Abstract
Case summary
A 7-month-old intact male Maine Coon cat was presented because of intermittent pyrexia, hyporexia and lethargy. On abdominal palpation, both kidneys appeared to be enlarged with an irregular shape. Haematology and serum biochemistry showed anaemia, hypoalbuminaemia and hyperproteinaemia. Ultrasonographic findings included multiple perirenal cyst-like lesions, heterogenous cortices with cortical striation and a medullary band sign, as well as renomegaly. The cat was euthanased and a limited necropsy of the kidneys revealed pyogranulomatous lesions, which were confirmed to be related to feline infectious peritonitis (FIP) by immunohistochemistry.
Relevance and novel information
To the authors’ knowledge, this is the first published report describing perirenal cyst-like lesions of the kidneys related to FIP.
Case description
A 7-month-old intact male Maine Coon cat presented to a local veterinarian with pyrexia and mucopurulent nasal and ocular discharge. Treatment with doxycycline (11 mg/kg q12h) was initiated without further diagnostic tests or examinations. The cat developed additional clinical signs of lethargy and anorexia and was presented by the owner to the AniCura Albano Animal Hospital, Sweden, for a second opinion. At the time of presentation, the cat had had a history of intermittent pyrexia for 1 week and had been treated with doxycycline for 6 days without any improvement.
On presentation to the AniCura Albano Animal Hospital, the cat was lethargic with pale mucous membranes, mild tachypnoea (respiratory rate 40 breaths/min) and mucopurulent nasal and ocular discharge. On abdominal palpation, both kidneys were enlarged with an irregular shape. Cardiac and pulmonary auscultations were unremarkable, and body temperature was normal (38.5° C). The cat showed mild ataxia and experienced a seizure of short duration (<1 min) with myoclonus and unconsciousness during the visit. The cat was initially treated with intravenous fluid (Ringer-acetat; Fresenius Kabi) and flow-by oxygen.
Haematology showed moderate anaemia (haematocrit 17.6%, reference interval [RI] 30–52; red blood cells 4.49 × 1012/l, RI 6.5–12.2) and lymphopenia (1.4 × 109/l, RI 1.5–7). Biochemistry revealed elevated serum amyloid A (38.7 mg/l, RI <10), hypoalbuminaemia (albumin 19.8 g/l, RI 27–37), hyperproteinaemia (total protein 108 g/l, RI 57–80) and normal glucose (5.2 mmol/l, RI 3.4–8.5), creatinine (58.1 µmol/l, RI <160), urea (5.8 mmol/l, RI 4–13), phosphate (2.08 mmol/l, RI 1–2.2), alanine aminotransferase (0.84 µkat/l, RI <1.2), alkaline phosphate (0.23 µkat/l, RI <1) and bile acids (4.4 µmol/l, RI <20). The cat tested negative for feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) on the SNAP Combo plus test for FIV/FeLV (IDEXX).
An abdominal ultrasound was performed using a Logiq9 11L linear transducer (GE Healthcare). The cat was awake during the examination. Both kidneys measured approximately 6 cm in length with an irregular shape caused by multiple perirenal cyst-like lesions. The outline of the cyst-like lesions was smooth, and they were filled with anechoic to echogenic fluid and had a diameter in the range of 0.5–4 cm. Some of the cyst-like lesions were confluent with each other and it could not be determined whether these were of cortical or perirenal origin. The cyst-like lesions contained soft tissue septations and debris (Figure 1). Doppler examination was not performed, hence a distinction between debris and vascularised tissue could not be made. The renal cortices were heterogeneously hyperechoic, with cortical striation and a hyperechoic medullary band measuring 1.8–2.5 mm in width (Figure 2). The perirenal fat was hyperechoic with a mild amount of anechoic retroperitoneal fluid interspersed. Mild abdominal lymphadenomegaly and a small amount of anechoic peritoneal effusion were present. The peritoneal effusion pockets measured < 5 mm in two regions.

(a,b) Multiple confluent cyst-like lesions adjacent to the renal cortex with the presence of soft tissue septations and debris (*)

Heterogenous hyperechoic cortex with cortical striation and hyperechoic medullary band
It was not possible to aspirate the peritoneal fluid; however, aspiration from one of the cyst-like lesions revealed a clear yellow sticky fluid.
Based on available information, the major differential diagnoses were feline infectious peritonitis (FIP), lymph-oma or infection with FeLV or FIV. As the FeLV/FIV SNAP test was negative, the most likely differential was considered to be FIP. Minor differential diagnoses included other infectious agents (such as Toxoplasma, Clamydophila felis, Mycoplasma haemofelis or Mycoplasma felis) or meningoencephalitis (infectious and non-infectious). The owner declined further diagnostic tests, such as analysis of the fluid, electrophoresis and urinalysis, and the cat was euthanased at the owner’s request. No neurological and ocular examinations were performed.
Because of financial constraints, only a limited necropsy of the kidneys was performed. The necropsy revealed a renal parenchyma interspersed with nodular to diffuse aggregates of granular white dense tissue. The changes were more pronounced in the left kidney. Subcapsular cyst-like lesions with a diameter of 1.5–4 cm with multifocal adhesions to the renal cortex were present in both kidneys. A cut section of the kidneys revealed that cystic spaces did not involve the renal parenchyma (Figure 3).

Cut surface of the left kidney. Replacement of the renal parenchyma with nodular to diffuse white aggregates (pink arrow). Presence of multiple remnants of the cystic structures (green arrows)
A histological examination of the kidney displayed a diffuse inflammatory and necrotising process, mainly present in the renal interstitium, which was composed of numerous macrophages mixed with viable and degenerated neutrophils (ie, pyogranulomatous inflammation [Figure 4a]), with a few lymphocytes and plasma cells. The inflammation was surrounded by areas of necrosis (Figure 4b) often mixed with fibrin deposits. Between the areas of inflammation and necrosis, hypereosinophilic remnants of suspected vascular structures were present. Expansion of the area between the capsule and renal parenchyma with the presence of necrosis and fibrin deposition was noted. In conclusion, the changes were interpreted as moderate to severe multifocal chronic pyogranulomatous nephritis with vasculitis.

(a) Microscopic image of the affected kidney showing severe involvement of the renal parenchyma by the inflammatory process (H&E, × 62). (b) Higher magnification of the affected kidney showing pyogranulomatous inflammation centred on necrosis and fibrin accumulation with remnants of vascular structures (H&E, × 280). H&E = haematoxylin and eosin
A histopathological diagnosis of marked diffuse pyogranulomatous and necrotising nephritis was made. After the histological examination, an automated immunohistochemical examination (Autostainer Link 48; Agilent Technologies Sweden) was performed using monoclonal primary antibody FIPV3-70 (Bio-Rad) with specificity to the nucleocapsid of FIP virus types I andII, which was visualised using a polymer detection system (Dako EnVision+/HRP, Mouse, HRP. Mouse, Immunohistochemistry Visualization, K400111-2; Agilent Technologies Sweden) followed by application of the chromogen diaminobenzodine (DAB) (Dako Liquid DAB+ Substrate Chromogen System, K346811-2; Agilent Technologies Sweden). Immunohistochemical examination showed the presence of feline coronavirus (FCoV) antigen within the tissue in association with the lesions (Figure 5).
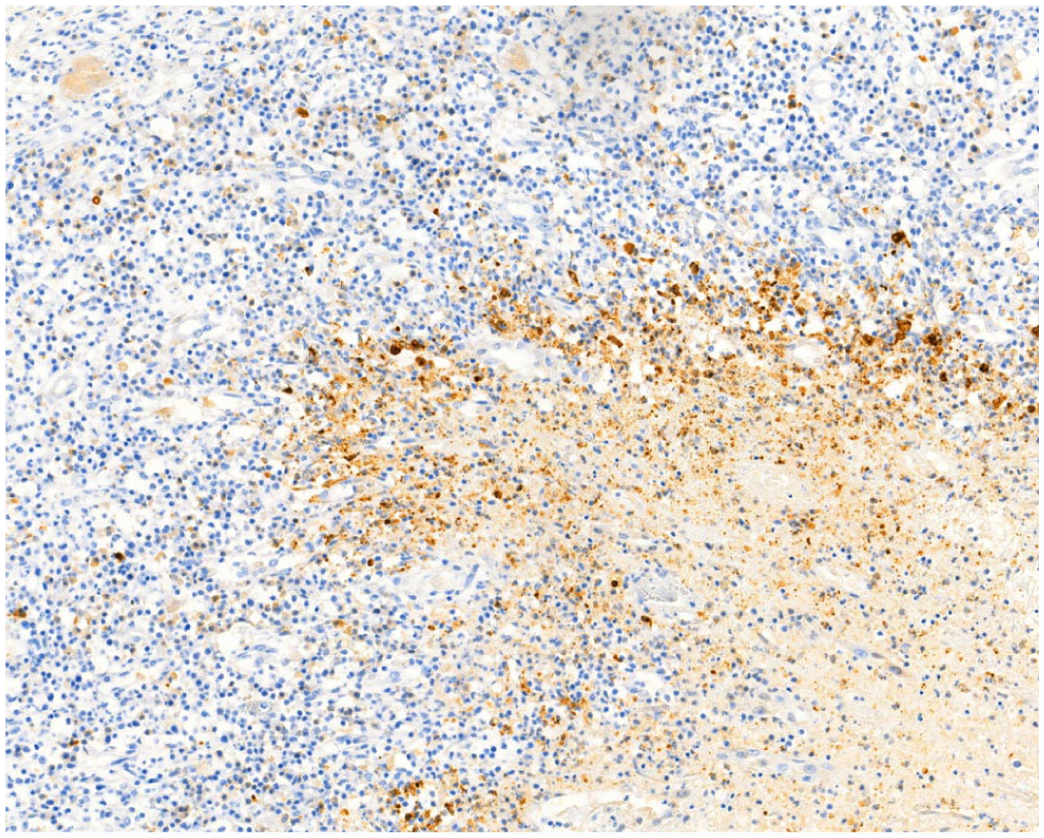
Immunohistochemical staining for feline coronavirus (FCoV) antigen in affected kidney showing the presence of marked positivity for FCoV antigen in the areas surrounding and within the inflammatory process (diaminobenzodine, × 282)
A post-mortem genetic test for the most prevalent polycystic kidney disease (PKD) gene was performed on formalin-fixed paraffin-embedded tissue at the Swedish University of Agricultural Sciences, yielding a negative result.
Discussion
To the best of our knowledge, this is the first report describing perirenal cyst-like lesions associated with FIP. Non-specific sonographic renal changes have previously been described in cases of FIP, including renomegaly, cortical striation, hyperechoic cortex, loss of corticomedullar definition, cortical nodules and a medullary rim sign.1–5 Occasionally, a thin hypoechoic subcapsular rim has been described.1,2,5 Commonly reported sonographic changes related to FIP also include anechoic or echogenic peritoneal effusion and non-specific changes in the liver, spleen, lymph nodes and intestines.1,2 In our case, there was a strong clinical suspicion of FIP. Histopathological changes associated with FIP often include pyogranulomatous inflammation, but the final diagnosis is based on immunohistochemistry of affected tissue.4,6 The histopathological findings of pyogranulomatous inflammation in this case are consistent with those described in FIP and immunohistochemistry confirmed the diagnosis.
Perirenal changes have previously been described, most commonly with perinephric pseudocysts in older cats with varying underlying chronic renal disease.7–9 The sonographic and histological appearances in our case do not correspond to the description of perinephric pseudocysts. In addition, the aspirated fluid from the cyst-like lesion in our case had a clear sticky yellow appearance. Although no further analysis was conducted, the macroscopic appearance of the fluid corresponded to that previously described in cats with FIP, which is often a protein-rich modified transudate or exudate.4,6 In cases of perirenal pseudocysts, the fluid consists of a transudate with a low cell count and low protein concentrations.7–9 Thus, the appearance of the fluid in our case does not correspond to previous descriptions of perinephric pseudocysts.
Cystic renal changes are most often described in association with PKD. 5 PKD is characterised by multiple cystic lesions located within the renal cortex,5,10 but not, as was seen in this case, adjacent to the cortex. Although sonographic examination of the kidneys is considered to have high specificity and sensitivity for the diagnosis of PKD, the disease is slowly progressive, and genetic testing is considered to be the diagnostic method of choice in young animals. Therefore, a post-mortem genetic test was performed and the cat was negative for the PKD1 gene, which is considered to be the most prevalent. 10
A medullary rim sign has previously been reported in cases with a wide range of different renal pathologies but is also considered to be a normal finding.3,11–13 Cordella et al 11 proposed that the term ‘medullary band sign’ should be used when the width of the rim exceeds 2 mm, as seen in our case. In their study, a medullary band sign was frequently more associated with kidney disease. 11 This correlates well with our case of pyogranulomatous nephritis related to FIP.
Renal and perirenal sonographic changes were present in the cat in this report, but without signs of azotaemia. Urinalysis was not performed, hence there is a limitation regarding evaluation of renal function. To reach a final antemortem diagnosis, further tests would have been required. For example, FCoV RT-PCR, cell count and protein concentration in fluid from multiple perirenal cyst-like lesions and the peritoneal effusion would have been of great value because of the high suspicion for FIP.
The fact that neurological and ophthalmic examinations were not performed is acknowledged to be a limitation of this case report. Other tests, such as electrophoresis and MRI of the brain, may have yielded useful information. A full necropsy, including histopathology of all the organs including the lymph nodes and brain, might also have provided more information.
Conclusions
Herewith we describe unusual ultrasonographic changes of the kidneys and perirenal area associated with FIP, confirmed by histopathology and immunohistochemistry. When perirenal cyst-like lesions are present, FIP should be considered as a differential diagnosis.
Footnotes
Acknowledgements
Thanks to Åsa Ohlsson at the Swedish University of Agricultural Sciences for their contribution to the genetic testing for the PKD1 gene.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
