Abstract
Objective
In this study, we describe the rationale, supported by preliminary data, for a novel, digital, immersive virtual reality (VR)-based mind–body approach, designed to reduce bedtime arousal in adolescents with insomnia.
Methods
Fifty-two high-school students (16–20 years; 32 female) with (N = 18) and without (N = 34) DSM-5 insomnia symptoms engaged with 20 min of immersive VR-guided meditation and paced breathing (0.1 Hz) (intervention condition) and 20 min of quiet activity (control condition), right before bedtime, on two separate evenings.
Results
The intervention resulted in acute autonomic and cortical modulation (p < 0.05), leading to reduced physiological arousal (↓heart rate, ↓cortisol) compared with the control condition, with similar effects in adolescents with and without insomnia. No significant changes were detected for cognitive arousal levels. During the intervention, all participants were able to achieve the targeted 0.1 Hz breathing rate, and the majority experienced no discomfort associated with the VR exposure. However, 30–40% of the participants experienced some trouble slowing down their breathing.
Conclusions
The study provides supporting preliminary evidence for the mechanism behind a novel VR-based digital approach, designed to regulate psychophysiological arousal levels by acting on neurocognitive and autonomic pathways. Further studies (e.g. randomized clinical trials) are needed to evaluate the isolated and synergistic effects of its components (e.g. VR vs. VR + paced breathing), and its efficacy, acceptance, and feasibility in alleviating insomnia symptoms in adolescents.
Introduction: rationale for a virtual reality-based mind–body approach to target hyperarousal in adolescent insomnia
Insomnia disorder
Poor sleep is ubiquitous in modern society. About one-third of the general population complain of nocturnal insomnia symptoms (i.e. difficulty in falling asleep and/or maintaining sleep). In about 6–10% of people, these symptoms are frequent and persistent and impair daytime functioning, fulfilling the clinical criteria for an insomnia diagnosis. 1 Insomnia disorder is particularly common and frequently originates in adolescence, reaching as high as 18.5% (Diagnostic and Statistical Manual of Mental Disorders, DSM-5) in this age group, 2 and frequently persists into adulthood. Sex differences in insomnia also emerge in adolescence, toward a greater prevalence of the disorder in older adolescent girls. 3 This sex difference is maintained in adulthood. Adolescence is a vulnerable period of biobehavioral maturation, and a number of factors are associated with the increased susceptibility to insomnia in adolescence, including changes in the homeostatic sleep pressure and the endogenous circadian oscillator (e.g. a normative delay in circadian phase and slower accumulation of homeostatic sleep pressure), maturation/alteration of central and autonomic nervous systems, increasing school obligations and social activities (e.g. early school start times, social, academic, and cultural pressures).4–7 Insomnia can be detrimental for adolescents’ biobehavioral development, and when considering that many health conditions in adulthood have their origin in adolescence, this developmental period offers a key target for prevention.
Insomnia in adolescence is a major public concern; it manifests as a primary disorder, as well as a comorbidity of other mental conditions (e.g. depressive disorders), and is a risk factor for mental and physical health problems,8–10 including suicidality. 11 Biological mechanisms underlying these associations are not fully elucidated. For example, there is a complex bidirectional relationship between insomnia and mood disorders, with insomnia as a risk factor but also a comorbid symptom of mood disorders in adolescents. 12 The relationship between sleep disturbances and both somatic and mood disorders could be mediated via multiple pathways, including altered dopamine functioning, inflammatory processes and autonomic or hypothalamic–pituitary–adrenal [HPA] axis dysregulation. Alterations in these physiological processes could also interact with factors specific to the adolescent context, such as changing cognitions and social factors and biobehavioral maturation. 13 Overall, insomnia in adolescence is poorly characterized and underrecognized, with limited treatment options, in a group notoriously difficult to reach and engage. Please refer to de Zambotti and colleagues, 14 for further discussion about diagnosis, impact and treatment of insomnia disorder in adolescence.
Cognitive behavioral therapy for insomnia (CBTi), which is a multicomponent approach incorporating sleep hygiene education, relaxation training, stimulus control, sleep restriction and cognitive therapy, is recommended first-line treatment for insomnia disorder.15,16 In addition to proven efficacy in both adults and adolescents 17 in its traditional and digital (internet-based) forms, 18 CBTi has proven to be superior in the long-term management of insomnia, and with fewer side effects than pharmacotherapy. However, a large proportion of insomnia sufferers do not respond to CBTi even if this behavioral intervention is combined with pharmacotherapy. For example, about 40–50% of insomnia patients do not achieve clinical remission. 19 The specific factors responsible for CBTi treatment success are still largely unknown. CBTi is also less accessible for individuals with subclinical symptomatology (majority of cases) due to limited availability and scalability. The lack of accessibility may increase the transition to clinical disorder if left untreated. Another hurdle is a lack of engagement and motivation complying with CBTi requirements, which is particularly challenging in certain age groups (e.g. adolescence).
Given this information, we propose a novel digital mind–body approach that targets adolescent insomnia. The approach is founded on the empirical evidence supporting hyperarousal as a key pathophysiological mechanism underlying the disorder. The proposed approach has been designed to mainly target (downregulate) physiological and cognitive components of hyperarousal, using guided meditation virtual reality (VR) immersion and slow breathing. Rationale and preliminary testing of the underlying mechanism of the approach are described.
Behind insomnia: Hyperarousal interferes with an individual's falling asleep and staying asleep processes
Insomnia is a complex, heterogenous disorder with multivariate etiology, and idiosyncratic manifestation. Insomnia is particularly complex in adolescence due to its overlay with normally occurring developmental biobehavioral (e.g. sleep and ANS maturation, shifting in circadian preferences) and psychosocial (e.g. increasing autonomy and personal independence, peer relationships) changes. In addition, insomnia in adolescence presents a dynamic symptomatology and an etiology that remains poorly understood.14,20 Although the pathophysiology of insomnia is not fully elucidated, 21 literature in adults and adolescents indicates that insomnia is a condition of psychophysiological hyperactivation (referred to as hyperarousal), involving the upregulation of several biopsychological domains including cognitive, emotional, autonomic (ANS) and central (CNS) nervous systems, both day and night. These domains can be characterized by excessive worry, anxiety, intrusive thoughts, muscle tension, cardiac acceleration, elevated cortical activation, brain and whole-body metabolism, which are more pronounced under insomnia-specific circumstances (e.g. at bedtime when people attempt to sleep).4,22–25
Hyperarousal can negatively affect an individual's process of falling asleep, altering the biobehavioral de-arousing processes normally occurring across the wake-to-sleep transition,26,27 frequently resulting in prolonged time spent falling asleep. Hyperarousal is also implicated in reducing sleep quality and disrupting sleep continuity, possibly via modulation of an individual's nighttime arousal level. It can also be reflected in the upregulation of other sleep bioprocesses including ANS and cardiovascular (CV) function.14,22,28,29 Hyperarousal, particularly its cognitive manifestation, has also been included in several models of insomnia.30,31
Given its centrality, the interfering role of hyperarousal in the falling asleep and staying asleep processes has been surprisingly overlooked at the mechanistic level. One method to examine the role of hyperarousal is by experimentally manipulating bedtime arousal levels, either by acutely upregulating (e.g. via stress) or downregulating (e.g. via relaxation) arousal. These strategies have been shown to alter (disrupting/promoting) the falling asleep and the overnight sleep processes. For example, our group reported that stress-induced arousal upregulation at bedtime acutely modulated presleep arousal levels (increases in cortisol and heart rate [HR], reduced vagal ANS modulation) and resulted in altered sleep ANS function (elevated HR and reduced cardiac ANS functioning across the night) and sleep EEG (elevated electroencephalographic [EEG] 15–23 Hz activity, reflecting cortical CNS hyperarousal) in women approaching menopause. These physiological changes were greater in women presenting clinical insomnia. 32 These results were similar to Hall et al., 33 who used a similar presleep stress manipulation protocol in younger men and women (at bedtime, participants were told that they would be asked to give a speech the next morning, and that their HR and blood pressure (BP) will be monitored and the speech performance evaluated for content and quality).
In contrast to stress, we found that bedtime audiovisual biofeedback relaxation (arousal downregulation) acutely reduced presleep arousal levels (decreased HR and increased total HR variability [HRV]), resulting in improvements in indices of polysomnographic sleep continuity (fewer awakenings and fragmentation) and a reduction in HR during rapid eye movement and non-rapid eye movement sleep, in midlife women self-reporting insomnia symptoms. 34 Similarly, Sakakibara and colleagues 35 found that 20-min of presleep HRV biofeedback resulted in improved overnight sleep vagal ANS function (increases in high-frequency components of pulse rate variability, reflecting sleep ANS arousal downregulation), in healthy adults. Distraction at bedtime has also been proven effective in reducing cognitive arousal in insomnia. For example, imagery distraction (imagining interesting and engaging but also pleasant and relaxing situations) at bedtime, compared to “general distraction” and “no instruction,” has been shown to reduce distress related to unwanted thoughts and perceived sleep onset latencies. 36
Cognitive behavioral strategies and mind–body techniques targeting sleep and insomnia: The role of hyperarousal
Although not directly targeting hyperarousal, arousal downregulating effects of commonly used cognitive behavioral strategies and mind–body techniques are implicated in their therapeutic effects. For example, Sunnhed and Jansson-Fröjmark evaluated the mediating effects of presleep arousal, unhelpful beliefs about sleep and maladaptive sleep behaviors on the treatment efficacy of CBTi, in adults with chronic insomnia.37,38 They found that reduction in these mediators accounted for 50–70% of the variance in the main CBTi treatment outcomes. 38 Similar findings have also been reported by others, 39 showing that presleep arousal and dysfunctional beliefs mediated the reduction in insomnia severity and that both presleep arousal and sleep-related worry mediated the improvements in sleep efficiency (from sleep diaries) following CBTi.
Interestingly, despite not being originally designed for sleep, the use of mind–body practices (e.g. bedtime meditation, imagery, presleep yoga exercises, progressive muscle relaxation or autogenic training) as sleep aids are increasingly being used. However, limited evidence exists about their efficacy, 40 and the specific mechanisms and rationale underlying the effectiveness of these techniques in improving sleep are still largely unknown. Ong and colleagues 41 showed decreasing presleep arousal together with a reduction in insomnia severity and perceived total wake time (from diaries) following eight weeks of mindfulness-based stress reduction or mindfulness-based therapy for insomnia, compared with a self-monitoring control condition. In the SENSE study (a randomized controlled trial evaluating if cognitive behavioral and mindfulness-based intervention can prevent the emergence of major depressive disorder in adolescence presenting sleep problems and anxiety symptoms), Blake and colleagues 42 showed that seven weeks of cognitive behavioral and mindfulness-based intervention improved self-reported presleep arousal and other sleep outcomes from actigraphy (shorter sleep onset latency) and diaries (increased sleep quality and efficiency; reduced anxiety). Importantly, in the study of Blake and colleagues, 42 presleep arousal mediated the improvements in perceived sleep quality and anxiety. In another study, 43 the authors investigated the psychological processes associated with health-related outcomes following eight weeks of mindfulness-based stress reduction. They showed that the changes in rumination, unwanted intrusive thoughts, thought suppression, experiential avoidance, emotion suppression and cognitive reappraisal (all constructs related to cognitive arousal), accounted for a significant portion (up to 32%) of the negative correlation between change in mindfulness and change in sleep disturbance following the mindfulness-based stress reduction program. Reductions in presleep cognitive arousal have also been found to correlate with improvement in sleep following four weeks of mindfulness, in 15–75-year-old participants. 44 Similarly, presleep arousal mediated the pre-to-post cognitive behavioral and mindfulness-based intervention-related changes in sleep quality, in a group of adolescents. 45
Most of these mind–body techniques (e.g. guided meditation, mindfulness, biofeedback, yoga) are multicomponent interventions, relying on some form of breathing exercises (e.g. slow breathing), focused attention (e.g. toward self and/or environment), as well as body motion, metacognition and other processes. 46 Interestingly, among these different components, focused attention and breathing relaxation are among the most common aspects that could be considered transversal across different mind–body techniques. Given the impact of these mind–body techniques on sleep, it is possible that cognitive (focus attention/distraction) and ANS (respiratory-driven vagal stimulation) pathways46,47 may be strongly implicated in the presleep arousal downregulation and the sleep-promoting effect of these techniques.
Most of these techniques do not specifically nor explicitly target sleep-related hyperarousal. Also, the extent to which these strategies affect an individual's arousal levels, which domains of arousal are involved, and the biopsychological pathways implicated in the arousal modulation remain largely unknown. However, this evidence still highlights hyperarousal as a key factor in the treatment efficacy of these techniques.
Toward the development of an immersive VR-guided meditation and paced breathing approach to target bedtime hyperarousal
Building on initial work from our group,34,48,49 we have developed a new modular, flexible approach to downregulate bedtime arousal. This approach could be standardized, self-delivered, and integrated into a stand-alone digital health solution, which would account for the multidimensionality of the hyperarousal construct commonly found elevated in individuals with insomnia. Our hyperarousal-targeted downregulation approach is based on immersive VR-guided meditation and paced breathing. Specifically, we have implemented 1) nature-based immersive VR-guided meditation to stimulate focus attention and distraction, 50 which modulates cognitive and cortical CNS arousal (neurocognitive pathway), and 2) paced breathing (at 0.1 Hz) to directly stimulate vagal functioning (ANS pathway), which downregulates ANS, CV and hypothalamic–pituitary–adrenal (HPA) axis activity.46,47 Figure 1 gives a schematic representation of the rationale behind the proposed approach.

Hypothesized rationale, main pathways, and target outcomes underlying the use of immersive virtual reality (VR)-guided meditation and paced breathing to downregulate psychophysiological arousal. The depicted independence of the pathways allows better clarity of the main conceptual framework, reflecting an oversimplification of the highly complex interplay and effects between and within biopsychological systems. HR, heart rate; HRV, HR variability; HF, high frequency; HPA, hypothalamic–pituitary–adrenal; EEG, electroencephalography.
Within this conceptualization, we have adapted a simplified model initially proposed by Taylor and colleagues, 51 which engages two pathways: neurocognitive and autonomic. The neurocognitive pathway reflects the engagement of top-down mechanisms, which includes areas of brain like the insula and other frontal areas like the anterior cingulate cortex, prefrontal cortex (PFC). These areas are engaged during meditation and other mental processes (e.g. sustained attention), leading to altered central processing of bodily sensations (e.g. pain), reduced cognitive arousal and increased EEG cortical activity. 52 The ANS pathway, in contrast, engages bottom-up mechanisms in which include changes in autonomic functioning (HR, HRV; shifting the sympathovagal balance toward vagal predominance), CV (BP) and stress-related responses (cortisol) are observed following acute and long-term practice of slow-paced breathing. A key driver of these peripheral effects is due to widespread vagal innervation. 51 Although studies are currently underway to understand the neurobiological aspects of these mind–body interventions, it is important to note that the beneficial effects on mental and physical health through these interventions are likely due to the bidirectional interaction of top-down and bottom-up mechanisms (e.g. integrative psychophysiological framework). For example, slow breathing can also change cortical functioning and arousal, which could be initiated by top-down processes like the shifting of attention toward active control and modulation of breathing.
The current study was designed as an exploratory, pilot study to investigate the extent to which a single application of nature-based VR-guided meditation and paced breathing acutely reduces psychophysiological activation at bedtime in adolescents with and without DSM-5 53 insomnia symptoms. The acute effects of the experimental manipulation were assessed across different dimensions of arousal (cognitive, cortical, ANS, CV, endocrine), which were measured via noninvasive multimodal data acquisition methods (questionnaires, electroencephalography [EEG], electrocardiography [ECG], sphygmomanometer BP, saliva cortisol). Details and specifications about the system and procedures used in the study are provided in “Material and methods.” The choice and rationale behind each of the specific active components of the intervention are outlined in sections 1.4.1, 1.4.2, and 1.4.3.
Virtual reality
Although still in its early stages, VR is gaining attention as a therapeutic tool for mental health, reducing the burden of anxiety, phobias, and stress-related disorders. 54 VR consists of a computer-generated projection of visual/acoustic 3D immersive interactive realities. VR systems are now commercially available as standalone head-mounted displays (HMDs) (e.g. Oculus Quest 2) for less than $300 USD, which makes the clinical use of these devices more feasible. The VR experience is characterized by the technologically mediated level of sensory fidelity provided by a VR system (immersion) and by the user's subjective and phenomenological response to a VR system (presence, “being there” in a place when events are really happening). 55 VR systems allow one to disconnect from the “real” world by simulating and controlling interactive environments (continuous stream of synthetic sensory information) targeting specific psychophysiological states and/or therapeutic effects.56–58 VR can also create positive emotional states, which can be ideal for individuals with anxiety and depression 59 or issues with emotional regulation. 60
The advantages of VR include increasing patients’ motivation and engagement, the use of safe environments, the interactivity possibilities and the high level of personalization offered by VR. 61 For instance, learning relaxation techniques has been proven to benefit from controllable immersive settings inspiring a high degree of presence. 62 Immersive solutions can be easily enriched with engaging game-like features to motivate users. These advantages are particularly relevant for adolescents, who often have extensive experience with digital technologies and may prefer gamification of activities, which can increase adherence and engagement. For example, the popularity of Pokémon GO, an augmented reality mobile game, among adolescents was an effective tool to promote physical activity, social interaction and psychological functioning. 63 Also, the immersive features of VR, likely driven by an increased sense of presence in the VR world, have been shown to be superior to less immersive systems (e.g. computer screens) across several intervention settings aiming at improving relaxation. 64 In addition, the literature on the advantages of VR over traditional lower tech solutions like screens is progressively receiving support from studies evaluating the impact of a fully immersive VR setting on processes belonging to the class of biofeedback. 65 For example, Juliano et al. 66 performed an experiment that suggested how immersive VR may lead to bigger improvements in human-technology integration processes like embodiment and performance, compared to computer monitors. Both immersive solutions of VR and lower tech displays like computer screens can be enriched with engaging game-like features to motivate the user. 65 However, VR can ease the burden of the user in terms of sustained attention while they learn to self-regulate their own physiological functions. This effect depends on the VR capability to erase any distraction from the real surroundings while it leads the user's mental focus toward appealing stimuli that facilitate the biofeedback exercise. In this way, the resulting immersive setting is totally tailored to enhance the continuous effort of maintaining the user's attention on a challenging task without any disruptive intrusion from the real context, unrelated to the exercise itself. Such contextual facilitation could not be achieved through any traditional display since it cannot summon the same degree of presence in the user experience. This last point was confirmed in studies like that of Rockstroh et al. 67 In this work, the authors also discussed how successful the design choice can be of combining biofeedback systems with VR instead of traditional monitors since it increased not only the sensation of presence but also the perceived restorativeness of the experience.
In terms of sleep, negative cognition, low mood, worry and rumination are several psychological factors affecting the bedtime experience and interfering with the sleep processes of insomnia sufferers. In our conceptualization, 48 VR immersion can be used (1) to restore a positive sleep environment by “moving” the individual away from their bedroom, which may be associated with “conditioned arousal” due to repeated associations between poor sleep and bedroom environment. 68 (2) VR immersion can also be used as a powerful distractor shifting the individual's thoughts away from worry and rumination. For example, across different studies, VR immersion, particularly using nature-based scenes, 69 acutely improves mood states and produces measures of relaxation (e.g. reduction in perceived and objective markers of stress including electrodermal activity, HR and BP).70–73 Additionally, VR has been proven to be a powerful distractor due to its ability to draw attention away from the external world (and even their internal sensations). Numerous studies have reported VR to reduce pain and distress related to various medical procedures (e.g. burn injuries undergoing wound care, and physical therapy).50,74 For example, in healthy individuals, both presence and immersion in VR and interactivity have been related to increases in pain threshold and tolerance and reduction in pain intensity when acutely exposed to pain stimulation. 50
Intriguingly, VR has been successfully and repeatedly used as a distractor during burn injury treatments among children, adolescents and adults, 50 showing a 35–50% reduction in procedural pain. 75 VR as a distractor has also been used in other conditions such as cancer treatments, traumatic brain injury treatments, surgical and presurgical procedures as well as dental treatments, showing a reduction in treatment-related distress.
Guided meditation
Meditation covers a wide range of complementary and alternative medicine techniques encompassing different styles (e.g. focused attention, open monitoring), originating from different traditions (e.g. Zen Buddhism, Yoga, Hinduism, Tantra), being context-dependent (e.g. religion) or context-free, and having different purposes and outcomes. 76 With this in mind, meditation has been linked to several positive acute and long-term effects, including self-regulation of mood and emotions, enhancement in overall relaxation and health benefits, 77 and is increasingly used as an adjunctive treatment for mental disorders. 78 The specific mechanisms and pathways for which different meditation techniques achieve these effects are largely unknown. However, as outlined above, focus, attention and breathing components are often shared across techniques.
Furthermore, meditation has recently been successfully integrated into VR. For example, Seabrook and colleagues 79 investigated the integration of mindfulness practice in VR. Participants “suggested that the use of VR helped them to focus on the present moment by using visual and auditory elements of VR as attentional anchors.” In our approach, “guided meditation” is an integrated part of the VR breathing control experience. It was specifically chosen to increase the engagement with the VR environment while maintaining the focus on breathing, guiding individuals to repeatedly shift attention (awareness) to inner (breathing) and outer (VR environment) information. The overall goal was to maximize the passive interaction with the environment and direct attention toward the desired aspects of the experience (the script used in the current study includes meditation components and is outlined in Figure 3).
Slow-paced breathing
The use of breathing as a healing and/or health-promoting resource has been applied for centuries and used in Eastern cultures (frequently linked to spirituality), while over the last century it has also gained popularity in Western cultures and transversal within the field of integrative complementary and alternative medicine. 80 Slow breathing (usually defined as breathing at a frequency below 10 breaths/min) techniques are widely used as relaxation tools in several health applications and disease conditions. In the current implementation, we used slow-paced breathing, with the following components: controlled (voluntary regulation of internal bodily states or enteroception; individuals are instructed to control the mechanics of the breathing, frequency and inspiration/expiration ratio); slow (target breathing frequency: 0.1 Hz); abdominal (use of the abdomen instead of the chest, to facilitate slow breathing and increase tidal volume); and paced (conscious synchronization of breathing to an external pacer; inhaling and exhaling according to a specific pre-set rhythm). The aim of this approach was to directly enhance ANS (and particularly vagal) functioning.
Slow controlled respiration (isolated or integrated with biofeedback paradigms) at ∼0.1 Hz (though reflecting resonance frequency) has been linked to several immediate effects, including reduced chemoreflex response to hypercapnia and hypoxia, increased arterial oxygenation and improved ventilation efficiency, changes in hemodynamics (e.g. increases in venous return), increased amplitude of BP and HR oscillations, enhanced low-frequency HRV (maximization of respiratory sinus arrhythmia) and baroreflex sensitivity, increase vagal activity and vagal dominance;.80,81 Sustained short-term and long-term physiological effects have also been documented. For example, in healthy adults, Lehrer and colleagues 82 showed that approximately 30-min of HRV biofeedback (targeting maximization of respiratory sinus arrhythmia, achieved by maintaining slow breathing rate at ∼0.1 Hz) increased HRV and baroreflex gain, not only while performing slow breathing but also in the post-session assessment, as compared to the pre-session baseline. Long-term cumulative effects on baroreflex gain were also evident at the 10th weekly session, with resting baroreflex gain being higher than the first session (an effect that was not influenced by the respiratory patterns).
In addition to the direct physiological effects of slow breathing, less studied in the impact of slow breathing on an individual's mental function (e.g. increases in relaxation and reduction of anxiety, increases in pleasantness and improvement of mood), cortical activity (e.g. increases in EEG Alpha and reduced EEG Theta power) and activities in other cortical and subcortical areas involved in breathing and ANS controls (functional magnetic resonance imaging study, 83 meditation-type effects, and anti-inflammatory effects.80,81,84 In these cases, isolating breathing-specific, baroreflex, vagal and/or cognitive mediated (e.g. focused attention) effects is challenging.
Pilot testing of a VR-based mind–body approach to target hyperarousal in adolescent insomnia
This work is part of a larger clinical trial (1R01HL139652-01) aiming at investigating the pathophysiology of insomnia in adolescence, which examined the acute experimental manipulation (upregulation via psychosocial stress anticipation; downregulation via relaxation) of the presleep arousal levels and its impact on the subsequent nighttime sleep and CV function. Here, we focus on the rationale and the immediate effect of the presleep arousal downregulation.
Material and methods
Sample
Fifty-two 10–12th grade Junior and Senior high-school students (16–20 years; 32 female) were recruited from local high schools and the community of the San Francisco Bay Area via flyers, word of mouth and social media advertising (e.g. Facebook, Nextdoor). Adult participants gave informed consent to participate, and minors provided informed assent along with informed consent from a parent/legal guardian. The study was carried out in accordance with the World Medical Association Declaration of Helsinki ethical principles for research involving humans. The study was approved by Advarra Institutional Review Board. Please refer to Yuksel et al., 85 for a detailed characterization of the sample.
All participants underwent an in-person, semistructured clinical interview for DSM-5 53 insomnia disorder with a CA licensed clinical psychologist. Six participants met full DSM-5 criteria for current insomnia disorder (significant difficulty initiating sleep, and/or significant difficulty maintaining sleep, and/or waking too early and having difficulty returning to sleep, on at least three nights per week, with a duration of ≥3 months despite adequate opportunity to sleep, along with significant distress or impairment). Twelve other participants met all DSM-5 insomnia criteria, except that they were below the DSM-5 threshold for insomnia duration and/or frequency. All of them were assigned to the insomnia sufferers’ group (N = 18). Participants who reported having no sleep difficulties and no previous history of other DSM-5 mental disorders were assigned to the good sleepers’ group (N = 34). For all participants, exclusion criteria were past-history of, or currently having, severe medical (e.g. cancer, epilepsy, heart diseases, diabetes) and/or current DSM-5 mental (e.g. major depressive disorder) conditions, currently taking medications known to affect sleep (e.g. hypnotics), HPA axis and/or ANS functioning (e.g. antihypertensives), presence of breathing-related and/or movement-related sleep disorders (confirmed by in-lab clinical polysomnography), traveling across time-zones (past month), pregnancy (females). Eleven women self-reported taking hormonal birth control or fertility drugs (over the past three months). Female participants were studied irrespective of the menstrual cycle phase.
As part of the screening process, trait measures of depression (the Beck Depression Inventory, BDI-II) 86 and anxiety (the State-Trait Anxiety Inventory, STAI-Y2) 87 were collected. Prior experience (self-reported) with VR (question: “What is your prior experience with VR?,” choices: “I have tried VR before,” “I own a VR system,” “I never tried VR”) and breathing relaxation (question: “Have you ever tried to use slow breathing techniques (known also as diaphragmatic, deep or belly breathing)?”, choices: “Yes, I use it frequently,” “I tried it before,” “I never used it”) was also assessed. Objective measures of height and weight were obtained, and body mass index was calculated.
All recordings were performed, before (March 2020) COVID-19 pandemic-related restriction orders (e.g. stay-at-home orders) took place. Sample characterization is provided in Table 1.
Sample characterization for the pilot study.a

Mean and standard deviation (or incidence, when appropriate) are provided for good sleepers and insomnia sufferers.
Significant (p < 0.05) group differences.
BMI: body mass index; BDI: Beck Depression Inventory; STAI: State-Trait Anxiety Inventory; VR: virtual reality.
Main laboratory procedures and experimental design
All recordings took place at the SRI International Human Sleep Research Lab. Prior to the main study, all participants underwent an in-lab full clinical polysomnography assessment. On that night, standard EEG, electromyography, bilateral electrooculography, ECG, finger oximetry, nasal airflow, thoracic and abdominal breathing (piezoelectric bands) and leg movement measurements were taken via Compumedics Grael® system (Compumedics, Abbotsford, Victoria, Australia). Sleep recordings, breathing and leg movement events were scored according to the American Academy of Sleep Medicine guidelines, 88 to confirm the absence of sleep-disordered breathing and leg movement disorders, and to familiarize participants with the sleep lab environment and staff.
In the main experiment, participants completed two nonconsecutive, counterbalanced (based on sequential participants’ IDs), evening sessions in which they engaged in 20 min of nature-based VR-guided meditation and paced breathing (intervention session) or 20 min without an intervention (control session), before bedtime. During the control session, participants did a quiet activity of their choice (e.g. watching TV or reading). Forty-two adolescents completed both evening sessions (22 participants had the intervention session preceding the control session). Five participants completed only the active session, while five participants completed only the control session due to COVID19-related protocol deviations (e.g. suspension of in-person measurements following stay-at-home orders related to the pandemic). Participants’ bedtimes were determined based on their usual sleep schedules based on self-reports, and there were no significant differences in the timing of the start of the evening intervention (22:36 ± 0:50) and control (22:28 ± 0:49) sessions (p > 0.05). Participants were asked to refrain from consuming alcohol and caffeine after 3 pm of each recording day. At lab entry, all participants were tested for recent alcohol/drug use. All participants registered 0.0% for breath alcohol content (S75 Pro, BACtrack Breathalyzers), and tested negative at the urine drug test (10 Panel iCup drug test, Instant Technologies, Inc.). All recordings took place on week nights.
Before the experiment, all participants received a training session. They were instructed on how to perform paced breathing (slow diaphragmatic breathing) and use the VR equipment. During the practice session, participants progressively performed paced breathing at 8, 7 and 6 breaths/min (they were asked to synchronize their breathing to an external pacer, for 1-min for each of the breathing conditions), and to confirm if they were comfortable performing slow breathing at 6 breaths/min (targeted breathing rate). Participants were also instructed how to activate a nature-based VR environment and control VR elements (they were exposed to the same VR setting and environment used in the main experiment; see “Experimental apparatus and nature-based VR-guided meditation and paced breathing procedure”).
Before, during and after the 20 min intervention or control sessions, measures of psychophysiological arousal (cognitive, cortical CNS, ANS, CV and endocrine) were taken (see “Self-report measures” for details).
Experimental apparatus and nature-based VR-guided meditation and paced breathing procedure
Briefly, the intervention session consisted of continuously performing slow diaphragmatic breathing, following an external audio pacer, while experiencing a relaxing nature-based VR environment, guided via a prerecorded meditation script (Figure 3), for a total duration of 20 min. During the session, participants were sitting on an armchair with an Oculus Rift (Facebook Technologies, LLC.) headset on, and an Oculus touch controller on the dominant hand (as part of the structured intervention, participants were instructed to activate VR elements at a specific time within the 20-min experience). Several sensors were placed on the participant for physiological monitoring (Figure 2). The lab staff controlled the entry and the exit into the VR environment using an Oculus mini controller, as well as participants’ compliance with the protocol.

Experimental setting used to deliver nature-based immersive virtual reality (VR)-guided meditation and paced breathing experience, and to collect physiological data. Participants were comfortably sitting on an armchair in a soundproof and temperature-controlled bedroom at the SRI International Human Sleep Research Laboratory. Sensors and equipment used for physiological data collection were attached to the participants, including electroencephalography (EEG) and electrocardiography (ECG) sensors, thoracic and abdominal piezoelectric breathing bands, and Portapres technology on the left hand to measure beat-to-beat blood pressure (BP). All sensors and equipment were connected to a Compumedics Grael® system (Compumedics, Abbotsford, Victoria, Australia) to assure synchronized data acquisition. All sessions were supervised by sleep lab staff members, who controlled proper breathing, compliance with the procedure, and started and ended the intervention. A subset of physiological data has been analyzed in the current manuscript. Nature Treks VR screen image courtesy of Greener games (https://www.greenergames.net).

Structured nature-based virtual reality (VR)-guided meditation and paced breathing intervention session. Nature Treks VR images courtesy of Greener Games (https://www.greenergames.net).
The Nature Treks VR app Version 1.71 (Greener Games; https://www.greenergames.net) was used to provide an immersive nature-based VR environment (forest in orange sunset). EZ-AIR PLUS Windows application Version 1.0 (Biofeedback Federation of Europe; Thought Technology Ltd) was used to provide a constant breathing sound, at a target rate of 6 breaths/min (inhale duration: 3 s; hold in duration: 0.5 s; exhale duration: 6 s; hold out duration: 0.5 s), for the participants to follow. Prerecorded breathing audio files for inhaling (person breathing in through the nose) and exhaling (person breathing out through the mouth) were used for paced breathing. The guided meditation script was delivered via a pre-recorded audio file. All sound sources (Nature Treks VR, EZ-AIR PLUS and guided meditation) were played directly through the Oculus headphones.
Before the session, the following specific instructions were also given to assure proper breathing to avoid over-breathing: “It is important that you feel comfortable while breathing. Please breathe low and slow; As you breathe out, keep your lips half-closed and blow the air out through your mouth very slowly; Please take in smaller inhalations, extend the exhalations and slow down the very beginning of the exhalation; Please try to relax your shoulders and breathe through your abdomen only; When you inhale let the air fill the abdomen (you should feel your left hand pushed up; your stomach moves out against your hand and no movement of the chest), pause, and let your abdomen fall as you breathe out. Breathe in slowly through your nose and out slowly through your mouth.” The 20 min intervention session was standardized (Figure 3).
Psychophysiological arousal state assessment
Measures and timing for data collection are displayed in Table 2.
Main dependent variables obtained before, during (intervention session only) and after the evening experimental intervention (nature-based virtual reality-guided meditation and paced breathing) or control (quiet activities) sessions.a

Measures of cortical and cardiac autonomic arousal were processed for the 5-min resting-state periods (participants lying still in bed with eyes open) preceding and following the 20-min experimental sessions, and during the 20-min intervention session. Automatic sphygmomanometer blood pressure readings were taken at the end of the 5-min resting-state periods. Saliva cortisol was sampled once right after the 20-min experimental sessions. Self-report measures were collected via a secure web survey tool (REDCap), on a tablet, before and after the 20-min experimental sessions.
PSAS: Pre-Sleep Arousal Scale; DISS: Daytime Insomnia Symptom Scale; EEG: electroencephalography; HPA: hypothalamic–pituitary–adrenal; HR: heart rate; HRV: HR variability; VLF: very low frequency; LF: low frequency; HF: high frequency; SBP: systolic blood pressure; DBP: diastolic blood pressure; CNS: central nervous system; ANS: autonomic nervous system; CV: cardiovascular.
Self-report measures
Cognitive arousal. The Pre-Sleep Arousal Scale (PSAS), 89 a 16-item questionnaire, was used to evaluate state cognitive arousal levels. Items were rated on a five-point Likert scale (“Not at all”, 1 to “Extremely”, 5), evaluating the severity of the symptoms. A total score for the cognitive arousal subscale was derived (higher scores indicate greater arousal).
Alertness, positive and negative mood and sleepiness. The Daytime Insomnia Symptom Scale (DISS), 90 a 19-item 0–100 mm (“Very Little” to “Very Much”) visual analog scale (VAS), was used to evaluate state alert cognition (five items: forgetful, clear-headed, able to concentrate, how much of an effort is it to do anything and alert), negative mood (five items: anxious, stressed, tense, sad and irritable), positive mood (five items: relaxed, energetic, calm, happy and efficient), sleepiness/fatigue (three items: fatigued, sleepy and exhausted). A total score for each of the subscales was derived.
Physiological measures
ECG and EEG data were collected via Compumedics Grael® system (Compumedics, Abbotsford, Victoria, Australia) and processed using MATLAB R2018b (MathWorks Inc, MA, USA).
Cortical. Surface scalp EEG was collected from frontal (F3/4), central (C3/4) and occipital (O1/2) derivations (256 Hz sampled) referred to as the contralateral mastoid, according to the international 10–20 system. Submental electromyography and bilateral electrooculography signals were also recorded, and sleep and wake states were determined based on visual analysis of EEG, electrooculography and electromyography 30-s epochs following the American Academy of Sleep Medicine rules. 88 None of the participants fell asleep during the evening experimental sessions. EEG signals were high (0.3 Hz) and low (50 Hz) pass filtered for resting-state EEG data analyses. After filtering, corrupted segments (i.e. muscle activity and eye movement artifacts) were identified by visual inspection and via independent component analysis 91 using the EEGLAB MATLAB toolbox. 92 Power spectral density was calculated for each EEG channel in 2 min bins (to match the ECG HRV analysis) using the Welch technique 93 with a Hanning window of 1024 sample points and an overlap of 512 points. Power spectral density was calculated for each channel over the range of 0.03–50 Hz. The following measures were derived: EEG absolute power (µV2/Hz) in theta (>4 Hz to ≤8 Hz), alpha (>8 Hz to ≤12 Hz), sigma (>12 Hz to ≤15 Hz), beta (>15 Hz to ≤30 Hz) and gamma frequencies (>30 Hz to ≤50 Hz) across frontal (F), central (C) and occipital (O) derivations.
Autonomic. ECG was recorded in a modified D2 Einthoven configuration via Ag/AgCl Meditrace surface spot electrodes (512 Hz sampled). The ECG signal was digitally filtered with a fourth-order Butterworth bandpass filter (upper cutoff: 0.5 Hz; lower cutoff: 25 Hz). Datapoints with a value more than 10 SDs away from the mean were identified as outliers and were replaced by interpolating the remainder of ECG samples. The frequency-domain HRV measures were calculated during the 5-min resting-state periods (two consecutive 2 min bins; the first 30 s length period and the last ∼30-s length data were excluded to minimize artifacts) preceding and following the 20 min experimental intervention and control manipulation, as well as across the 20 min nature-based VR-guided meditation and paced breathing intervention session (10 consecutive 2-min bins). R peaks and normal beat-to-beat HR were calculated using customized algorithms, and frequency-domain measures derived as per standard practice. 94 The following measures were analyzed: HR (bpm), HRV LF power (ms2), LF peak frequency (Hz; reflecting breathing frequency in the condition of slow-paced breathing at 0.1 Hz), HF power (ms2; reflecting vagal modulation under controlled breathing conditions), HF peak frequency (Hz; reflecting breathing frequency under normal spontaneous breathing within 0.15–0.4 Hz frequency ranges) and total power (ms2; reflecting ANS flexibility).
Endocrine (HPA axis). Saliva samples were collected for cortisol analysis using Salivette swabs (Salimetrics, Carlsbad CA, USA). Samples were collected right after the 20 min of arousal downregulation (or quiet activities). Samples were frozen at −80°C for subsequent analysis by the Salimetrics’ Saliva Lab (Salimetrics, Carlsbad, CA) using the Salimetrics Salivary Cortisol Assay Kit (Cat. No. 1-3002), without modification to the manufacturers’ protocol. Samples were thawed to room temperature, vortexed and then centrifuged for 15 min at ∼3000 RPM (1500 × g) right before the assay was performed. The sample test volume was 25 µl of saliva per determination. The inter- and intra-assay coefficients of variation were 6.0% and 4.6%, respectively, with an assay range of 0.012–3.0 µl/dL and a sensitivity of 0.007 µg/dL, meeting the manufacturers’ criteria for accuracy and repeatability (https://salimetrics.com/rigor-and-reproducibility-lab-results/). Absolute measures of cortisol (nmol × L−1) were obtained.
Cardiovascular. Systolic (SBP) and diastolic (DBP) BP measures were taken via upper arm automatic sphygmomanometers (Omron 10 Series, Omron Healthcare, Inc.) from the participants’ non-dominant arm, while lying in bed. Participants were simply instructed to stay as still as possible, refrain from speaking and/or engaging in any activity, breathe as usual, keep arms relaxed at their side and keep eyes open and look at a preset mark (adhesive dot) on the ceiling.
Assessment of potential side effects and compliance with the nature-based VR-guided meditation and paced breathing intervention session
Given the known association between VR immersion and cybersickness
95
and the potential side effects associated with slow breathing (e.g. hyperventilation), participants were asked the following morning to rate any potential discomfort and difficulties experienced while engaging in the bedtime intervention session (
Statistical analyses
BDI, cortisol, PSAS cognitive, HRV (LF power, HF power and total power) and EEG measures were log-transformed [log(x + 10)] to improve normality. Shapiro–Wilk test was used to assess normality of the data. Chi-square tests (Yates corrected) were used to compare distributions for sex, race, ethnicity, prior experience with VR and slow-paced breathing, between insomnia sufferers and good sleepers. Between-subjects one-way ANOVAs with factor group (between, insomnia sufferers vs. good sleepers) were used to analyze demographic and sample characteristics. Repeated measures ANOVAs (RM-ANOVAs) with factors group (between, insomnia sufferers vs. good sleepers), condition (within, intervention session vs. control session), and group × condition, were used to analyze the user experience and compliance, PSAS cognitive, and cortisol levels. RM-ANOVAs with factors group (between, insomnia sufferers vs. good sleepers), condition (within, intervention session vs. control session), time (within, pre vs. post manipulation), condition × time, and condition × time × group, were used to analyze autonomic (HR, HRV LF power, HF power, HF peak frequency, and total power), cortical (EEG absolute power in theta, alpha, sigma, beta and gamma frequencies) and cardiovascular (SBP and DBP) measures. In the intervention session, additional RM-ANOVAs were used to evaluate cortical and autonomic responses to the intervention, with factors group (between, insomnia sufferers vs. good sleepers), time (within, baseline pre-session and 1-to-10 2 min periods of the 20 min of nature-based VR-guided meditation and paced breathing session) and group × time. Adjusted univariate test for repeated measures (Greenhouse–Geisser [G-G] corrections) was applied, and epsilon (ɛ) and adjusted p-values were provided. Bonferroni correction was used for post hoc tests. Analyses were run using STATISTICA 64 (Dell Inc.) Version 13. Significance was set at p < 0.05.
Results
Self-report: Cognitive arousal, cognitive alertness, positive and negative mood as well as sleepiness
None of the measures showed significant group, condition or group × condition effects.
Autonomic, cardiovascular, and hypothalamic–pituitary–adrenal axis function
Resting-state HR was significantly lower (all p's < 0.001) in the post nature-based VR-guided meditation and paced breathing intervention session, compared to its pre-intervention level, and compared to its levels preceding and following quiet activities (condition × time interaction: F1,40 = 9.27, p = 0.004, pη2 = 0.19). No significant HR differences were detected in the baseline resting-state periods preceding the intervention and the quiet activities. No between-groups differences in HR was detected. In contrast, frequency-domain HRV analysis showed that insomnia sufferers had overall significantly lower HRV HF power compared to good sleepers (group main effect: F1,40 = 4.57, p = 0.039, pη2 = 0.10), with no group differences in HRV LF power and total power, suggesting reduced vagal activity in insomnia sufferers. No significant condition or time effects were detected for HRV HF. HRV HF peak frequency did not show any significant group, condition or group × condition effects, suggesting no confounding effects of breathing on HRV outcomes (Figure 4). None of the models were significant for sphygmomanometer SBP and DBP.

Mean and standard errors of heart rate (HR) (top panel) and HR variability (HRV) high-frequency (HF) power (bottom panel) during the resting state periods preceding (t1) and following (t2) the control session (20 min of quiet activities) and the intervention session (20 min of nature-based VR-guided meditation and paced breathing), in good sleepers (n = 28) and insomnia sufferers (n = 14). Main results: HR decreased in response to the intervention in good sleepers and insomnia sufferers. In contrast, no significant HR changes were noted in the pre-to-post quiet activities (condition × time interaction). Overall, insomnia sufferers had significantly lower HRV HF power (vagal activity) compared to controls (group main effect).
As expected, while performing nature-based VR-guided meditation and paced breathing, compared to the preceding resting-state period, HRV LF strongly increased (driven by the shifting in breathing rate toward the targeted 0.1 Hz), together with increases in HRV VLF and HF. See Table 3 and Figure 5, for significant time main effects and graphical representation of the changes. HRV responses did not differ between good sleepers and insomnia.

Graphical representation of very low (VLF), low (lf), and high (hf) frequency heart rate variability (HRV) in the resting state periods preceding (pre), during, and following (post) the 20 min nature-based virtual reality (VR)-guided meditation and paced breathing experience, in good sleepers (n = 28) and insomnia sufferers (n = 14). Data are displayed as mean and standard errors.
Significant changes in heart rate variability (HRV) while participants performed nature-based virtual reality-guided meditation and paced breathing (intervention), compared to the pre-intervention baseline resting state.

Significant (p < 0.05) differences (Bonferroni).
VLF; very low frequency; LF: low frequency; HF: high frequency.
Cortisol levels were lower after engaging with 20 min of nature-based VR-guided meditation and paced breathing (1.08 ± 0.68 nmol/L), as compared to the post 20 min of quiet activities (2.02 ± 2.53 nmol/L), in groups with and without insomnia (condition main effect: F1,40 = 13.44, p < 0.001, pη2 = 0.25) (Figure 6).

Cortisol levels detected after the evening control session (20 min of quiet activities) and after the evening intervention session (20 min of nature-based virtual reality-guided meditation and paced breathing), in good sleepers (n = 28) and insomnia sufferers (n = 14). Main results: In the whole sample, cortisol levels were significantly reduced in the post-intervention session, compared to post quiet activities (condition main effect).
Cortical function
No significant differences in cortical EEG activity were detected in the analyses of the resting state periods preceding and following the 20 min intervention and control sessions (Table 4).
Mean (standard deviation) of resting-state electroencephalographic (EEG) activity (µv2/hz) before and after participants engaged with the 20 min virtual reality (VR)-guided meditation and paced breathing experience (intervention) or quiet activities (control), in good sleepers and insomnia sufferers.
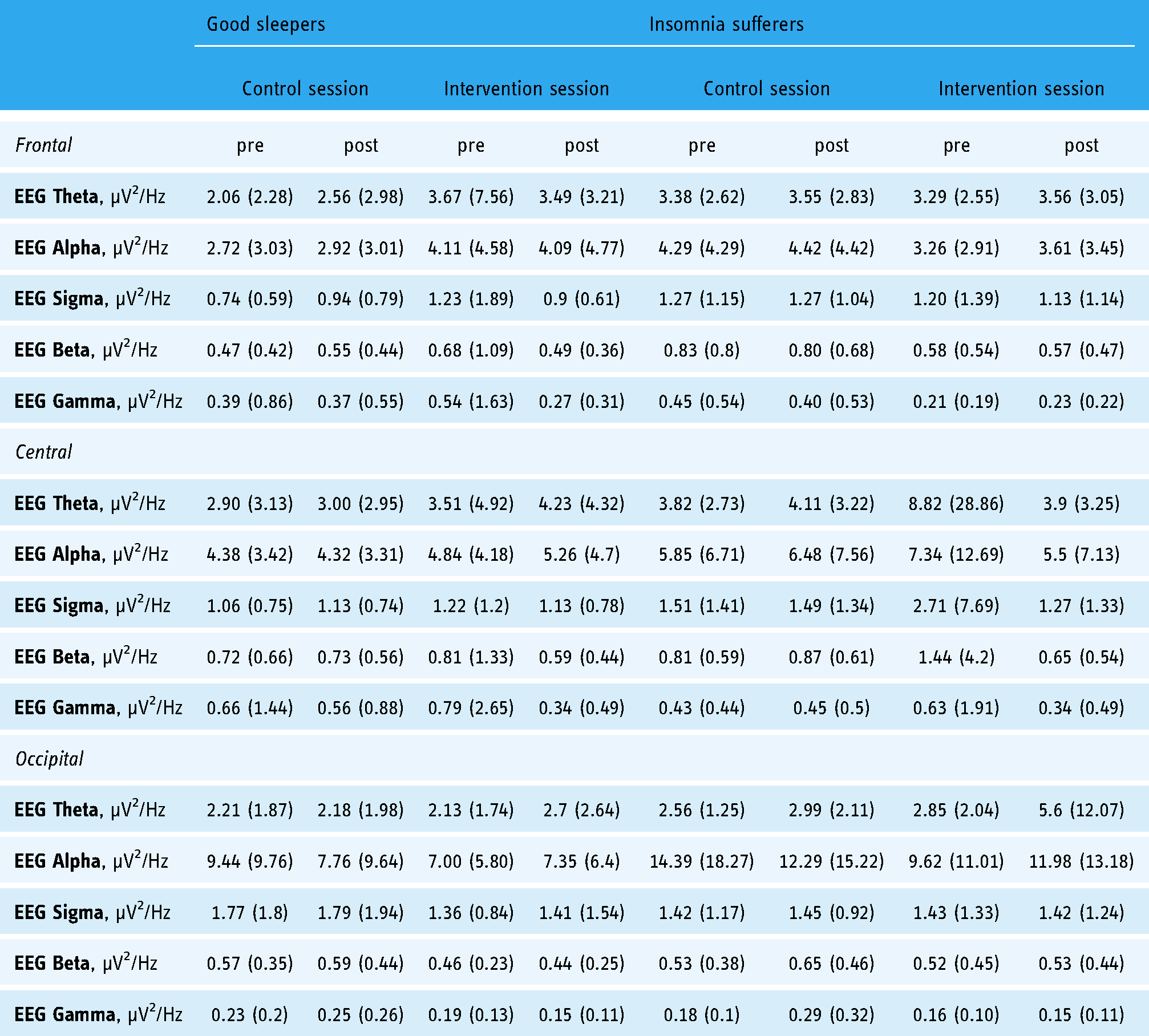
However, significant EEG changes were detected while participants were performing nature-based VR-guided meditation and paced breathing compared to the resting state baseline preceding the intervention. EEG alpha (occipital) power reduced during the intervention, while EEG sigma (central and occipital), beta (frontal, central and occipital) and gamma (occipital) powers increased. See Table 5 and Figure 7, for significant time main effects and graphical representation of the changes. No other significant group, time or group × time effects were detected.

Graphical representation of cortical electroencephalographic (EEG) activity in the theta, alpha, sigma, beta and gamma frequency ranges, across scalp location (frontal, central and occipital) in the resting state periods preceding (pre) and following (post), and during the 20 min nature-based virtual reality (VR)-guided meditation and paced breathing experience, in good sleepers (n = 28) and insomnia sufferers (n = 14). Data are displayed as mean and standard errors.
Significant changes (time main effects) in electroencephalographic (EEG) activity while participants engaged with the 20 min virtual reality (VR)-guided meditation and paced breathing experience (intervention), as compared to the pre-intervention EEG resting-state baseline period.

Significant (p < 0.05) differences (Bonferroni).
Prior experience with VR and slow breathing, side effects and compliance with the intervention
There were no group differences in the prior experience with VR and slow breathing techniques, with most adolescents being naive to VR and not habitually engaging in slow breathing exercises (Table 6).
Prior experience with a virtual reality (VR) system and with slow breathing techniques, in good sleepers (n = 34) and insomnia sufferers (n = 18).

During the 20 min experimental intervention session, the evaluation of HRV LF peak frequency (reflecting breathing frequency under forced slow-paced breathing condition) confirmed that participants complied with the instructions, by maintaining the target breathing frequency (HRV LF peak frequency, mean ± SD: 0.098 ± 0.011 Hz), across the entire session (Figure 8).

Heart rate variability (HRV) low-frequency (lf) peak frequency (hz) across the 20 min of nature-based virtual reality-guided meditation and paced breathing. HRV LF peak frequency is defined by the frequency with the highest power within the LF range (0.05–0.15 Hz), reflecting breathing frequency under forced breathing within a low-frequency range.
Post-intervention session survey outcomes indicated that most of the participants perceived low discomfort associated with the VR immersion, while slowing down the breathing (target: 6 breaths/min across the entire 20 min of intervention) was experienced as challenging by 30–40% of the sample (Figure 9).

100% stacked graph of following morning survey outcomes of participants perception (rating on 1–5 Likert scale: 1, strongly disagree | 2, disagree | 3, neutral | 4, agree | 5, strongly agree) for potential discomfort in experiencing virtual reality and in performing slow breathing.
Discussion
The current study highlights the rationale and preliminary testing of a novel mind–body approach designed to reduce bedtime psychophysiological arousal levels in adolescent insomnia. Results highlight the underlying psychophysiological mechanisms, that is, hyperarousal-targeted downregulation, and pathways of the approach. Outcomes of this preliminary study are discussed in the context of the intervention's rationale, by separating the physiological effects occurring while actively engaging in the 20 min of VR-guided meditation and paced breathing session, from those reflecting pre to post-intervention changes in resting-state psychophysiology, in relation to quiet activities.
Ultimately, this study may lead to the development of novel mind–body treatments for adolescent insomnia using a VR-based digital health paradigm that could be standardized and self-delivered, targeting key pathophysiological mechanisms underlying the disorder. This approach may be particularly relevant for adolescents, who commonly prefer digital solutions and gamification of activities.
Autonomic and cortical function while experiencing the nature-based VR-guided meditation and paced breathing
Irrespective of the group, during the intervention (while actively engaging in VR-guided meditation and paced breathing), we observed increases in HRV (reflecting ANS vagal activation), increases in EEG sigma, beta and gamma powers, and reductions in EEG alpha power (reflecting attentional processes), as compared to their baseline levels.
The acute HRV changes were most likely due to the strong respiratory-driven enhancement in HRV LF activity during slow-paced breathing. HRV LF activity reached ∼68% of the total power under slow-paced breathing during the intervention. At the same time, it shared ∼38% of the HRV total power under normal breathing during the resting state period preceding the intervention. Respiration is known to affect HRV via respiratory sinus arrhythmia (the variation in the inter-beat intervals according to inspiration and expiration patterns), with HRV being higher when breathing at slower rates and progressively reducing for faster breathing rates.96,97 The HRV changes during controlled slow-paced breathing are likely to be entirely vagally mediated. Convincing data on the ANS sources of HRV under slow-paced breathing are provided by Kromenacker et al. 98 They evaluated HRV activity under sympathetic β-adrenergic blockade (esmolol), vagal muscarinic-cholinergic blockade (glycopyrrolate) and placebo (saline) across different slow breathing rates, ranging from 4 to 9 breaths/min. They found that increases in HRV peak power under slow breathing conditions were almost entirely suppressed by a vagal blockade. In contrast, no differences in HRV were found when comparing sympathetic blockade and placebo. Thus, while under normal breathing conditions (breathing rates falling within conventional 0.15–0.40 Hz frequency ranges), HRV HF power is generally considered and used as an index of vagal activity, under slow breathing conditions (<10 breaths/min, with breathing rates usually falling within 0.05–0.15 Hz frequency ranges), increases in HRV LF activity can be imputed to increases in vagal trafficking. Overall, while the meditative component of the intervention and the immersive VR experience can also acutely modulate ANS function,64,99 possibly via relaxation/stress reduction effects, we believe that if they have an impact, their contribution would be minimal compared to a much more substantial effect of respiratory-driven ANS modulation.
The analysis of cortical EEG activity showed significant suppression in occipital EEG alpha power, likely to reflect attentional information processing,100–103 while performing the intervention compared to the baseline resting-state period. Of relevance, EEG alpha desynchronization has also been reported in response to full VR immersion, 103 reflecting a shifting in attention in the human–environment interaction in complex VR environments. Like our results, it was accentuated in parietal–occipital regions. Also, similar to our findings of pronounced EEG alpha drops in the first half of the VR-based intervention, in the study of Magosso et al., 103 alpha desynchronization was less pronounced toward the end of VR immersion experience, interpreted as declining in the attentional demand of the environment when participant get used to it. However, while in Magosso et al., 103 the results were based on static VR experience only, in the second half of our intervention, participants had some form of minimal engagement with the VR (see Figure 3), which may also affect EEG expression by involving sensorimotor processing. Modulations in EEG alpha (decreases), as well as beta power (increases), were also shown to play a considerable role in attentional control related to video gaming.104,105
In addition to alpha suppression, we found increases in EEG high-frequency synchronization, including elevated EEG gamma activity in response to the intervention, which can also reflect attentional processes,106,107 as well as perceptual information processing during meditation. 108 Although increases in EEG theta power, a frequency associated with EEG alpha suppression 100 would also be expected, high interindividual variability in dominant theta frequencies may have masked these potential effects. 100
The mind–body approach tested here is complex due to the nature of its interactive multicomponents, that is, VR immersion, guided meditation and breathing control. The dynamics of EEG information processing occurring across the 20-min of intervention, as well as its relation to specific cognitive and autonomic aspects of the intervention, is yet to be determined (see limitation “Limitation, future directions, and considerations”). For example, in addition to strong ANS modulation, slow breathing may also directly affect EEG activity. 81 Also, specific cortical processes are differently influenced and expressed according to numerous factors. For example, it has been shown that EEG alpha may respond differently (increases vs. suppression) according to if a processing task requires externally versus internally (e.g. visual imagery, mental arithmetic) directed attention, passive versus active engagement, relevant versus irrelevant information processing; different effects are also expected across different brain areas, sensor modality, task demand, and specific EEG frequencies (standard bands, sub-bands, individuals-specific dominant frequencies).100–103
Resting-state pre-to-post intervention psychophysiological changes
In the analyses of pre-to-post manipulation resting-state psychophysiology (intervention and control sessions), significant changes were detected for HR, which was reduced in the post-intervention compared to the pre-intervention levels and compared to the levels preceding and following the control session (indicating ANS arousal downregulation). Similarly, cortisol levels were lower after engaging with nature-based VR-guided meditation and paced breathing, compared to post quiet activities, indexing a reduction in HPA activation. Overall, the strong vagal stimulation occurring while participants were performing paced breathing combined with the relaxing effect of immersive VR-guided meditation may have accounted for these relaxation responses.
In a previous study, our group used immersive audiovisual respiratory biofeedback at bedtime (continuously across the wake-to-sleep transition) in women self-reporting insomnia symptoms. 34 Like the current study, the intervention led to acute HR reductions. Outcomes were particularly relevant when considering that most of the participants engaged with the intervention for <10 min, and that the HR downregulation effects were evident throughout the night. Like in the current study, de Zambotti and colleagues 34 previously showed that the HRV changes were limited to the period in which participants performed respiratory biofeedback, most likely due to the direct effects of slow breathing on HRV. Although these acute HRV changes are likely to be vagally mediated (see above), HR is under the dual sympathetic and vagal influence, 109 and relaxation-driven tonic sympathetic inhibition may also account for the HR changes. VR relaxation physiological effects (e.g. HR reduction, changes in electrodermal activity) have been reported by others, 64 even in the absence of breathing-related ANS modulation. Interestingly, Repetto et al. 110 showed that a multisession and multicomponent (relaxation, meditation, exposure therapy) VR-based program to target generalized anxiety resulted in significant HR reduction, and that pre-to-post sessions changes in HR were evident when the program was including or not HR biofeedback, suggesting that significant HR changes are achieved even without directly targeting HR suppression (no biofeedback). On the other hand, Blum et al. provided evidence of several advantages of VR across multiple indices of relaxation when combined with traditional HRV respiratory-driven biofeedback, 111 but not for ANS indices more directly modulated by changes in breathing patterns.
No significant changes were found in cortical and/or perceived cognitive arousal followed the VR-guided meditation and paced breathing intervention session. Several reasons may account for these results. Cognitively, the complexity of the intervention, involving switching attention among its breathing, VR, and meditative components, may require more extensive practice to reduce its associated cognitive load. Similarly, despite high compliance with the targeted 6 breaths/min during the 20 min intervention session, slowing down the breathing was experienced as challenging by 30–40% of the sample, which may be translated into the high cognitive effort. More practice sessions (currently, only a single practice session was used) may overcome this issue. It is also possible that multiple sessions are needed to effectively and significantly modulate cognitive arousal, and its EEG correlates. In addition, cognitive and cortical arousal suppression may be more evident under higher baseline state levels of cognitive arousal. A complex pattern of EEG activation in response to a similar nature-based VR-guided meditation experience has been shown by Tarrant et al., 112 in adults self-reporting generalized anxiety disorder symptoms. Like our study, the authors failed to show changes in mean absolute EEG alpha and beta frequencies (targeted frequency of interest) in response to the VR session versus the control session.
Adolescent insomnia and hyperarousal
In the current study, state-level markers of psychophysiological arousal were collected in the evening in the context of the experimental paradigm aimed at modulating the individuals’ arousal levels. To our knowledge, this is the first study providing evidence of reduced cardiac vagal activity in adolescent insomnia, in line with some but not all28,113,114 literature in adults providing evidence of ANS hyperarousal (across different ANS indices from microneurography, beat-to-beat BP, HRV, impedance cardiography) in insomnia sufferers.14,24,25,28,115–117 On the other hand, our sample of adolescents with insomnia did not significantly differ from good sleepers in other baseline measures of cognitive arousal or EEG measures thought to reflect cortical hyperarousal and previously shown to be altered in adults with insomnia.28,118 The only other study evaluating EEG activity in adolescent insomnia is by Fernandez-Mendoza and colleagues, 4 with authors finding evidence of elevated EEG beta activity in adolescents with insomnia symptoms, during the process of falling asleep and while asleep. Different factors may account for these results. It is possible that in-lab state (current) levels of arousal (vs. trait measures), high night-to-night variability in the manifestation of arousal, timecourse of arousal (e.g. wake vs. sleep, 24 h manifestation) and dynamics and complexity of different arousal dimensions (e.g. cognitive hyperarousal is pronounced at bedtime with distinct qualitative and quantitative aspects characterizing the insomnia experience 119 may account for the lack of consistency in the expression of this key trait feature of insomnia when snapshots of data are taken.
Limitation, future directions and considerations
The characterization of insomnia in adolescents is poorly understood, 14 and limited options exist for treatment. Although this study provides preliminary evidence supporting the rationale (hyperarousal targeted downregulation) of this new mind–body approach, further studies are needed to prove its efficacy on the targeted outcomes, that is, sleep measures and insomnia symptoms.
In the current study, the single-time use of the intervention was tested for rationale. However, the extent of the effectiveness in arousal downregulation needs to be evaluated over time. For example, sustained effects of VR as a distractor in pain processing were previously shown by Rutter et al., 120 over eight weeks of use. Optimal timing (e.g. evening, bedtime) and duration (e.g. fixed vs. adaptive time, longer or shorter session) of the intervention also needs to be evaluated. Furthermore, these aspects are intertwined with user-centered design issues: even considering how far, nowadays, the advances in VR for therapy and rehabilitation moved from the weaknesses described in Rizzo and Kim, 121 the user experience can certainly be improved in such a field. Novel visualization options should be explored to minimize the impact of removing a VR headset on the user experience (e.g. general-purpose low-cost CAVE systems). 122 Overall, efforts to evaluate the user experience and the usability of digital enhancement of sleep quality currently target really specific applications – for example, sport. 123 Nevertheless, user-centered evaluation methods can be based on the assessment of systems with similar scope and functions, as in the case of applications for relaxation and meditation.124,125 Interestingly, recent works explored the process of falling asleep in multiuser environments, 126 highlighting the importance of meaningful social settings: such observation suggests the opportunity to consider contextual factors beyond the nature-inspired quality of the virtual location.
It has been shown that cybersickness may affect both cognitive127,128 and physiological 128 processes. However, these studies used VR navigation that required users to accelerate and brake in the VR environment (e.g. virtual ride on a roller coaster), whereas, in our VR experience, user motion was limited to a stationary semi-passive (the only action required was to slowly grab and release few VR elements from a stationary position; see Methods) observation of nature-based scenario and elements. Overall, less than 10% of our sample experienced some level of VR-associated discomfort (e.g. vertigo, blurred vision). However, our retrospective (the next morning) assessment of cybersickness may have been affected by recall bias. Ultimately, standardized repeated assessment of cybersickness right before and after the intervention is needed to test its potential effect on intervention-related cognitive and physiological processes.
More insight is needed in the measurement, characterization and separation of different multimodal and multidimensional components of hyperarousal, in relation to the pre-intervention arousal levels (baseline psychophysiological state) and the extent of the intervention-driven arousal downregulation. Hyperarousal has both trait and state features. It can be both a cause (acutely affecting sleep and/or daytime insomnia symptoms, or a vulnerability factor in the development of the disorder 129 as well as a consequence of insomnia. Thus, the potential therapeutic effect of the proposed intervention may be determined by the timing in which the intervention is delivered within the individual's insomnia – hyperarousal loop.
Another limitation of the study design is the lack of a true control condition. The intervention was compared to a matched period of no intervention; however, future studies should consider a more controlled comparison condition. For example, the use of the same setting with the participant wearing a non-operating VR headset, and not engaging in any activities could be more ideal as a true control. Along the same lines, independent activation of one or more intervention components (VR immersion, and/or guided meditation, and/or paced breathing) is also necessary to evaluate isolated versus synergistic effects of the approach, on different arousal dimensions and processes (e.g. attention toward VR environment vs. self, slow breathing-related CNS changes). 81 For example, the isolated activation of each intervention component can result in ANS and CNS modulation. Additional intervention-component specific and synergic effects should also be considered. For example, breathing exercises can affect both processes of presence and embodiment in VR under appropriately designed conditions,130,131 suggesting their potential synergy with interactive scenarios for our goals. Thus, the next step is to isolate the contribution of VR, meditation and breathing by adding experimental conditions that test their isolated and synergistic effects (e.g. VR vs. VR + paced breathing, VR vs. VR + meditation). Also, while the current intervention was delivered in an open-loop mode, closed-loop feedbacks (biofeedback) linking VR and physiological measures have been previously used by us and others34,111 and may confer additional therapeutic advantages. Other factors known to potentially affect the user experience (e.g. use of HDM vs. liquid crystal display) should be also considered to advance understanding of the true therapeutic advantage of using VR versus non-VR settings, within a benefit/cost analysis approach.
Some biases may have been introduced when considering that the manipulation conditions (intervention vs. control) were delivered on two different days. However, nights were scheduled based on the availability of participants and staff. Thus, the recording sessions happened on a “random” day within a weekly schedule. Also, we controlled for several factors. At each lab visit, all participants had to register 0.0% for breath alcohol content and test negative on the urine drug test. Participants’ bedtimes were determined based on their usual sleep schedules determined from self-reports, and there were no significant differences in the timing of the start of the evening intervention and control sessions. Also, we did not find any significant differences (all p's > 0.05) in the pre-intervention baseline resting-state assessment across the different psychophysiological measures collected (cognitive arousal, cognitive alertness, positive and negative mood, and sleepiness, autonomic, cardiovascular and cortical indices).
Hyperarousal is not only a therapeutic target in adolescent insomnia. Presleep hyperarousal has been shown to mediate the relation between stress and sleep (time spent falling asleep) in non-insomnia adolescents, with coping strategies moderating this relationship. 132 Our study found that the intervention had hyperarousal downregulation effects in good sleepers and insomnia sufferers, suggesting the potential use of this approach beyond insomnia, as a possible relaxation strategy.
VR systems are now commercially available at relatively low-cost as stand-alone HMD, leading toward a wide use of VR in our daily lives. Driven by the gaming industry, and partially accelerated by the COVID-19 pandemic, the broad adoption of VR platforms for education, training (e.g. surgical training, safety courses), health and wellness (e.g. meditation, social interactions, treatments like pain relief, rehabilitation) are opening new markets for VR applications. Within the need of implementing evidence-based digital therapeutics in the healthcare system (particularly for mental health care), the potential of self-administering VR-based therapeutics offers an attractive alternative to the traditional routes and potentially decreases cost and burden for individuals, payers and providers.
Footnotes
Authors’ note
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Acknowledgements
The authors thank Quimby Lee and Teji Dulai for their support and effort in the data collection process and study execution.
Conflict of interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MdZ is a co-founder and Chief Scientific Officer (CSO) of Lisa Health Inc. MdZ and FCB have ownership of shares in Lisa Health. MdZ and FCB have received research funding unrelated to this work from Noctrix Health, Inc., Verily Life Sciences LLC., and Lisa Health Inc.
Contributorship
CRediT author statement. MdZ: Conceptualization, Methodology, Formal analysis, Visualization, Project administration, Funding acquisition, Writing – original draft, Writing – review & editing; DY: Conceptualization, Methodology, Formal analysis, Visualization, Project administration, Writing – review & editing; OK: Formal analysis, Writing – review & editing; GB: Conceptualization, Writing – original draft, Writing – review & editing; NA: Investigation, Writing – review & editing; LV: Investigation, Writing – review & editing; CK: Conceptualization, Visualization, Writing – review & editing; FB: Conceptualization, Project administration, Funding acquisition, Writing – review & editing.
Ethical approval
The study was approved by Advarra Institutional Review Board (Pro00042640).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Heart, Lung, and Blood Institute, (grant number R01 HL139652), and by the Cincinnati Children's Research Innovation/Pilot Funding Program for CK. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Guarantor
MdZ.
