Abstract
An 8-y-old Pygora doe was presented to the University of California–Davis, Veterinary Medical Teaching Hospital because of non-healing facial swelling of 2-wk duration. The lesion grew despite medical treatment, causing discomfort masticating, little-to-no airflow from the right nasal passage, and led to euthanasia. On gross examination, a large facial mass with a draining tract through the skin and hard palate was identified. On section, the mass was brown-pink, homogeneous, and friable. Abscess-like masses were identified in the lungs and kidney. Histopathology of the face, including oral and nasal cavities, salivary glands, and lymph nodes, as well as the lung and kidney lesions, revealed large areas of necrosis with numerous wide ribbon-like, mostly aseptate, fungal hyphae consistent with zygomycetes. PCR for fungal organisms performed on formalin-fixed, paraffin-embedded tissue from the face identified Lichtheimia corymbifera (formerly Absidia corymbifera) of the order Mucorales and an Aspergillus sp. The lesion was suspected to have started either as a fungal rhinitis or dental feed impaction, subsequently spreading to the face and systemically to the lungs and kidney. We describe here the lesions associated with facial mucormycosis in a goat and present a literature review of L. corymbifera infection in veterinary species and fungal infections in goats.
Keywords
An 8-y-old female Pygora goat was presented to the University of California–Davis, Large Animal Veterinary Medicine Teaching Hospital because of a 2-wk history of non-healing facial swelling, inappetence, and lethargy. The goat lived outdoors as a pet with 2 ponies and 4 other goats. The referring veterinarian was seen the day after the facial swelling was noticed, and the goat was treated with unknown doses of injectable steroids (drug unknown), furosemide, and penicillin, but the mass continued to grow and impeded the goat’s breathing and food consumption. Upon presentation, the goat was febrile, tachycardic, tachypneic, with absent right nasal airflow and enlarged mandibular and parotid lymph nodes. She was euthanized given the poor prognosis.
On postmortem examination, the facial mass significantly expanded the right side of the face. Externally, a 2-cm, circular, depressed, ulcerated focus exposed the underlying soft tissue. The right upper first molar was very loose, and a defect through the hard palate allowed visualization of deeply impacted feed material toward the nasal cavity (Fig. 1). This defect communicated with another defect medially from the periodontal defect that went through the hard palate (Fig. 2). On section, the mass was firm-to-friable, homogeneously pale-pink to tan, extending rostrally to 4 cm from the tip of the nose and caudally to the cranial pharynx. The cribriform plate was intact. The right nasal cavity was mostly occluded by the mass, but the midline was not shifted (Fig. 2). The right mandibular lymph node was firm and enlarged and, on section, had a central, irregular, pale-tan, soft mass with a thick capsule. All lung lobes had 3–5, pale, 0.5–1.5-cm, round-to-ovoid, firm nodules that, on section, were well-demarcated, and slightly oozed soft, white inspissated material. The kidneys had 5–6 similar but smaller, < 5-mm, nodules. Impression smears of the facial mass revealed numerous fungal hyphae in a background of cell debris and minimal mixed inflammation.
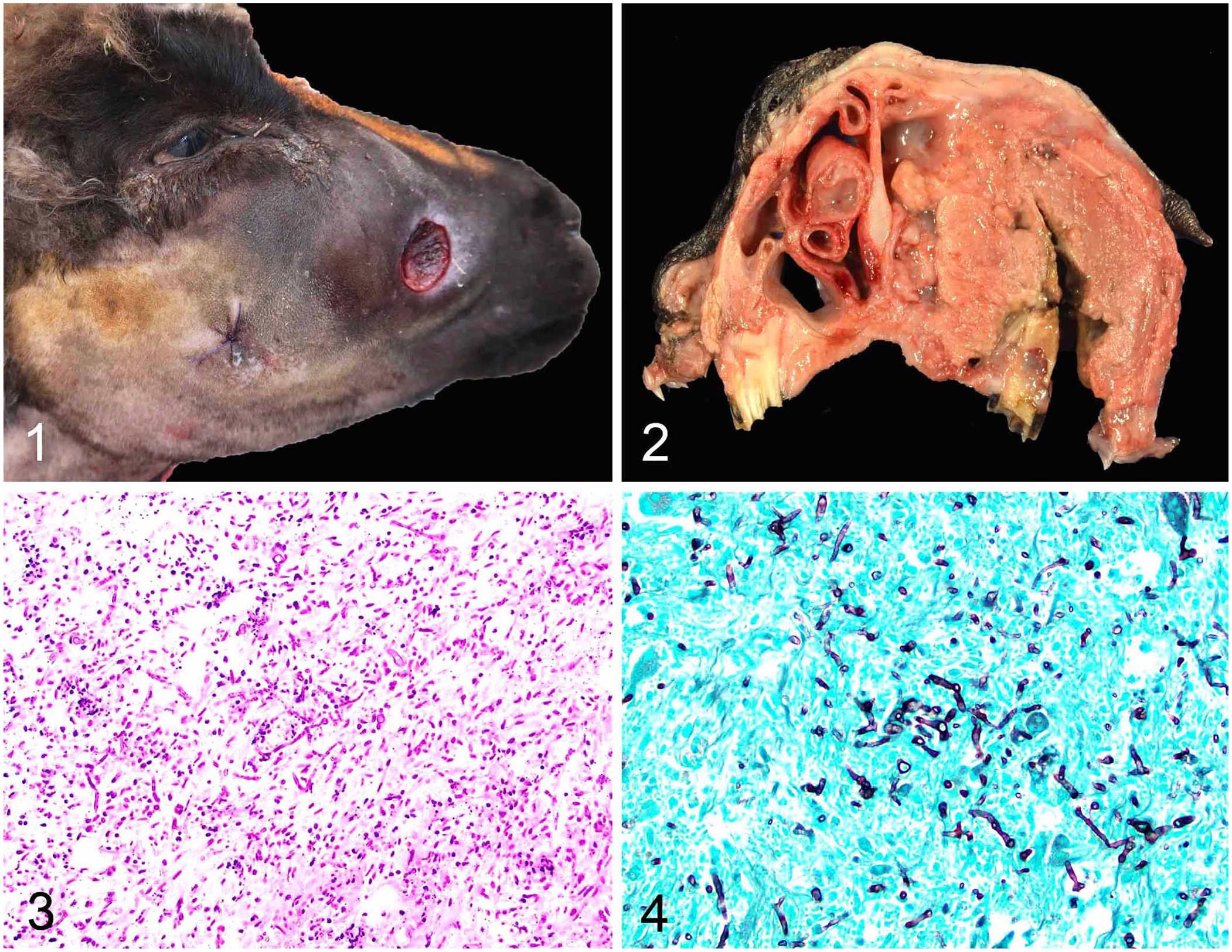
Gross and histologic features of facial mucormycosis in a goat.
Histologically, the oral mass was composed of large areas of mixed liquefactive and coagulative necrosis with numerous 4–15-μm wide, non–parallel-walled, fungal hyphae with occasional septa, with bulbous swelling and non-dichotomous branching (Figs. 3, 4). Neutrophils, macrophages, and multinucleate giant cells were restricted to the periphery of the necrotic area. Smaller fungal granulomas and vasculitis with hyphae were seen in the lungs, lymph node, and kidney.
Scrolls from formalin-fixed, paraffin-embedded sections of a section of the facial mass were submitted for fungal identification to the Washington Animal Disease Diagnostic Laboratory (Pullman, WA, USA). After DNA extraction, the D1D2 region of the 28S ribosomal RNA gene was amplified by PCR using universal fungal primers. The sequence of the PCR amplicon (GenBank OR730874) matched that of Lichtheimia corymbifera (100% sequence identity with GenBank MT312852) in the order Mucorales, formerly Absidia corymbifera. Additionally, a block of affected tissue was sent to the University of Florida Molecular Fungal ID Laboratory (Gainesville, FL, USA). Following DNA extraction, the resulting sequences obtained with panfungal PCR targeting the internal transcribed spacer (ITS) region matched L. corymbifera (GenBank OR667126) with 100% identity with L. corymbifera CBS 429.75 (GenBank NR_111413.1). An additional sequence targeting the 28S large subunit ribosomal region (LSU) matched Aspergillus sp. (GenBank OR920939) with 97.9% identity with Aspergillus assiutensis CBS 132773 (GenBank NR_070052.1).
Fungal and bacterial cultures were not pursued because the tissue was not available during further workup.
Previously, the order Mucorales was classified under the phylum Zygomycota as they reproduce by zygospores. Recent molecular phylogenic studies led to the abandonment of this phylum. Instead, pathologic zygomycetes have been regrouped under phylum Mucoromycota and Zoopagomycota.1,39 Under Mucoromycota is the order Mucorales and under Zoopagomycota is the subphylum Entomophtoromycotina, and the orders Basidiobolales and Entomophthorales which include the genera Basidiobolus and Conidiobolus, respectively. 1 Mucormycosis refers to a group of diseases caused by fungi in the order Mucorales. Major pathogens in this order include Mucor, Rhizopus, Lichtheimia, and Rhizomucor. Differentiating mucormycosis and entomophthoromycosis is difficult in the literature because these have been jointly called zygomycoses.
Reports of mucormycosis or zygomycosis in goats are limited despite this condition being reported in a wide variety of species. 33 A case report from Sudan describes 2 goats with mucormycosis from the same village. 26 In both of these cases, the lesions were primarily in the forestomach, suggestive of feed contamination, and in one case, spread to the lungs and liver. The diagnosis of mucormycosis was based on the histologic appearance of the fungi; further testing was not performed to identify the agent. In a report from Denmark of 27 goats with mycotic mastitis all suspected to be caused by Aspergillus fumigatus given its immunohistochemical reaction against a polyclonal antibody, one goat did have intralesional hyphae that either reacted to the A. fumigatus antibody or Rhizopus arrhizus antibody, suggestive of a dual infection. 17 Interestingly, although there are reports of entomophthoromycosis in sheep, caused mostly by the species Conidiobolus, 10 we were unable to find confirmed or suspected cases of natural mucormycosis in sheep when searched in PubMed and Google.
The fungal organism identified in our case through 2 independent molecular tests was Lichtheimia corymbifera. Fungal and aerobic culture on fresh tissue may have provided additional support to this diagnosis and may have identified coinfections that were not identified with molecular testing. L. corymbifera, like other mucormycetes, is a ubiquitous, saprophytic, filamentous fungus globally found in various substrates including soil, decaying vegetation, and unprocessed and processed foods, such as soybeans and flour, and is also a recognized opportunistic pathogen. 39 In the veterinary literature, L. corymbifera has been implicated most often in bovine abortion,19,34 but also as a cause of subcutaneous, lymph node, and cerebral lesions of cattle,18,29,36 systemic infection in horses,14,21,37 lymphadenitis in pigs, 38 peritonitis in a dog, 7 systemic infection in Scottish red deer, 28 pulmonary infection in Danish farmed deer, 16 and systemic infection in bank voles, 3 a pigeon, 30 a parrot, 5 and recently, marine mammals. 15
We retrieved no cases of rhinofacial mucormycosis that led to systemic mycosis in a probable immunocompetent goat caused by L. corymbifera in a search of Google and PubMed, using search terms “goat” or “caprine” and “mucormycosis” or “Mucorales” or “zygomycete,” suggesting that this condition has not been reported in the goat. An interesting observation during our search was that many of these case reports documented dual infection of L. corymbifera with Aspergillus spp.,14,16,17,19,37 a feature that we observed: one of the 28S sequences matched Aspergillus sp. We did not find reports of coinfection in other mucormycetes during our review. Although L. corymbifera infection with or without aspergillosis appears to be uncommon in veterinary species, future epidemiologic studies or pathogen studies may be of value to determine whether there is a synergistic effect between these 2 pathogens. Mucormycosis caused by other species of mucormycetes has been reported in many different veterinary species and is briefly presented elsewhere.15,33
In our case, the lesion extensively involved the maxillary molar, hard palate, and nasal cavity, similar to the lesions described in rhinofacial entomophthoramycosis caused by Conidiobolus spp. in sheep, 10 rarely goats, 23 and people, 1 and rhino-cerebro-orbital mucormycosis of people. 31 Additionally, secondary vascular dissemination to the local lymph nodes, lungs, and kidneys was identified. The extensive involvement of the nasal cavity in our case supported a primary fungal rhinitis, although a primary tooth root abscess due to dental disease and facial spread was also considered. Based on the presentation in other species, a nasal route of entry is suspected.
It is important to mention that there was no evidence of immunosuppression in our case clinically or on postmortem examination, although functionality of the immune system is admittedly difficult to determine on a postmortem basis aside from assessing disruption or depletion of major lymphoid organs and the presence of concurrent hematologic neoplasia or infectious diseases. An incidental thymoma was noted on gross examination and confirmed histologically; however, there is likely no correlation of the thymoma with the fungal disease in our case, given that thymomas in goats are quite common, generally incidental, and have not been reported to alter or disrupt the function of the systemic leukocytic population.
In general, fungal infections in goats are uncommon, even compared to sheep, and most are localized to the skin or oronasal cavity. The most common fungal infection, similar to other species, is due to cutaneous fungi, dermatophytes, and Malassezia.27,41 Common dermatophytes include Trichophyton verrucosum or T. mentagrophytes. Infection by dermatophytes and Malassezia spp. occur due to local dysbiosis of the skin, concurrent bacterial skin infections, or from penetrating or erosive trauma.12,27,41 Although Malassezia spp. infection typically incites localized or generalized seborrheic or exudative dermatitis typical of dermatomycosis, M. furfur has additionally been implicated as a cause of mastitis in goats in southern Florida. 12 Cryptococcosis, although still quite uncommon to rare, appears to be one of the most frequently reported fungal infections in goats and is reviewed in a 2020 article. 10
Aspergillosis, one of the most significant and common fungal diseases of veterinary species, has only been confirmed and reported in 2 goats in a report from Egypt as a cause of pneumonia, 24 in a goat with Aspergillus pneumonia with concurrent cholangiocarcinoma, paratuberculosis, and peritonitis, 11 in a mastitis outbreak from a Danish farm in which 27 of 73 goats were affected, 17 and in a case report of facial mycosis from Brazil. 8 Reports on Aspergillus or other fungal abortion in goats were difficult to find; one case reported was due to Candida spp. 25
Other reports of fungal disease in goats are more sporadic. Histoplasmosis has been reported in a goat with multicentric lymphoma from Texas, USA, 32 as well as adiaspiromycosis caused by Emmonsia spp. 20 A few cases of Pneumocystis pneumonia have been described and reviewed in goats, compared to cattle, sheep, and other mammals. 40 Blastomycosis with dermatophilosis has been described in Nigeria. 4 Exserohilum rostratum, a dematiaceous fungus, plant pathogen, and cause of an outbreak of fungal meningitis in people associated with contaminated methylprednisolone injections, 35 has been reported as a cause of rhinitis in a goat in Brazil. 6 As previously mentioned, there is a report of Conidiobolus facial mycosis in 2 goats from Brazil, 23 but none of basidiobolomycosis based on PubMed and Google searches. Systemic fungal infection tentatively identified as sterile fungi, a group of fungi without reproductive cells or conidia, that also involved the nasal cavity was reported in a Tur goat. 13 Fungus-like organisms such as oomycetes and algae have also been rarely reported. There is one report of oomycete Pythium sp. infection in a goat in Brazil in which the organism caused ulcerative dermatitis and necrotizing rhinitis with deformation of the nasal cavity. 9 There have been 2 cases in Brazil in which the pathogenic algae, Prototheca wickerhamii, was reported to cause rhinitis and facial lesions.2,22
Footnotes
Acknowledgements
We thank Dr. Marietta Barro for taking and uploading gross images to the UC Davis pathology database.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
