Abstract
Two 4-year-old spayed female Siamese cats were seized by the British Columbia Society for the Prevention of Cruelty to Animals after confinement to an abandoned housing unit without food for 9 weeks. One cat was found dead, and the second was euthanized within 24 hours due to neurologic deterioration despite therapy. Polioencephalomalacia of the caudal colliculus, hepatic lipidosis, cachexia, and congestive heart failure with cardiomyocyte atrophy were identified in both cats through postmortem examination and attributed to a prolonged period of starvation. Brain lesions were likely the result of thiamine deficiency (Chastek paralysis), which can be associated with both malnutrition and liver disease. This case highlights the importance of thiamine supplementation during realimentation of cats with hepatic lipidosis. Heart failure resulting from cachexia may have contributed to the death of the first cat and the morbidity of the second cat.
Keywords
Two 4-year-old spayed female Siamese cats were seized by the British Columbia Society for the Prevention of Cruelty to Animals after being confined in an abandoned housing unit for 9 weeks. The cats had access to toilet water, but there was no food on site. One cat was found dead. On presentation to a veterinarian, the second cat was laterally recumbent and 10% to 12% dehydrated with significant muscle wasting. The cat was dull and depressed but responded to touch with muscle fasciculations and seizure-like behavior. Rectal temperature was 37.1
Complete blood cell count demonstrated moderate hemoconcentration (hematocrit, 49.2%), schistocytosis, and microcytosis. Serum biochemistry revealed elevated alanine aminophosphatase (448 U/l), alanine aminotransferase (749 U/l), total bilirubin (76 µmol/l), and urea (19.4 mmol/l). Cholesterol was also elevated (6.1 mmol/l), and total thyroxine was depressed (<6 nmol/l). Sodium (159 mmol/l), potassium (4.4 mmol/l), phosphorous (1.97 mmol/l), calcium (2.68 mmol/l), albumin (36 g/l), and globulins (34 g/l) were within normal limits. Urine collected by cystocentesis was negative for ketones and had normal specific gravity (1.045), magnesium ammonium phosphate crystalluria, bilirubinuria, proteinuria, pyuria, hematuria, and bacteriuria. Assays for feline leukemia and feline immunodeficiency virus were negative (SNAP FIV/FeLV Combo Test; IDEXX Laboratories, Westbrook, ME, USA).
Intravenous fluids (0.9% NaCl) were administered overnight at a rate of 12 ml/h. Despite improved hydration status and serum levels of urea, hepatic enzymes, and total bilirubin, the following day, the cat continued to decline clinically. She developed forelimb extensor rigidity, hypokalemia (3.1 mmol/l), hypophosphatemia (0.54 mmol/l), hypocalcemia (1.81 mmol/l), hypoalbuminemia (18 g/l), and bilateral mydriasis and was euthanized due to poor prognosis.
At necropsy, both animals were in excellent condition and demonstrated similar lesions. Abundant internal fat stores persisted in the face of generalized muscle wasting. The lungs were congested and edematous. The livers were enlarged, pale yellow, and friable, with a highlighted acinar pattern.
The history, clinical signs, and clinicopathologic changes suggested feline hepatic lipidosis (FHL) secondary to starvation in both cats. The urinalysis findings in the second cat were consistent with a urinary tract infection and believed to be incidental. Differential diagnoses for the neurologic and motor deficits in the second cat (muscle fasciculations, seizure-like activity, extensor rigidity, mydriasis, and ptyalism) included hypomagnesemia, hypokalemia, hypophosphatemia, hepatic encephalopathy, thiamine deficiency (TD), and toxicosis.
Hypomagnesemia affects roughly one-third of FHL patients. 2 Clinical signs include muscle weakness and impaired mentation that worsens with crystalloid therapy and fluid volume expansion. 2 Magnesium levels were not measured in either cat. Hypokalemia and hypophosphatemia are also common in FHL patients, with a similar clinical picture to hypomagnesemia. 2 The electrolyte disturbances seen in the second cat likely resulted from fluid volume expansion. The apparent hypocalcemia was probably a consequence of hypoalbuminemia.
Hepatic encephalopathy is a potential outcome of liver disease and can lead to ptyalism, ataxia, and depressed mentation. However, the syndrome is not commonly identified in FHL patients. 3 High fasting blood ammonia and ammonium phosphate crystalluria can differentiate hepatic encephalopathy from electrolyte disturbances and other neurologic conditions. 3
TD can occur in cats secondary to starvation or malnutrition. 2 Thiamine is a coenzyme essential to cerebral and cerebellar glucose metabolism and synaptic nerve transmission. 2 In carnivores, TD presents clinically as central vestibular ataxia, abnormal postural reactions, cervical ventroflexion, mydriasis, hypothermia, seizures, and/or coma. 2,4
Representative tissue samples from both animals were fixed in 10% buffered formalin, processed routinely for sectioning, and stained with hematoxylin and eosin. Brain sections examined were: the medulla oblongata at the central canal, obex, and peduncles; cerebellum at ventral median fissure; rostral colliculus; hippocampus; thalamus at the interthalamic adhesion; basal nuclei; occipital cortex; temporoparietal cortex–roof of the lateral ventricle; and the frontal cortex. Lung sections were additionally stained for iron with Perls’ Prussian blue, and liver sections were stained for lipid with oil red O. Both cats demonstrated similar histologic lesions.
There was marked, diffuse, macro-, and microvesicular hepatocellular cytoplasmic vacuolation with bile stasis (Fig. 1). Oil red O stain confirmed that the hepatocellular vesicles contained lipid.
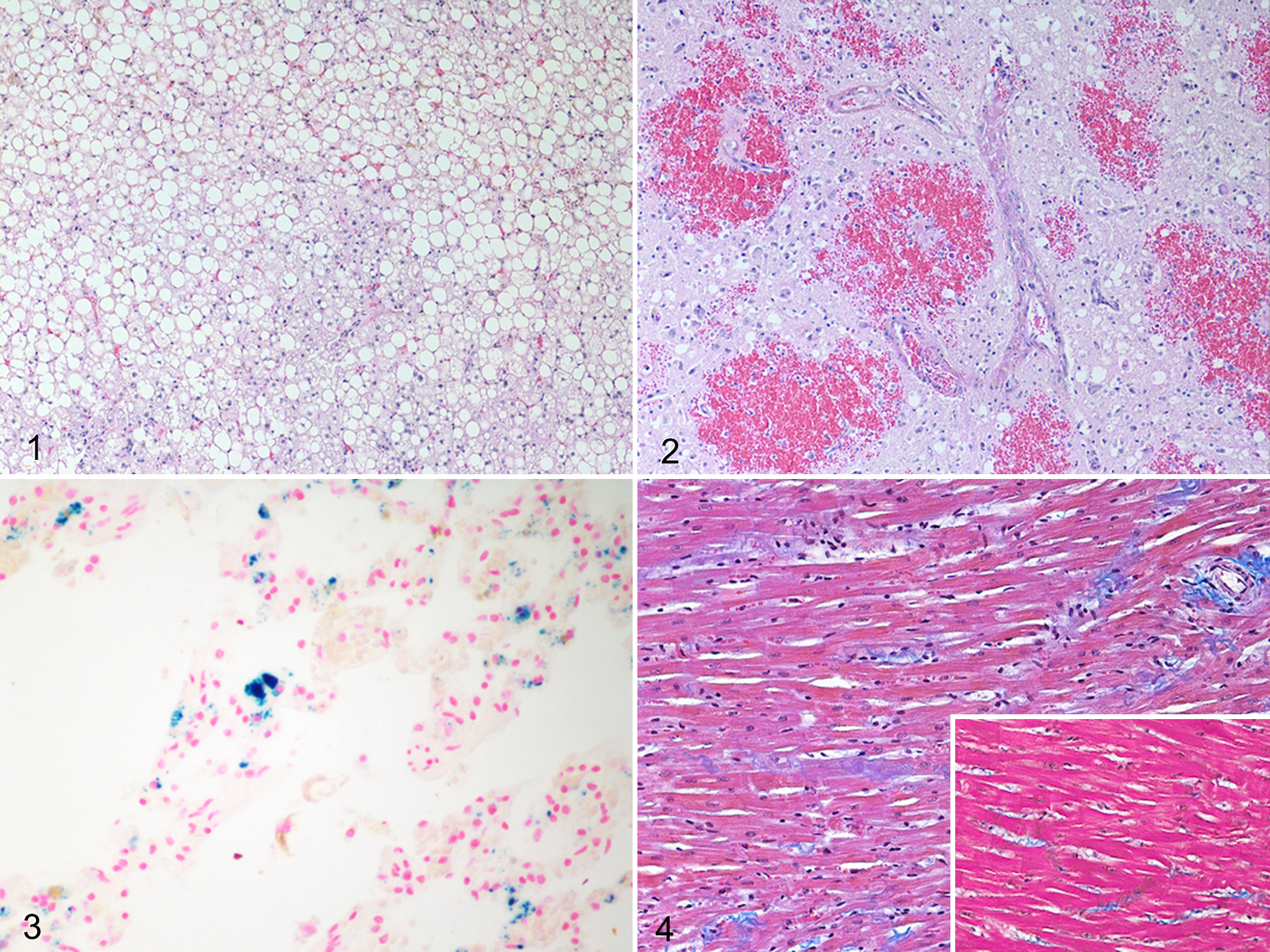
Within the caudal colliculus, there was prominent gliosis with severe spongiosis, rarefaction of the neuropil, and hemorrhage. Degenerate and necrotic neurons, spheroids, reactive astrocytes, and necrotic cellular debris were numerous. There was occasional fibrinoid vascular degeneration; affected vessels were lined by hypertrophic endothelial cells and surrounded by edema, fibrin, and erythrocytes (Fig. 2). Similar but less severe changes were present in the superficial gray matter of the dorsal parietal cortex of the second cat.
The lungs were diffusely congested with alveolar and interstitial edema, and increased numbers of iron-laden alveolar macrophages were demonstrated by Perls’ Prussian blue stain (Fig. 3). There was decreased skeletal myofiber diameter, and the myocardium had reduced cardiomyofiber diameter and increased prominence of interstitial connective tissue (Fig. 4) when compared with 2 age-matched control cats that died of noncardiac disease without protein malnutrition (Fig. 4, inset).
The diagnosis was FHL, polioencephalomalacia, skeletal and cardiac muscle atrophy, and congestive heart failure. All of the pathology was attributable to the prolonged period of starvation noted in the history. The collection of lesions could not be explained by any of the other differential diagnoses alone.
FHL is most commonly recognized in previously obese animals that have undergone a period of anorexia or starvation. 1 The retained fat stores in these cats following 9 weeks of food deprivation suggested that they were obese at the time of their abandonment. As obligate carnivores with high requirements for protein and essential fatty acids, cats are limited in their ability to adapt urea cycle enzymes to reduced protein intake and adjust protein metabolic pathways for conserving nitrogen. The result is rapid protein malnutrition in anorexic cats. 2 The amino acids taurine, arginine, and carnitine are necessary for bile acid conjugation and urea cycle function and for the transport, β-oxidation, and egress of fatty acids from the liver. Depletion of these amino acids contributes to cholestasis, increased blood ammonia, and accumulation of triglycerides in the liver, which are the defining features of FHL. 2
Treatment of FHL involves fluid therapy containing a fortified B-complex vitamin solution (including thiamine); realimentation with a protein-replete feline diet; supplementation of L-carnitine, taurine, vitamin K, and vitamin E; and antioxidant therapy. 2 While most FHL patients require a high-protein diet to facilitate mobilization of accumulated triglycerides in the liver, patients with hepatic encephalopathy require short-term low-protein alimentation. 1
The polioencephalomalacia was most likely caused by TD. FHL patients may be particularly susceptible to TD due to the liver’s role in storage and activation of water-soluble vitamins. 2 TD can result from chronic anorexia, a thiamine-deficient or thiaminase-containing diet, meat preserved with sulfur dioxide, malabsorption, increased renal losses, or carbohydrate loading in the face of marginal thiamine stores. 2,7 Clinical signs of TD are easily mistaken for hepatic encephalopathy. 2 Consequently, it would be useful to diagnose TD antemortem and differentiate it from other neurological conditions.
The defining neurologic lesion associated with TD in carnivores (also known as Chastek paralysis) 4 is bilaterally symmetrical hemorrhagic encephalomalacia of the periventricular gray matter of the brain, often involving the caudal colliculi, the medial vestibular, lateral geniculate, habenular, oculomotor, accessory vestibular, cuneate, and red nuclei and less commonly the mammillary bodies, accessory cuneate nucleus, superior colliculi, nucleus reuniens, ciliary nucleus, and the occipital cortex. 4
The pathogenesis of these lesions in carnivores is unclear, but impaired neuronal energy metabolism, lactic acidosis, and accumulation of glutamate have all been implicated. 7
TD can be verified antemortem by response to supplementation. Magnetic resonance imaging can also aid in the diagnosis by revealing the characteristic symmetrical pattern of hemorrhage in vulnerable gray matter regions of the brain. 7 Serum thiamine can be measured by microbiological assay or indirect assay of enzymes requiring thiamine as a cofactor. 6 A direct method of thiamine evaluation using high-pressure liquid chromatography in ruminants has also been evaluated for cats. 5
In humans, TD typically presents as heart failure. Interestingly, both cats had iron-laden alveolar macrophages, evidence of chronic passive pulmonary congestion. Heart failure has been experimentally induced in rodents fed a thiamine-deficient diet. Both cats had evidence of cardiomyocyte atrophy, consistent with one study of experimentally induced TD in rats. 8 Alternatively, the reduced cardiomyofiber diameter could have resulted from catabolism secondary to prolonged protein malnutrition. The reduced skeletal myofiber diameter is also consistent with protein malnutrition. In humans, protein deficiency and subsequent catabolism are known to result in cardiac atrophy and decreased blood pressure, heart rate, blood volume, and stroke volume. 9 This decline in cardiac function is often masked by a proportional reduction in overall metabolic activity, and heart failure may only manifest clinically upon a sudden increase in metabolic demand, such as refeeding. 9 Sudden death may also occur in obese individuals who undergo rapid weight loss without high-quality protein supplementation, and it is attributed to heart failure secondary to cardiac atrophy. 9 Accordingly, heart failure due to TD, protein-energy malnutrition, or a combination of the two may have contributed to the natural death of the first cat and to the general malaise of the second.
This case highlights TD as a differential diagnosis in cats suffering from FHL that perish or develop neurological disease.
Footnotes
Acknowledgements
We thank Dr Mark Steinebach and the British Columbia Society for the Prevention of Cruelty to Animals for their work in this case.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
