Abstract
Fungi are among the most common infectious agents affecting the skin of animals. The skin can serve as a port of entry for fungal infections, which can eventually become disseminated. In some regions of the world, oomycetes, such as Pythium and Lagenidium, are also responsible for a significant number of severe cutaneous infections. Histologic evaluation of fungal morphology, including size, shape, septation, branching, and budding characteristics, combined with the distribution of inflammatory infiltrates within different skin layers can potentially identify etiologic agents, guiding selection of antifungals and additional diagnostics. Fungal infections of the skin surface are typically caused by Malassezia and rarely Candida, with opportunistic fungi also capable of colonizing the skin surface, especially when the barrier is broken. Folliculocentric infections, caused by dermatophytes, result in mild to severe inflammation and can occasionally penetrate deep into the skin. A wide range of fungi, including agents of hyalohyphomycosis, phaeohyphomycosis, and dimorphic fungal infections, as well as oomycetes, result in nodular cutaneous and subcutaneous lesions. With the occasional exception of dimorphic fungi, fungal speciation often requires cultures performed on fresh tissues. However, molecular techniques such as pan-fungal polymerase chain reaction on paraffin blocks is becoming an increasingly useful tool to distinguish between cutaneous fungal pathogens. This review focuses on describing the clinical and histologic features of the most common fungal and oomycete infections affecting the skin of animals, divided according to distribution patterns of lesions and fungal or oomycete morphology.
Keywords
Numerous fungal species are known to inhabit the skin surface of humans and animals, and these vary across different body sites and hosts. 84 The first encounter with fungal organisms occurs at birth during passage through the vaginal canal. 44 Relative to human skin, which is mainly colonized by Malassezia spp, the skin of animals is inhabited by highly diverse fungal communities. In dogs, predominant fungal genera identified on the skin surface include Alternaria, Cladosporium, and Epicoccum, 59 which are likely acquired from the environment. In addition, variable proportions of Malassezia spp, including Malassezia globosa, Malassezia restricta, and Malassezia pachydermatis, have been identified in healthy canine skin. 61 Similarly, Cladosporium, Alternaria, and Epicoccum are some of the most abundant genera identified in the skin of cats. 58
These microorganisms are believed to contribute to skin health; however, an imbalance, either primary or secondary, in these microbes may lead to the development or aggravation of skin lesions. For instance, canine patients with otitis have microbial dysbiosis with increases in the yeast M. pachydermatis and increased Staphylococcus and Streptococcus. 4 Devon Rex and Sphynx cats, which often suffer from seborrheic dermatitis, have higher carriage rates of Malassezia spp, even in the absence of skin lesions. 75
Several fungal and oomycete species can penetrate through skin defects and are responsible for causing superficial and/or deep infections, occasionally resulting in systemic disease. Mycotic infections have become more prevalent over the years, with novel fungal pathogens being identified frequently.15,56 One of the reasons for the increased number of fungal infections, especially opportunistic infections, is the higher number of immunosuppressed individuals being treated for cutaneous or systemic immune-mediated diseases or receiving chemotherapy treatments. 15 In addition, in some regions of the United States and other countries, oomycete infections, such as those caused by Pythium insidiosum, can occur in immunocompetent individuals, resulting in severe cutaneous infections often requiring surgical excision and frequently failing to respond to antifungal treatments.
General practitioners and dermatologists use multiple tools to confirm common cutaneous fungal infections, including signalment, clinical history, geographic location, and travel history, as well as clinical appearance and distribution of lesions. Diagnostic methods that are commonly used in practice include Wood’s lamp examination, trichography, and Sabouraud dextrose agar or dermatophyte test medium (DTM) cultures as well as direct impression smears for superficial skin infections, such as infections with Malassezia spp. 65 Identification of subcutaneous and deep infections often requires collection of skin biopsies for fungal culture and histological examination. 65 While insufficient for speciation in most cases, histologic evaluation of fungal morphology in skin biopsies can provide important clues to the pathologist in narrowing the list of potential etiologic agents and guide the selection of additional diagnostics. 35 Histochemical techniques are frequently employed to complement this evaluation, most commonly the periodic acid-Schiff (PAS) reaction and Grocott’s methenamine silver (GMS) stain. Fungal cell walls contain polysaccharides that form aldehyde groups upon oxidation and can be detected by both stains. 92 In the PAS reaction, periodic acid oxidizes the polysaccharides, and the resultant aldehydes react with the Schiff reagent to produce a bright magenta hue. 92 In a GMS procedure, silver ions bind to aldehyde groups and are reduced, imparting a black color to the fungal cell wall. 92 While generally thought to require more technical precision than PAS, GMS is more sensitive and will detect nonfungal oomycete organisms, in contrast to PAS. 10 GMS also has the benefit of staining both living and nonviable fungi, while PAS will only work consistently on living fungi.10,92
As fungi are widespread in the environment, and thus are frequent skin contaminants, it is crucial to correlate organisms cultured from skin lesions with the morphologic appearance of fungal elements identified in the tissue to confirm the pathogenic significance of the fungi.35,44 Thus, it is imperative for pathologists to be familiar with the morphology of common fungal and oomycete agents, not only to confirm culture results but also to be able to distinguish among different types of infections and assist clinicians in choosing the best treatment for different fungal diseases. This review will focus on describing the clinical and histologic features of the most common fungal and oomycete infections affecting the skin of animals, divided according to distribution patterns of lesions and fungal or oomycete morphology. This information is summarized also in Tables 1 and 2.
Typical morphologic features of fungi causing superficial, folliculocentric, or deep dermal to subcutaneous infections in animals.
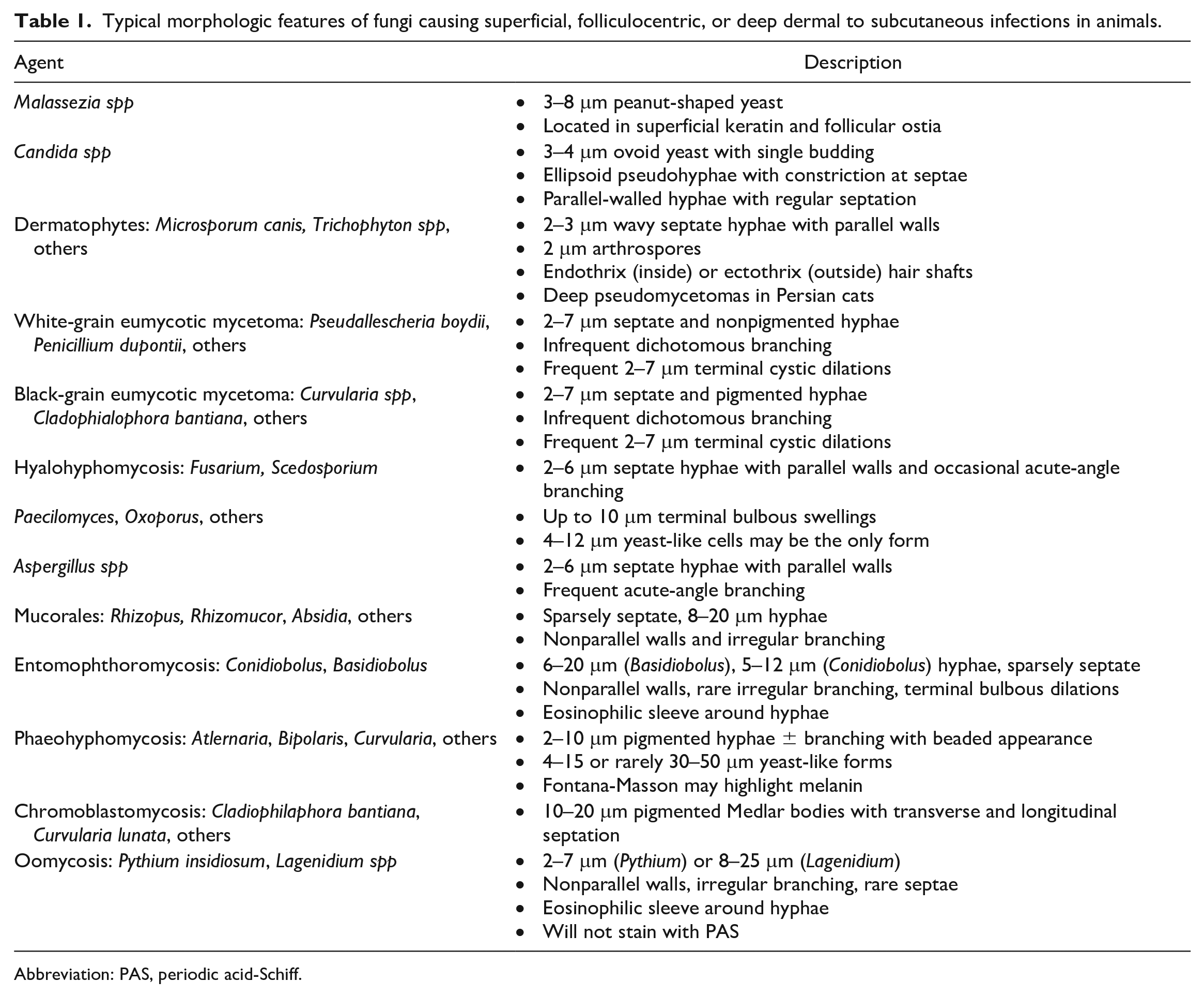
Abbreviation: PAS, periodic acid-Schiff.
Typical morphologic features of dimorphic fungi causing cutaneous infections in animals.
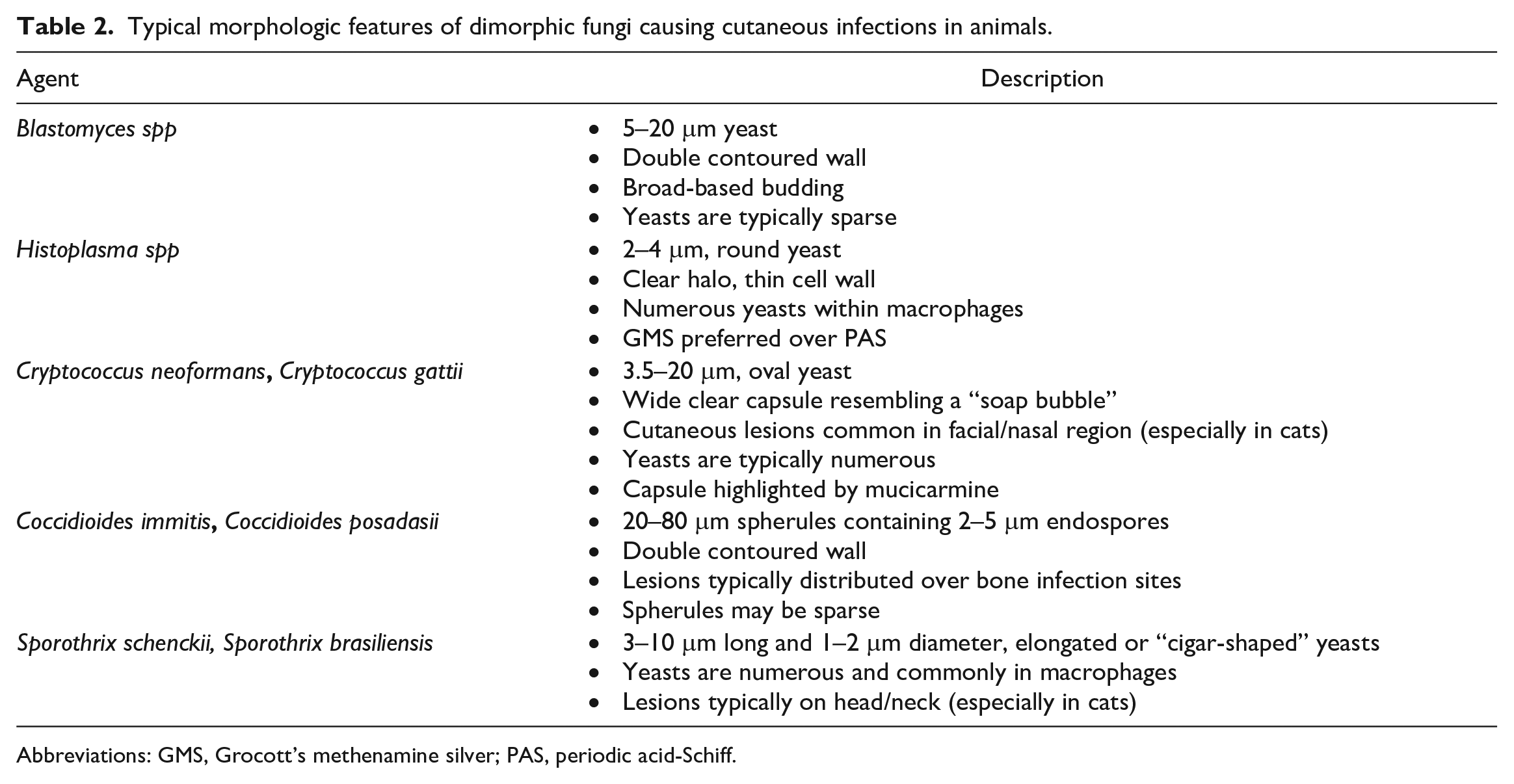
Abbreviations: GMS, Grocott’s methenamine silver; PAS, periodic acid-Schiff.
Superficial Fungal Infections
Malassezia spp
Dermatitis associated with Malassezia spp, typically M. pachydermatis, is common in dogs and comparatively rare in cats, horses, and ruminants.34,89,90 Malassezia spp are often lipid-dependent yeasts and ubiquitous commensal microorganisms in many domestic species that may act as secondary, opportunistic pathogens.13,36 The pathogenesis of Malassezia dermatitis is still debated, but hypersensitivity disorders, cornification defects, and endocrinopathies are common underlying predispositions.8,52 Numerous studies indicate a role of hypersensitivity toward yeast antigens in dogs, with the same suspected to be true for horses.39,70,90 Therefore, the number of yeasts required to induce dermatitis appears to be highly variable. In cats, while Malassezia overgrowth has been reported in various dermatopathies, including allergic dermatitis, Malassezia-associated dermatitis is frequently a sign of severe underlying systemic disease and has been reported in cats with thymoma-associated exfoliative dermatitis, paraneoplastic alopecia, and superficial necrolytic dermatitis.24,27,55
Clinically, Malassezia-associated dermatitis presents as alopecia with greasy to waxy scales/crusts and a characteristic musty odor.34,36 With chronicity, the skin can become hyperpigmented and lichenified. 36 The ventral neck or abdomen, intertriginous regions, pinnae, feet, and skin folds are commonly affected sites in dogs and cats, while horses typically demonstrate intertriginous involvement.34,90 Otitis externa is particularly common in dogs and occurs in up to 70% of dogs with Malassezia-associated dermatitis. 23 Rare reports in cattle also indicate a propensity for otitis externa, while a similar presentation to dogs has been described in a goat.22,79 A unifying feature of this condition in all species is pruritus.34,90 Predisposed dog breeds include the West Highland White Terrier, Cairn Terrier, Dachshund, and Newfoundland, among others. 34
Histologic findings include moderate to marked epidermal hyperplasia involving the stratum spinosum to around 10 layers thick, extending to the follicular infundibula, typically with characteristic scalloping or “festooning” of the epidermal margin (Fig. 1a). 34 This is accompanied by mixed parakeratotic hyperkeratosis with multifocal mounds of keratin or crust formation, a mixed dermatitis involving eosinophils and lymphocytes, and lymphocytic exocytosis of predominantly T lymphocytes. 52 Hyperkeratosis is the most consistent finding in cats, which typically lack the festooning described in dogs. 53 Unfortunately, while epidermal festooning may be suggestive, the histologic appearance is not specific for Malassezia-associated dermatitis and can be seen in any chronic allergic dermatitis. Further complicating a histologic diagnosis is the low sensitivity of histology compared with cytology for the detection of yeasts, with approximately 30% of biopsies reported to be falsely negative. 55 Yeasts are best identified in the superficial keratin, particularly in follicular ostia, and may be seen in clusters or adhered to keratinocytes.34,90 The yeasts are 3–8 μm in diameter, exhibit monopolar budding, and are classically peanut-shaped (Fig. 1b; Table 1). A PAS reaction can aid in identifying the yeasts in keratin layers (Fig. 1b, inset).
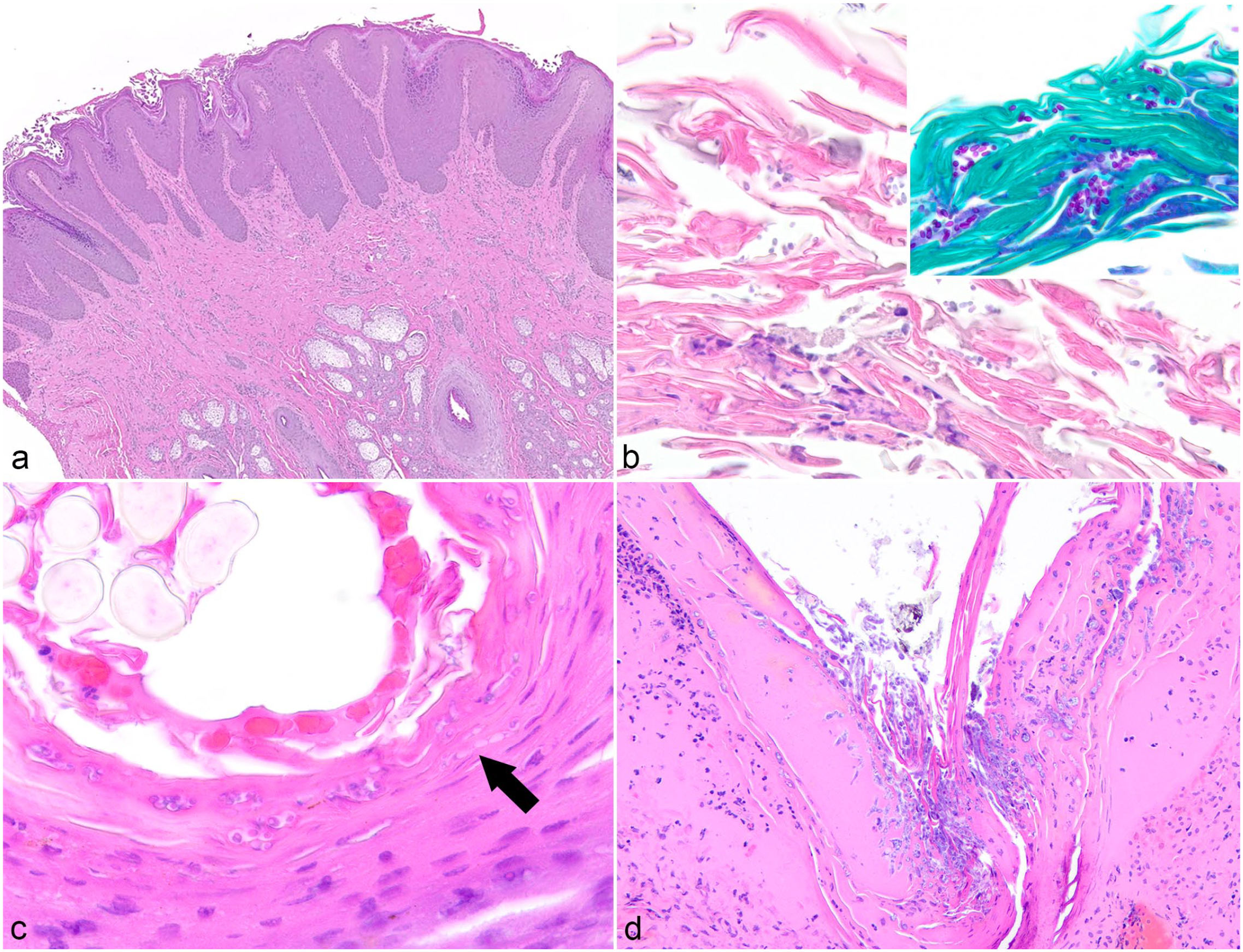
Histopathologic features of superficial fungal skin infections in domestic animals. Hematoxylin and eosin. (a) Haired skin, dog. Hyperplasia with scalloping or festooning of the epidermis and hyperkeratosis is typical of Malassezia spp–associated dermatitis. (b) Haired skin, dog. Malassezia pachydermatis yeasts can typically be found in the superficial keratin. Inset: The yeasts are characteristically peanut-shaped and can be highlighted with periodic acid-Schiff reaction. (c) Candida spp–associated dermatitis in the footpad of a dog. Yeasts (circle) and pseudohyphae with constriction at the septae (arrow) can be identified superficially. (d) Haired skin, dog. Numerous fungal hyphae are identified superficially in a serocellular crust. The absence of hyphae and associated inflammation in the underlying tissue indicates this is a contaminant.
Candida spp
Candida spp are common commensal microorganisms in the gastrointestinal and urogenital tracts of both humans and domestic animals. Cutaneous candidiasis, most frequently associated with Candida albicans, is extremely rare in domestic animals. 30 Infections are usually opportunistic and are secondary to a combination of underlying immunosuppression secondary to immunosuppressive drug therapy or debilitating systemic disease, such as diabetes mellitus or hyperadrenocorticism, alongside disruption of the normal cutaneous/mucosal barrier function and endogenous microbiome, as occurs with prolonged antibiotic treatment.68,71 Candida spp are a well-recognized cause of urinary tract infections in dogs and cats with diabetes mellitus, for example. 15 Vulvovaginal candidiasis has also been reported in horses following administration of a synthetic progestogen. 66
Most reports of cutaneous or mucocutaneous candidiasis describe young pigs or foals, in which the condition is known as “thrush.” The typical presentation involves formation of pale, pseudomembranous plaques with ulcers on the tongue and gingiva. 57 Other affected sites in domestic animals, including horses, dogs, and pigs, are the oral mucocutaneous junction and distal extremities, with less common locations being the perineum, vulva/scrotum, external ear canal, and intertriginous sites.30,34,90 Erosions and ulcers are frequently covered by gray, malodorous, partially adherent exudate with variable pruritus.30,89 Histologically, candidiasis is associated with the formation of large areas of superficial pustulation containing predominantly neutrophils accompanied by serocellular crust formation, spongiosis, and erosion to ulceration.30,71 Candida spp are unique among the cutaneous opportunistic fungal pathogens in that the organism grows superficially as yeasts, hyphae, and pseudohyphae simultaneously (Fig. 1c). Yeasts are 3–4 μm in diameter, ovoid, and exhibit single budding (Table 1). 6 Pseudohyphae are elongated ellipsoid cells with constrictions at the septae, while the parallel-walled true hyphae have regular septation without constriction. 6 The presence of hyphae may lead to suspicion of superficial pustular dermatophytosis, although the addition of budding yeasts should steer the pathologist toward the identification of Candida spp. Organisms will be present in crusts or pustules, and a PAS reaction will assist in highlighting the various morphologies of Candida spp in tissues.
Surface Contaminants
Fungi are ubiquitous in the environment and, as such, are both part of the normal cutaneous microbiome and frequent contaminants in fungal culture. Numerous commensal species present on the skin of normal animals may serve as opportunistic pathogens. In horses, common cutaneous microbiota include Alternaria and Cladosporium, which are reported causes of phaeohyphomycosis, as well as Fusarium and Aspergillus. 91 In cats, the most commonly isolated fungi are Cladosporium and Alternaria. 58 Mucorales spp are also widely present in the environment and can be frequently found on the surface of animal skin or hair. 34 Hyphae may therefore be observed on the surface of keratin or crusts on a biopsy but do not automatically represent pathogens (Fig. 1d).
Several factors must be considered before ascribing a histologically evident fungus to disease, the most important of which is the presence of fungal elements in tissue. A fungus identified or isolated from the skin surface or exudate may represent a contaminant, while fungi observed within or cultured from tissue, particularly an aseptically collected biopsy specimen, are far more likely to be pathogenic. The species of fungus is also key to consider as if even one colony of a dimorphic fungus (ie, Blastomyces or Histoplasma) is cultured, or if such fungi are observed histologically, it is highly likely to represent a pathogen. On the other hand, isolation of a few colonies of a saprophytic, opportunistic fungus or observation of non-dermatophytic hyphae on the surface without accompanying inflammation or presence of the agent in tissue is likely to represent contamination rather than a fungal dermatitis.
Folliculocentric Fungal Infections
Dermatophytosis, also known as ringworm or tinea, is a superficial skin infection involving keratinized structures, such as the stratum corneum, hair, and nails. These infections have a worldwide distribution with increased prevalence in high humidity areas. 69 The most common dermatophyte genera include Microsporum, Trichophyton, and Epidermophyton, with certain species adapted to humans (anthropophilic), animals (zoophilic), or the environment (geophilic) (Table 3).26,54 One of the most common species that causes infections in dogs and cats is Microsporum canis. 69
Common dermatophyte species in domestic animals (reproduced from Mauldin & Peters-Kennedy, 2016). 54

The genera include Microsporum and Trichophyton.
+=uncommon; ++ common; +++ most frequent isolate(s)
Abbreviation: n/r, not reported.
Affected dogs and cats present with a variety of clinical lesions, including alopecia, scales, crusts, erythema, and less frequently papules or pustules, often with an asymmetrical distribution pattern. 69 In ruminants and horses, lesions tend to be characterized by circular areas of alopecia with crusting and scaling.34,90 Dermatophytic lesions can be mild and self-limiting in many animal species; however, dermatophytic infections can be easily spread to humans and other animals, with outbreaks commonly occurring in shelters and confined animals. 69 The nonspecific nature of skin lesions may be difficult to distinguish from other common skin diseases, such as superficial pyoderma and demodicosis. As such, dermatophytosis can often be overdiagnosed, especially in dogs. 78 Persian cats and Yorkshire Terrier dogs appear to be predisposed to clinical dermatophytosis, with Persians often presenting with a more chronic and generalized disease compared with other cat breeds. 69
First and foremost, it is important for clinicians and pathologists to recognize that histology is not as sensitive as culture for identification of dermatophytes and thus should not be the diagnostic test of choice in suspected cases of dermatophytosis. 34 However, histology can be useful in cases where dermatophytosis is not initially suspected and in patients with deep skin infections. Histologically, the pattern of lesions may vary depending on the animal species and region of the body affected (haired skin versus nails). In general, dermatophytes infect hair shafts, resulting in areas of alopecia, and thus the most common pattern of histologic lesions is perifolliculitis, folliculitis, and furunculosis (Fig. 2a). 54 The inflammatory infiltrates are often composed of neutrophils, macrophages, lymphocytes, and plasma cells. When follicles rupture, the inflammation becomes more severe and may become predominantly pyogranulomatous with eosinophils often resulting in a “foreign body reaction,” in part due to release of keratin and hair shafts into the dermis (Fig. 2b). 54 Acantholytic cells may be seen within follicles and the follicular ostia. Hair shafts within intact and nonintact follicles are often infiltrated by numerous narrow, 2- to 3-μm diameter, negatively staining, frequently wavy, septate hyphae with parallel walls and basophilic, approximately 2 μm in diameter, round arthrospores inside (endothrix) or on the edges of hair shafts (ectothrix) (Fig. 2c; Table 1). 34 Hair shafts entrapped within crusts should also be evaluated as this occasionally may be the only location in which dermatophytic organisms are observed. In patients with perifolliculitis, folliculitis, and/or furunculosis where no obvious organisms are observed, it is crucial for pathologists to run special stains such as PAS or GMS to identify dermatophytes within lesions, as these may be easily missed if only routine stains are used. Orthokeratotic and parakeratotic hyperkeratosis with serocellular crusting is often observed. 54
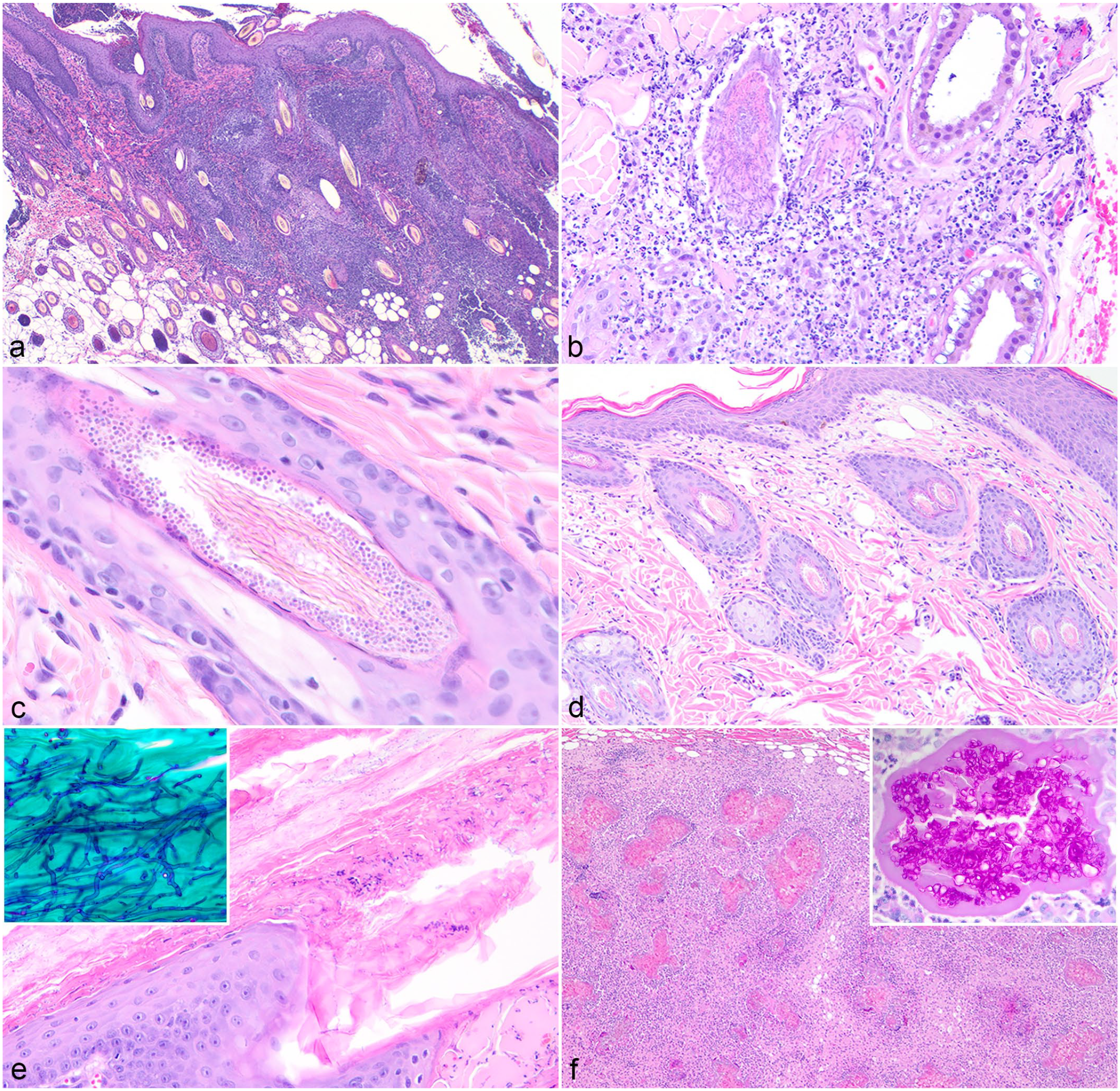
Histopathologic features of cutaneous dermatophytosis in domestic animals. Hematoxylin and eosin. (a) Folliculitis and furunculosis associated with Microsporum gypseum infection in a dog. (b) Pyogranulomatous inflammation secondary to furunculosis in a dog with M. gypseum infection. (c) Numerous arthrospores surround and hyphae infiltrate a hair shaft in a cat with dermatophytosis. (d) Minimal to mild perifolliculitis in a cat with dermatophytosis. (e) Haired skin, cat. Dermatophyte hyphae may be observed exclusively in the stratum corneum rather than the typical location of hair shafts. Inset: Periodic acid-Schiff (PAS) reaction highlights narrow dermatophyte hyphae within the stratum corneum. (f) Dermatophytic pseudomycetomas, typically seen in Persian cats and caused by Microsporum canis, are composed of pyogranulomatous inflammation surrounding large central aggregates of hyphae. Inset: PAS stain highlights the densely tangled hyphae.
In cats with dermatophyte infections, it is not uncommon to see mild perifollicular infiltrates of lymphocytes, histiocytes, and occasionally neutrophils and plasma cells (Fig. 2d), and in disseminated cases, dermatophyte hyphae may extend beyond hair follicles and infiltrate the stratum corneum. 34 In infections caused by Trichophyton mentagrophytes and Microsporum persicolor in dogs, dermatophytes may be found exclusively in the stratum corneum (Fig. 2e). 54
In rare cases, dermatophytes may penetrate into the dermis and form nodules within the skin, including dermatophytic mycetomas, also known as pseudomycetomas and kerion. 65 The nodular lesions of dermatophytic pseudomycetomas are mainly seen in Persian cats, which exhibit subcutaneous nodules with draining tracts and intralesional tissue grains. 34 Histologically, these lesions are characterized by pyogranulomatous inflammation with large central aggregates of thick-walled hyphae forming a “mycetoma” (Fig. 2f). 34 In kerion, the pyogranulomatous inflammation surrounds a central hair shaft with fungal hyphae and spores. 69
Fungal Infections Causing Cutaneous and Subcutaneous Nodular Lesions
General Features
The following sections describe cutaneous infections caused by a variety of opportunistic, saprophytic fungi. Given the wide distribution of such fungi in nature, histologic demonstration of the organism in tissue with accompanying inflammation is required to associate these fungi with disease. Cutaneous infections typically develop due to traumatic implantation or, less commonly, are secondary to disseminated disease.13,34,96 Defects in the immune response are often present or suspected in cases of systemic disease with opportunistic fungi and in some cases of cutaneous infections. The frequency of such cutaneous infections has perceptibly increased over the past 2 decades following the introduction and widespread use of cyclosporine in dogs.15,56 Such a trend has also been observed in human medicine, and the risk of opportunistic fungal infection increases with simultaneous use of multiple immunosuppressive medications in both humans and animals.15,56 Natural causes of immunosuppression, including diabetes mellitus, retroviral infections, and lymphoid neoplasia, have been documented as predisposing factors in cats.15,56,101 German Shepherd dogs are also noted to be predisposed to disseminated aspergillosis and hyalohyphomycosis due to a suspected familial immunodeficiency.15,97
Cutaneous infections by opportunistic fungi typically present as grouped nodules or masses in the dermis and/or subcutis that tend to enlarge and coalesce over time.15,34,96 Lesions may become ulcerated and develop fistulous draining tracts. Histologically, inflammation is typically pyogranulomatous to granulomatous and may involve a significant component of eosinophils, depending on the fungal genera.15,64,96 These fungi typically grow as hyphae in tissues, and PAS or GMS stains can be helpful in highlighting the fungi and their structural features to aid in classification. While clues may be obtained as to the taxonomic category of opportunistic fungi by observing histologic morphology, it is critical to note that histopathology alone cannot speciate in cases of opportunistic fungal infections. In contrast, the dimorphic fungi, which act as primary pathogens, are typically distinct enough morphologically to allow for a diagnosis based on histology alone.
Before describing the major categories of opportunistic fungal cutaneous infections, it is important to review the appropriate terminology which is oftentimes confusing and frequently changes. Hyalohyphomycosis refers to all opportunistic, non-dermatophytic fungal infections caused by saprophytic fungi that grow as nonpigmented hyphae in tissue. Phaeohyphomycosis is caused by pigmented dematiaceous fungi, which grow as hyphae and yeast-like cells. The term chromoblastomycosis is sometimes used to refer to pigmented fungi that are seen predominantly as large, rounded, thick-walled cells called sclerotic bodies or Medlar bodies. The two are occasionally lumped together under the largely historical term, chromomycosis. Opportunistic fungi may also result in the formation of large nodules containing distinctive tissue granules or grains, called eumycotic mycetomas. Confusingly, some of the same fungal species that may cause either hyalohyphomycosis or phaeohyphomycosis may alternatively be agents of eumycotic mycetomas. The eumycotic mycetoma, however, is classified separately based on its characteristic appearance and not on the inciting fungal species. Finally, the phylum Zygomycota has recently been reclassified into the phyla Mucoromycota, which includes the subphylum Mucoromycotina (Mucor, Rhizopus, etc), and Zoopagomycota, which includes the subphylum Entomophthoromycotina (Conidiobolus, Basidiobolus). 94 Clinical, histologic, and fungal morphologic features of each of the above categories will be summarized in the following sections.
Eumycotic Mycetoma
Mycetomas are dermal to subcutaneous nodules that exhibit 3 characteristic features: tumefaction, grains or granules within the exudate, and a draining tract, although the third feature is not always present.34,96 They may be caused by certain types of either bacteria (actinomycotic) or fungi (eumycotic). The tissue grains represent large aggregates of the infectious organisms, fungal hyphae in the case of eumycotic mycetoma. In animals, eumycotic mycetomas are usually solitary and located on the distal limbs or face, particularly the nostrils and lip commissures in horses.90,96 The nodules are relatively large (1–10 cm in diameter), firm, and may ulcerate. Over time, progressive fibrosis imparts a firm, tumor-like appearance to the eumycotic mycetoma.16,90 The portal of entry for fungi is thought to be wound contamination or direct implantation, and this lesion is most commonly reported in subtropical to tropical climates. 90 Eumycotic mycetomas may be classified as “black-grain” when caused by pigmented hyphae or “white-grain” when the hyphae are nonpigmented. Numerous fungal species have been reported as causes of eumycotic mycetoma. White-grain mycetomas are most often associated with Pseudallescheria (Scedosporium) boydii and Penicillium (Thermomyces) dupontii, while black-grain mycetomas are typically caused by Curvularia lunata, Curvularia geniculata, Cladophialophora bantiana, and less commonly Phialophora oxyspora and Madurella spp.3,16,90,95 Interestingly, white-grain mycetomas in dogs are typically intraperitoneal with possible connection to the body wall and formation of draining tracts, presumably from a penetrating wound or surgical procedure in the preceding months or years.15–16 Black-grain mycetomas, however, are typically in the skin or subcutis.
The nodules are composed of (pyo)granulomatous dermatitis and panniculitis with fungal hyphae present in 0.2–5 mm diameter, central aggregates which comprise the clinically evident tissue grains (Fig. 3a–c). Hyphae are 2–7 μm in diameter and septate with infrequent dichotomous branching and frequent formation of 5- to 20-μm diameter, terminal cystic dilations (Table 1).16,30,90 These dilations, called chlamydoconidia, are commonly seen at the outer edge of the tissue grain with the septate hyphae tangled centrally (Fig. 3d). The fungi may or may not be pigmented and are held together by eosinophilic, amorphous material representing Splendore-Hoeppli reaction. 34 Surrounding the fungal aggregates is a zone of degenerate neutrophils further surrounded by macrophages and variable numbers of multinucleated giant cells with variable enclosing fibroplasia. The pathologist may diagnose an eumycotic mycetoma based on this characteristic histologic appearance and specify whether the fungi are pigmented or not. However, speciation requires culture or polymerase chain reaction (PCR).
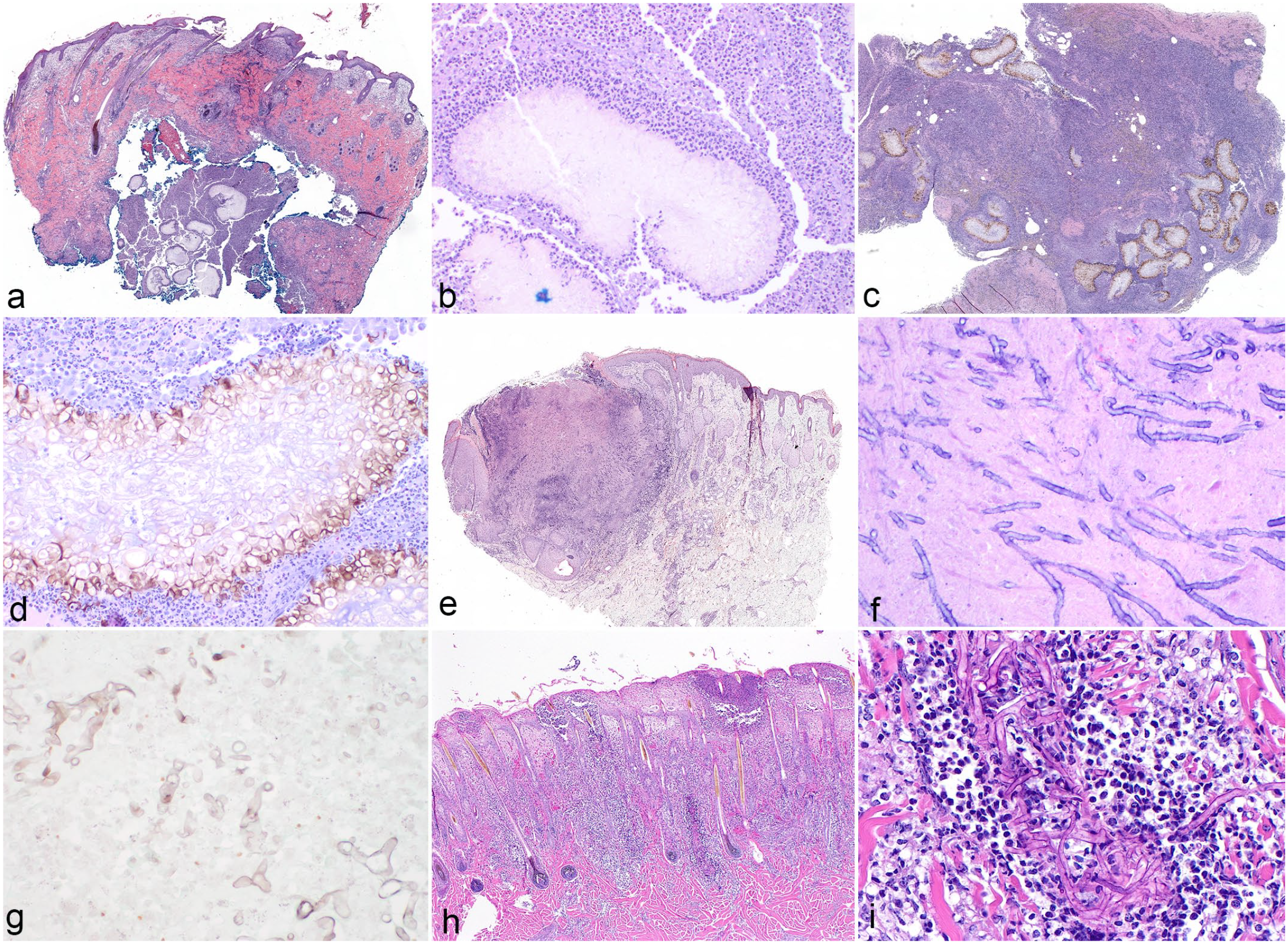
Histopathologic features of nonpigmented and pigmented eumycotic mycetomas and cutaneous hyalohyphomycosis in domestic animals. (a) White-grain eumycotic mycetoma due to Pseudallescheria (Scedosporium) spp in a horse. Mycetomas are composed of pyogranulomatous dermatitis and panniculitis with central aggregates of hyphae that form the clinically evident tissue grains. Hematoxylin and eosin (HE). (b) Higher magnification view of (a) demonstrating the dense arrangement of nonpigmented hyphae in tissue grains. HE. (c) Black-grain eumycotic mycetoma due to Curvularia spp in a horse. Note the dense aggregates of pigmented fungal hyphae surrounded by inflammation. HE. (d) Higher magnification of (c) demonstrating terminal dilations, or chlamydoconidia, located at the edge of a tissue grain. HE. (e) Necrotizing and pyogranulomatous dermatitis and panniculitis due to Aspergillus spp in a horse. Note the ulceration and expansion of the underlying tissue by inflammatory cells. HE. (f) Higher magnification of (e) demonstrating the typical septate, parallel-walled hyphae with acute-angle branching (arrows) of Aspergillus spp HE. (g) Dermatitis due to Rhizopus microsporus infection in a cat. Broad, pauciseptate hyphae with acute- and irregular-angle branching are typical of the Mucorales fungi. Grocott’s methenamine silver. (h) Haired skin, aborted bovine fetus. Ulcerative and hyperkeratotic dermatitis, folliculitis, and furunculosis associated with Saksenaea erythrospora infection. HE. (i) Higher magnification of (h) demonstrating the nonparallel walls, sparse septations, and irregular branching of Mucorales fungi. HE.
Hyalohyphomycosis
Nonpigmented fungal hyphae may cause nodular cutaneous lesions without the distinct tissue grain formation of mycetomas, falling under the general term hyalohyphomycosis. Various fungi are associated with cutaneous infections, including Fusarium and Scedosporium, with systemic disease also reported for Acremonium, Geomyces, Lemontospora, Oxyporus, Paecilomyces, Penicillium, Rasamsonia, and Talaromyces.5,16 Both immunocompromised and immunocompetent animals may develop nodular to ulcerative, dermal to subcutaneous lesions, most often on the distal extremities, or systemic disease. 15
Lesions are composed of neutrophilic to pyogranulomatous inflammation with numerous nonpigmented fungal hyphae seen extracellularly and within macrophages and multinucleated giant cells.15–16 Occasionally, hyphae may invade blood vessels and cause thrombosis, a change which is more frequently reported with fungi of the order Mucorales or Aspergillus spp. 49 Hyphae are typically narrow (2–6 μm wide) and basophilic with regular septation, parallel walls, occasional acute-angle branching, and up to 10-μm diameter bulbous swellings either at the terminus or between septations (Table 1).42,43,64,96 Yeast-like cells may be observed and rarely may be the only morphology present. These structures are 4–12 μm in diameter and result from conidiogenesis, a process that typically requires the presence of air, but which several fungi can achieve within tissue, including Fusarium, Paecilomyces, and Scedosporium. 100 Importantly, when nonviable, hyphae may appear negatively stained and pauciseptate, and they may be confused with oomycetes. However, the prominent eosinophilic inflammation common in oomycete infection is not typical of hyalohyphomycosis. 34
Aspergillosis
Aspergillus spp, although nonpigmented, have been typically classified separately from agents of hyalohyphomycosis. Their morphologic features of narrow, parallel-walled, regularly septate hyphae with acute-angle branching combined with their common association with rhinitis or systemic disease in dogs or cats are considered by many to be sufficient to achieve a diagnosis of aspergillosis. However, there has been a recent debate as to whether aspergillosis can be definitively distinguished from hyalohyphomycosis histologically, as recent studies have described cases where hyphae morphologically consistent with Aspergillus spp on histology were determined to be other genera based on culture or PCR.16,17 In addition, the recently named Aspergillus caninus is morphologically different from Aspergillus terreus and Aspergillus deflectus, presenting as 5- to 30-μm diameter yeast-like structures without visible hyphae. 101 Aspergillus vitricola has also been reported as a cause of a cutaneous nodule in a cat in which the fungi appeared as round, yeast-like cells. 7 Therefore, it may be more prudent to consider Aspergillus spp among the agents of hyalohyphomycosis and confirm histologic suspicions with culture or PCR, as certain hyalohyphomycotic fungi are known to be resistant to common antifungal medications, such as itraconazole, that are mainstays of treatment for aspergillosis. 15
Reported cutaneous infections in small animals are typically, though not always, an extension of sinonasal or systemic disease.7,46 Skin lesions in aborted bovine fetuses, typically caused by Aspergillus fumigatus, are also well described and are characterized by localized or widespread, gray to yellow plaques representing a hyperkeratotic dermatitis with superficial fungal hyphae. 89 Ulcerated nodules on the scrotum and medial thighs or face in association with rhinitis have been reported in goats, as has generalized papular dermatitis in pigs.18,89 However, it is important to note that Aspergillus is found widely in the environment and can be part of the normal microbiome, and demonstration of the fungus in tissue is required to associate it with cutaneous disease (Fig. 3e). Morphologically, Aspergillus spp typically appear with 45° branching (Fig. 3f), while hyalohyphomycotic agents such as Fusarium or Paecilomyces more commonly exhibit variations in widths and both 45° and 90° angle branching (Table 1). 49 Mucorales fungi also tend to exhibit irregular walls and 90° angle branching but are typically wider and sparsely septate (Fig. 3g; Table 1). However, as stated above, Aspergillus spp may resemble other nonpigmented fungi histologically and vice versa, and diagnosis should always be confirmed by culture or PCR, whenever possible.
Mucormycosis
Another category of nonpigmented fungal disease typically categorized separately from hyalohyphomycosis is mucormycosis, caused by fungi of the order Mucorales. 94 These ubiquitous saprophytes are part of the normal microflora and are more frequently isolated in subtropical to tropical climates, including in the United States around the Gulf of Mexico.34,90 Genera reported to cause disease in animals include Rhizopus, Rhizomucor, Absidia, Saksenaea, and Cokeromyces. Cutaneous infection in dogs and cats is very rare with a report of Saksenaea vasiformis causing an ulcerated mass on the front limb in a dog and Mucor spp causing a focal area of nodular dermatitis over the muzzle in a cat.83,99 There are also rare reports in horses due to Absidia corymbifera with single or multiple, ulcerative and granulomatous lesions on the limbs and face. 37 As with Aspergillus spp, Mucorales fungi have been reported to cause ulcerative and hyperkeratotic dermatitis in aborted bovine fetuses (Fig. 3h). 48 Infections appear to be more common in cattle, although disease frequently involves forestomach and abomasal ulceration or placentitis, rather than the skin. 89
Inflammation associated with Mucorales fungi is typically pyogranulomatous to necroulcerative, frequently with fungal vascular invasion and thrombosis. 89 Mucorales hyphae are typically broad, measuring 8–20 μm in diameter, with nonparallel walls, irregular branching, and sparse septations (Fig. 3i; Table 1). 16 While this appearance should be suggestive to the pathologist, it is not definitive and may resemble other hyalohyphomycotic fungi or oomycetes.
Entomophthoromycosis
A final category of infection caused by nonpigmented fungal hyphae (hyalohyphomycosis) is entomophthoromycosis. The primary causes of this condition are Conidiobolus spp and Basidiobolus spp, which are currently classified in the phylum Zoopagomycota which comprises part of the former Zygomycota phylum. 94 As with eumycotic mycetoma and mucormycosis, infections are more common in subtropical to tropical climates, and the portal of entry for these saprophytic fungi is wound contamination or extension from gastrointestinal or respiratory disease.30,102 Conidiobolus typically forms nodules on the external nares and within the nasal passages in horses, dogs, and rarely cats, resulting in nasal discharge, facial deformity, and exophthalmos with possible extension into adjacent skin or the brain.16,38,90,102 Conidiobolus coronatus is one of the most common infectious causes of rhinitis and sinusitis in horses.67,102 Basidiobolus ranarum most commonly causes solitary, large (up to 50 cm), ulcerative skin lesions on the chest, trunk, head, or neck in horses. 90 Basidiobolus has also been associated with ulcerative skin lesions in dogs resembling those of pythiosis in addition to gastrointestinal and disseminated disease. 30
Inflammation is pyogranulomatous to granulomatous and frequently ulcerative in the case of Basidiobolus. Numerous eosinophils may also be observed in addition to central areas of necrosis.38,102 The inflammation is centered on basophilic to poorly stained fungal hyphae embedded in eosinophilic, amorphous material forming eosinophilic sleeves around the hyphae.10,32 Hyphae are broad, with B. ranarum being usually slightly larger at 6–20 μm in diameter and C. coronatus being 5–13 μm in diameter (Fig. 4a, b; Table 1).29,38,102 Both are sparsely septate with rare irregular branching, nonparallel walls, and terminal bulbous dilations (Table 1). 29 The morphology of the hyphae and the presence of an eosinophilic sleeve around these structures resemble oomycete infections. Both GMS and PAS should highlight Conidiobolus and Basidiobolus spp; however, PAS typically does not stain the cell walls of oomycetes.
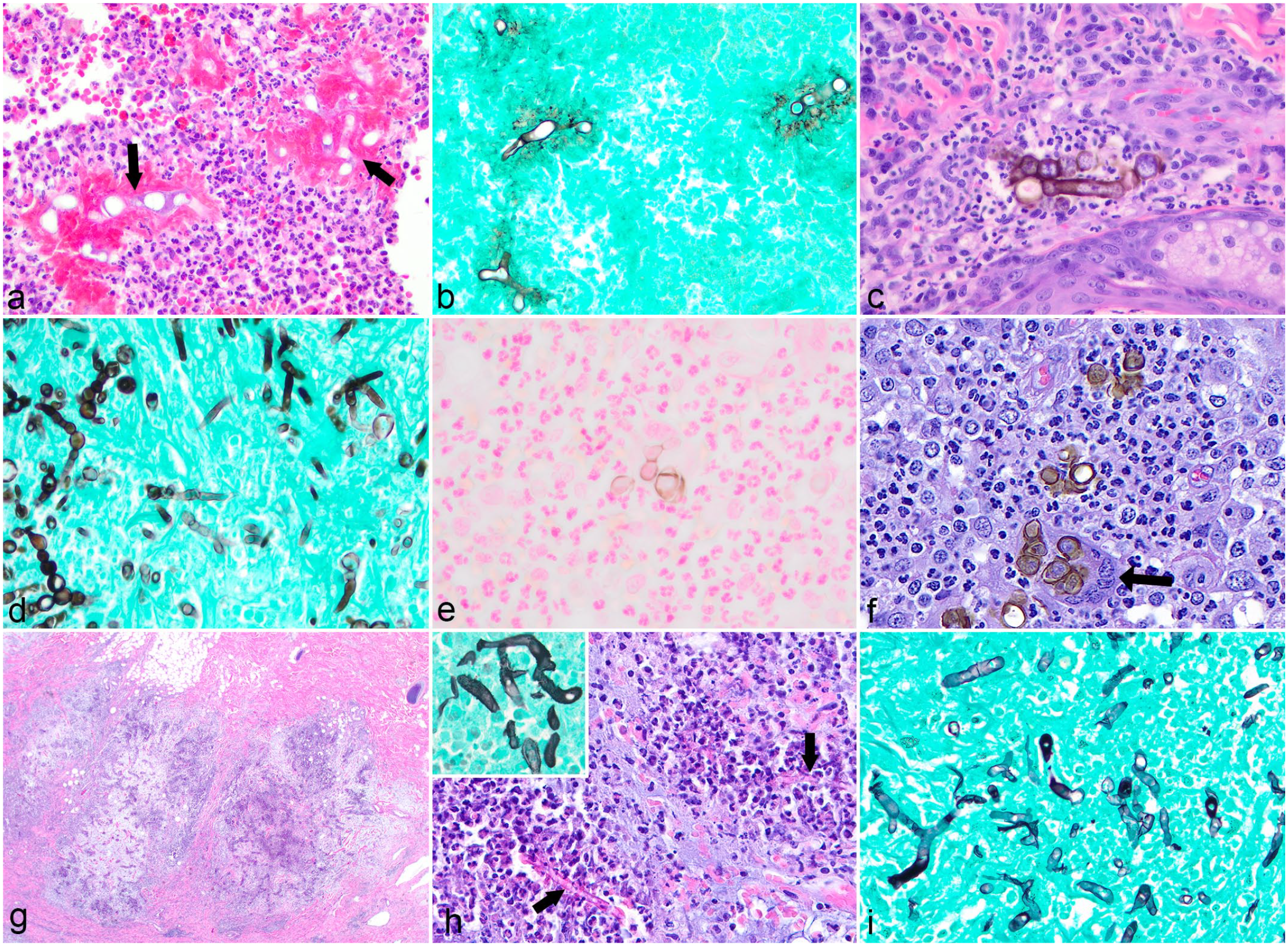
Histopathologic features of cutaneous entomophthoromycosis, phaeohyphomycosis, and oomycosis in animals. (a) Pyogranulomatous dermatitis due to Conidiobolus coronatus in a horse. Note the broad, sparsely septate hyphae with terminal bulbous dilations surrounded by a thick eosinophilic “sleeve” (arrows). Hematoxylin and eosin (HE). (b) A Grocott’s methenamine silver (GMS) stain highlights the pauciseptate nature of the hyphae typical of the Entomophthorales fungi. (c) Phaeohyphomycosis due to Curvularia geniculata in a dog. Septated, occasionally pigmented, fungal hyphae are embedded within pyogranulomatous inflammation. HE. (d) A GMS stain in the dog from (c) demonstrates the beaded, or toruloid, appearance of the hyphae. (e) Phaeohyphomycosis due to Paraconiothyrium spp in a dog. Fontana-Masson stain can highlight melanin in hyphae when the pigment is not visible with HE. (f) Chromoblastomycosis due to Tetarosphaeriaceae spp in a horse. Note the round, pigmented sclerotic bodies surrounded by inflammatory cells. Several yeasts are being engulfed by a multinucleated giant cell (arrow). HE. (g) Cutaneous oomycosis due to Lagenidium spp in a dog. Note the well-demarcated foci of eosinophilic and pyogranulomatous inflammation within the deep dermis and subcutis. HE. (h) Pythium insidiosum in a dog. Negatively staining hyphae are surrounded by a thin layer of eosinophilic material and degranulated eosinophils (arrows). An inset shows the hyphae with nonparallel walls highlighted in GMS (i) A GMS stain from the dog in (g) demonstrates the irregular branching typical of oomycetes. Note the similarity in appearance to Conidiobolus coronatus in (b).
Entomophthorales respond better to medical therapy in comparison with oomycetes because traditional antifungal agents target ergosterol, an important component of the fungal cell wall. 31 Thus, it is important to confirm the cause of these infections and differentiate it from oomycosis, as it has implications in response to treatment and prognosis.
Phaeohyphomycosis and Chromoblastomycosis
Cutaneous/subcutaneous infection caused by pigmented fungal hyphae that does not result in the formation of tissue grains is termed phaeohyphomycosis. Over 60 genera of fungi have been described as causative agents in humans and animals, with the most commonly reported including Alternaria, Bipolaris, Cladophialophora, Curvularia, Exophiala, and Fonsecaea.2,20,22,50 Lesions result from wound contamination and the commonly affected sites are the nose and digits, areas more frequently in contact with soil.15,90 Both immunocompetent and immunosuppressed animals may be affected, although dissemination to multiple distant skin sites, lymph nodes, or other internal organs is more commonly associated with immunosuppression.15,30
As with other opportunistic fungal infections, inflammation is pyogranulomatous to granulomatous but without significant numbers of eosinophils.2,15 In horses, cutaneous nodules caused by Alternaria alternata have been reported to frequently exhibit prominent lymphoplasmacytic inflammation with follicle formation as the predominant pattern. 96 Hyphae are 2–10 μm in diameter with or without branching and are frequently accompanied by round, yeast-like forms measuring 4–15 μm, and rarely up to 30–50 μm, in diameter (Fig. 4c; Table 1).9,63,90 Hyphae commonly have a toruloid, or beaded, appearance with constrictions at the septae (Fig. 4d). 9 Fungal forms are seen both extracellularly and within macrophages and multinucleated giant cells. Hyphae may be dense and pigmented enough to result in clinically pigmented lesions resembling melanoma. 15 Importantly, however, fungi may be poorly or not visibly pigmented with routine hematoxylin and eosin staining but may appear so in culture. 63 Fontana-Masson stain can highlight melanin within fungal cell walls in tissue sections when pigmentation is not histologically evident, aiding in the diagnosis of phaeohyphomycosis (Fig. 4e). 98
Chromoblastomycosis is a subset of pigmented fungal infections defined by the presence of round to ellipsoid-shaped, 10- to 20-μm diameter, pigmented forms with a thick cell wall and prominent transverse and longitudinal septation called sclerotic bodies or Medlar bodies (Fig. 4f; Table 1). 80 Small numbers of hyphae may also be observed, although the sclerotic bodies comprise the majority of fungal forms present. Various species have been reported in association with chromoblastomycosis in humans, including several species that will also cause phaeohyphomycosis, such as C. bantiana, Exophiala spinifera, A. alternata, C.lunata, and Fonsecaea pedrosoi. The yeast-like cells seen in phaeohyphomycosis are distinguished from sclerotic bodies by the fact that they have thinner walls, show septation in a single plane, and are typically arranged in chains. 80 The factors that determine whether a particular fungus will cause phaeohyphomycosis versus chromoblastomycosis remain undefined. Most reports in veterinary medicine are from amphibians or horses.1,41,87,96 As with phaeohyphomycosis, the associated inflammation is pyogranulomatous.
Oomycosis
Although not classified as fungi, oomycetes share many clinicopathological features with fungi within the order Entomophthorales (Conidiobolus spp and Basidiobolus spp), which makes differentiation between these organisms difficult and behooves their discussion here. 31 Pythiosis and lagenidiosis, collectively known as oomycosis, are emerging diseases of temperate, tropical, and subtropical climates caused by the globally distributed oomycetes Pythium and Lagenidium, respectively.31,76 These fungal-like eukaryotic microorganisms belong to the Kingdom Stramenopila and are phylogenetically more closely related to algae than fungi. They possess cellulose and β-glucan rather than chitin or ergosterol as the primary component of the cell wall or membrane, respectively. 31 Pythium and Lagenidium spp are found in warm, aquatic environments and are reported globally, most often in southeast Asia, Korea, Japan, eastern coastal Australia, New Zealand, and South America with the highest incidence in Brazil, Costa Rica, and the Caribbean (Haiti). In the United States, it is more prevalent in Gulf Coast states and in the southeastern, midwestern, and western regions of the country.31,74
P. insidiosum can affect the skin, gastrointestinal tract, eyes, and, in some cases, nasal cavity of animals. 31 Pythiosis is mostly reported in dogs and horses, especially in the Americas. 31 However, sporadic cases occur in cattle, cats, and sheep.19,77,82 Animals that became infected often have a history of recurrent exposure to warm freshwater habitats, although a significant percentage of animals have not had known exposure to these environments. Affected animals often are immunocompetent and healthy. 31 P. insidiosum zoospores have a special tropism toward animal hair, wounds, damaged skin, intestinal mucosa, and plant leaves. Zoospores encyst on the nonintact tissues and secrete amorphous glycoprotein, facilitating adhesion to the surface. 31 The geographic distribution of lagenidiosis, as well as the mode of transmission and predisposing features for disease, are very similar to pythiosis. 32 Infections caused by Lagenidium spp have been diagnosed mainly in dogs with a few cases reported in cats.32,81 In dogs, Lagenidium spp are responsible for progressive cutaneous, subcutaneous, and disseminated disease, resembling those caused by P. insidiosum. 32
Dogs with pythiosis may develop cutaneous and subcutaneous forms of the disease or gastrointestinal disease, and only rarely are these 2 manifestations observed in the same patient.21,34 Cutaneous lesions are characterized by severe nodular to diffuse dermatitis and panniculitis, consisting of nonhealing draining wounds and invasive masses with ulcerated surfaces. When draining lymph nodes are swollen, it usually indicates extension of infection.21,31 Pythiosis in cats is uncommon, likely due to their water aversion, but, when present, skin lesions resemble those observed in dogs. 93 With lagenidiosis, the frequent ulceration and regions of necrosis in cutaneous lesions can lead to a misdiagnosis of bacterial infection and erroneous treatment with antibiotics. 62 As with pythiosis, cutaneous lesions are progressive, locally invasive, and often fail to respond to therapy. 31 However, in contrast to pythiosis, gastrointestinal involvement is uncommon with regional lymphadenopathy being a more typical lesion.31,62 Lagenidium giganteum forma caninum is responsible for severe, progressive cutaneous disease, lymphadenopathy, pulmonary nodules, and vessel invasion. 31
Pythiosis, in contrast to lagenidiosis, is reported relatively frequently in horses, which typically develop skin lesions on the limbs, scrotum, or abdomen characterized by ulcerative nodules or tumor-like masses, often with necrotic areas and serosanguineous exudates, irregular margins, fistulous tracts, and irregular cavitations at the cut surface, known as “kunkers” (yellow-gray, hard concretions composed of necrotic debris with myriads of filamentous hyaline hyphae).86,90 In sheep, P. insidiosum can cause rhinitis, dermatitis, and gastroenteritis. Cutaneous lesions are characterized by dry or wet, variably sized (2–30 cm diameter) ulcers in haired skin. The subcutaneous tissue is yellow or dark-brown and surrounded by a thin, white, fibrous capsule. Interestingly, “kunkers” are not observed in sheep with P. insidiosum. 19 According to Carmo et al, 19 skin lesions may expand to adjacent tissues, including ligaments, tendons, nerves, and bones. As opposed to dogs and cats, livestock are much more likely to respond to treatment. 89
Another agent that shares antigenic and morphologic similarities with. giganteum f. caninum and other Lagenidium species was previously classified as Lagenidium karlingii; however, with after phylogenetic analysis, it has now been reclassified in a new genus as Paralagenidium karlingii. 33 P. karlingii has been associated with slowly progressive disease limited to cutaneous and subcutaneous tissues in dogs. It is associated with a more favorable prognosis and a better treatment response. 33
Histologically, skin lesions of both pythiosis and lagenidiosis are characterized by eosinophilic, granulomatous to pyogranulomatous inflammation with fibrosis, multifocal necrosis, and sometimes vasculitis, similar to the lesions seen with fungi of the Entomophthorales (Fig. 4g).31,34,62 P. insidiosum hyphae are broad (mean diameter, 4 μm; range, 2–7 μm) and rarely septate with nonparallel walls and irregular branching, and are found in necrotic centers of granulomas (Fig. 4h; Table 1). 31 These oomycetes are identified as clear spaces surrounded by an acellular eosinophilic material and are best visualized with a GMS stain (Fig. 4i).21,31 Because the organisms’ cell walls do not have chitin, the hyphae are poorly detected with PAS.31,51 Lagenidium hyphae are described to be bigger than P. insidiosum (2–7 μm), Conidiobolus spp (5–13 μm), and Basidiobolus spp (5–20 μm), measuring 8–25 μm in diameter (Table 1). They are negatively staining, ribbon-type, infrequently septate and have irregular nonparallel walls.31,32 The eosinophilic sleeve surrounding the hyphae appears to be thinner in Lagenidium spp compared with those observed in Conidiobolus spp and Basidiobolus spp which are often as wide as 5–10 μm and up to 25 μm, respectively (Fig. 4a, b). 32 It is the authors’ opinion that the size of these organisms varies in tissues, and size and morphology cannot be used to definitively differentiate between these genera, necessitating the use of culture or PCR.
Dimorphic Fungi
Unlike the opportunistic saprophytic fungi discussed in the previous sections, dimorphic fungi are characterized by their growth in culture as hyphae, but in tissue as yeasts or spherules. Two other differentiating factors are their classification as primary pathogens and the relative uniqueness of their morphologies, frequently allowing for a definitive diagnosis (at least to the genus level) on histopathology. The dimorphic fungi of known pathogenic significance to animals include Blastomyces spp, Histoplasma spp, Coccidioides spp, Cryptococcus spp, and Sporothrix spp.
All the pathogenic dimorphic fungi exist within an ecological niche from which they can be transmitted to animals or humans. Histoplasma spp typically reside in organic matter enriched with bird or bat feces. Within this genus, Histoplasma capsulatum is the primary cause of disease in animals and is distributed worldwide. Histoplasma duboisii is endemic to western and central Africa, where it causes lymphatic-associated disease with cutaneous and bone dissemination in humans and nonhuman primates, while Histoplasma farciminosum primarily infects equids and produces cutaneous to subcutaneous lymphatic lesions. 25 Blastomyces is typically isolated from moist, acidic, enriched soil near wooded regions with decaying vegetation. The predominant pathogen in this genus is Blastomyces dermatiditis, which has a range focused on the Ohio and Mississippi River valleys, the Great Lakes, and the Saint Lawrence River. 25 Coccidioides immitis and Coccidioides posadasii are soil-dwelling fungi endemic to the southwestern United States. Cryptococcus spp are globally distributed saprophytes found mainly in nitrogen-rich alkaline debris, most commonly soil contaminated with pigeon feces. 34 Of the numerous species, Cryptococcus neoformans (var. neoformans and var. grubii) and Cryptococcus gattii have been most frequently reported as causing illness in veterinary species. 11 Finally, Sporothrix schenckii and Sporothrix brasiliensis are typically associated with soil and decaying plant matter in tropical and subtropical regions with hyperendemicity reported in some areas (primarily Central and South America). 25
Cutaneous involvement of dimorphic fungal infections is uncommon, with systemic mycosis being most frequently diagnosed in dogs, cats, and occasionally large animals such as horses. 34 Most of these fungi invade immunocompetent hosts through inhalation of conidia (Histoplasma), spores (Blastomyces), blastospores (Cryptococcus), or arthroconidia (Coccidioides) which transform into pathogenic yeasts or spherules (Coccidioides) within the host nasal cavity or lungs. 54 Following phagocytosis by macrophages, hematogenous dissemination causes systemic disease with possible cutaneous involvement.34,54 Rarely, these fungi can be introduced into the skin through direct wound inoculation, a route more commonly taken by Coccidioides than by the other genera.25,40 With cryptococcosis in particular, cutaneous lesions are commonly located in the facial/nasal region of cats, and less commonly in dogs or small ruminants, and represent local extension from nasal infections.11,34 Sporothrix is an exception: cutaneous lesions are typically caused by puncture or wound contamination, rather than by inhalation. 12 Systemic dissemination can then occur, most commonly with S. brasiliensis. 85
Dermal and subcutaneous papules to nodules, which may ulcerate and occasionally produce fistulous draining tracts, are the typical cutaneous manifestations of dimorphic fungal infections. 34 Considering the tropism of Coccidioides spp for bone, these nodules are typically distributed over sites of bone infection. 28 Again, lesions of cryptococcosis typically involve the head, neck, ears, and bridge of the nose, with multiple firm subcutaneous nodules along the muzzle being the most common clinical presentation. 34 A similar distribution on the head and neck is observed with sporotrichosis in cats.25,34
Histologically, the dermis and subcutis contain nodular to confluent infiltrates of numerous epithelioid macrophages and variable numbers of neutrophils and multinucleated giant cells, frequently associated with discrete granuloma or pyogranuloma formation. 34 Yeasts or spherules are typically numerous in lesions of histoplasmosis, sporotrichosis, and cryptococcosis, while they are sparser in blastomycosis and coccidioidomycosis.25,34 In histoplasmosis, round, 2- to 4-μm diameter yeasts with a clear halo and a thin cell wall are observed in great numbers within the cytoplasm of macrophages (Fig. 5a, b). 40 Sporothrix appears fairly similar, with numerous elongated, 3- to 10-μm long and 2- to 4-μm diameter, “cigar-shaped” yeasts commonly seen within macrophages (Fig. 5c, d; Table 2).34,40 Yeasts are typically more abundant in feline than in canine lesions. Blastomyces spp are observed in smaller numbers, free or within macrophages (Fig. 5e). 34 Yeasts are spherical, approximately 5–20 μm in diameter with a double wall, and exhibit broad-based budding (Fig. 5e inset; Table 2). 40 Coccidioides spherules frequently exhibit endosporulation in which numerous basophilic endospores may be seen within the 20- to 80-μm, double contour walled spherules (Fig. 5f; Table 2).34,40 Spherules will be variably sized as they enlarge during the endosporulation process, eventually rupturing to release endospores into surrounding tissue. 28 As a note of caution, the 2- to 5-μm diameter endospores, once released, may be confused with yeasts or other organisms, and the presence of spherules is required for the confirmation of Coccidioides spp infection. 35 Cutaneous nodules of cryptococcosis typically contain numerous yeasts that frequently outnumber the inflammatory cells.11,34 Yeasts are refractile, spherical to oval, 3.5–20 μm diameter, and are surrounded by a wide clear capsule that forms a halo and imparts a “soap bubble” appearance at low magnification (Fig. 5g, h; Table 2).11,34 Occasional narrow-based budding may be observed. Mayer mucicarmine or Alcian blue will highlight the capsule. 11 An atypical form of cryptococcosis is also described in cats. In these atypical cases, the inflammatory infiltrates are composed of epithelioid macrophages, neutrophils, and eosinophils with rare acapsular yeasts within tissues, which may be easily missed and require special stains to be visualized (Fig. 5i). 73
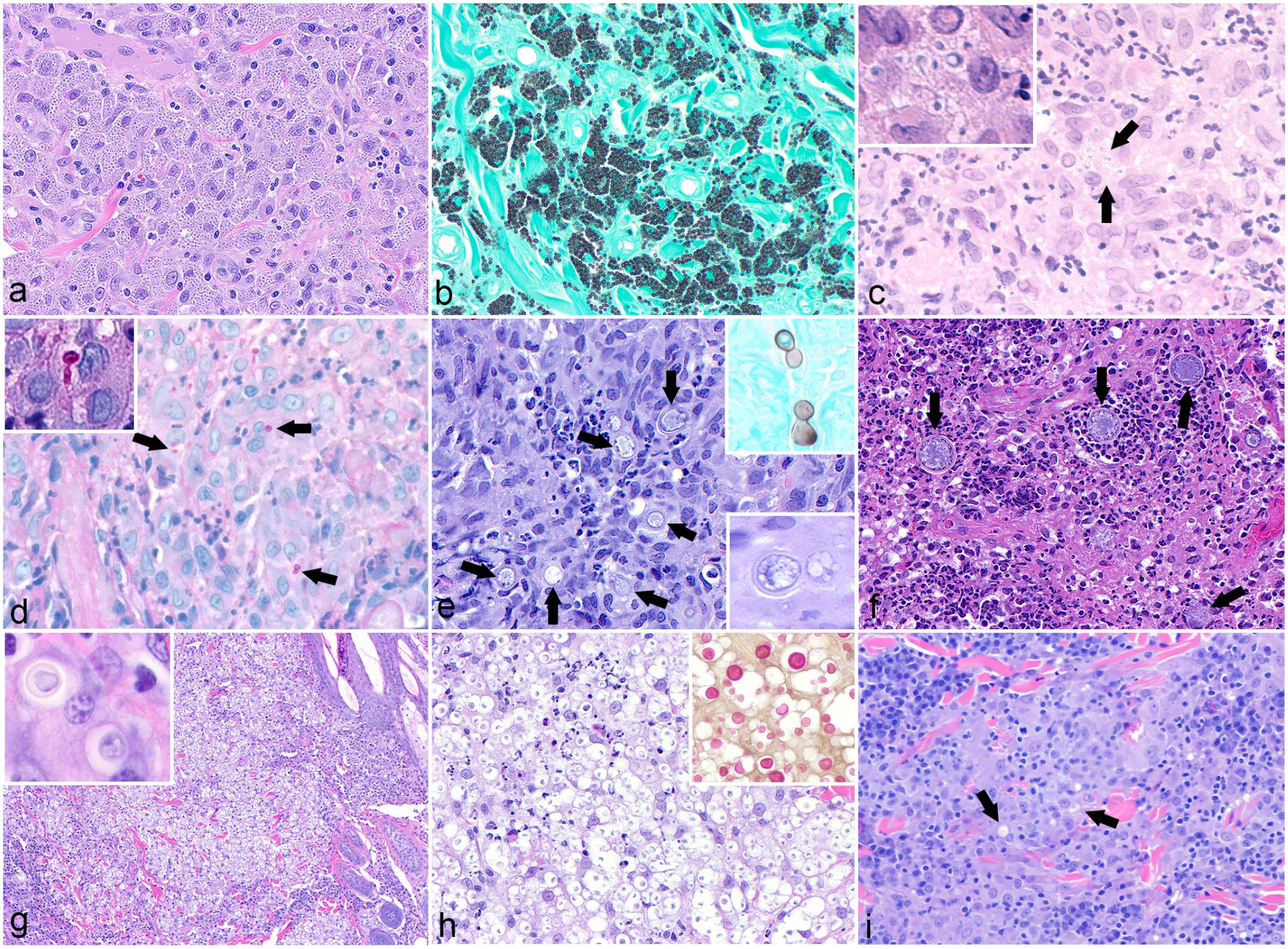
Histopathologic features of cutaneous infection with dimorphic fungi in domestic animals. (a) Dermatitis associated with Histoplasma capsulatum in a cat. The dermis is diffusely expanded by macrophages laden with numerous 2- to 4-μm diameter yeast. Hematoxylin and eosin (HE). (b) A Grocott’s methenamine silver (GMS) stain highlights the myriad yeasts within macrophages in the cat from (a). (c) Sporothrix schenkii–associated dermatitis in a cat. Small numbers of yeasts are observed within macrophages (arrows). An inset shows the thin cell wall and clear halo surrounding the yeast. Note the similarity in appearance to Histoplasma spp. HE. (d) A periodic acid-Schiff reaction highlights S. schenkii yeast (arrows), improving visibility over the HE stain in (c). An inset shows narrow-based budding of the yeast. (e) Cutaneous blastomycosis in a dog. Note the spherical yeast with a double-contoured wall within the center of a pyogranuloma (arrows). HE. Inset, top right: a GMS stain highlights yeasts exhibiting typical broad-based budding; Inset, bottom right: a higher magnification view of a single yeast clearly demonstrates the double-contoured wall. (f) Cutaneous Coccidioides posadasii infection in a llama. Note the large fungal spherules with double-contoured walls demonstrating endosporulation (arrows). HE. (g) Cutaneous cryptococcosis in a cat. Numerous yeasts, which outnumber inflammatory cells, expand the dermis and displace adnexal structures. An inset shows the wide clear capsule surrounding the yeast. HE. (h) A higher magnification view of (g) demonstrates the clear halo around Cryptococcus spp yeast, imparting a soap bubble appearance. HE. Inset: a mucicarmine stain highlights the capsule. (i) Granulomatous dermatitis in a cat. Rarely, Cryptococcus spp yeasts are acapsular and much more difficult to appreciate on histology (arrows). HE.
Both PAS and GMS stains can be used to highlight yeast, which is particularly useful in cases where organisms are sparse. Histoplasma yeast stain with PAS or GMS stains, although PAS often stains only the nucleus and in cases with a paucity of organisms, the yeast may be missed; thus, GMS is often preferred. 34 Mucicarmine may be used to highlight the capsule of Cryptococcus spp, which may be particularly useful in distinguishing atypical cryptococcosis from blastomycosis. Importantly, animals with cutaneous lesions should always be evaluated for potential underlying systemic/visceral disease.
Ancillary Tests for Fungal Identification and Pan-Fungal PCR on Histological Specimens and Cytological Preparations
Definitive diagnosis of fungal infections should be based on culture, cytology, and histologic evaluation. Fungal culture from clinical samples is considered the gold standard for fungal identification. Although fungal cultures may take weeks to provide results depending on the isolated organisms, the combination of fungal cultures with matrix-assisted laser desorption/ionization-time of flight (MALDI-TOF) has allowed for faster identification of fungi grown in culture. 45 Despite progress in improving the speed to diagnose fungal infections, culture of fungal organisms is still problematic, as some organisms may fail to grow if samples were frozen or not kept at appropriate temperatures, and culture of dimorphic fungi may pose a safety risk to laboratory personnel. 45 In addition, the biggest issue may be related to the fact that many veterinary practitioners fail to collect samples for culture while obtaining samples for histology. Another alternative in fungal diagnostics is the use of antigen testing, although these assays tend to be limited to certain fungal pathogens. 45
To overcome some of the issues related to fungal cultures and other diagnostic tests, the use of molecular assays from formalin-fixed paraffin-embedded (FFPE) tissues and cytology slides can be beneficial. PCR assays have been developed to target specific genera and species of fungi from clinical samples. Similarly, immunohistochemistry and in situ hybridization can be combined with histology to confirm the fungal organisms within histological specimens. The problem comes when the genus and species of interest are not known, which is especially common in hyalohyphomycosis. An alternative that has become more widely used is PCR using primers targeting conserved regions within the fungal genome. 47 These primers amplify hypervariable regions, which can then be sequenced and compared with reference databases, allowing for genus and species identification and classification of fungal infections. These tools can give results in a shorter period (within approximately 10–15 days) and allow for classification of a wide range of fungal organisms, enabling improved selection of antifungal treatments. 47 Similar PCR-based and sequencing assays are also used in combination with fungal cultures. When pure cultures are obtained, these can then be amplified and sequenced, enabling the identification of any unknown fungus. 45
In a previous study using a pan-fungal PCR targeting conserved regions within the internal transcribed spacer region, we obtained high-quality fungal DNA sequences from FFPE tissues in 69.5% of cases. 60 Sequences were compared with a database of fungal sequences to determine the genus and species of fungal organisms, and sequences and histologic identification matched in 61.7% of cases. 60 The same assay can also be used on cytologic preparations, which prove to be useful in identifying fungi from stained slides. 72 The sensitivity of the pan-fungal PCR assay performed on stained cytology slides was 67% overall and 86% when considering only slides with abundant fungi. The specificity was 62%, which was attributed to amplification of fungal DNA from environmental contaminants from control slides with no visible fungus and negative culture results, or from PCR reagents. 72
The pan-fungal PCR is a very sensitive assay that will amplify even small amounts of fungal DNA; therefore, it may be easy to amplify contaminating fungal DNAs that are present in the sample, the environment, or even in PCR reagents. 14 For this reason, it is important to use this assay as an adjunct to histology or cytology when fungi are observed in the samples.
Conclusions
In this review paper, we covered the primary clinical and histologic features observed in some of the most common fungal infections found in the skin of animals. By combining the clinical history with histological features and fungal morphology, and using appropriate special stains, pathologists should be able to better provide guidance to practitioners regarding the taxonomic category of fungal infections affecting the skin of animals. In several cases, such as those caused by dimorphic fungal infections, histology alone may be able to provide genus (and even species) level identification. However, in other opportunistic fungal or oomycete infections, particularly in hyalohyphomycosis, histology alone is often unable to confirm the taxonomic classification of the fungal organisms resulting in an infection. 88 In these cases, a thorough histological description should be followed by a recommendation for clinicians to pursue additional ancillary tests to better identify the causative agents, as this could have direct implications in the treatment and prognosis of the fungal infection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
