Abstract
Case summary
Toxoplasma gondii is a globally distributed apicomplexan protozoan infecting all warm-blooded animals. Cats are the definitive host, susceptible to clinical disease. In Spain, studies have shown the widespread presence of Toxoplasma IgG antibodies in cats but there are no published data on clinical toxoplasmosis in cats from Spain. We describe a case of fatal disseminated toxoplasmosis in a 7-year-old outdoor domestic shorthair cat. The cat, an active hunter, was presented with a 1-week history of malaise and acute deterioration over 24 h. Fever, tachypnoea and abdominal discomfort were detected on physical examination. No evidence of immunosuppressive disease or previous treatment could be found. A moderate increase in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) was found on serum biochemistry. Radiography revealed hepatomegaly and a small patchy pulmonary alveolar pattern in the caudal lung lobes. In 48 h, the AST and ALT levels were severely increased and the cat’s respiratory signs worsened. Parenteral clindamycin administration (25 mg/kg IV q12h) and supportive treatment could not prevent progression to death. Cytology from the liver and lungs revealed organisms morphologically consistent with T gondii. Indirect immunofluorescence antibody test Toxoplasma titres were 1:100 IgM and 1:100 IgG. PCR performed on scraped material from glass cytology slides was positive for T gondii in both lung and liver aspirates.
Relevant and novel information
To the authors’ knowledge, this is the first case of fatal disseminated toxoplasmosis in a cat described in Spain, confirmed by PCR. Fine-needle aspirates from the liver could assist in the diagnosis of fatal disseminated toxoplasmosis when liver enzymes are increased.
Case description
A 7-year-old spayed female outdoor domestic shorthair cat was presented after 1 week of inappetence and malaise. Blood tests (Table 1) and ultrasonography performed by the local veterinarian 4 days before presentation were unremarkable. The cat was an active hunter. The day before presentation, its condition deteriorated severely. On physical examination, the cat was mentally depressed, very weak and there was moderate tachypnoea of 40 breaths/minute (bpm). The cat’s temperature was 40°C and abdominal palpation elicited diffuse abdominal pain. Body condition score was 6/9.
Blood tests performed by the local veterinarian 4 days before presentation, in-house at presentation and 48 h after presentation
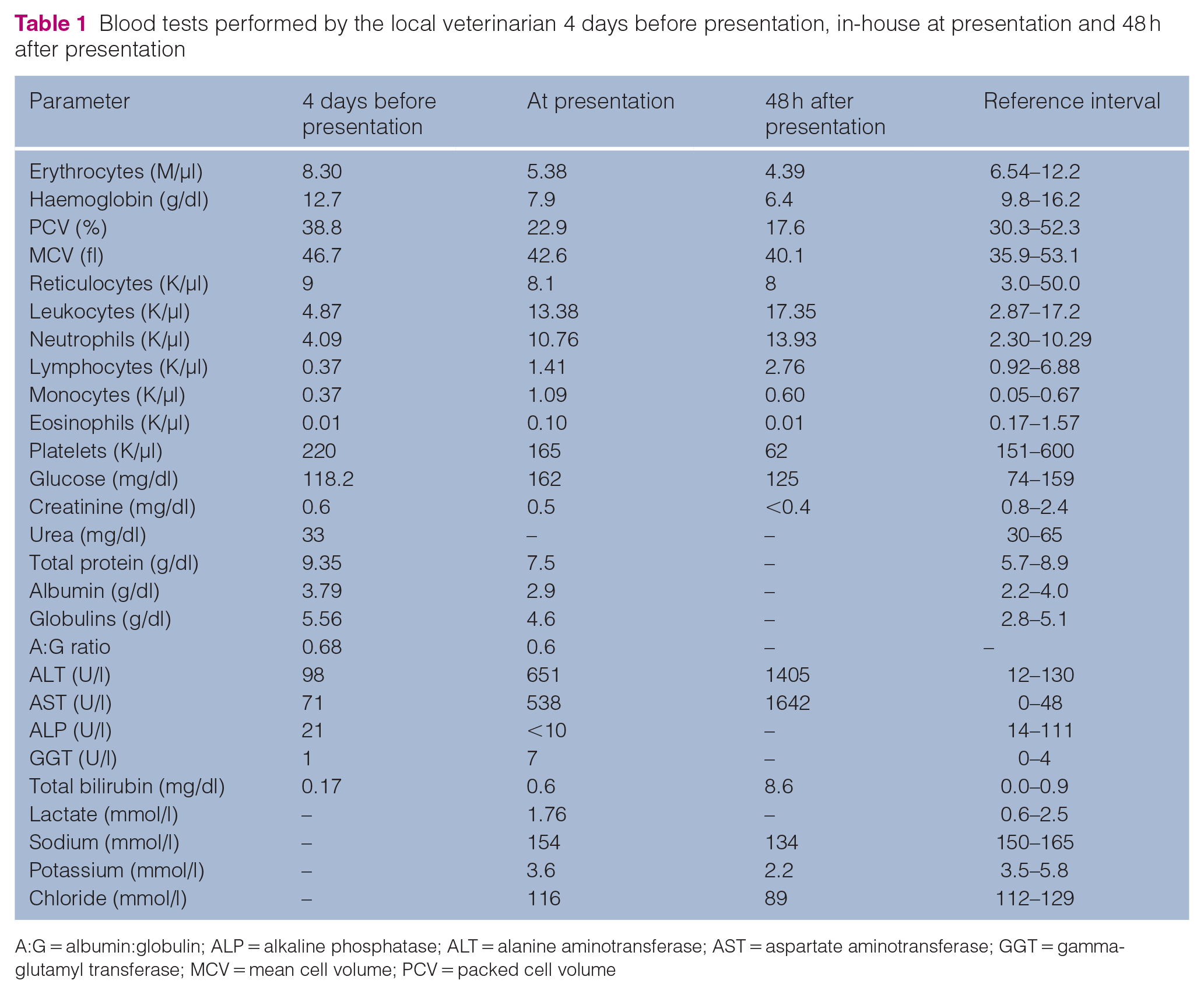
A:G = albumin:globulin; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase; MCV = mean cell volume; PCV = packed cell volume
In-house haematology and serum biochemistry tests revealed non-regenerative anaemia, leukocytosis and a moderate increase in alanine aminotransferase (ALT), aspartate aminotransferase (AST) and gamma-glutamyl transferase (Table 1). An in-house feline leukaemia virus (FeLV)/feline immunodeficiency virus (FIV) test (SNAP combo; IDEXX) was negative for both viruses. Radiography (Figure 1) revealed one small patchy pulmonary alveolar infiltrate in the caudal lung lobes and hepatomegaly. The cat was placed in an oxygen cage, fluid therapy was initiated and ampicillin 22 mg/kg IV q8h, clindamycin 16 mg/kg IV q12h, maropitant 1 mg/kg IV q24h and buprenorphine 0.02 mg/kg IV q8h were administered. After 24 h, there was clinical improvement, but the cat’s respiratory rate had worsened to 70 bpm and a gallop rhythm was detected. The patient’s temperature was 39.5°C. A second radiograph (Figure 2) revealed a ventral alveolar pattern with multiple focal pulmonary opacities. In-house B-type natriuretic peptide (BNP) test (SNAP feline proBNP; IDEXX) was performed as a functional cardiac biomarker of myocardial stress. The result was abnormal. Furosemide 1 mg/kg SC was administered and the clindamycin dosage was increased to 25 mg/kg IV q12h. The cat’s respiratory rate decreased to 40 bpm, and ultrasound-guided fine-needle aspiration (FNA) from the lung and liver under mild sedation were scheduled. Twelve hours later, the cat had acutely deteriorated. Severe icterus and an increased respiratory effort in spite of a stable respiratory rate were detected. The cat’s temperature was 38.9°C. In-house haematology and serum biochemistry tests (Table 1) revealed a three-fold increase in AST and ALT, severe hyponatraemia, hypokalaemia, hypochloraemia and moderate leukocytosis. A third radiograph (Figure 3) showed ventral lung consolidation and air bronchograms. Dexamethasone 0.1 mg/kg IV was administered. There was mild improvement in the cat’s respiration and stamina, but despite supportive therapy, it died 5 h later. Permission for a post-mortem examination was declined. However, the caregivers gave their consent to obtain post-mortem FNA from the lungs and liver. Cytology glass slides were stained with haematoxylin and eosin (H&E) and the presence of organisms morphologically consistent with Toxoplasma gondii was detected. Post-mortem review by an external pathologist described the presence of an ovated spherical structure 100 µm in diameter compatible with a T gondii tissue cyst and isolated crescent-shaped structures 4–6 µm in diameter compatible with T gondii tachyzoites-bradyzoites in the liver (Figures 4–6) and lungs (Figures 7 and 8). The slides of cytological samples stained with H&E were scraped with a microtomy blade. The scraped material was collected in a 1.5 ml conical plastic tube. DNA was extracted using the DNA mini-Blood Kit (QIAGEN Gmbh) following the protocol for tissues provided by the manufacturer. PCR for T gondii was performed in two successive amplifications in a single tube using 18S-5.8S rRNA ITS1 targeted primers. A region of 227 bp of the ITS1 of T gondii was amplified. The PCR products were analysed by electrophoresis through 1.5% (w/v) agarose gels in Tris-borate-EDTA buffer with GelRed Nucleic Acid Stain and the DNA fragments stained were visualised under ultraviolet light on a transilluminator. The PCR result was positive for T gondii in both liver and lung samples.

Radiograph taken at presentation. There is an ill-defined increase in opacity in the caudal lung lobes and oesophagus (white arrow), flattening of the diaphragm, cardiac silhouette spacing from the diaphragm and hepatic parenchyma size increase, with rounded borders

Radiograph taken 24 h (without approximately) after presentation. There is increased lung opacity in multiple ventral areas, small and ill-defined, an increase in the cardiac silhouette compared with presentation and an increase in abdominal distension

Radiograph taken 48 h after presentation, showing the presence of multiple areas of increased pulmonary opacity, with air bronchograms and loss of pulmonary vessel detail, affecting the ventral half of the lung parenchyma

Fine-needle aspiration of the liver, × 100. The hepatocellular population shows discrete to moderate degenerative changes. Some hepatocytes evidence vacuolated and foamy cytoplasm, with small accumulations of bile pigment granules (red arrow). There is a mixed cellular inflammatory component consisting of different maturation stages of lymphocytes and a small number of neutrophils (white arrowheads)

Fine-needle aspiration of the liver, × 100. Structures 4–6 µm in size, crescent-shaped, in a group of seven, compatible with Toxoplasma gondii (yellow arrow)

Fine-needle aspiration of the liver, ×100. Spherical ovated structure, 100 µm in size, filled with crescent-shaped structures, compatible with a Toxoplasma gondii tissue cyst (yellow arrow)

Fine-needle aspiration of the lung, × 100. Epithelial respiratory cells showing typical apical cilia (white arrow) can be observed. One crescent-shaped organism (yellow arrow) compatible with Toxoplasma gondii is present outside the cells

Fine-needle aspiration of the lung, × 100. Three to four crescent-shaped organisms (yellow arrow) compatible with Toxoplasma gondii can be found in the cytoplasm of the respiratory cells
The results of blood tests performed by an external laboratory arrived after the death of the cat (Table 2).
Blood tests performed at an external laboratory at presentation; the results arrived post mortem

A:G = albumin:globulin; BNP = B-type natriuretic peptide; IFAT = immunofluorescence antibody test
Discussion
T gondii is an apicomplexan coccidian protozoan with a global distribution. It can infect any nucleated cell and affect all warm-blooded animals. 1 Cats and other felids are the definitive hosts of its complex life cycle and release oocysts, the environmentally resistant form of the parasite, with all other warm-blooded animals acting as intermediate hosts. 2 Oocysts, which can be found in all environments, are ingested by intermediate hosts in which an asexual cycle is completed. Tachyzoites disseminate throughout the body and produce tissue cysts in which, probably for the life of the host, they persist as bradyzoites, mostly in neural and muscular tissues, but also in visceral organs, including the lungs, liver and kidneys. 3 The main source of infection for cats is ingestion of intermediate hosts or their tissues, either by hunting small prey or eating raw meat fed by humans.4,5 Other sources of infection are congenital6,7 and, probably, galactophorous.7,8 The parasite can either first undergo an asexual cycle, remaining in the cat as tissue cysts, or complete an enteroepithelial sexual cycle, producing oocysts that will again contaminate the environment. The relationship between T gondii and felids and their prey is ancient, meaning that evolutionary mechanisms have adapted both in a highly specialised relationship. 9 This could play a role in the rare presentation of clinical disease in cats: in a large institution located in Boston (USA), 100 cases of confirmed clinical toxoplasmosis were described over 40 years. 10 In Finland, 3% of a long series of cats positive for toxoplasma showed signs of clinical disease over 3.5 years. 11 The same rate was described in Denmark over a 2-year period. 12
Feline toxoplasmosis was first reported in 1942. 13 In 1954, the first clinically diagnosed case was published. 14 Different presentations have been described since in many countries,15 –45 as well as series of cases.6,8,46 –50 In Spain, studies have detected the presence of T gondii antibodies in 24.2–84.7 % of domestic cats, free-roaming cats and wild felids in different regions.51 –58 However, no cases of fatal disseminated toxoplasmosis in cats have been described in Spain.
Acute systemic toxoplasmosis is the result of overwhelming intracellular replication of tachyzoites. It mostly affects pulmonary, hepatic, central nervous system (CNS) and pancreatic tissues,25,28,59 arising from primary infection or through reactivation of tissue cysts present in the cat. Our case was an outdoor hunting cat, showing vague illness with acute deterioration 24 h before presentation. In reported cases, an immunosuppressive predisposing cause can be found, such as a renal transplant, oclacitinib, cyclosporine or prednisolone treatment, concurrent FIV/FeLV infection or feline infectious peritonitis infection.15,21,29,30,38,40,42,48,49 However, in other cases,18,22,27,44 including ours, a predisposing cause cannot be determined. It has been suggested that more pathogenic strains may be involved.15,23,25,35,59,60 As in other cases described, findings on physical examination included fever, tachypnoea, depression and diffuse abdominal discomfort on palpation. The detection of an increase in ALT, AST, leucocytosis and radiographic pulmonary patchy alveolar infiltrates, indicating hepatic and respiratory disease, has also been described in cases of fatal disseminated toxoplasmosis.10,18,21,22,27,40,44,46,48 Eliciting a pain response on abdominal palpation has been attributable to hepatitis, pancreatitis or discomfort from interference with respiration in those cats with advanced pneumonia. 10 In our case, acute hepatic disease and early pneumonia would be the most likely causes, since hepatomegaly and some pulmonary alveolar infiltrates could be detected on radiographs at presentation. Pneumonia is a common finding in fatal disseminated toxoplasmosis,22,28,46,61 being present in 97.7% of cats in a series of cases. 10 The most striking biochemical change was abnormal AST and ALT, which presented a massive increase over 48 h. We could not assess the presence of pancreatitis and there were no CNS clinical signs. Severe hypokalaemia, hypochloraemia, hyponatraemia were detected 48 h after presentation. That could reflect acute hepatic failure and severe respiratory disease.18,25,26,40,44,62 Acid-base disorders contributing to the electrolyte imbalances detected could not be determined. Furosemide administration 27 is an unlikely cause, since only a single low dose was administered. There was an acute fatal evolution despite treatment, similar to other cases described.10,18,21,27,38,40,44,50
A diagnosis of fatal disseminated toxoplasmosis is based on the presence of compatible clinical signs and demonstration of the organism. 59 Nevertheless, it is rarely confirmed ante mortem. In our case, FNA from the lungs and liver were performed post mortem, enabling the detection of organisms consistent with T gondii. Although cytology has been described for the diagnosis of toxoplasmosis using bronchoalveolar lavage (BAL) and other body fluids and tissues,10,16,19,22,23,25,27,29,31,32,34 –38,40,42,43,48,50 its application in detecting T gondii in liver parenchyma FNA has not previously been reported. Acute Toxoplasma hepatitis has been described, but no liver cytology was performed. 22 In one study, cytological observation of tachyzoites in BAL fluid proved more sensitive than histological examination of tissue samples. 63 It is important to distinguish T gondii from Neospora caninum25,27 and to note that our area is endemic for Leishmania infantum. 64 PCR is a useful technique for confirming T gondii infection59,65 and the use of fixed stained smears and scrape lysates as material for PCR has been described in the diagnosis of mycobacteria and pulmonary toxoplasmosis in cats.37,38,66,67 In our case, PCR performed on samples of lung and liver scraped from cytology slides allowed confirmation of fatal disseminated T gondii infection.
One limitation of our case is that molecular characterisation was not possible. In Spain, one study in cats revealed type I and type II strains of T gondii. 68
Serology has been used in the diagnosis of T gondii with varying results. An IgM titre of 1:64 or higher is indicative of active infection, as seen in our case (Table 2). The interpretation of IgG is more complex, as single positive titres indicate only exposure rather than infection. For a tentative diagnosis, multiple requirements are needed.4,10,17,27,41,47,59 Some confirmed cases may present negative titres.36,39,48 In acute cases, serology is of little use ante mortem as results often arrive after the cat has died, as occurred in our case.
Parenteral clindamycin treatment4,20,47 was initiated in this case; however, there was no clinical response. This lack of response may be due to overwhelming infection, parasite drug resistance or an antibiotic-associated decrease in the antitoxoplasmic activity of phagocytic cells, which are responsible for the control of T gondii. 69 Parenteral trimethoprim-sulfonamide was unavailable; other similar cases treated with combination protocols have also not survived.22,40,46,48,49 Some chronic cases treated with clindamycin, even at doses lower than described, have survived.18,33,22 Treatment with oral pyrimetamine or azithromycin was not an option in this case.
Dexamethasone was administered at a low anti-inflammatory dose to control parasite-related inflammation. 23 However, it is unlikely to have contributed to the poor outcome, as the cat’s condition had already deteriorated before administration. The use of prednisolone in the treatment of toxoplasmosis has been described without evidence of worsening disease or a predisposition to recrudescence.16,24
A second limitation of our case is that no post-mortem examination was allowed, which hindered the description of all affected organs. However, it is possible that myocardial disease was present. Upon admission, feline cardiac proBNP was within the normal range, but 24 h later, the in-house test returned abnormal results and a gallop rhythm was detected. In addition, ultrasensitive cardiac troponin I levels were mildly elevated at presentation (Table 2). Unfortunately, it was not possible to perform an echocardiogram. In a series of 100 cases, myocarditis was detected post mortem in 62.7% of the cats. 10 Another study on neonatal infections revealed myocardial lesions in 100% of kittens. 70
Conclusions
To the authors’ knowledge, this is the first reported case of fatal disseminated toxoplasmosis in a cat in Spain, confirmed using PCR. When suspecting fatal disseminated toxoplasmosis, FNA of the liver could assist in achieving an early diagnosis.
Footnotes
Acknowledgements
The authors would like to thank Histolab Veterinaria (Malaga, Spain) for the post-mortem review of the liver and lung FNA cytology samples.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
