Abstract
Case summary
Dynamic intrapelvic urinary bladder displacement (DIUBD) is an uncommon condition where the urinary bladder intermittently displaces caudally into the pelvic canal, resulting in episodic lower urinary tract obstruction. Although this phenomenon is recognized more commonly in canine patients with perineal hernias, documented feline cases remain rare. Trauma or pelvic fractures can compromise the strength and conformation of local musculature and fascia, permitting the bladder to slip caudally under changing pressure gradients. In this case, an 8-year-old spayed female domestic shorthair cat, previously affected by multiple healed pelvic fractures, developed progressive urinary and defecatory straining. Initial imaging, including abdominal radiographs and ultrasonography, confirmed a caudally displaced bladder occasionally reverting to a more cranial position. Blood work and repeated urinalyses indicated post-renal azotemia on re-presentation. Surgical exploration revealed a mobile bladder without a distinct muscular defect, prompting cystopexy to anchor the bladder in a stable cranial position. Postoperative monitoring showed rapid improvement in clinical signs and renal values, and follow-up imaging confirmed that the bladder retained its corrected orientation. This case supports the importance of thorough imaging and an early corrective procedure for cats with recurrent lower urinary obstruction and a history of pelvic fractures.
Relevance and novel information
Although DIUBD is more often recognized in dogs, this case indicates that it can similarly arise in cats with a history of pelvic trauma. Targeted imaging coupled with cystopexy can successfully address the intermittent obstruction.
Introduction
Dynamic intrapelvic urinary bladder displacement (DIUBD) refers to a scenario where the urinary bladder intermittently shifts caudally into the pelvic canal, rather than maintaining a consistent, intra-abdominal position.1,2 Although malposition of the bladder in small animals has been predominantly documented in association with canine perineal hernias, relatively few feline cases have been reported.1,3 The scarcity of this condition reported in feline patients could impact on the diagnosis of DIUBD in cats presenting with episodic urinary obstruction. Prompt recognition of DIUBD is important as obstruction and incomplete bladder emptying can lead to complications such as hematuria, bladder wall inflammation and renal compromise from back pressure. 4 In humans, weakened pelvic musculature can cause intermittent displacement of organs in this region. 5 Although anatomy and clinical presentations differ across species, the underlying issue of compromised and inadequate support is comparable. This report details an 8-year-old spayed female domestic shorthair cat presenting with progressive stranguria and ultimately diagnosed with DIUBD. Cystopexy was performed to secure the bladder in a stable cranial position with no recurrence of stranguria episodes 6 months postoperatively.
Case description
An 8-year-old spayed female domestic shorthair cat arrived at an emergency veterinary referral hospital because of progressively worsening tenesmus, stranguria, vocalizing during urination attempts and incomplete defecation for several weeks, with a marked decline in the last 24 h. Reportedly, the cat’s history included multiple healed pelvic fractures, likely associated with trauma before adoption at 2 years of age, a non-reducible umbilical hernia and a previous tail amputation due to historical self-mutilation. Although the client noted a mildly altered gait since adoption, no prior urinary obstruction episodes had been observed. At the referral facility, the cat appeared bright, alert and responsive while maintaining a hunched posture. The cat had a body condition score of 5/9 6 and exhibited a heart rate of 180 bpm, respiratory rate of 36 breaths/min and a rectal temperature of 100.2°F (37.9°C). A chemistry panel revealed a creatinine level of 0.9 mg/dl (reference interval [RI] 1.0–1.6) and an elevated blood urea nitrogen of 34.2 mg/dl (RI 15–32). and a venous blood gas showed a negative base excess of –9.1 (RI –4 to 4), indicating mild metabolic acidosis. A cystocentesis-based urinalysis produced a urine specific gravity (USG) of 1.030 (RI ⩾1.030), pH of 6 (RI 6.0–7.5) and evidence of 2+ granular casts. Abdominal radiographs demonstrated an intrapelvic bladder and multiple healed pelvic fractures, while an abdominal ultrasound documented intrapelvic urinary bladder, an umbilical hernia and mild bilateral pyelectasia. Initially diagnosed with idiopathic cystitis, the cat was discharged with analgesics including buprenorphine (0.01 mg/kg transmucosally q8h) and robenacoxib (1.5 mg/kg PO q24h for 3 days).
Later that day, the owner brought the cat to the veter-inary teaching hospital because of continued vocalization and failure to urinate. On examination, the cat was tachypneic with intermittent open-mouth breathing after straining, painful on perineal palpation and had a rectal temperature of 100.2°F (37.9°C). Venous blood gas showed a mildly elevated creatinine of 2.03 mg/dl (RI 1.0–2.7), raising concern for urinary obstruction. A 15 cm, 3.5 Fr MILA indwelling urinary catheter was placed. Thoracic radiographs were unremarkable. Initial lateral abdominal radiographs showed a cranially positioned bladder with the catheter tip abutting the bladder wall and no urine output. After repositioning the catheter, follow-up radiographs revealed caudal bladder displacement into the pelvic canal and colonic distension (Figure 1). Urinalysis showed USG 1.046, pH 7.0, 1+ protein, 3+ blood, 20–99 red blood cells/high-power field (HPF), 0–3 white blood cells (WBCs)/HPF, 0–1 WBC casts/low-power field, and no bacteria or crystals. Hospital treatments included Plasma-Lyte-R (2 ml/kg/h IV), buprenorphine (0.02 mg/kg PO q8h), gabapentin (10 mg/kg PO q12h), robenacoxib (1.5 mg/kg PO q24h) and prazosin (0.125 mg/kg PO q12h). Although catheterization provided partial relief, straining and lethargy persisted. Repeat ultrasound showed a caudally displaced bladder with perivesicular free fluid, rectal wall edema (Figure 2) and mild bilateral hydronephrosis. Sedated rectal exam revealed intact perineal support structures. All radiographic and ultrasonographic images were captured and interpreted by a board-certified veterinary radiologist.
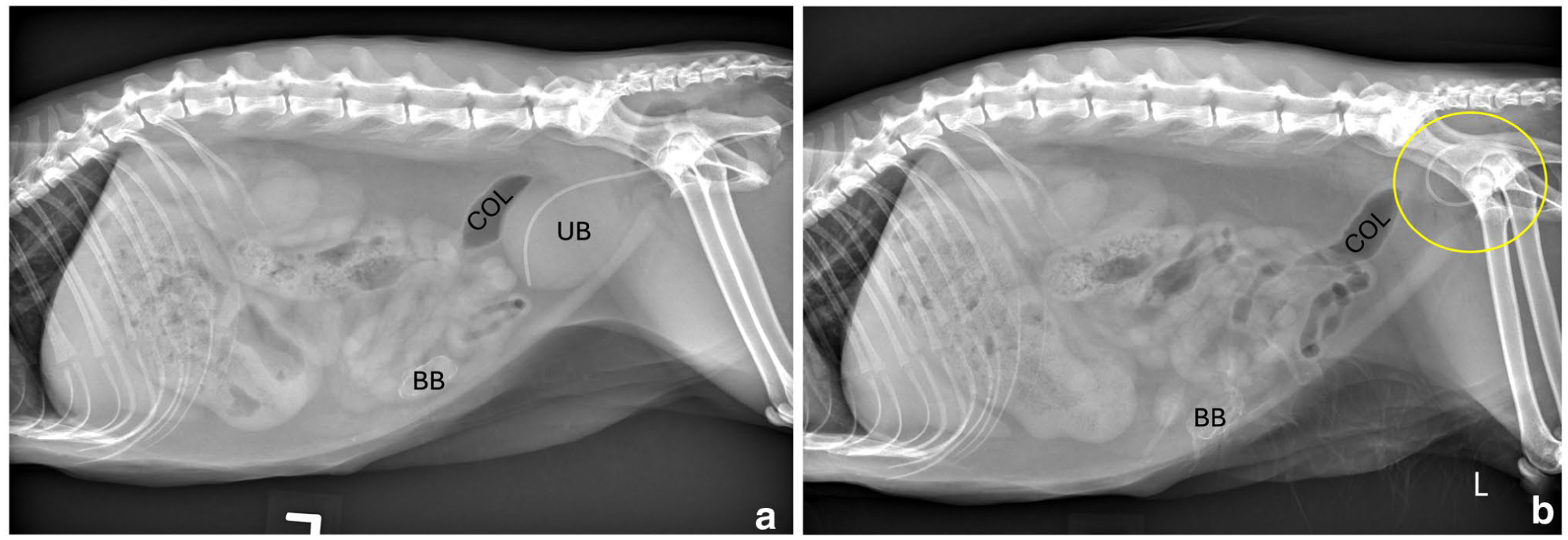
Left lateral abdominal radiographs of a cat after urinary catheter placement (a) and after drainage of the UB (b). (a) Lateral radiograph demonstrating a moderately distended UB in the typical intra-abdominal location. (b) Repeat radiograph after UB drainage demonstrates the radiopaque urinary catheter retroflexed within the pelvic canal, with the distal tip directed caudally and no distinct visualization of the UB, suggestive of intrapelvic bladder displacement (circled). The colon remains normal in position and diameter across both views. Additional findings include an incidental BB, moderate gastric distension with gas and soft tissue/fluid opacity, mild cortical irregularity of the left kidney and ventral spondylosis deformans at the lumbosacral junction. BB = Bates body; COL = colon; L = left; UB = urinary bladder
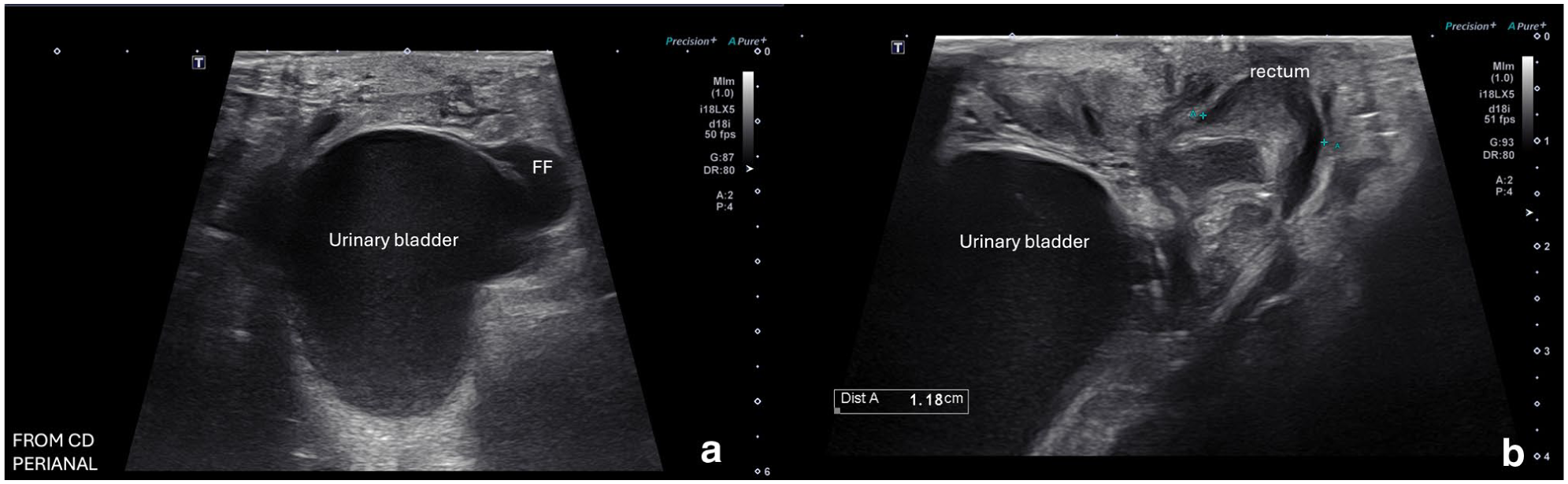
Perineal ultrasound images of the pelvic cavity obtained in a caudal-to-cranial orientation: (a) transverse plane image demonstrating the urinary bladder positioned within the pelvic canal surrounded by a small volume of anechoic FF; and (b) longitudinal plane image showing the urinary bladder in close apposition to a moderately thickened, hypoechoic rectal wall, consistent with mural edema. FF = free fluid
As a result of persistent obstructive signs and confirmation of bladder mobility on imaging, the cat underwent an exploratory laparoscopy with a board-certified veterinary surgeon and in-training resident. Under general anesthesia, controlled and monitored by a board-certified veter-inary anesthesiologist, a 3 cm right paramedian, caudal abdominal incision was made using monopolar electrocautery. A single-incision laparoscopic surgery (SILS) port with three 5 mm cannulae was inserted, and the abdomen insufflated to 8 mmHg with CO₂. Laparoscopic examin-ation using a 5 mm, 0° laparoscope revealed a caudally displaced and erythematous bladder with no evidence of necrosis. The bladder was noted to have increased mobility and was visualized to displace caudal and torse along the longitudinal axis causing obstruction of the uretero-vesicular junction. No other abnormalities of the caudal pelvic diaphragm and urinary bladder, including the ventral ligament, were appreciated.
A laparoscopic-assisted cystopexy was then performed. The SILS port was removed and stay sutures using 4-0 Biosyn were placed on the bladder apex for retraction (Videos 1–3 in the supplementary material). A 2 cm partial-thickness incision was made in the ventral bladder seromuscular layer and aligned with a matching incision on the right paramedian body wall. A cystopexy was performed using 4-0 Maxon in a combination of interrupted and cruciate patterns. The body wall was closed in three layers: 3-0 Maxon (body wall), 4-0 Biosyn (subcutaneous) and 4-0 Biosyn intradermally (skin). A bupivacaine liposome injectable suspension (0.4 ml/kg, Nocita) was infused peri-incisionally into closing layers. The urinary catheter was removed after closure. The cat recovered without complications.
Postoperatively, fluid therapy was maintained, metha-done (0.02 mg/kg IV q6h) was used for analgesia and was subsequently switched to transmucosal buprenorphine (0.01 mg/kg q8h) and gabapentin (12.5 mg/kg PO q12h). By day 3, the cat’s appetite had returned, creatinine had decreased to 1.6 mg/dl (RI 1.0–2.7) and stranguria had resolved. The cat was discharged with activity restriction and scheduled for recheck in 14 days. At follow-up, the owner reported normal urination, with radio-graphs confirming that the bladder remained in a cranial position. The owner also reported that the cat had returned to her normal playful self, without any note of continued tenesmus, and has been doing well for approximately 6 months after surgical intervention.
Discussion
Feline DIUBD literature is limited, so context from other species is informative. In small-breed dogs, DIUBD has been reported as a cause of intermittent obstruction. A case series of three dogs described resolution after cystopexy, but the sample is small, so efficacy cannot be generalized. 2 In humans, severe pelvic organ prolapse can displace the bladder and cause partial obstruction, although urinary incontinence is more common than obstruction. 5 The absence of incontinence in this cat is plausible given species differences in urethral length and periurethral support, the intermittent and directional nature of displacement, and preserved sphincter competence despite episodic urethral angulation.
Compromised pelvic support can permit abnormal bladder displacement in animals and humans.3,5 Cats are not susceptible to the postpartum or menopause-related pelvic floor weakness seen in humans,7,8 but trauma may produce a functionally similar instability that permits dynamic caudal movement across the pelvic brim. 3 Current feline reports on bladder malposition emphasize perineal hernias, traumatic ruptures or congenital variants such as intrapelvic bladder or urethral hypoplasia. 1 In this cat, there was no perineal laxity or pelvic diaphragm defect. Prior pelvic fractures likely weakened support structures so that increases in intra-abdominal or intravesical pressure permitted caudal displacement. 2 This resulted in intermittent urethral kinking and clin-ical signs of stranguria and hematuria.
The interval between historical trauma and onset of urinary signs is biologically plausible. Potential mechan-isms include pelvic fracture malunion with progressive canal narrowing, scar remodeling with loss of tissue compliance, deconditioning of pelvic support musculature and later emergence of precipitating factors such as constipation or tenesmus. These mechanisms could convert a compensated state into intermittent obstruction over time. From a pathophysiologic standpoint, reduced pelvic support combined with transient increases in abdominal strain allowed the bladder apex to slide caud-ally and angle the urethra. Repeated partial outflow obstruction likely caused mucosal irritation and hematuria, with incomplete emptying contributing to bilateral hydronephrosis. This is conceptually consistent with cystocele-associated obstructive uropathy in humans.4,7
In cats with a history of trauma and recurrent lower urinary signs, variable bladder positioning on imaging should raise suspicion for DIUBD. Static lateral radiographs may appear unremarkable if the bladder is cranially positioned at the time of imaging; therefore, when DIUBD is suspected, sequential radiographs or provocative studies during attempted voiding are preferred. Manual voiding cystourethrography or positive-contrast retrograde vaginourethrocystography can capture displacement in real time.1,2 If laparoscopic or open explor-ation is pursued, observing abnormal bladder mobility without an associated hernia supports the diagnosis and allows definitive treatment. Cystopexy anchors the bladder in a consistent cranial position to prevent recurrence; reinforcement of pelvic soft tissues can be considered when intraoperative findings indicate tearing or laxity.
Since the diagnosis relied on serial static imaging and intraoperative findings without dynamic contrast studies, and functional assessments such as cystometry or urethral pressure profiling were not performed, this case lacks generalizable applicability. These omissions limit diagnostic certainty and preclude physiologic characterization of outflow resistance. Undiagnosed DIUBD may exist in other cats, and the roles of factors such as mild colonic distension and perineal edema remain unclear. Larger case accrual using standardized dynamic imaging and functional testing is needed to define prevalence, risk factors and whether pelvic floor reinforcement improves outcomes beyond cystopexy alone.
DIUBD in a cat bears conceptual similarities to humans with pelvic organ prolapse, even though the etiological triggers differ. 5 Trauma-induced pelvic weakness in cats is conceptually similar to displacement-induced obstruction described in women with severe pelvic organ prolapse. 5 In both species, advanced ima-ging can be the definitive tool for identifying organ displacement, such as with MRI or dynamic defecography in humans, approaches that may be adapted for complex cat cases. 1 Furthermore, while the immediate prognosis after cystopexy is favorable, owners should be informed that any new pelvic trauma, ongoing straining behaviors or progression of underlying tissue damage could the-oretically lead to a future recurrence.
Conclusions
DIUBD in cats, although uncommon, carries a risk of intermittent urethral obstruction and adverse renal consequences. Identifying the displacement early through repeated imaging can avert persistent stranguria and protect renal function. This cat’s case underscores that a history of pelvic fractures should prompt veterinarians to consider DIUBD among other differential diagnoses. By employing targeted imaging and proceeding to cystopexy once diagnosis is established, clinicians can re-store bladder position and prevent further lower urinary obstruction. As this condition garners more attention, timely recognition and surgical correction may become more routine in clinical practice.
Footnotes
Acknowledgements
The authors would like to acknow-ledge the family who allowed us to publish this case as well as the veterinary nurses and assistants who helped to care for the cat throughout her hospitalization within the critical care unit.
Supplementary material
The following files are available as supplementary material:
Videos 1–3: Laparoscopic exploration confirmed dynamic caudal displacement and axial torsion of the urinary bladder, resulting in ureterovesicular obstruction and adjacent free fluid near the urinary bladder.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
