Abstract
Case summary
This report describes a case of gastric mucormycosis in a young Ragdoll cat with a 5-day history of vomiting. Physical examination detected mild dehydration and tenderness was elicited on abdominal palpation. The results of blood work-up and radiographic study were unremarkable; however, abdominal ultrasonographic examination revealed multiple hyperechoic neoformations at the level of the pyloric antrum, which were confirmed on endoscopic examination. Non-septate hyphae of irregular diameter with a branched appearance were observed on cytology, and histological examination revealed severe diffuse necrotising and granulomatous gastritis with the presence of intralesional fungal hyphae indicative of mucormycosis, which was confirmed by PCR tests. Antifungal therapy with ketoconazole in addition to supportive treatment temporarily improved the clinical condition. Lethargy, fever and abdominal effusion developed in the following days. Cytological examination of abdominal fluid was compatible with septic peritonitis and, given the severity of the condition, euthanasia was opted by the owners. Post-mortem examination confirmed septic peritonitis resulting from perforation of the gastric wall at one of the neoformations of the pyloric antrum.
Relevance and novel information
To the authors’ knowledge, this is the first reported case of gastric mucormycosis in a cat. Previous literature includes a case of mucormycosis in a Persian cat affecting only the duodenum. In both the Persian cat and the cat described here, gastrointestinal mucormycosis disease progressed rapidly and was fatal.
Introduction
Mucormycosis is a fungal disease caused by genera of the order Mucorales, previously known as Zygomycota. 1 This order includes genera such as Rhizopus, Absidia, Mucor, Cokeromyces and Saksenaea. 2 Although the earliest reports of mucormycosis in animals date back to the late 1960s, 3 reported cases are still relatively rare in the veterinary literature. In human medicine, mucormycosis is more frequently observed, and has been identified as an emerging and frequently fatal condition, particularly in diabetic and immunocompromised patients. 4
This case report is the first, to the authors’ knowledge, to document gastric mucormycosis caused by Rhizopus microsporus in a cat.
Case description
A 9-month-old spayed female Ragdoll cat was presented with acute persistent vomiting over the past 5 days. On one occasion, haematemesis was also noted. Furthermore, reduced appetite and lethargy were observed. The cat had an unremarkable past history and had been on a raw rabbit-based biologically appropriate raw food (BARF) diet for the past 3 months. Clinical examination revealed moderate dehydration (7%), hyperthermia (39.9°C), pale mucous membranes with a capillary refill time of approximately 3 s and mild discomfort on abdominal palpation. The body condition score was 4/9 and body weight was 4.2 kg. Haematological and biochemical tests were within normal limits, and the cat tested negative for feline immunodeficiency virus (FIV) antibody and feline leukaemia virus (FeLV) antigens. Radiographic examination was unremarkable; however, abdominal ultrasonographic examination revealed multiple rounded neoformations at the pyloric antrum with increased echogenicity and mild mesenteric lymphadenomegaly. The rest of the abdominal cavity appeared normal.
Fluid therapy and omeprazole were initiated to stabilise the clinical condition. An endoscopic examination was carried out thereafter, which confirmed several neoformations at the pyloric antrum, ranging in size from 0.5 cm to 2 cm, with a smooth surface, sessile base and hard-elastic consistency (Figure 1). The duodenum appeared normal. During the procedure, cytological brushing (Olympus BC-201C-1006) and biopsies using 1.8 mm fenestrated forceps (Olympus FB-231 D) were performed.
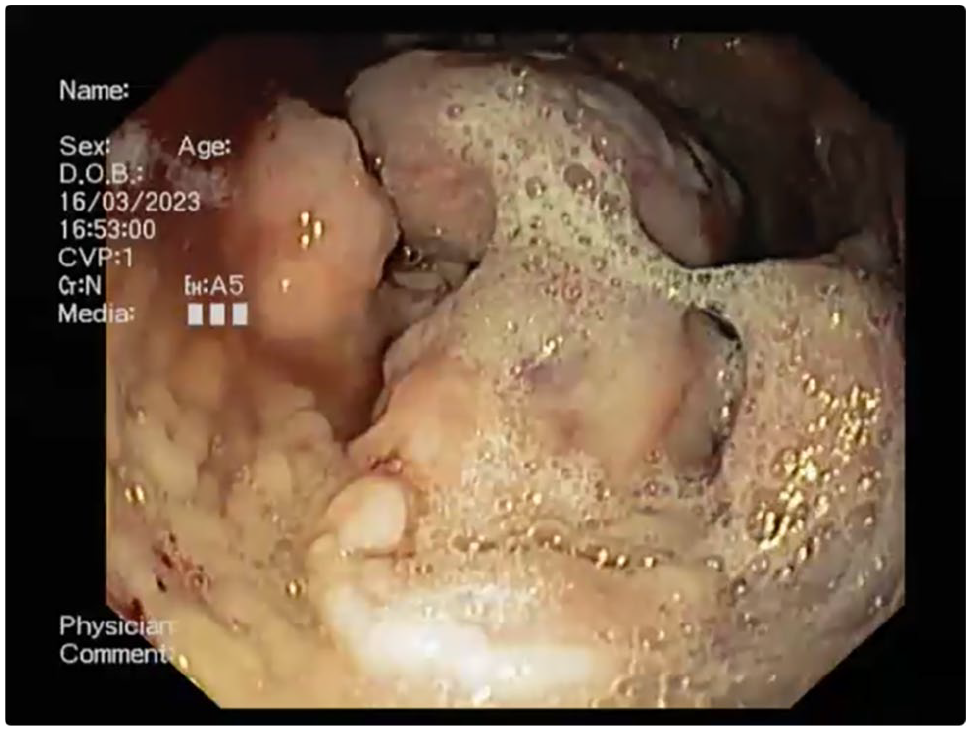
Gastric neoformations seen on endoscopy
Cytological analysis revealed mucosal cells in small clusters, mixed inflammatory cells with a predominance of neutrophils and macrophages, and occasional large, non-septate hyphae with an irregular diameter and branched appearance. Pending histological results, fluid therapy and omeprazole were continued. Additionally, maropitant, sucralfate and antifungal therapy with ketoconazole at 20 mg/kg q12h was initiated. Treatment with amphotericin B was declined because of its excessive cost and concerns about side effects. In the days following the endoscopy, the cat’s condition improved slightly, as characterised by partial restoration of appetite and reduced episodes of vomiting. However, hyperthermia and lethargy persisted.
Histological examination revealed severe diffuse necrotising and granulomatous gastritis with intralesional fungal hyphae, consistent with mucormycosis (Figure 2a). Biopsies of the gastric body and duodenum were normal (Figure 2b). PCR examination confirmed the presence of Rhizopus microsporus using ITS3 and ITS4 primers, with NL1 and NL4 sequenced by Basic Local Alignment Search Tool (https://blast.ncbi.nlm.nih.gov).
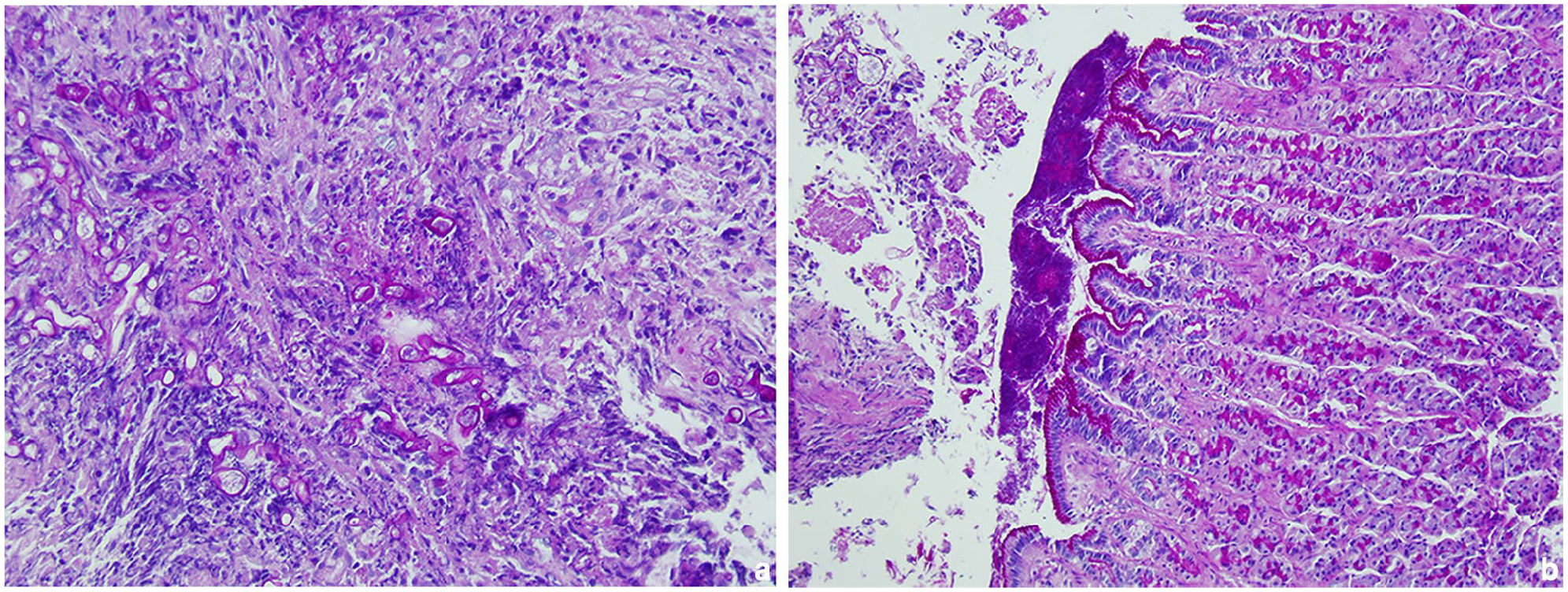
Histology of gastric biopsies stained with haematoxylin and eosin showing inflammatory lesions secondary to the presence of Rhizopus microsporus
Because of the limited clinical response to ketoconazole and its poor efficacy in mucormycosis,5 –7 surgical excision and amphotericin B therapy were proposed. However, the day before surgery, the cat became severely lethargic and showed marked hyperthermia (40.3°C) and dehydration (10%), as well as abdominal effusion. Abdominal ultrasonographic examination revealed signs of peritonitis, and cytology of abdominal fluid revealed septic neutrophilic inflammation with phagocytosis of both cocci and rods, but no fungal elements were detected in the exudate. No cultures or additional PCR tests were performed. Given the cat’s critical condition, the owner elected euthanasia.
Post-mortem examination confirmed septic peritonitis as a result of perforation of the gastric wall at one of the neoformations in the pyloric antrum (Figure 3). Histological examination of the stomach, duodenum, ileum, colon, spleen, liver, kidney and lung was performed; however, the presence of mucormycosis was confirmed only in the gastric biopsies.

Stomach as seen on necroscopy. It is evident that there is perforation of the gastric wall and fungal granulomas
Discussion
Mucormycosis is a disease caused by opportunistic fungi that are found in soil, decaying materials and food. 8 In the veterinary literature, systemic forms have been reported in horses 9 and cattle, 10 as well as dogs. 11 In cats, the disease has been reported in various organs, including the skin, brain, nasal and duodenal areas.12 –14 Gastrointestinal mucormycosis, though rare in both human and veterinary medicine, presents with high mortality rates, reaching up to 85% even with early diagnosis in humans.7,8,15 Recent increases in cases of gastrointestinal mucormycosis in humans suggest the emergence of more virulent fungal strains. 16 In humans, the stomach is the most commonly affected site, followed by the colon, ileum, duodenum and jejunum.4,17 Notably, gastric mucormycosis has not been documented in veterinary cases. The current case aligns with the age of onset described in the only other case of duodenal mucormycosis in a cat: 9 months in the present case compared with 7 months in the previous report. 14 By contrast, humans typically develop this disease at a later age, though cases in adolescents have been reported. 18
In humans, gastrointestinal mucormycosis is often linked to the ingestion of contaminated food, such as fermented products or herbal remedies. 4 The current patient had been on a BARF diet, including raw rabbit with bones, in the 3 months preceding the onset of clinical signs. Given that the patient was an indoor cat, accidental ingestion of contaminated soil or grass seems unlikely. We speculated that the pathogen entered via the food.
In humans, gastrointestinal mucormycosis is more common in immunocompromised individuals,8,16 with uncontrolled diabetes mellitus, solid organ or stem cell transplantation, haematological malignancies and major trauma as the first causes. 19 Cases have also occurred in patients without evident immunodepression. 16 The present patient, similar to others described in feline and canine studies,13,14 showed no signs of immunodepression; serology for FIV and FeLV was negative, and the cat was in good health before presentation. Other fungal diseases, such as aspergillosis, also do not necessarily involve systemic immunodepression as a predisposing factor. It is hypothesised that individual susceptibilities and environmental conditions contribute to the development of mycosis. 20
In human medicine, mucormycosis progresses through stages: colonisation, infection and vascular invasion, potentially leading to systemic dissemination and septic peritonitis. 21 Gastric wall perforation and subsequent septic peritonitis are reported complications.18,22,23 The current patient experienced gastric wall perforation about 2.5 weeks after the onset of fever, which coincides with human case reports.5,23 This raises the suspicion that mucormycosis may be underdiagnosed, particularly in fatal septic peritonitis where the primary diagnosis remains unclear. 24 The present patient had a single episode of haematemesis, which resolved with symptomatic treatment, unlike the severe bleeding often reported in human cases.
Initial haemato-biochemical examinations were non-specific, although lymphocytosis and anaemia developed following peritonitis. Although these changes are consistent with mucormycosis in humans, lymphocytosis could also be related to septic peritonitis. Abdominal ultrasonographic examination revealed multiple intragastric neoformations, a finding also seen in humans but less commonly than circumferential thickening or ulceration. 14 Cytological examination of ascites did not reveal fungal elements; however, we did not perform culture or PCR on the effusion. It is possible that cytology alone is unlikely to detect mucormycosis in effusion, suggesting it may be underdiagnosed as a primary cause of septic peritonitis. Endoscopic findings of multiple round lesions in the stomach of a young cat are unusual, with differential diagnoses including neoplasms, mycotic and parasitic granulomas. Cylospiruria is rare, 25 and phytiosis has not been described in European cats; 26 therefore, mucormycosis should be considered in the differential diagnoses of intragastric neoplasms. Confirmation, as demonstrated in this case, requires endoscopic examination along with cytological and histological analysis.
Histological diagnosis is often performed by laparotomy in humans, and cytological examination by brush was diagnostic in the present case. This method provides rapid results, but histological confirmation, including periodic acid–Schiff staining and fungal typing by culture or PCR, remains essential. Despite advances, only 25% of human patients achieve an ante-mortem diagnosis. 16
Veterinary literature offers limited information on treatment for mucormycosis. In one duodenal case, diagnosis was achieved post mortem with no attempted therapy beyond surgical excision. 14 In our case, although treatment led to a modest improvement in the clinical condition, the decision to avoid more aggressive antifungal options, such as liposomal amphotericin B, was influenced by economic constraints. In human medicine, prompt and aggressive treatment is emphasised, which includes addressing predisposing factors, performing surgical debridement and administering antifungal therapy. 16
Conclusions
Although mucormycosis is infrequent and likely underdiagnosed, this study highlights the importance of considering it in the differential diagnosis of septic peritonitis in young cats. Owing to the high mortality associated with this disease, prompt surgical intervention is likely to be crucial for improving outcomes. Early and accurate diagnosis, followed by timely surgical treatment, may significantly impact the prognosis of mucormycosis in feline patients.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
