Abstract
Case summary
A 12-year-old spayed female domestic shorthair cat with a weight of 4.5 kg and a body condition score of 3/9 presented with anorexia and vomiting. Feline hepatic lipidosis was diagnosed based on blood tests and ultrasound examination. Naso-oesophageal tube feeding was initiated, but hypersalivation persisted despite supportive care. As a result of poor feeding tolerance, a percutaneous endoscopic gastrostomy (PEG) tube was placed for long-term enteral nutrition, with endoscopic confirmation of gastric placement. Two hours after the procedure, blood staining on the PEG tube bandage suggested bleeding at the insertion site. Abdominal ultrasonography and CT revealed splenic perforation. A laparoscopic partial splenectomy was performed successfully and the cat recovered without complications. Follow-up laboratory tests demonstrated a gradual improvement in haematological parameters, including haematocrit and total bilirubin. The cat remained clinically stable without further complications.
Relevance and novel information
This report details the first documented case of splenic perforation during PEG tube placement in a cat, successfully managed through laparoscopic partial splenectomy. Minimally invasive laparoscopy has demonstrated efficacy in addressing rare complications, including splenic perforation associated with PEG tube placement in cats.
Introduction
Hepatic lipidosis is a common liver disease in cats, requiring aggressive nutritional support. 1 Naso-oesophageal or nasogastric tubes are commonly used for short-term nutritional needs, whereas oesophagostomy or gastrostomy tubes are preferred for long-term support. Percutaneous endoscopic gastrostomy (PEG) tube placement, which is recognised as safe and effective in humans, dogs and cats, ensures reliable long-term feeding.2 –5 However, despite its general safety, PEG tube placement can lead to rare but serious complications, such as injury to intra-abdominal organs (eg, spleen, liver, small intestine, colon) and massive haemorrhage.5 –9 This report presents a case of splenic perforation during PEG tube placement that was successfully resolved through laparoscopic partial splenectomy.
Case description
A 12-year-old spayed female domestic shorthair cat presented with anorexia and vomiting. The cat had no significant medical history but experienced weight loss over the past 2 months after relocation stress. Blood tests indicated elevated hepatic enzyme activity (alkaline phosphatase 141 U/l, alanine aminotransferase 474 U/l, aspartate aminotransferase 76 U/l and total bilirubin 2.7 mg/dl). Ultrasonography indicated hepatic lipidosis, characterised by diffuse mild hepatomegaly with blunt margins, uniformly increased hepatic echogenicity, increased attenuation of the ultrasound beam and poor visualisation of the intrahepatic vessel borders. Liver fine-needle aspiration or biopsy was not pursued owing to owner concerns about stress and lack of a definitive diagnostic approach. A presumptive diagnosis of primary hepatic lipidosis was established based on a history of stress-induced anorexia and vomiting, blood test results and the absence of ultrasonographic indicators of secondary hepatic lipidosis. A 5 Fr naso-oesophageal tube was placed, and the cat was started on a liquid diet at 25% resting energy requirement (RER), increasing to 100% over 4 days. Medications included vitamin K (1 mg/kg SC q24h), maropitant (1 mg/kg SC q24h), metoclopramide (0.5 mg/kg SC q12h), cobalamin (250 µg/cat SC once), metronidazole (15 mg/kg IV q12h), esomeprazole (1 mg/kg IV q24h), liver supplement (SAMYLIN; VetPlus) (1 tablet PO q24h) and L-carnitine (330 mg tablet PO q24h). Metronidazole was given, despite antimicrobial concerns, for suspected gastrointestinal inflammation, based on mild small intestinal corrugation. During hospitalisation, vomiting decreased, and total bilirubin reduced to 0.6 mg/dl. To mitigate hospitalisation-induced stress, the cat was discharged with a naso-oesophageal tube after 5 days. The owner was instructed to administer liquid nutrition every 6 h (to 100% RER) and oral medications (ursodeoxycholic acid 7.5 mg/kg, silymarin 10 mg/kg, metoclopramide 0.5 mg/kg, metronidazole 15 mg/kg, esomeprazole 0.75 mg/kg, all q12h). Two days after discharge, the cat’s condition deteriorated, presenting with vomiting and worsening bloodwork (alkaline phosphatase 304 U/l, total bilirubin 1.4 mg/dl), necessitating readmission. Despite using a naso-oesophageal tube for feeding, the cat experienced ongoing salivation, indicating the need for a more stable and effective form of nutritional delivery. To ensure adequate nutrition and address the salivation, a PEG tube was placed using a 16 Fr G-tube (MILA). The procedure was conducted without immediate complications, and an endoscopic examination confirmed correct PEG tube placement in the stomach. Two hours after the procedure, blood staining on the PEG tube bandage indicated bleeding at the insertion site. Coagulation testing was not performed because of vitamin K therapy initiated at presentation, but a more proactive diagnostic strategy should have been considered for a comprehensive evaluation. Abdominal ultrasonography and CT revealed splenic perforation (Figure 1). Exploratory laparoscopy was planned to address the ongoing bleeding and deterioration of vital signs. The initial haematocrit was 36.8%, decreasing to 33.8% with confirmed bleeding, 31% before laparoscopy, accompanied by pale mucous membranes and low systolic blood pressure (108 mmHg, measured by Doppler), and 27.5% after partial splenectomy. Bradycardia and hypotension during anaesthesia were managed with glycopyrrolate (10 µg/kg) and noradrenaline (norepinephrine) (0.5 µg/kg/min).

(a–c) Abdominal ultrasonography. Part of the splenic parenchyma and capsule were damaged, and the PEG tube was perforating the spleen. (d,e) Abdominal CT image demonstrating splenic perforation by the PEG tube (zoomed-in oblique reconstructive image in panel e). (f) Abdominal ultrasonography showed stable postoperative changes in the spleen. PEG = percutaneous endoscopic gastrostomy
Procedural details
General anaesthesia was induced after premedication. A 1 cm incision was made caudal to the umbilicus and a 5 mm trocar and cannula were inserted via the modified Hasson technique. Carbon dioxide was insufflated at 8–10 mmHg to establish pneumoperitoneum. An additional port was placed 5 cm cranially at the midline. A 5 mm, 30°, 29 cm laparoscope (Olympus) was introduced for laparoscopic exploration. Laparoscopic examination revealed an enlarged liver with rounded edges, a mottled appearance and yellow discolouration, consistent with hepatic lipidosis (Figure 2).10 –12 The PEG tube perforated the spleen (Figure 3). A laparoscopic partial splenectomy was performed using a bipolar vessel-sealing device. The PEG tube was repositioned using a laparoscopic grasper. The abdomen was inspected for bleeding and the procedure was concluded.

Exploratory laparoscopy. Gross examination revealed an enlarged liver with rounded edges, a mottled appearance and yellowish discolouration
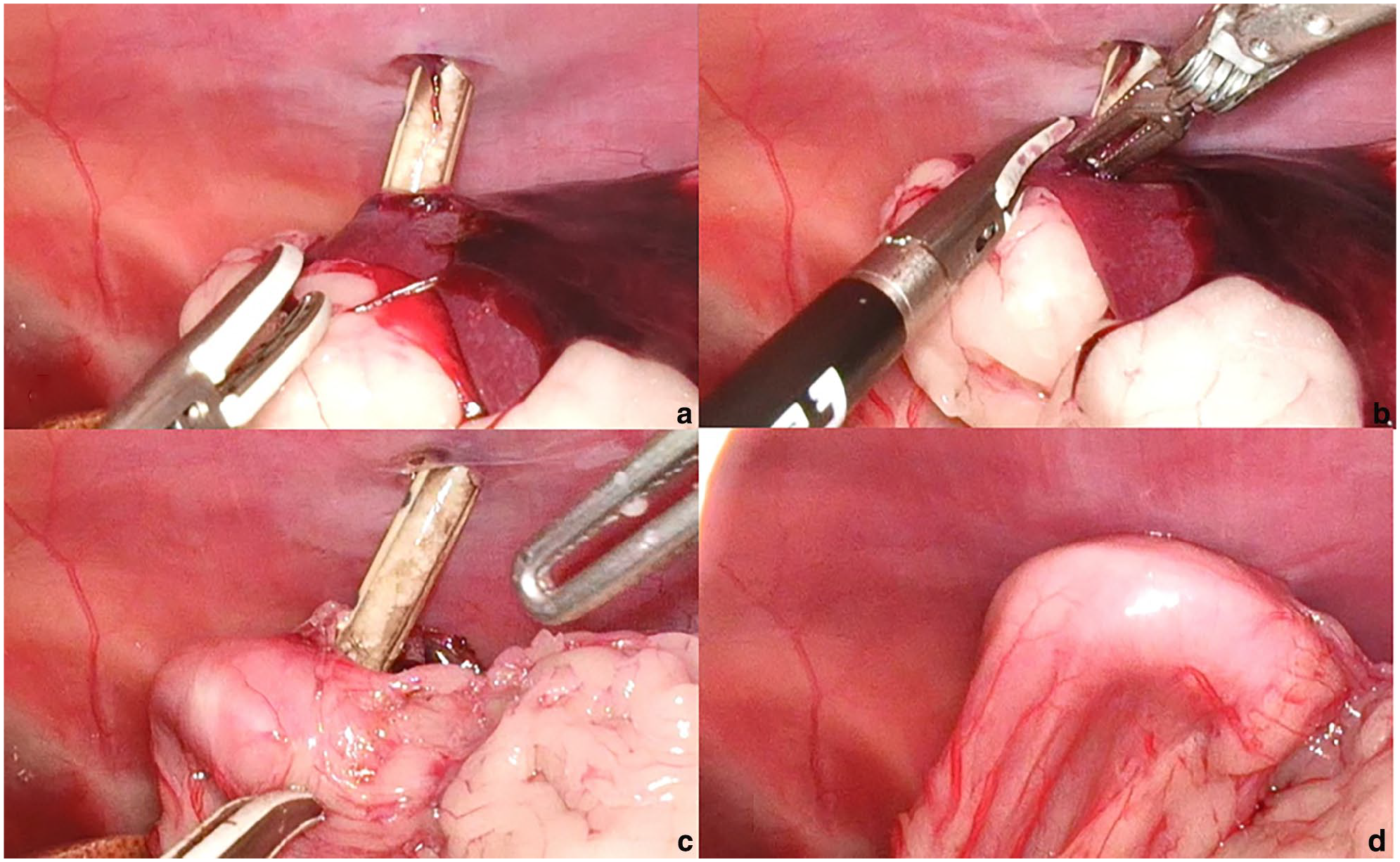
(a) During the placement of the PEG tube, the spleen was perforated and ended up in the stomach. (b) Using a bipolar vessel-sealing device, the penetrated spleen was partially resected in a wedge shape. (c) Removal of the part of the spleen that was trapped between the stomach wall and the abdominal wall was confirmed. (d) Through traction on the PEG tube outside the abdominal wall, the stomach wall was pulled outward, resulting in direct contact with the abdominal wall. PEG = percutaneous endoscopic gastrostomy
Management and outcome
Postoperative recovery was uneventful, with no recurrence of complications such as bleeding at the tube insertion site or the spleen (Figure 1f). Tramadol (1 mg/kg IV q8h) was prescribed for pain. The cat’s condition stabilised as its haematocrit increased from 27.5% to 31.4%, leading to discharge the next day with amoxicillin-clavulanate (12.5 mg/kg PO q12h). The PEG tube provided essential nutritional support, meeting 100% of the RER. Royal Canin Gastrointestinal pouches were initially used owing to soft stools, followed by a RECOVERY diet four times a day. Transitioning from a liquid diet (naso-oesophageal tube) to wet canned food (PEG tube) improved stool consistency and hepatic lipidosis signs, including bilirubinuria, lethargy and weakness. Four days after replacement of the PEG tube, alkaline phosphatase (41 U/l), alanine aminotransferase (51 U/l), aspartate aminotransferase (22 U/l), gamma-glutamyl transferase (4 U/l) and total bilirubin (0.2 mg/dl) normalised. The white blood cell count increased to 27.02 ×109/l, then normalised to 14.3 ×109/l on postoperative day 5. Haematocrit reached 33.5% during follow-up. Platelet counts remained normal. Complete normalisation of all blood test parameters was achieved on postoperative day 13. Oral medication was maintained with ursodeoxycholic acid and silymarin. On postoperative day 31, a MIC-KEY tube (0120-16-1.2; Avanos) was used to replace the PEG tube with minimal adjustment. A follow-up visit on postoperative day 45 showed no distress, normal blood tests and appetite, indicating successful treatment. All medications had been discontinued. Despite the completion of treatment, the owner decided to maintain the MIC-KEY tube because of concerns about the recurrence of anorexia and for convenience, opting to keep it in place for as long as possible. The cat has remained healthy with the MIC-KEY tube in place for 4 months after treatment. The tube will remain in place for as long as the owner wishes. The cat’s weight increased by 500 g compared with the initial visit, reaching 5 kg, with an improved body condition score of 5/9.
Discussion
Hepatic lipidosis in cats requires aggressive nutritional support, commonly via a PEG tube placed for long-term care. An oesophagostomy tube allows faster insertion, lower cost and reduced invasiveness. 13 However, this cat’s persistent hypersalivation, which was unresponsive to antiemetic and prokinetic therapy and exacerbated by nasogastric tube placement, suggested oesophageal irritation. The use of an oesophagostomy tube in this cat posed a risk of further irritating the oesophageal mucosa, potentially worsening the hypersalivation and discomfort. In contrast, a PEG tube, with a larger luminal diameter and transabdominal positioning, mitigated the risk of obstruction and emesis-related complications. Considering the owner’s preference for minimising stress during recovery, a transition to a MIC-KEY gastrostomy tube was planned after PEG placement. This option provided multiple benefits, including reduced risk of dislodgement, elimination of extensive bandaging, and enhanced overall comfort and safety for the patient during recovery.
The most common indications for PEG tube placement are hepatic lipidosis and oral neoplasia in cats, and oesophageal disease in dogs.3,4 Although PEG tube placement is generally considered safe,2 –5 several studies in human and veterinary medicine have documented complications associated with PEG tube placement, classified as minor (eg, peristomal inflammation, fistula formation, tube dislodgement, pneumoperitoneum) and major (eg, bleeding, visceral injury, aspiration pneumonia, peritonitis).2,3,5 –9 During the PEG tube placement procedure, the cat was positioned in right lateral recumbency and the stomach was insufflated with air to provide clear visualisation and safe access for the PEG tube. The insertion site was carefully verified using endoscopic transillumination and skin imprinting through finger palpation to minimise the risk of organ injury. The PEG tube was placed without deviation from the standard procedure, and no immediate complications were identified. Despite these precautions, 2 h after the procedure, bleeding was observed at the insertion site. Bleeding was primarily noted at the PEG tube insertion site, although intra-abdominal bleeding should also be considered. Postoperatively, a complete blood count and ultrasonography are recommended to assess for bleeding or related complications. No previous studies in the veterinary literature report splenic perforations caused by PEG tube placement in cats. In human medicine, factors such as a history of abdominal surgery, abnormal anatomy and significant obesity have been identified as potential risk factors for complications during PEG tube placement.2,6 –9 However, none of these factors were observed in this cat. In investigating the cause of these complications, inadequate stomach insufflation was hypothesised to have positioned the spleen cranially, resulting in perforation. Proper gastric insufflation during PEG tube placement ensures cranial positioning of the liver and caudal displacement of the spleen and colon, reducing the risk of organ injury. This hypothesis is supported by a study involving 47 dogs. 14 This found that insufficient stomach insufflation caused the spleen to move cranially. If adequate gastric insufflation is not achieved, the procedure should be aborted and alternative access options should be assessed. Percussion, by contrasting dull and hollow sounds, could be used to assess the adequacy of gastric insufflation. Similarly, human cases have reported instances of PEG tubes being inadvertently placed through the colon because of inadequate stomach insufflation. 8 Transcolonic misplacements are particularly common in patients with a history of abdominal surgery or excessive obesity, in addition to inadequate gastric insufflation. 7 To prevent such complications, the ‘safe tract’ method uses a saline-filled syringe to avoid colon penetration and confirm accurate placement.7,15
While laparoscopic splenectomy has been well documented in dogs and cats, laparoscopic partial splenectomy has been reported only in dogs.16–19 Although there is limited literature on laparoscopic partial splenectomy in cats, minimally invasive splenectomy via laparoscopy is effective in reducing surgical time and complications. 19 Indications for laparoscopic partial splenectomy typically include idiopathic splenomegaly, splenic cysts, benign tumours, metastases, splenic infarcts, iatrogenic injuries, haematological diseases and focal splenic tumours.20,21 Studies comparing open surgery and laparoscopic splenectomy have shown that laparoscopy offers significant advantages, such as lower intraoperative complication rates, shorter surgical duration and quicker patient recovery. In addition, unlike total splenectomy, partial splenectomy preserves splenic function, which is beneficial.16,20,21 Based on these findings, we prioritised laparoscopy over open surgery to correct these complications. Laparoscopy confirmed that the PEG tube had caused splenic perforation, which was subsequently corrected through partial splenectomy and PEG tube repositioning. The cat remained clinically stable, with no further complications observed.
This case report is subject to diagnostic limitations, including the absence of histopathological confirmation via fine-needle aspiration or biopsy and the lack of coagulation testing before PEG tube placement.
This case is clinically significant in emphasising the need to recognise rare, life-threatening complications associated with PEG tube placement in cats and to maintain vigilant patient monitoring, as exemplified by the successful management of splenic perforation via minimally invasive laparoscopic partial splenectomy.
Conclusions
During placement of the PEG tube, a rare complication of splenic perforation was encountered and was successfully treated using laparoscopic partial splenectomy. Ensuring adequate gastric insufflation and vigilant postoperative monitoring are crucial to prevent and address complications. Minimally invasive laparoscopy has been shown to be effective in managing rare complications, such as splenic perforation, associated with PEG tube placement in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
