Abstract
Objective
Lifestyle interventions are increasingly becoming an integrated part of nonalcoholic fatty liver disease (NAFLD) management. Electronic lifestyle interventions may be able to expand the access and utility of this approach. This study aimed to synthesize the evidence for the effects of electronic-based lifestyle interventions on weight, anthropometric, and liver enzyme measurements in patients with NAFLD.
Methods
Medline, Scopus, and Web of Science were searched up to February 2023. Clinical trials investigating the effects of electronic lifestyle interventions on weight, body mass index (BMI), waist circumference (WC), and liver enzymes in NAFLD patients were reviewed. After reviewing full-text articles, seven clinical trials were included in the systematic review.
Results
Two articles included telephone calls, one was based on text messaging, two studies were based on web-based lifestyle modifications, and two used mobile apps. Except for one, all other six studies indicated a significant impact on weight loss. BMI was reported in six of seven studies. Except for one, BMI was significantly reduced in the group receiving e-health. WC was reported in four studies, which indicated a significant reduction in the e-health intervention group. Alanine transaminase (ALT) was reported in all the included studies. Except for two, others demonstrated a significant improvement in ALT in the e-health intervention groups. As reported in four studies, Aspartate transaminase (AST) significantly decreased in the group receiving e-health interventions, except in one study.
Conclusions
The results support applying electronic lifestyle interventions in NAFLD patients to reduce weight, BMI, WC, AST, and ALT.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is one of the most common causes of chronic liver disease worldwide. NAFLD is usually characterized by hepatic steatosis in the presence of no other cause of secondary hepatic fat accumulation (e.g. excessive alcohol consumption).1,2 In NAFLD, hepatic steatosis is common without evidence of inflammation. Nonalcoholic steatohepatitis (NASH) is more severe than NAFLD. In NASH, hepatic steatosis is associated with lobular inflammation and apoptosis, which can lead to fibrosis and cirrhosis.2–4 In a meta-analysis study by Riazi et al. 5 in 2022, the overall prevalence of NAFLD worldwide was estimated to be 32.4%. Statistics also show that the prevalence of NAFLD has increased from 25.5% in or before 2005 to 37.8% in 2016 or later.
The underlying mechanism of NAFLD development is not fully understood. It has been suggested that the development of NASH is a two-step process: first, the deposition of fat in the liver and, second, the oxidation of fatty acids. 6 Scientists believe that insulin resistance (IR) is the primary pathophysiological mechanism 7 that is directly associated with obesity, especially abdominal obesity. 8 Among the risk factors of NAFLD, the most important are obesity, low physical activity (PA), and type 2 diabetes. 9
Although scientists are constantly in search of innovative treatment methods in the field of liver diseases, such as gene therapy 10 or novel liver-specific ligands, 11 still, the first-line treatment for NAFLD is lifestyle interventions. 12 In most interventions, weight loss between 7% and 10% can improve fibrosis, steatosis, and inflammation of liver cells. While exercise can improve hepatic steatosis and metabolic indices even without weight loss, combination interventions seem to be the most effective strategy for improving NAFLD.13–15 Current guidelines for the management of NAFLD recommend 150 minutes per week of moderate exercise16,17 and reducing caloric intake between 750 and 1000 kcal per day or 30% of normal intake, to improve IR, hepatic steatosis, and fasting glucose and reduce free fatty acids.18,19 The combination of both interventions can cause improvements in liver histology and liver enzymes and is associated with weight loss of 4.2% to 10.6%.14,20
Lifestyle interventions are an integrated approach to the therapeutic management of NAFLD.6,18 However, the success of such interventions, such as weight loss, depends on the intensity of nutrition counseling and the frequency of referrals to nutritionists and exercise therapists. 21 This treatment method is associated with limitations such as intensive and costly resources and a high wastage rate due to accessibility and scalability. 22 Electronic lifestyle interventions, providing healthy lifestyle counseling, education, and support through the Internet, websites, mobile applications, short message services (SMS), or computers, have recently become increasingly popular for weight loss.23,24 In a study that aimed to evaluate the effect of electronic lifestyle intervention on 108 NAFLD patients for 6 months, 55 patients in the intervention group showed a significant reduction in weight, waist circumference, blood pressure, and liver enzymes level. 25 By reducing barriers associated with frequent nutrition and exercise therapy sessions, mobile apps and other electronic lifestyle interventions can potentially expand the access and utility of lifestyle interventions.
To our knowledge, there has been no systematic review investigating the efficacy of e-health interventions on weight loss, anthropometric, and liver enzyme measurements in NAFLD patients; this systematic review aimed to investigate the effects of electronic lifestyle interventions in NAFLD patients.
Methods
This systematic review was consistent with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement 26 and was not registered in PROSPERO. The PRISMA checklist is provided as Supplementary file 1.
Search strategy
Systematic searches of clinical trials published until 22 February 2023 were independently conducted by two authors (NS, SS) on Medline (via PubMed, National Library of Medicine), Scopus, and Web of Science. The search strategy was designed in accordance with the database orientations using Boolean operators (AND, OR), parenthesis, quotation marks, and asterisks. The search strategy used in Medline is presented in Supplementary file 2.
Furthermore, a manual reference check was conducted on the identified articles to find further relevant studies.
Screening and eligibility of records
The population, intervention, comparison, outcome, and study design (PICOS) strategy was used to identify inclusion criteria (Table 1). The title and abstract of all identified articles were independently screened by two authors (NS, HB). Clinical trials that assessed the effect of electronic lifestyle interventions on NAFLD were selected. The full text of selected articles was read and assessed regarding compliance with established eligibility criteria. Cohen's kappa statistics was used to measure the author’s agreement on the inclusion of studies using IBM SPSS Statistics 25. Discrepancies were resolved by discussion with a third researcher (MGM).
PICOS criteria for inclusion and exclusion of the studies.

BMI: body mass index; LFTs: liver function tests; NAFLD: nonalcoholic fatty liver disease.
Data extraction and synthesis
The following variables were considered in data extraction: title, authors, year, country, study aim, population features (body mass index (BMI), age, number of participants, experimental design, intervention (type of intervention, frequency, and duration), the interventionist, follow-ups, and main results.
Risk of bias assessment
The five-point JADAD score was used independently by two authors (NS, SS) to assess the quality of included studies. Discrepancies were resolved by discussion with a third researcher (MGM). The five domains of the JADAD score included being randomized, appropriately describing of randomization, being double-blind, appropriately describing blinding, and explanation of withdrawal and dropouts. The quality of non-randomized clinical trials was assessed using the methodological items for non-randomized studies (MINORS) score. It includes 12 items, four of which are only considered in the comparative studies. The items are scored 0 (not reported), 1 (reported but inadequate), or 2 (reported and adequate). The maximum score is 16 for non-comparative and 24 for comparative studies.
Outcomes
The clinical outcomes of interest were weight, BMI, WC, and liver function tests. The mean differences for each outcome were used for the presentation of the results.
Analysis
As intervention methods and outcome measures were heterogeneous, meta-analysis was not considered in this study, and we used a narrative approach to summarize findings.
Results
A total of 1056 articles were found from the three databases (Figure 1). After removing duplicate studies, 602 articles remained for screening of the titles and abstracts, and 578 articles were excluded. Then, 24 probably related articles remained for full-text review. Then, we performed a snowball search to find additional articles. After reviewing full-text articles, we found that two studies have overlaps in the study population27,28: one reporting the results per protocol and the other intention to treat analysis. As we aimed to assess the effect of an electronic health intervention on liver enzymes, besides weight and anthropometric measurements, we selected Huber et al.’s 27 study. Another two studies also had overlaps in the study population.29,30 As the study by Mazzotti et al. included a larger sample size and reported liver enzymes and waist circumference besides weight changes, we selected this study. Finally, seven studies were included in the systematic review. According to Cohen's kappa statistics (Ƙ = 0.903, % of agreement =95.8 %), the two reviewers (NS and HB) have “almost perfect agreement” to include studies. 31 As presented in Table 2, of the included studies, four were randomized controlled trials (RCTs), two were single-arm pre-post trials, and the other was a comparative non-randomized clinical trial. Studies were published during 2016–2022 and conducted in Iran, China, the USA, Italy, Singapore, and Germany. NAFLD was confirmed in all patients using imaging or biopsy.

The PRISMA flow diagram.
Studies evaluating the effect of e-health lifestyle interventions on anthropometric and liver enzyme measurements in NAFLD patients.

I: intervention group; C: control group; NAFLD: nonalcoholic fatty liver disease; BMI: body mass index; RCT: randomized controlled trial; PA: physical activity; N: number; WB: web-based; GB: group-based.
*JADAD score.
MINORS score.
MINORS score considering the comparative group.
Two articles included telephone calls, 32 one was based on text messaging, 33 two studies were based on web-based lifestyle modifications,27,29 and two used a mobile app. 25 As there was a limited number of articles, we described all with details as follows:
In Javanmardifard et al.’s 32 study, both intervention and control groups received dietary advice from a nutritionist, orally and in written form. Patients were also requested to perform moderate PA for 30 minutes/day for 4–5 days a week to increase heart and respiratory rates. They also received a training pamphlet. Telephone calls in the intervention group (n = 30) continued for 12 weeks; two calls/per week in the first month and once a week for the rest. Each call was 15–20 minutes, focusing on adherence to diet and PA advice, and if a patient did not adhere, finding the reason and providing a solution. During the study, the control group (n = 30) only received routine follow-up visits by a specialist. As presented in Table 3, they found that, in the intervention group, weight and BMI significantly decreased after 12 weeks, although in the control group, changes were non-significant. Regarding liver enzymes, ALT and AST significantly fell in the intervention group (p < 0.001), while they had a non-significant increase in the control group (p = 0.7). The mean change of ALT and AST during the study was significantly different in the two groups.
Reported outcomes in studies evaluating the effect of e-health lifestyle interventions on anthropometric and liver enzymes measurements in NAFLD patients.
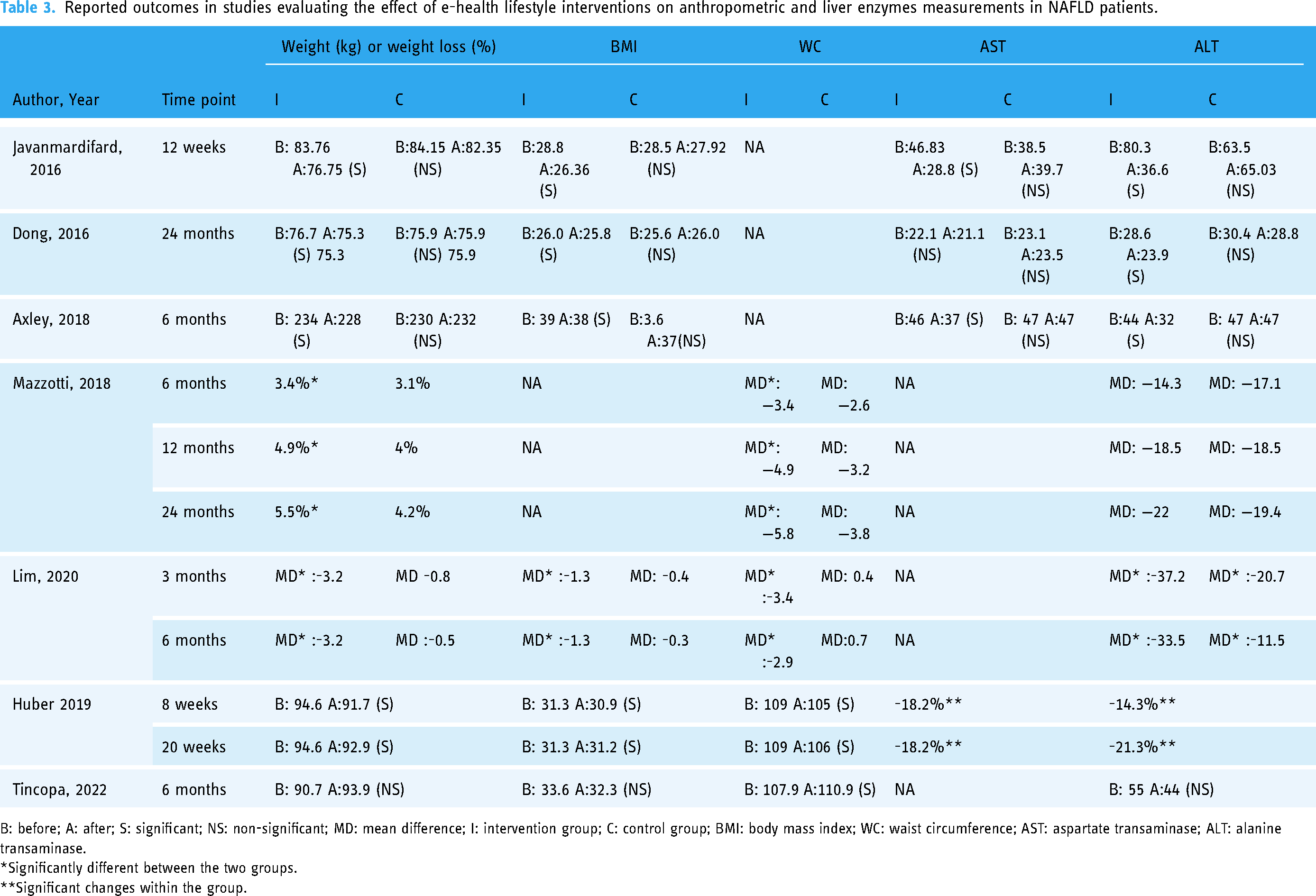
B: before; A: after; S: significant; NS: non-significant; MD: mean difference; I: intervention group; C: control group; BMI: body mass index; WC: waist circumference; AST: aspartate transaminase; ALT: alanine transaminase.
*Significantly different between the two groups.
**Significant changes within the group.
In the RCT conducted by Dong et al., 34 all participants received diet and PA counseling from two professional physicians (one dietitian and one exercise physiologist). Participants in the intervention group also received a phone visit from doctors every 3 months for 2 years, concerning health guidance on diet and exercise. Patients in the control group did not receive periodic calls. As presented in Table 3, they found that in the intervention group, weight and ALT significantly fell, while BMI and AST had a non-significant decrease. In the control group, changes were not statistically significant. Comparison of parameters between the intervention and the control group after the 2-year lifestyle intervention showed that ALT was significantly lower in the intervention group (23.95 ± 12.5 vs. 28.78 ± 20.88, p = 0.02). There was no significant difference in weight and AST. BMI and WC were not reported.
In Axley et al.’s 33 study, all patients received standard care for their liver disease, besides detailed instructions on diet and PA for weight loss. The intervention group (n = 13) also received three uni- and bi-directional text messages every week for 6 months. The text messages domain included NAFLD education, goal setting, motivation, nutrition, exercise, behavior change, stress management, and overcoming barriers. They found that patients in the text messaging group experienced a significant weight loss (−3.1 Kg, p = 0.03), while the control group gained weight (+0.8 kg, +=0.45). The BMI significantly reduced in the intervention group, while it had no change in the control group. The mean change in weight and BMI during the study were significantly different in the two groups (p = 0.023 and 0.019, respectively). Regarding liver enzymes, ALT and AST significantly decreased in the intervention group (−12.5 ± 5.3, p = 0.03 and −9.3 ± 4.2, 0.04), while they had no significant changes in the control group (see Table 3).
In Mazzotti et al.’s 29 study, patients were asked to engage in the group-based intervention (GBI) program (n = 438), which consists of group counseling on a healthy diet and habitual PA. There were five 120-minute weekly sessions chaired by physicians, dietitians, and psychologists. The first four sessions were about energy balance, nutrients and weight control, the food pyramid and portion sizes, food shopping and labels, and PA. The final session covered behavioral strategies to lose weight and maintain weight loss. Patients who could not participate in the GBI group, mainly because of job, time, or distance constraints, engaged in the web-based intervention (WBI) program. The first four sessions were similar to the GBI program. The final session was a role-game measuring adherence to previously learned objectives. All sessions included an introductory message, online questionnaires, and 25–35 engagement slides. There was no limitation for repeating the individual sessions, and patients were able to contact the clinic in an offline mode. As presented in Table 3, in the WBI group, weight significantly decreased during 6, 12, and 24 months of follow-ups (all p-values < 0.001). At the three time points, weight loss was significantly higher in the WBI group. The number of patients achieving 10% weight loss at any time point was not significantly different between the two groups. In the WBI group, waist circumference showed significantly more decline at all-time points. The ALT decreased significantly in both groups, but there was no significant difference between them.
In the study conducted by Lim et al., 25 patients in the intervention group received dietary and PA advice in a single face-to-face session with a nutritionist. Then, they were remotely followed up by a mobile app for 6 months. This app tracked their diet and PA and induced behavioral changes. Dietitians could monitor the user's input and progress and provide real-time feedback and encouragement. Participants in the control group received standard care consisting of dietary and PA advice from a nurse trained in nutrition counseling in a single face-to-face session. As presented in Table 3, participants in the intervention group lost significantly more weight than those in the control group at 3 and 6 months (p < 0.001). The mean difference in weight loss between the groups was 2.3 kg at 3 months and 2.6 at 6 months. The weight loss percentage at 3 months was 3.7% in the intervention group, while it was 0.9% in the control group (p < 0.001). The weight loss percentage at 6 months was 4.4% in the intervention group and 0.6% in the control group (p < 0.001). Reductions in BMI were also significantly greater in the intervention group at 3 and 6 months. Participants in the intervention group had a greater decrease in WC at 3 and 6 months; the mean difference was 3.8 and 4.1 cm, respectively. Both ALT and AST levels decreased markedly at 3 and 6 months, regardless of the intervention. However, there was a significantly more significant reduction in the intervention group.
In Huber et al.’s 27 study, a sports therapist introduced the patients to a web-based platform, in which patients received an 8-week individualized training program, 3–5 sessions per week. Patients had frequent interactions with a counselor. Besides, a discussion board and chat room were established for peer support. Patients who had completed more than 70% of training sessions were included in the analysis. Patients have a significant weight loss of 0.9% after 8 weeks of intervention (p = 0.009). Considerable weight loss was maintained after 12 weeks of follow-up (p = 0.046). BMI significantly decreased from 31.3 to 30.9 (p = 0.006) during 8 weeks. WC was also reduced from 109 cm to 105 cm (p = 0.006). The decrease in BMI and WC maintained significant after 12 weeks. ALT and AST decreased by 14.3% (p = 0.002) and 18.2% (p < 0.001) during the 8-week intervention and continued to decline until the end of follow-up by 21.3% and 18.2%, respectively (p = 0.001) (see Table 3).
In the study by Tincopa et al., 35 participants received a FibBit to encourage self-monitoring and provide real-time feedback for PA, along with weekly tailored step count goals (10% increase per week with a maximum increase of 800 steps per week to a maximum of 10,000 steps per week) and motivational messages via email. For the first 3 months, weekly personalized feedback was provided to each participant. For the last 3 months, it was biweekly. Study staff also provided nutritional assessments and dietary counseling at baseline and at month 3. A total of 33 participants completed the study. As presented in Table 3, weight, BMI, and ALT did not significantly change. WC significantly increased after 6 months. Around 50% of patients had a reduction in weight, and 18.2% of the population had >=5% weight loss.
Discussion
Currently, weight loss is the most effective strategy to treat NAFLD. Even if the patient is not obese, weight reduction may be beneficial. As e-health lifestyle interventions have the potential to reach many individuals and are relatively low-cost, they would be practical approaches to addressing weight reduction in NAFLD patients. Although we found only seven articles regarding the effect of electronic lifestyle interventions on weight reduction in NAFLD patients and improvement in liver enzymes, the results supported the use of e-health lifestyle interventions. Except for one, all the included studies indicated a significant impact on weight loss. BMI was reported in six of seven studies. Except for one, BMI was significantly reduced in the group receiving e-health interventions in all the other studies. Waist circumference was reported in four studies, which indicated a significant reduction in the e-health intervention group. ALT was reported in all the included studies. Except for two, others demonstrated a significant improvement in ALT in the e-health intervention groups. As reported in four studies, AST significantly decreased in the group receiving e-health interventions, except in one study.
Weight loss improves serum insulin levels, liver enzymes, and liver histology, so it is recommended as the primary therapy for most NAFLD patients. 14 Accessible and cost-effective strategies are necessary to provide a large-scale impact on weight loss. By electronic health interventions, the barriers of frequent face-to-face nutrition visits and PA programs are mitigated, and the costs are significantly reduced. 36 Through the e-health system, individualized goals can be set, and diet and PA can be monitored. Users can also receive personalized guidance. The results of this systematic review support the use of e-health interventions to reduce weight in NAFLD patients. This is in accordance with the results of other meta-analysis studies on the effect of e-health interventions on weight loss in overweight and obese adults, 37 children, 38 and diabetic patients, 39 which showed a significant effect.
The intervention groups across the studies found a great diversity of e-health lifestyle strategies, including telephone calls, text messaging, and web-based and mobile applications. Except for one study that only focused on PA, others included both nutritional and PA advice. On the other hand, the interventions were also diverse, such as regular assessment of PA and nutritional intakes, counseling, and providing feedbacks and motivational messages. This diversity makes it difficult to infer which intervention is more effective in reducing weight and improving liver enzymes in NAFLD patients.
The issue of who implemented the interventions in the studies is of great importance. In the included studies, nutrition counseling was provided by nutritionists, dietitians, physicians, or study staff. Earlier studies have demonstrated that high-intensity weight-loss counseling by primary-care physicians resulted in non-clinically significant weight loss, while dietitian-delivered care was effective regardless of the level of intensity. 40 Besides, most of the included studies continued the interventions for a short-term period (less than 6 months), followed up for a maximum of 24 months. In e-health interventions, attribution is a crucial issue, as we typically face a steep decline in usage over time and have poor completion rates. 41 So, it is impossible to conclude the efficacy of e-health interventions for long-term weight loss in NAFLD patients. As well, adherence to diet or PA program was self-reported in these studies, which raises the possibility of bias.
Although the results of this systematic review added to the existing knowledge of NAFLD management, it included a small number of clinical studies, which reduce the generalizability of the results. As there was heterogeneity regarding the type of intervention, duration and frequency of contacts, and the interventionist providing e-health care, the optimal e-health intervention for managing weight in NAFLD patients would not be concluded. Besides, compliance, long-term benefits, and cost-effectiveness should be considered to introduce the most effective strategy. Finally, although we have followed PRISMA guideline for this systematic review, the study protocol was not registered in PROSPERO.
Conclusion
Web-based digital health interventions were reported to be effective interventions in promoting weight loss, BMI, and WC reduction and improving liver enzymes in adult NAFLD patients.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231187597 - Supplemental material for The effects of electronic-based lifestyle interventions on nonalcoholic fatty liver disease: A systematic review
Supplemental material, sj-docx-1-dhj-10.1177_20552076231187597 for The effects of electronic-based lifestyle interventions on nonalcoholic fatty liver disease: A systematic review by Najmeh Seifi, Hossein Bahari, Sanaz Soltani, Mahya Nikoumanesh, Mojtaba Hajipoor, Gordon A. Ferns and Majid Ghayour-Mobarhan in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076231187597 - Supplemental material for The effects of electronic-based lifestyle interventions on nonalcoholic fatty liver disease: A systematic review
Supplemental material, sj-docx-2-dhj-10.1177_20552076231187597 for The effects of electronic-based lifestyle interventions on nonalcoholic fatty liver disease: A systematic review by Najmeh Seifi, Hossein Bahari, Sanaz Soltani, Mahya Nikoumanesh, Mojtaba Hajipoor, Gordon A. Ferns and Majid Ghayour-Mobarhan in DIGITAL HEALTH
Footnotes
Acknowledgements
The support provided by Mashhad University of Medical Sciences (MUMS) to conduct this study is highly acknowledged.
Contributorship
NS and MGM equally contributed to the conception and design of the research; NS, SS, MN, and MH contributed to the methodology and investigation; NS and HB drafted the manuscript; GAF and MGM contributed to reviewing and editing; MGM supervised the project. All authors critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research will be funded by the vice chancellery for research of Mashhad University of Medical Sciences (MUMS).
Guarantor
MGM.
Informed consent
N/A. As a systematic review, not including human subjects, patient consent was not required in this study.
Availability of data
The materials used in this review, such as data collection form and data extracted form are available by request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
