Abstract
Background
Adherence to antipsychotic medication is critical for bipolar disorder (BPD), major depression (MDD) and schizophrenia (SCZ) patients. Digital tools have emerged to monitor medication adherence along with tracking general health. Evidence on physician or patient preferences for such tools exists but is limited among caregivers. The study objective was to assess preferences and willingness-to-pay (WTP) for medication adherence monitoring tools among caregivers of SMI patients.
Methods
A web-based survey was administered to caregivers of adult SMI patients. Twelve discrete choice questions comparing adherence monitoring tools that varied across two attribute bundles: (1) tool attributes including source of medication adherence information, frequency of information updates, access to adherence information, and physical activity, mood, and rest tracking, and (2) caregiver monthly out-of-pocket cost attribute were administered to caregiver respondents. Attributes were parameterized for both digital and non-digital tools. Random utility models were used to estimate caregivers’ preferences and WTP.
Results
Among 184 study-eligible caregivers, 57, 61 and 66 participants cared for BPD, MDD, and SCZ patients, respectively. Caregivers highly preferred (odds ratio (OR): 7.34, 95% confidence interval (CI): 5.00–10.79) a tool that tracked medication ingestion using a pill embedded with an ingestible event market (IEM) sensor and tracked patients’ physical activity, mood, and rest than a non-digital pill organizer. Additionally, caregivers were willing to pay $255 per month (95% CI: $123–$387) more for this tool compared to a pill organizer.
Conclusion
Caregivers of SMI patients highly preferred and were willing to pay more for digital tools that not only measures medication ingestion but also tracks general health.
Keywords
Introduction
In 2019, an estimated 13.1 million adults in the United States (U.S.) were diagnosed with a serious mental illness (SMI). 1 SMI includes bipolar disorder (BPD), major depressive disorder (MDD) with psychotic symptoms, treatment-resistant depression, schizophrenia (SCZ), anxiety disorders, eating disorders, and personality disorders with severe functional impairment.1,2 Among them, BPD, MDD and SCZ are commonly diagnosed conditions. 2 The estimated annual prevalence rates of BPD, MDD, and SCZ are 1.5%, 10.4% and 1.1%, respectively.3–5 BPD is an affective disorder characterized by dramatic shifts in mood, energy, and activity levels. 6 MDD is a mood disorder characterized by feelings of sadness, hopelessness, reduced appetite, anxiety, sleep disturbances, and suicidal thoughts. 7 SCZ is characterized by symptoms ranging from hallucinations, delusions, reduced emotional expression, motor impairment and cognitive disability. 8 Further, the estimated annual economic burden of BPD, MDD and SCZ in the U.S is $195 billion, $325 billion and $155 billion, respectively9–11 with 70–80% of burden attributed to indirect costs that include productivity loss or unemployment by patients or caregivers, missed time at school, medication adherence issues and caregiving support.9,12,13
Patients with SMI face several mental, behavioral and emotional challenges that lead to poor social support, unemployment, medication adherence issues, repeated psychiatric hospitalizations, and susceptibility to other substance use disorders. 1 The burden of SMI is not only borne by the patient, but also by caregivers, often unpaid, who spend an average of 32 h a week caring for patients with SMI. Several factors contribute to caregiver burden including lack of social or peer support, limited financial support for their caregiving duties, longevity of caregiving responsibilities, lack of caregiving education, and patients’ poor medication adherence, contributing both to caregiver burden and indirect healthcare costs.12–14
The feasibility and efficacy of digital health interventions across disease areas is well documented. Digital health interventions have been shown to improve disease management, 15 patient mental and physical health,16–19 and patient adherence to medications. 20 Thus, such interventions have the potential to reduce the burden typically borne by informal caregivers of patients with SMI. In recent years, several health technologies and tools have been developed to measure medication adherence to aid in SMI medication management, including mobile phone applications (apps) that track patients’ self-reported intake of medications,21–23 medical event monitoring systems (MEMS) that electronically track the opening of pill bottles through a device in the cap, 24 smart pill dispensers that electronically counts the number of pills dispensed, 25 and drug-device combination products that directly track medication ingestion. 26 While these technologies have demonstrated effectiveness in improving medication adherence and are highly valued by patients and providers in therapeutic areas including chronic diseases and autoimmune diseases,27–29 the value that caregivers of patients with SMI place on these adherence monitoring tools has not yet been investigated.
The study's aim was to evaluate the value of digital adherence monitoring tools among informal caregivers of SMI patients by estimating their preferences and willingness to pay (WTP) for these tools compared to non-digital adherence monitoring tools.
Methods
Study population
To evaluate the value to caregivers of different health technology tools designed to aid in monitoring medication adherence, a web-based online survey was administered to informal caregivers who care for patients diagnosed with SMI. An informal caregiver was study-eligible if he/she was: (1) at least 18 years old; (2) caregiver to a patient diagnosed with one of the three commonly diagnosed SMI conditions -BPD, MDD or SCZ; (3) caregiver to a patient receiving a second-generation oral atypical antipsychotic medication; (4) proficient in English; (5) not formally paid to be a caregiver, (6) able to complete informed consent; and (7) believed that his/her patient had medication adherence issue(s).
Survey instrument
A discrete choice experiment (DCE) was designed to assess caregivers’ preferences for adherence monitoring tools. DCE surveys present respondents with a series of choices with varying attributes and levels to determine how much preferences and WTP vary based on the attributes of interest. These methods are well established in the literature and endorsed by the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) Conjoint Analysis Good Research Practices Task Force for Pharmacoeconomic Research.30–32 As per the ISPOR guidelines, the DCE was designed by identifying all available health technology tools used to monitor medication adherence (referred as technology comparators in the study), by identifying the attributes and their levels that vary between the technology comparators, and by developing the DCE choice sets.
Digital and non-digital health technology tools used to monitor medication adherence currently available in the market were identified through a targeted literature review.23–26,33,34 Among digital health technology tools, four technology alternatives were identified: (1) smart pill bottles; (2) smart pill dispensers; (3) mobile apps; and (4) a pill embedded with an ingestible event market (IEM) sensor. A smart pill bottle, such as a MEMS cap, is a cap that fits on standard medicine bottles and records the date, time and number of times the bottle is opened or closed. 24 A smart pill dispenser automatically sorts and organizes pills for daily use, and a few models dispense the pills mechanically. 25 Smart pill bottles and smart pill dispensers share common features in which both track the frequency at which either a pill bottle is opened, or medication is dispensed.25,35 Therefore, among the two, only the smart pill dispenser was included as a tool alternative in the DCE. Mobile apps that help caregivers with their duties are commonly used. Some apps remind patients when medication needs to be taken, while others provide external monitoring and send adherence-related data to health care providers or caregivers.36,37 Other health-related information is also included in the app and reported to caregivers. Pills embedded with IEM sensors are new digital health tools being utilized in patients with SMI. 26 The sensor embedded in the pill is activated once the medication has been ingested and emits a signal to a patch worn by the patient. The patch is connected to a smartphone app or dashboard via Bluetooth that records the signal upon medication ingestion. Along with capturing medication ingestion information, this technology also tracks other health-related measures, including physical activity (number of steps per day) and rest (time spent resting). Within the smartphone app or dashboard, the patient can also self-report their mood. In turn, caregivers can track these measures to assist with their caregiving duties.
Among non-digital health technology tools, two alternatives were identified: (1) health diaries, and (2) non-digital pill organizer. Health diaries and their use in monitoring medication adherence in mental illness patients have been described in literature, 34 however given that they are open tracking sources and lack consistency in reporting information across individuals, only the non-digital pill organizer was included as a non-digital technology alternative in the study. Overall, four adherence monitoring tools were included as technology alternatives in the study: (1) smart pill dispenser, (2) mobile app, (3) pill embedded with IEM sensor, and (4) non-digital pill organizer (see Table 1).
Individual and bundled attributes for technology alternatives used in the discrete choice experiment.
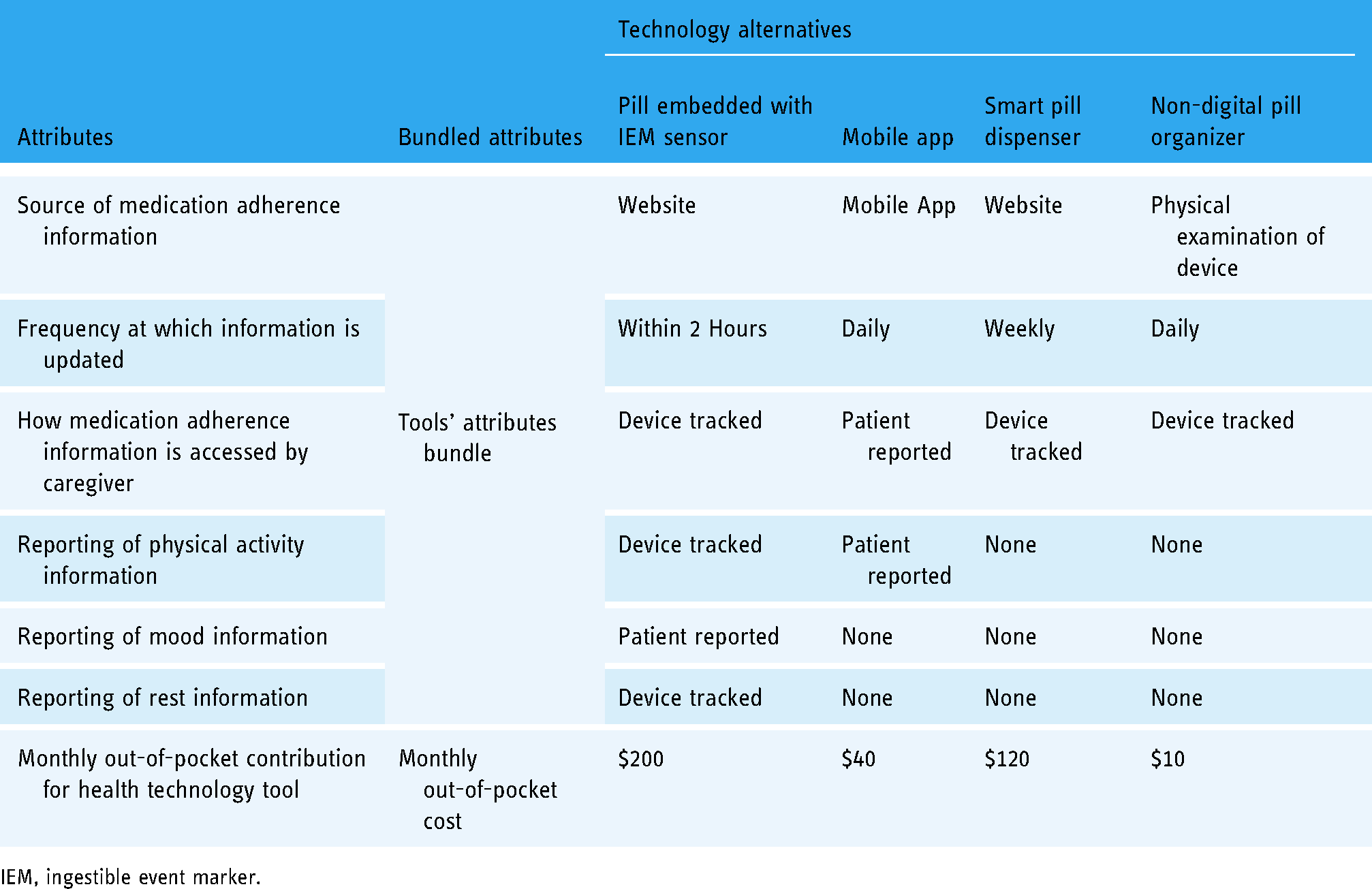
IEM, ingestible event marker.
To identify attributes and associated levels for each of the four selected technology tools, product descriptions of the four technology alternatives were obtained from the literature.23–26,34 Subsequently, attributes with differences in attribute levels between the four technology tools were selected. The final attributes included- (1) source of medication adherence information; (2) frequency at which the information is updated by the tool; (3) how medication adherence information is accessible to the caregiver; (4) reporting of patients’ physical activity, mood, and quality of rest information; and (5) caregivers’ monthly out-of-pocket cost for access to the health technology tool.
After identifying the technology alternatives, and attributes and associated levels, DCE choice sets comparing two technologies in each choice set were designed using aforementioned attributes without specifying the name of the tool. In many DCEs, choice sets are developed by randomizing all identified attributes across the treatment/technology alternatives to assess participants’ preferences for specific attributes. In this study, the four non-price attributes are likely inter-related and caregiver preferences are likely to exhibit complex attribute interactions. For instance, it is possible that measuring medication adherence digitally may be of low value if this information is not delivered in a timely manner; similarly measuring medication adherence information rapidly may be of low value if it is based on self-reported data. To adjust for non-linear preferences, the four non-price attributes including- source of medication adherence information, frequency at which information is updated by the tool, how medication adherence information is accessible to the caregiver, and reporting of patients’ physical activity, mood and quality of rest information were combined into the tools’ attributes bundle (see Table 1). Each choice set comparing two technology alternatives involved randomizing the tools’ attributes bundle and caregiver monthly out-of-pocket cost. The levels for monthly out-of-pocket cost attribute were $10, $40, $120, $200 and calibrated to non-digital pill organizer, mobile app, smart pill dispenser and pill embedded with IEM sensor, respectively. Non-zero dollar amounts were defined for monthly out-of-pocket cost attribute levels as previous studies have shown that free products were not valued the same as non-free goods 38 (See Figure 1). The DCE was designed using the AlgDesign software package for R based on the D-efficiency criteria.

Example of discrete choice scenario administered to caregivers in the study.
In addition to the DCE module, the survey instrument included an eligibility screener, and additional modules to obtain information on caregiver sociodemographic characteristics, their current use of medication adherence monitoring tools, and their relationship with the patient with SMI that they cared for. The entire survey instrument was designed to be completed in about 20 min.
Data collection
The survey was fielded to a convenience sample of caregivers. We partnered with a survey vendor to recruit potentially eligible participants from their proprietary panels. Participants are recruited into panels through (1) offline methods including phone and in-person recruitment at academic and professional conferences, (2) online methods including targeted e-mail campaigns, social media, direct marketing, and banner placements, (3) using special interest groups, industry associations, and affinity groups, and (4) referrals from other panel members. An invitation to participate in the survey was sent to all participants in the vendor's panels. Participants who expressed interested were directed to provide their informed consent. Participants who provided informed consent were then asked a series of questions to determine their eligibility. All eligible caregivers were given access to the full survey to provide their responses.
Sample size calculations were conducted using the method described from Louviere et al. 39 and it was estimated that n = 147 caregivers would be sufficient to power the analysis. Equal sample quotas of caregivers were set based on type of SMI diagnoses of the patient for whom they provided care. All eligible caregivers who successfully completed the survey were compensated for their time and effort. The Advarra Institutional Review Board reviewed and approved all study procedures.
Statistical analysis
Data collected from participants’ responses to the survey instrument were checked for lexicographical preferences. Descriptive analyses were conducted and mean and standard deviations (SD) for continuous variables, and frequencies and proportions for categorical variables were presented. A random parameters logit model was used to estimate the utility weights. A preference weight (reported as odds ratio (OR)) for each technology tool was estimated using the responses to DCE choice sets in the survey. WTP (reported as USD) for each adherence monitoring tool was calculated using the ratio of estimated utility weight of the tools’ attributes bundle and the disutility weight of the monthly out-of-pocket cost attribute. Furthermore, preference shares for the four adherence monitoring tools were assessed.
Preferences and WTP for adherence monitoring tools may differ across caregivers’ subgroups due to the heterogeneous nature of who decides to become a caregiver for a patient with SMI. To address this, subgroup analyses were conducted to evaluate how caregiver preferences and WTP for adherence monitoring tools varied based on (1) SMI diagnosis of the patient, (2) annual household income, and (3) relationship with the patient.
Results
Sample selection
Of the 1320 individuals who indicated interest in participating in the study, 693 failed to either provide informed consent or did not meet the inclusion criteria. Additionally, 246 caregivers who agreed to participate were excluded once a priori sample quotas for each SMI type were met. Of the 381 caregivers who were given full access to the survey, 176 did not provide complete responses and were excluded. The initial study sample included 205 caregivers who met the study criteria and completed the survey. Twenty-one of these respondents who either took unusual time (<4 min and >2 h) in completing the survey (n = 16) or who reported their patients taking LAIs along with oral atypical antipsychotic medication (n = 5) were further excluded resulting in the final sample of 184 caregivers with complete information. The final 184 analyzed is still well above the 147 required for our study to be sufficiently powered. No caregivers were detected as having lexicographic preferences.
Sample description
A majority of caregivers were female (59%), Caucasian (69%), married (69%), had some college education (72%), and reported >$75,000 mean annual household income (47%). One-third of caregivers were parents of the patients with SMI, 30% were spouses/partners, 14% were children, and 22% were other types of family members. It should be noted that we did not purposively exclude non-family members from participation in the survey, and potential participants could also include friends, neighbors, or other unpaid caregivers. However, the analytic sample ultimately included participants who reported being related to the individual with SMI. Among the 184 caregivers, 57 (31%), 61 (33%) and 66 (36%) provided care to patients diagnosed with BPD, MDD and SCZ, respectively. Sixty four percent of the caregivers reported using medication adherence tools, and among them, 85% used the tools daily. In addition, although not mutually exclusive, a majority of caregivers reported using a non-digital pill organizer (63%), followed by diary (46%), smart pill organizer (44%), mobile app (30%) and paper and pencil (28%) (Table 2).
Caregiver sociodemographics, caregiving characteristics, and utilization of adherence monitoring tools, N = 184.

IEM, Ingestible Event marker; MEMS, Medication Event Monitoring System.
Family member other than spouse/partner, parent, or child.
Not mutually exclusive.
118 caregivers reported using adherence-medication monitoring tools, and used as denominator.
Caregivers’ preferences for digital adherence monitoring tools
Overall, caregivers highly preferred digital adherence monitoring tools as compared to non-digital. The disutility weight of the out-of-pocket cost was 0.0078 (95% confidence interval (CI): 0.011, 0.0038). Caregivers of patients with SMI highly preferred (OR 7.34, 95% CI: 5.00–10.79) a pill embedded with IEM sensor and were willing to pay $255.04 (95% CI $123.21–$386.86) per month more for this tool than a non-digital pill organizer. Similarly, caregivers had 2.64 times higher preference (95% CI: 1.90–3.70) with a WTP $124.50 (95% CI: $48.18–$200.81) per month more for a mobile app relative to a non-digital pill organizer. The smart pill organizer was found to be less preferred by caregivers (OR) 0.54 95% CI: 0.40–0.73) with a WTP of −$78.40 (95% CI: −$129.88, −$26.92) per month, which can be interpreted as needing to pay a caregiver $78.40 per month to use the smart pill dispenser (Table 3).
Caregivers’ preferences and willingness to pay for different health technology tools to monitor medication adherence.

IEM means Ingestible Event Marker; *P-value < 0.05.
When stratified by primary diagnosis, caregivers of patients with BPD and MDD highly preferred a pill embedded with IEM sensor (OR 7.47, 95% CI: 3.81–14.65 for BPD; OR: 9.79, 95% CI: 4.81–19.9 for MDD). They were willing to pay more for a pill compared to the non-digital pill organizer [$197.48/month (95% CI: $50.91–$344.04) among caregivers of BPD patients, and $227.76/month (95% CI: $66.04–$389.47) among caregivers of MDD patients]. Caregivers of patients with SCZ also highly preferred a pill embedded with IEM sensor (OR 6.71, 95% CI 3.29–13.69), but the WTP was not found to be statistically significant when compared to the non-digital pill organizer. The inability to find statistically significant results is likely because the subgroup analyses are underpowered. The power sample calculation found that 147 individuals per subgroup would be needed for the analyses to be fully powered. Further, subgroup analyses by relationship found that parents, spouses, or child caregivers of patients with SMI highly preferred a pill embedded with IEM sensor than a non-digital pill organizer. However, WTP was found to be significant only for children of the patient, where they were willing to pay $227.27 (95% CI: $15.78–$438.76) per month more for the pill with IEM than a non-digital pill organizer. Compared to a non-digital pill organizer, higher preferences and WTP for a pill embedded with IEM sensor was found among caregivers irrespective of income groups (Supplementary Table 1).
Based on these preferences, a plurality of caregivers preferred a pill embedded with IEM sensor. Specifically, 39.5% of caregivers preferred a pill embedded with IEM sensor followed by a mobile app (26.6%) and a non-digital pill organizer (23.8%) (Figure 2).

Preference shares of different health technology tools used to monitor medication adherence among caregivers in the study.
Discussion
To our knowledge, this is the first study to estimate preferences and WTP for medication adherence monitoring tools among caregivers of SMI patients. Previous studies have demonstrated the value of medication adherence measurement tools in other diseases40–43; however, evidence in the SMI space and in particular from the informal caregiver's perspective is limited. Effective psychopharmacotherapy requires patients with SMI to take their medication consistently. However, it is often difficult for caregivers to track medication adherence of these patients.44,45 Advancements in medical technology have resulted in the emergence of novel health technology tools that track medication adherence through data on pill ingestion. 26 In this study, caregivers highly preferred a pill embedded with IEM sensor that tracks not only medication ingestion but also tracks patients’ physical activity, quality of rest, and self-reported mood. They were willing to pay $255 per month more for this tool than the non-digital pill organizer.
Non-adherence to medication remains a major obstacle to improving health outcomes among patients with SMI, contributing to significant direct and indirect costs.13,46 Underlying SMI condition and symptom severity, social support, socioeconomic status, complexity of adherence interventions, and heterogeneous beliefs in measurement of adherence adds to the complexity of understanding medication non-adherence among these patients.45,47,48 Previous studies have demonstrated that digital health tools have the ability to improve medication adherence in several therapeutic areas including cardiovascular diseases, diabetes and other chronic conditions, and rare autoimmune diseases.40,41,49,50 Furthermore, evidence has been emerging on the value of adherence monitoring tools in clinical practice among patients and providers.27–29,51 However, evidence on the value of such tools from caregiver perspective is lacking, and the current study address this gap.
The study findings demonstrated that caregivers preferred and were willing to pay more for a digital health tool that tracked medication ingestion and SMI patients’ general health compared to a non-digital pill organizer. These findings are consistent with previous studies evaluating caregiver preferences for digital health tools for caregiving of individuals diagnosed with chronic diseases or behavioral conditions.52,53 A contingent valuation study of caregivers of patients with dementia found a positive WTP for new technologies that aid in conversations with their patients and provide medication reminders. 54 In addition, caregivers who were children of patients with SMI highly preferred and were willing to pay more for a pill embedded with IEM sensor and a mobile app, which can be attributed to their age. Younger individuals are likely to be more comfortable using and be willing to pay for digital adherence tools than non-digital tools.43,55 In general, all caregivers in the study, irrespective of their relationship with their SMI patient, highly preferred the pill embedded with IEM sensor to monitor medication adherence in their patients. Overall, the study findings demonstrated the value that caregivers place on digital adherence monitoring tools that track medication ingestion along with other general health measures.
Although evidence is emerging to support the value of digital health technologies in SMI and other disease areas, little research has been conducted on the broader economic impact of these interventions. More research is needed to better understand their benefit and sustainability to quantify their potential cost-savings.56,57 While this study does not directly estimate the broader economic impact of digital health tools used for medication adherence, the positive estimates of value indicates that digital health tools are could potentially have an impact on cost-savings or decreasing burden. Future research on the estimating the magnitude of burden reduction or cost-savings is generated from digital health tools is needed.
Ultimately, caregivers’ preferences and value for these technologies are only one stakeholder perspective on the overall value of digital health technologies. While this study was focused on caregivers, capturing patient input remains critical not only for the valuation and development of digital health technologies but to ensure long- and short-term engagement. Several studies have sought to incorporate patient input around specific features associated with digital health technologies across disease areas,58–61 suggesting that patients prefer digital health technologies primarily for its convenience and communication.62,63 In the SMI space, a recent qualitative study of patients with SMI, found that with respect to digital health tools, data privacy is a major concern for patients with SMI. 64 Additional work should be conducted to capture the perspectives of and preferences for features and attributes of digital health technologies among patients diagnosed with SMI.
Strengths and limitations
We believe that this study is the first to attempt to quantify caregiver WTP for digital health technology. The discrete choice experiment approach employed is a well-established methodology for quantifying preferences for digital health technology while removing external decisional influences. As such, this approach is better suited to determine preferences than a real-world study of treatment patterns. For example, a caregiver may not have a smartphone or access to internet; thus, the real cost of using an app would be higher for individuals without a smartphone or internet access compared to those with access to such technologies. Measuring the actual consumption behavior may not detect an individual's inherent preferences of the goods. The hypothetical choice sets presented in a DCE allows the observation of an actual choice, as the cost of the good is similarly experienced by all participants, which allows for a more accurate estimate of preferences.
The study has several limitations that merit discussion. First, this study bundled individual attributes into tools’ attributes bundle prior to randomization, which allowed us to account for nonlinearities between specific tool features. This approach, however, prevented us for being able to estimate the preferences and WTP for individual attributes separate from the tools’ attributes bundle. Second, the DCE methodology requires the respondent to make hypothetical decisions by evaluating the trade-offs between attributes in a choice set, which differs from real-world scenarios where a myriad of other factors influence preferences and demand. Despite this inherent limitation, the need for DCEs in health care technology assessment continues to grow and is one of the frequently used methodologies to generate evidence in treatment decision-making.65,66 Third, our study only examined U.S. caregivers of patients with SCZ, BPD or MDD, and thus the results may not be extrapolated to caregivers of patients with other types of mental illness, other chronic medical conditions or caregivers in other countries or paid caregivers. Fourth, our study only included caregivers of patients with SMI and receipt of second generation oral atypical antipsychotics as inclusion criteria, thus the results may not be extrapolated to caregivers of patients on other antipsychotic or antidepressant medications. Fifth, caregivers in our study were recruited through convenience samples from the survey vendor's proprietary panels, limiting the generalizability of the study findings to all caregivers who care patients with SMI. Related to the recruitment, while this study did allow for non-family informal caregivers to participate in the study, our analytical sample coincidentally ended up with only family informal caregivers. While family members are most likely to be informal caregivers, 67 we acknowledge our final sample may not be representative of informal caregivers in the real world setting.
Finally, the nature of our recruitment methods meant that respondent caregivers had access to the Internet. Further, the caregivers were generally highly educated, predominately White, and relatively high income. Thus, this group may not be fully representative of the US population of SMI caregivers. However, in our subgroup analysis, we did find that lower-income caregivers had a positive and statistically significant WTP for the pill with IEM. While our sample does draw upon participants with higher incomes, WTP would likely still remain positive if our sample had included more lower-income respondents. Despite these limitations, the study adds considerable value to the literature on caregiving, medication adherence among patients diagnosed with SMI and value of new technological advances in clinical decision-making.
Conclusion
Compared to a non-digital pill organizer, caregivers of patients with SMI had 7.3 times higher preference for a pill embedded with IEM sensor that tracks not only medication ingestion but also patients’ physical activity, quality of rest and self-reported mood, and were willing to pay $255 per month more for this tool. The study findings suggest that caregivers of patients with SMI place a high value on digital medication adherence monitoring tools. Future research studies could consider examining which specific aspects of new digital technologies are most valuable to the caregivers and identify opportunities and barriers to adoption of medication adherence monitoring digital tools in clinical care management.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076221084472 - Supplemental material for Are caregivers ready for digital? Caregiver preferences for health technology tools to monitor medication adherence among patients with serious mental illness
Supplemental material, sj-docx-1-dhj-10.1177_20552076221084472 for Are caregivers ready for digital? Caregiver preferences for health technology tools to monitor medication adherence among patients with serious mental illness by Felicia Forma, Kevin Chiu, Jason Shafrin, Dusica Hadzi Boskovic and S Phani Veeranki in Digital Health
Supplemental Material
sj-docx-2-dhj-10.1177_20552076221084472 - Supplemental material for Are caregivers ready for digital? Caregiver preferences for health technology tools to monitor medication adherence among patients with serious mental illness
Supplemental material, sj-docx-2-dhj-10.1177_20552076221084472 for Are caregivers ready for digital? Caregiver preferences for health technology tools to monitor medication adherence among patients with serious mental illness by Felicia Forma, Kevin Chiu, Jason Shafrin, Dusica Hadzi Boskovic and S Phani Veeranki in Digital Health
Footnotes
Acknowledgements
The authors would like to thank all participants who participated in the study. In addition, the authors would like to acknowledge Richard Murphy, Natalie Land, and Kelly Birch at PRECISIONheor for their assistance in drafting the manuscript.
Author contributions
All authors contributed to one or major sections of the study. The study was conceptualized and designed by FF, KC, JS, and PV. Survey administration, data collection, management and analyses, and results interpretation were conducted by KC and PV with support from JS, DH and FF. PV drafted the manuscript draft with support from KC, JS, DH, and FF. All authors contributed to manuscript revisions and approved the final manuscript draft.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PRECISIONheor received funding from Otsuka Pharmaceutical Development & Commercialization Inc. to conduct this research. KC is an employee of PRECISIONheor, PV and JS are former employees of PRECISIONheor. DH is an employee of Otsuka Pharmaceutical Development & Commercialization, Inc., and FF is a former employee of Otsuka Pharmaceutical Development & Commercialization, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Otsuka Pharmaceutical Development & Commercialization Inc.
Ethics approval
The study received approval from Advarra Institutional Review Board.
Informed consent
Informed consent was obtained from participants prior to enrollment and survey administration.
Trial registration
Not applicable, because this article does not contain any clinical trials.
Guarantor
KC.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
