood pressure; SDNN: standard deviation of normal-to-normal RR intervals; SE: sleep efficiency; TST: total sleep time.
Digital biomarkers of sleep
The gold standard for the measurement of sleep is polysomnography (PSG), which is usually performed in a sleep center or clinic. PSG is a combination of the assessment of sleep, as well as several related physiological functions. 59 To assess sleep itself, it is divided into stages that are based on patterns in brain activity (electroencephalography; EEG), eye movement (electrooculography; EOG), and muscle activity (electromyography; EMG). In addition to these three core components, cardiac monitoring is performed by the measurement of heart rate and rhythm (electrocardiography; ECG), as well as respiratory monitoring via the measurement of chest and abdominal movements (respiratory effort), oxygen saturation (SpO2), and nasal and oral airflow. Finally, limb movement and sometimes supplementary audio and video recordings can be tracked. Although PSG is considered to be highly accurate and important for diagnosing sleep disorders, the development of less intrusive alternatives that utilize wireless technology, new biomedical sensors, and a more data-analytical approach is a key challenge in sleep medicine. 60
Devices that utilize wearable sensor technology, often referred to as wearables, aim to bridge this gap, but are faced with the challenge to do so based on a less diverse set of inputs. Wearable-based sleep measurements are therefore usually “surrogate” or “proxy” indicators of sleep that attempt to provide an estimation of the time spent in each sleep stage, without using EEG, EOG, and/or EMG. A common approach to do so is to monitor for changes in the autonomous nervous system across sleep stages by measuring HRV patterns during sleep, as well as to track limb movements. Therefore, most wearables include an accelerometer that tracks wrist movement, as well as a PPG sensor that can be used to measure heart rate (HR), HRV, respiratory rate, and SpO2, and sometimes a temperature sensor. 61 A recent systematic review showed that wearables either use a subset of sensors that are used in traditional PSG or utilize alternative sensing modalities. 62 It concluded that EEG and PPG were the most commonly used sensing modalities. Although EEG-based systems tend to be more accurate (particularly for sleep stage estimation), PPG-based systems are more convenient to use and better suited for general long-term monitoring since those devices can be worn day and night and can therefore be used to collect a broader scope of digital biomarkers.
Wearable sleep trackers are often developed by commercial, consumer-facing companies that utilize proprietary algorithms that need to be independently validated. 63 The accuracy of wearables for sleep monitoring therefore is highly device-specific. Generally, PPG-based wearables can accurately differentiate sleep from wake but have trouble in identifying specific sleep stages.646566–67 Better utilization of currently available data may improve sleep stage estimation of PPG-based wearables in the near future, 68 but utilizing wearables to collect digital sleep biomarkers such as the Duration of the Sleep Episode (DSE), Total Sleep Time (TST), Waketime After Sleep Onset (WASO), Sleep Onset Latency (SOL), and Sleep Efficiency (SE) is already feasible. Despite the limitations of consumer-facing wearables, the underlying principle itself works: algorithms can be trained for the target group at hand, and be used to achieve relatively good performance.16,17
The performance of consumer-facing wearables tends to be assessed in the early part of their life cycle, as more novel iterations of the devices often follow on a year-by-year basis. As a result, the performance of most wearables is particularly assessed in relatively young and healthy subjects. 62 Currently, it is unknown to what degree the presence of CMD or taking related medication influences the accuracy of PPG-based sleep trackers. The accuracy of PPG-based wearables relies on volumetric changes in the blood flow, often in small arteries on the dorsal side of the wrist, which may be influenced by the presence of CMD and taking related medication. PPG-based wearable devices have been utilized to monitor sleep in cardiovascular patients, 69 but ideally their performance needs to be assessed in a broader range of CMD patient populations to improve generalizability for this target population.
Although the field of studies that investigate associations between wearable-based sleep measurements in CMD is still emerging, some studies show that wearable-based sleep measurements can be used to identify the risk of developing CMD (Table 1). For instance, a cross-sectional study in 482 adults who wore a Fitbit Charge HR for 3 to 11 days found that having a higher TST was associated with a lower Body Mass Index (BMI) (β = −5.7×10−03, P = 0.040) and levels of total cholesterol (β = −1.5×10−03, P = 0.043). 70 The same study showed that having a high SE was associated with a lower BMI (β = −1.1×10−01, P = 0.040), waist circumference (β = −4.1×10−01, P = 0.009), waist-to-height rotation (β = −2.5×10−03, P = 0.008), as well as higher levels of HDL cholesterol (β = 9.5×10−03, P = 0.046). Another study in 692 healthy adults who wore a Fitbit Charge HR for 3 to 5 days algorithmically generated high-resolution phenotype scores based on TST, SE, and sleep stages, which predicted composite scores for blood pressure (systolic and diastolic), obesity (waist circumference and BMI) and lipids (triglycerides and total-, HDL-, and LDL cholesterol). 71 Although this current body of knowledge particularly assesses cross-sectional associations, improving the availability and accuracy of wearable sleep trackers may contribute to more longitudinal assessments of wearable-based sleep measurements and CMD-related outcomes in the future.
Another example of digital biomarkers for diagnosing sleep disorders is the measurement of HRV during sleep for the detection of OSA, which is associated with disturbances in autonomic nervous function. The parasympathetic function was significantly decreased in patients with OSA, which was more serious in patients with severe OSA. 72 Timely recognition of OSA can help patients with CMD to reduce related symptoms, and may potentially contribute favorably to the future course of the CMD itself. In addition, PPG measurements may diagnose decreased respiratory rate and hypoxia that can result from OSA, highlighting the multi-parameter potential of wearable technology in the diagnosis of disease.
Digital biomarkers of circadian rhythm
The measurement of circadian rhythms in humans is complex. Rhythms in core body temperature, cortisol, and melatonin have been used as markers for the circadian phase of the central clock (SCN), but they require highly controlled circumstances. 73 The gold standard for circadian phase determination in humans is the assessment of dim light melatonin onset (DLMO). During dim light, melatonin concentrations increase beyond a certain threshold, typically happening in the evening or early night. 74 Traditionally, the determination of this threshold requires the collection of several salivary samples 5–6 h before the usual bedtime of an individual. As light suppresses the secretion of melatonin, sampling must be performed under controlled dim light conditions. This makes DLMO determination complicated and complex. 75 Over the past few years, progress has been made to determine a person's internal time from blood samples. This technique, which is based on a small set of blood-based gene transcripts, requires only one blood sample taken at any time during the day instead of multiple saliva samples and no need for a dim-light environment during sampling and its accuracy is similar to that of the DLMO.76,77
Yet, despite the advantages of blood sampling versus salivary DLMO, it is still a complicated procedure that is inconvenient for larger-scale applications in clinical or consumer-facing settings. Moreover, whether predicting DLMO using one blood sample works similarly well in patients or cohorts with known lower circadian amplitudes and/or in the presence of internal circadian misalignment (e.g. in the elderly or in shift workers) remains to be elucidated. 77 Furthermore, estimation of DLMO is done by questionnaires (Morningness-Eveningness Questionnaire (MEQ) and Munich ChronoType Questionnaire (MCTQ)), yet these questionnaires are not accurate enough to base timed treatments on. 78 Accurate assessment of the SCN phase (i.e. internal time) is important for, for example, effective timing of chrono-therapy, sleep-wake therapeutics, light treatment, and melatonin administration. 79 Exploration of continuous digital biomarkers of circadian rhythm could therefore be helpful.
Actigraphy can be considered a digital biomarker for circadian rhythmicity. Actigraphy can be used to detect the rhythms in rest-activity patterns that are reflective of sleep-wake patterns in relatively undisturbed sleep conditions. Results of actigraphy recordings correlate well with currently used markers for the circadian phase of the SCN, like the rhythms of melatonin and core body temperature. 80 When individuals try to sleep in an unfavorable circadian phase, these recordings show disturbed sleep patterns; hence, they could support the diagnosis of delayed or advanced sleep disturbances in people doing shiftwork or having jet lags. However, rest-activity rhythms, measured by wearables, are susceptible to masking effects like social obligations and may not neccesarily show the endogenous rhythm of the SCN. 81
A few studies have already utilized wearable sensors in order to link circadian rhythm-related measurements to the risk of CMD (Table 1). A recent study utilized data from a large cohort of 103,712 adults who wore an accelerometer (Axivity AX3 triaxial) for 7 days at the baseline and were subsequently monitored for CVD incidence for 5.7 (±0.49) years. 82 The study showed that compared to individuals with a sleep onset between 10:00 and 10:59 p.m., individuals with a sleep onset before 10:00 p.m. (OR 1.24, P < 0.005), between 11:00 and 11:59 p.m. (OR 1.12, P = 0.04), or after 12:00 a.m. (OR 1.25, P = 0.03) had an increased risk of CVD incidence. A smaller study in 83 working adults who wore a Fitbit Charge 2 for 21 days also showed that steps-based inter-daily stability of circadian activity was positively associated with HDL cholesterol (β = 5.4 per 10% change, P = 0.005) and triglycerides (β = −6.8 per 1000 steps, P = 0.01), indicating that being physically active at similar times on a daily basis may positively contribute to lowering the risk of developing CMD. 83
Digital biomarkers of nocturnal heart rate and heart rate variability
Both resting HR and HRV are important predictive physiological parameters in CMD. In epidemiological studies, higher resting HR independently predicts all-cause mortality and cardiovascular events as a reflection of autonomic imbalance and decreased HRV acts as a prognostic factor for CMD.84,85 Resting HR and HRV have associations with other cardiometabolic processes and disorders, such as vascular stiffness, hypertension, and diabetes.85,86 Resting HR and HRV are correlated and similarly influenced by acute physiological stressors (e.g. physical activity, alcohol intake, menstruation, and sickness), although resting HRV tends to be a more sensitive biomarker for these outcomes. 87 It has been shown that nocturnal HR and HRV have greater prognostic value than daytime or 24-h HR and HRV, most likely explained by the fact that there is less interference from external artifacts, such as physical activity (Table 1).88,89 After adjustment for conventional cardiovascular risk factors and medication use, increased nocturnal HR predicted all-cause mortality (hazard ratio = 1.17, P = 0.007) and cardiovascular events (hazard ratio = 1.17, P = 0.026) over 76 months in a population with no apparent cardiovascular disease. 89 Also, decreased nighttime HRV (hazard ratio = 0.68, P = 0.005), but not 24-h HRV (hazard ratio = 0.87, P = 0.514), after correction for cardiovascular risk factors, predicted stroke in an apparently healthy cohort with a follow-up of 76 months. 90 Multiple other studies have shown the prognostic value of decreased nocturnal HRV and elevated nocturnal HR for cerebrovascular disease.90919293–94 Interestingly, nocturnal HRV (P = 0.002) and not daytime HRV (P = 0.097) appears to be an important mediating factor in the association between OSA and cerebral small vessel disease. 88 Other studies have focused on the associations between nocturnal HR and HRV and pathophysiological processes that drive CMD, such as glycemic control 95 and inflammation, 96 indicating that nocturnal HR and HRV could act as early indicators of disease progression.
Most of the studies currently utilize clinical instruments to collect nocturnal HR and HRV, limiting their applicability as digital biomarkers in everyday life. Yet, accurate remote monitoring of HR and HRV by ECG and PPG is rapidly entering healthcare allowing for improved at-home monitoring and early detection of CMD utilizing nocturnal HR and HRV.
Digital biomarkers of nocturnal blood pressure and blood pressure variability
Traditionally, blood pressure (BP) is measured via a cuff, but recent advances in cuffless BP measurement methods now also utilize techniques such as Pulse Transit Time (PTT), Pulse Wave Analysis (PWA), or PPG. 97 Additionally, BP variability has gained attention over the last years and is increasingly seen as a valuable prognostic and predictive biomarker for cardiovascular and mortality outcomes.98,99 While this often refers to long-term BP variability, determined based on BP data from sequential clinical visits over months to years, ambulatory BP monitoring allows for a more granular view based on mid-term and short-term BP variability that is collected over a period of hours to weeks. 100
Since the introduction of ambulatory BP monitoring, nocturnal BP monitoring has been clinically accepted and incorporated in the European Society of Hypertension guidelines as a useful practice, especially for identifying and assessing nocturnal hypertension and non-dipping.50,101 Various studies have shown that nocturnal BP management beyond daytime blood pressure, is important to prevent cardiovascular events, as well as organ damage such as chronic kidney disease. 50 For example, a meta-analysis of four prospective cohort studies on a hypertensive population found that average nighttime BP was a better predictor of mortality in hypertensive people than daytime blood pressure (Table 1). 102 Nighttime SBP, but not daytime SBP, predicted cardiovascular death (hazard ratio = 1.34, P < 0.01), cardiovascular disease (hazard ratio = 1.36, P < 0.001), coronary heart disease (hazard ratio, P < 0.01), and stroke (hazard ratio = 1.38, P < 0.01). Also, nighttime BP variability demonstrated greater prognostic value than daytime BP variability (Table 1). For example, the Ambulatory Blood Pressure International Study, a meta-analysis of eight prospective cohort studies, showed that nocturnal BP variability was associated with higher cardiovascular and all-cause mortality, which was not the case for daytime BP variability. 103 An increased standard deviation of nocturnal SBP was associated with a higher number of cardiovascular events (hazard ratio = 1.48, P = 0.0003), higher cardiovascular mortality (hazard ratio = 1.83, P = 0.0082), and all-cause mortality (hazard ratio = 1.83, P = 0.0003). Current advancements in cuffless BP monitoring technologies will further facilitate the establishment of digital biomarkers of nocturnal blood pressure to improve prognosis, prediction, and possibly even diagnosis of cardiometabolic outcomes.
Digital biomarkers of nocturnal glucose levels and glucose variability
The measurement of nocturnal blood glucose and its variability has become possible using continuous glucose monitoring (CGM), adding great value to diabetes management. Nocturnal hypoglycemia is less well detected than daytime hypoglycemia, because people are asleep, posing a serious risk for “dead-in-bed” syndrome. CGM allows for the timely detection of nocturnal hypoglycemia which is associated with a decreased risk of major adverse cardiovascular outcomes, such as non-fatal stroke and cardiovascular death. 104 Also, hypoglycemia was associated with arrhythmic risk, both in people with and without diabetes, possibly explained through the fact that hypoglycemia causes QT prolongation (Table 1).105106107–108 Severe nighttime hypoglycemia, based on laboratory glucose measurements, predicted QT prolongation in people with diabetes (odds ratio = 2.17, P = 0.01) and people without diabetes (odds ratio = 4.07, P = 0.008) presenting at the emergency department. 108 Studies utilizing CGM have been less conclusive. Nocturnal hypoglycemia in type 2 diabetes appeared to have a stronger association with several arrhythmias than daytime hypoglycemia, including bradycardia (incident rate ratio = 8.42, P = 0.02), atrial ectopic activity (incident rate ratio = 3.98, P = 0.04), and ventricular premature beats (incident rate ratio = 3.06, P < 0.01). 106 Alternatively, others found that, in people with type 1 diabetes, diurnal, but not nocturnal, time spent in hypoglycemia and hyperglycemia was associated with arrhythmia risk (incident rate ratio = 1.08 [95% CI: 0.99–1.18] and incident rate ratio = 1.07 [95% CI: 1.02–1.13] per 5 min, respectively. 109 The number of hyperglycemic episodes during daytime was also significant (incident rate ratio = 2.03 [95% CI: 1.21–3.40]). 109 Studies with larger sample sizes are required to further verify the utility of nocturnal glucose levels in predicting cardiometabolic outcomes. Nevertheless, monitoring nocturnal glucose levels under real-world conditions is crucial to timely detect nighttime hypoglycemia and its associated poor sleep quality. 110
In addition, nocturnal hyperglycemia is associated with micro- and macrovascular complications (Table 1). Type 2 diabetes patients with cardiovascular autonomic neuropathy presented with higher average nocturnal glucose levels than those without (134 vs 118 mg/dL, P = 0.017), which was not observed for average glucose levels during daytime. 111 Furthermore, average nighttime glucose, especially nighttime hyperglycemia (glucose ≥ 161 mg/dL), was significantly associated with common carotid artery intima-media thickness and branchial ankle pulse wave velocity. 112 Altogether, since the introduction of CGM technology, nocturnal monitoring of glucose has become possible, providing further insight into disease progression and management, utilizing sleep as a window for cardiometabolic health.
Future outlook
This review outlined the pathophysiological interaction of poor sleep, circadian misalignment, and CMDs and how they can be monitored using (nocturnal) digital biomarkers to aid the management and prevention of CMDs. These digital biomarkers, especially when monitored simultaneously and continuously, can facilitate remote patient management and early identification of cardiometabolic risk. The monitoring of 24 h rhythms of these digital biomarkers delivers relevant information especially in nocturnal conditions, offering sleep as a window of health monitoring. At the same time, the monitoring of 24 h rhythms of digital biomarkers offers the opportunity for users to evaluate the impact of behavior or treatment on digital biomarker outcomes offering an opportunity for self-management. Additionally, digital biomarkers could drive personalized sleep and cardiovascular medicine, for example guiding digital cognitive behavioral therapy, 113 light therapy, and other chronotherapeutic approaches. 114 Identifying disruptions helps clinicians to target high-risk periods, such as morning peaks in cardiovascular events, improving prevention and treatment strategies in prescribing, for example, anti-hypertensives.115,116 Ultimately, feedback on sleep quality is envisioned to help individuals improve their sleeping habits or seek professional help timely to identify the underlying causes of poor sleep. Current evidence and adoption of digital biomarker-driven management of sleep and circadian rhythm in CMD is limited. The adoption of digital health technologies in healthcare is a challenging task related to multiple factors, including thorough clinical evidence generation, regulatory compliance, and user engagement. Overall, current evidence is often based on retrospective cohorts, or relatively small pilot studies. Sufficiently powered prospective studies are required to ensure clinical validation of digital biomarkers that exploit sleep as a window for cardiometabolic health. Additionally, as a general point of attention, digital biomarkers must be carefully evaluated for bias and equity, as the accuracy or the accessibility of wearable sensors may be suboptimal for some groups. 117 For example, PPG-based sensors may perform differently depending on skin color 118 and access to wearable sensors is greater in those with digital literacy and socioeconomic resources. 117 Nevertheless, the field of digital medicine is rapidly expanding and frameworks are now proposed to ensure its evidence-based and meaningful development involves all relevant stakeholders.119,120
Conclusion
Disturbed sleep and circadian misalignment represent the next frontier of controllable risk factors of CMD. This narrative review described for the first time how inadequate sleep and circadian misalignment are linked to various CMDs, including cardiovascular disease and type 2 diabetes. It is envisioned that the potential of connected sensor technologies can be unlocked to let digital biomarkers enhance remote and preventative healthcare and improve sleep, circadian rhythms, and cardiometabolic health for many.
Footnotes
Contributorship
WJB, JEO, and SW conceived the manuscript; WJB, JEO, DS, and HJV wrote the manuscript; and all authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The work was funded by an internal TNO Early Research Program Digital Health Measurements.
Guarantor
WB.
