Abstract
Pneumocystis jirovecii pneumonia, typically an opportunistic infection, is commonly associated with risk factors such as low CD4+ lymphocyte count, underlying malignancies, organ transplantation, or immunosuppressive medications. However, occurrences in healthy individuals without known risk factors are exceptionally rare and sparsely documented. In our retrospective analysis of a 42-year-old male without past medical history at Abderrahmane Mami Hospital, Tunisia, Pneumocystis jirovecii pneumonia was diagnosed. The patient presented with fever, productive cough, hemoptysis, and a decline in general health. Clinical examination revealed fever and hypoxemia, and imaging studies demonstrated bilateral necrotic alveolar opacities. Despite empirical antibiotics, nonresponse necessitated bronchoscopy, confirming Pneumocystis jirovecii. Treatment with oral Sulfamethoxazole-Trimethoprim yielded excellent outcomes. This case highlights the potential occurrence of Pneumocystis jirovecii pneumonia in immunocompetent individuals, underscoring the importance of direct microbiological methods in assessing suggestive clinical and radiological features.
Keywords
Learning points
Pneumocystis jirovecii Pneumonia in Immunocompetent Patients: This case report highlights a significant finding that Pneumocystis jirovecii pneumonia, traditionally associated with immunocompromised individuals, can also affect healthy immunocompetent patients without underlying comorbidities. This challenges the conventional understanding of the disease’s epidemiology and emphasizes the importance of considering Pneumocystis jirovecii pneumonia in the differential diagnosis, even in patients without apparent immune deficiencies.
Optimal Outcomes in Pneumocystis jirovecii Pneu-monia for Immunocompetent Patients through Early Recognition and Prompt Intervention: In the context of Pneumocystis jirovecii pneumonia in immunocompetent patients, the severe clinical presentation, often necessitating respiratory assistance, contrasts with a noteworthy observation of relatively low mortality rates when prompt and appropriate treatment is administered. This underscores the pivotal significance of early diagnosis and intervention in achieving favorable outcomes, potentially reducing morbidity and optimizing healthcare resource utilization. Healthcare providers should exercise heightened diagnostic vigilance when encountering clinical symptoms and imaging findings suggestive of the condition, regardless of the patient’s immune status. Emphasizing a proactive approach to treatment initiation is paramount for improved patient prognosis and overall healthcare management, highlighting the critical role of timely intervention in optimizing outcomes for immunocompetent individuals with Pneumocystis jirovecii pneumonia.
Introduction
Pneumocystis pneumonia (PCP) is an opportunistic, pathogenic fungal infection. It is a leading cause of morbidity and mortality in immunosuppressed patients. 1
For several decades, many studies have been focused on PCP in Human Immunodeficiency Virus (HIV)-infected populations. More recently, a significantly increased incidence of PCP in non-HIV immunosuppressed populations—such as malignancies, organ transplantation, or immunosuppressive medications—has been noticed.2,3 However, a few studies are interested in PCP in patients without any risk factor.1,4,5
We report a case of PCP in a healthy immunocompetent patient.
Case report
A 42-year-old man, nonsmoker, without past medical history especially personal or family history of inherited immunodeficiency disorders or autoimmune diseases, was admitted to the hospital complaining of hemoptysis, dyspnea on exertion, fever, weight, and appetite loss for 45 days. On examination, the patient was febrile at 38.5°C and hypoxemic (Oxygen saturation on a nasal cannula at 1 L was 91%). The chest X-ray revealed bilateral and extensive alveolar opacities (Figure 1).

Admission chest X-ray.
Laboratory results indicated the presence of inflammatory syndrome (white blood cell: 11,800/mm³, C-reactive protein: 220 mg/L), hemoglobin: 11.7 g/dL, hepatic cytolysis, and cholestasis. Sputum and blood culture did not yield any organisms. Serology for Mycoplasma pneumoniae, Legionella pneumophila, and Chlamydia pneumoniae were negative. Pneumococcal antigen urinary testing was negative. The HIV antibody test was negative. Serum rheumatoid factor was positive (64 UI/mL). Antinuclear antibodies, anti-cyclical Citrullinated Peptide antibodies, and antineutrophil cytoplasmic antibodies were negative.
The lymphocyte subpopulations CD4, CD8, and CD4/CD8 were carried out and were normal (1050/mm3, 480/mm3, 2.18%, respectively) .The serum levels of immunoglobulin M, G, and A were within the normal ranges (1.7 g/L, 12 g/L, 2.3 g/L, respectively).
Computed tomography (CT) (Figure 2) revealed multiple diffuse consolidations associated with nodules sometimes displaying necrosis, along with enlarged necrotized bilateral mediastinal and hilar lymph nodes and pericardial effusion.

Computed tomography images show multiple nodules of varying sizes and bilateral lymph nodes (arrowheads).
The patient was treated with amoxicillin-clavulanic acid 1 g × 3 daily for 7 days. On the fifth day, bronchoscopy with bronchial aspiration was performed for cytology, BK testing, and bacteriological examination, and it was normal. The bacteriological study of bronchial fluid has been performed, excluding specific testing for Pneumocystis. Both direct examination and culture were unable to identify any microorganisms. Dyspnea escalated (relying on 5 L per minute with a high-concentration mask and subsequently necessitating noninvasive ventilation due to hypercapnia), while the fever remained. The chest film showed an extension of alveolar opacities (Figure 3). Inflammatory syndrome increased (white blood cell: 16,800/mm³, C-reactive protein: 343 mg/L). Antibiotic therapy switched to imipenem 1 g × 3 daily, vancomycin 1 g × 2 daily for 12 days for both, and levofloxacin 500 mg × 2 daily for 2 days, then 500 mg daily for 10 days.

Chest X-ray after 10 days.
A second bronchoscopy with bronchoalveolar lavage (BAL) and protected distal sampling was conducted due to ongoing fever and the advancement of alveolar opacities. It revealed inflammation throughout the entire bronchial tree mucosa. The protected distal sampling showed a polymorphic pattern. Bronchial biopsy showed an inflammatory mucosa. The bronchial fluid was examined by optic microscopy. It contained inflammatory but not neoplastic cells. A study of bronchial fluid by Grocott’s methenamine silver (GMS) on cellblock revealed a few Pneumocystis jirovecii organisms (Figure 4(a) and 4(b)).
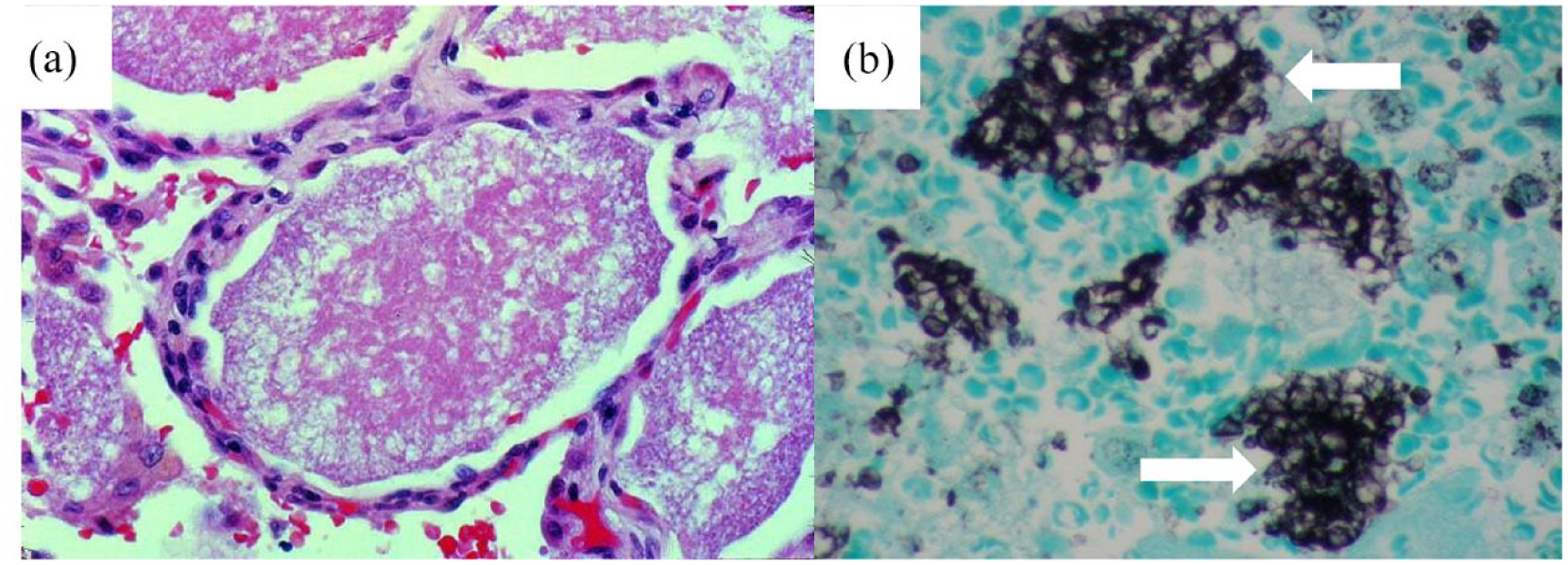
(a) Alveolar spaces filled with pink, foamy amorphous material composed of proliferating fungi and cell debris (hematoxylin and eosin stain (HE)×40). (b) Cysts are black (white arrowheads) with the Grocott–Gömöri’s methenamine silver (GMS) stain (GMS×20).
The diagnosis of PCP was established. The patient was, therefore, treated with oral Sulfamethoxazole-Trimethoprim 1600 mg per day in four doses and dexamethasone 40 mg twice per day (which was gradually tapered off) for 3 weeks.
The patient exhibited clinical improvement (afebrile, oxygen saturation: 95% on room air) and biological improvement (white blood cell count: 14,000/mm³, C-reactive protein: 23 mg/L). He was discharged home without complications.
The patient was seen at the outpatient clinic 1 month after discharge from the hospital, and he reported no respiratory complaints. Subsequently, he was lost to follow-up.
Discussion
This report describes a case of PCP in adults without known underlying immunosuppressive illness. In literature, PCP is uncommon in immunocompetent subjects.6,7 More rarely, few cases have been reported in immunocompetent without any underlying diseases like our patient.
The source and reservoir of infection in humans have not been yet identified, which makes understanding the mechanisms of Pneumocystis infection a crucial epidemiological challenge. 8 It is thought to result either from a de novo infection or from reactivation of a latent childhood infection. 9
Diagnosis of PCP is based on many arguments, including radio-clinical suspicion, patient risk factors, and confirmed by sputum studies, BAL fluid findings, or lung biopsies. 10
Radiological presentation is various. A chest X-ray will typically reveal diffuse bilateral peri-hilar interstitial infiltrates. Sometimes, it may show solitary or multiple nodules which may become cavitating, as well as the case of our patient. Chest CT may show ground glass attenuation or cystic lesions with high sensitivity. 10
Pneumocystis cannot be cultured. Thus, a definitive diagnosis is made by the detection and identification of the organism by polymerase chain reaction assays of respiratory specimens, histopathological staining (especially GMS staining), or fluorescein antibody staining. Tests in this manner require microscopic examination of a patient’s sputum or BAL fluid.6,10
In fact, GMS is a special stain to detect fungi. Fungal morphology on GMS is usually not specific enough to allow definitive species identification. However, it can be very useful when Pneumocystis is suspected on hematoxylin and eosin stain (HE).11,12
The Open-lung biopsy is the most invasive procedure with the highest sensitivity and specificity for diagnostic confirmation. 13
Treatment should be initiated in front of clinical suspicion of PCP in patients with known risk factors. 10 It depends on the clinical severity. In mild cases, treatment can be performed with oral therapies on an outpatient basis unless patients cannot tolerate oral therapies. In moderate or severe cases; patients require corticosteroids and a drug regimen that may only be given intravenously.14,15
Our patient was treated with a conventional dose of Sulfamethoxazole-Trimethoprim and he completely recovered without complications. Kosaka et al. showed that comparable survival rates were found between non-HIV-PCP treated with a conventional dose (15–20 mg/kg/day) and a low dose of Sulfamethoxazole-Trimethoprim (<15 mg/kg/day). However, the low-dose regimen is better tolerated and associated with fewer side effects. 16
Over the past two decades, the mortality rates of PCP have dropped because of earlier diagnosis and treatment. PCP has an even worse prognosis in patients without HIV infection, with mortality rates over 50%. Generally, the mortality rates are much higher when there is an underlying lung disorder and in patients who require mechanical ventilation.14,17
The same finding has been reported by Kim et al. 18 They published in 2021 a study based on the comparison of clinical characteristics and prognostic factors for mortality between immunocompromised (IC) and nonimmunocompromised (non-IC) patients treated for PCP. They showed that patients with PCP non-IC were older and had higher mortality rates and delayed anti-PCP treatment was associated with increased 90-day mortality rates. In this study, the majority of non-IC patients have underlying illnesses but this did not affect the mortality rate. 18
To our knowledge, no study has compared the characteristics of healthy immunocompetent patients to those of immunocompetent patients with comorbidities or to IC patients. Table 1 summarizes all the previous cases reported in the literature of healthy immunocompetent patients with PCP.
Summarize all previous cases of healthy immunocompetent patients with Pneumocystis jirovecii pneumonia across the literature.
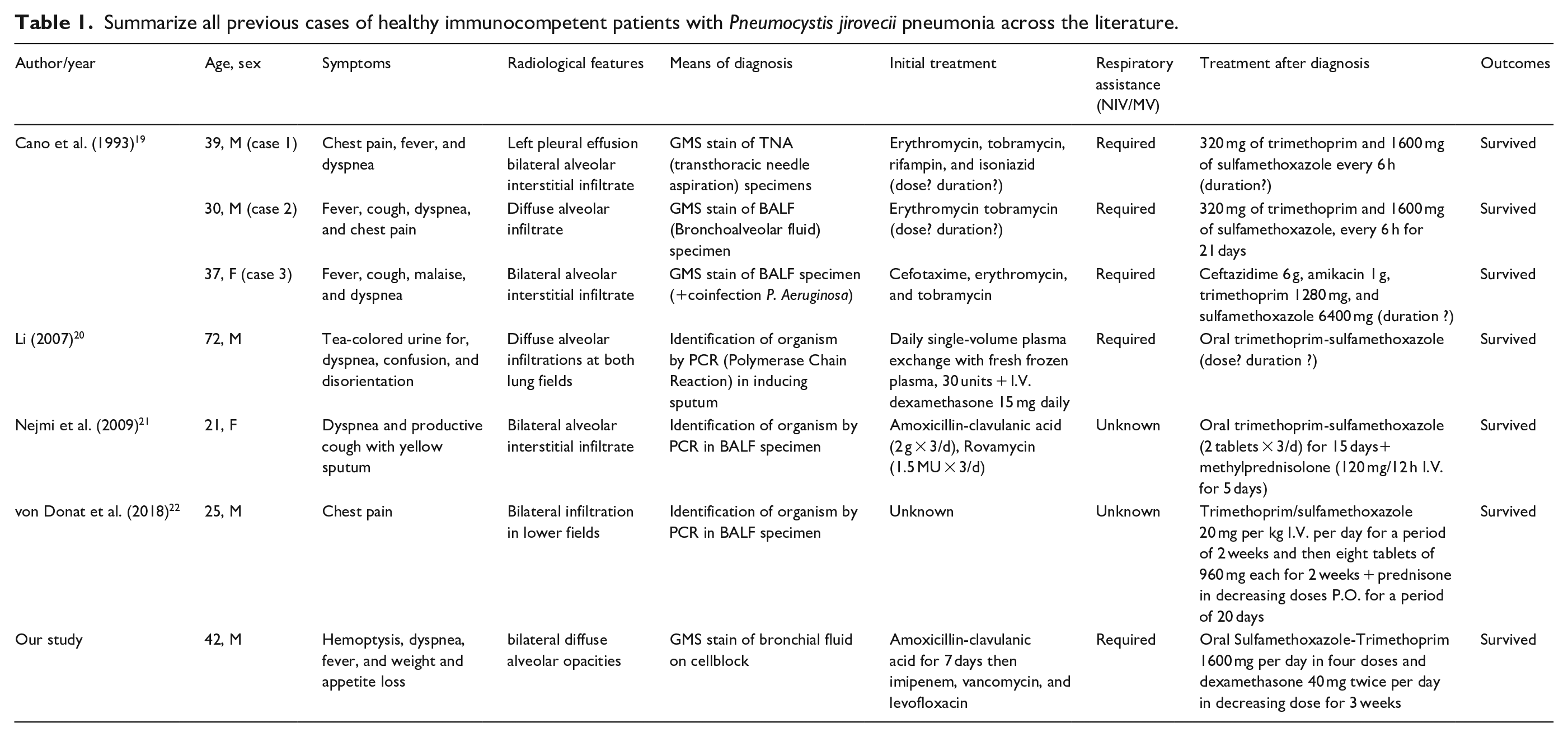
It has been noticed that despite the gravity of clinical presentation (requiring generally respiratory assistance), mortality was exclusively nil.
Conclusion
In conclusion, clinicians should be attentive to the possibility of PCP in healthy immunocompetent patients. If clinical symptoms and imaging features suggest a reasonable likelihood of PCP, then diagnostic confirmation and early treatment for PCP should be provided as soon as possible, regardless of the immune status, to provide the best outcomes.
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
H. R., and H. K. contributed to conceptualization; H. R., C. M., Y. H., and H. K., contributed in data collection; H. R., C. M., and H. K. contributed in the first writing of the original draft; Y. H., I. Z., and C. M. contributed to reviewing and editing; H. G. and S. M. contributed to validation. All authors read and approved the final article.
Availability of data and materials
The authors declare that any datasets used can be accessed.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The authors declare that appropriate written informed consent was obtained for the publication of this article and accompanying images.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
