Abstract
Case series summary
A 2-year-old female spayed Abyssinian cat was evaluated for lethargy and inappetence that first occurred approximately 4 days prior. In addition, urination had not been observed by the owner for 5 days. A Lilium species plant had been brought into the house approximately 5 days before initial evaluation, and intoxication was therefore suspected. Bloodwork revealed anemia, severe azotemia and hyperkalemia. As the cat was anuric with severe azotemia, hyperkalemia and fluid overload, intermittent hemodialysis was recommended. Attempts were made to place a hemodialysis catheter into the external jugular veins bilaterally, but after initial successful venipuncture, the instrumentation would not pass into either vein. During fluoroscopic angiography, no internal jugular veins or external jugular veins caudal to the thoracic inlet were visualized and venous drainage from the head occurred via the vertebral veins. Owing to the anomalous anatomy in the cervical region, a hemodialysis catheter could not be placed into either jugular vein. Alternative sites for the placement of an extracorporeal catheter were discussed with the owner, but humane euthanasia was elected.
Relevance and novel information
To the authors’ knowledge, this is the first report of a cat with bilateral external jugular vein anomalies resulting in blind endings that did not communicate with the vena cava. This was discovered during attempts to place a hemodialysis catheter for the management of anuric renal failure secondary to Lilium species intoxication. While this anatomical variation is likely uncommon, it is an important differential to consider when faced with challenging external jugular vein catheterizations in feline patients.
Case series description
A 2-year-old female spayed Abyssinian cat was evaluated at the University of California-Davis Veterinary Medical Teaching Hospital (UCD-VMTH) after it was diagnosed with anuric renal failure secondary to Lilium species intoxication. The cat first showed signs of lethargy and decreased appetite approximately 4 days before presentation. In addition, urination had not been observed by the owner during the previous 5 days. Before this episode, the cat had no prior history of any medical problems. A toxic lily plant had been brought into the house approximately 5 days before evaluation, and the cat had access to the plant.
The cat had been taken to the referring veterinarian before presentation at the UCD-VMTH, and bloodwork at that time revealed severe azotemia with a creatinine of 30.2 mg/dl (reference interval [RI] 0.3–2.4 mg/dl) and a blood urea nitrogen (BUN) of 231 mg/dl (RI 16–35 mg/dl).
On physical examination at the UCD-VMTH, the cat was quiet, alert and responsive, and weighed 3.7 kg. The cat’s temperature was 98.6°F (RI 100.5–102.5°F) and the pulse was 200 beats/min (RI 180–220 beats/min) with a respiratory rate of 40 breaths/min (RI 16–30 breaths/min). The hydration status was considered mildly overhydrated. The remainder of the physical examination was unremarkable.
The next morning, results of a complete blood count and biochemistry panel were available. The cat had a mild non-regenerative anemia (hematocrit 26.6%, RI 30–50%, reticulocytes 5900/µl). A biochemistry panel revealed multiple abnormalities, including severe azote-mia with creatinine of 34.1 mg/dl (RI 1.1–2.2 mg/dl) and BUN of 259 mg/dl (RI 18–33 mg/dl), hyperphosphatemia (16.9 mg/dl, RI 3.2–6.3 mg/dl), total hypercalcemia (11.3 mg/dl, RI 9–10.9 mg/dl), hyperkalemia (7.2 mEq/l, RI 3.8–5.4 mEq/l), hypoproteinemia (5.7 g/dl, RI 6.6–8.4 g/dl), hypoglobulinemia (2.4 g/dl, RI 2.8–5.4 g/dl), increased aspartate aminotransferase (86 IU/l, RI 17–58 IU/l) and increased creatinine kinase (8900 IU/l, RI 73–260 IU/l). Urinalysis revealed urine-specific gravity of 1.009, glucosuria (100 mg/dl, RI negative), proteinuria (30 mg/dl, RI negative) and microscopic hematuria with 50–100 rbc/hpf (RI 0–2 hpf). Urine culture was negative for bacterial growth.
Three-view thoracic radiographs were obtained and demonstrated that the cardiac silhouette was rounded on the lateral projections with a base-wide appearance on the dorsoventral projection. The pulmonary vasculature was distended with branches extending further into the periphery than expected. A patchy interstitial pulmonary pattern was noted. The combination of radiographic findings was most suggestive of fluid overload and/or left-sided congestive heart failure. An echocardiogram was performed, and demonstrated no overt evidence of primary cardiac disease; however, the left ventricle, and right and left atria were mildly dilated, suggesting fluid overload.
A full abdominal ultrasound examination (iE 33 Ultrasound; Philips Healthcare Solutions) was performed. The kidneys measured within normal limits (left kidney 3.3 cm in length, right kidney 4.0 cm in length) but were noted to have bilaterally thickened, hyperechoic cortices. There were irregularities along the cortical margins. There was a moderate volume of anechoic effusion within the retroperitoneal space bilaterally.
As no other cause of acute kidney injury was identified, lily intoxication was suspected due to the recent exposure. As the cat was anuric with severe azotemia, hyperkalemia and fluid overload, initiation of renal replacement therapy with intermittent hemodialysis was recommended. An intravenous catheter was placed in the cephalic vein, and anesthesia was induced with 8 mg/kg propofol administered intravenously. After intubation, the cat was maintained under anesthesia with isoflurane in oxygen.
The cat was transferred to the hemodialysis suite and was placed in left lateral recumbency. The cervical neck region was clipped free from hair, prepared with aseptic technique and draped. An 18-gauge over-the-needle catheter (Becton, Dickinson and Company) was inserted into the right external jugular vein. The catheter was advanced over the needle into the jugular vein and the needle was removed. A strong flash of blood was obtained from the catheter, and the blood was able to be aspirated. A J-tip guidewire (Spring-wire Guide: 0.032-inch diameter; Teleflex) was placed in the catheter, but the guidewire was unable to be advanced further than approximately 4 cm distally. A 9-Fr dilator (Teleflex) was advanced over the guidewire into the jugular vein for several centimeters and then exchanged for a 16-cm-long 8-Fr double-lumen catheter (ARROWgard Blue PLUS Two-Lumen CVC; Teleflex). However, the catheter was unable to be advanced further than approximately 10 cm and no blood was obtained at this location. A physical obstruction to the guidewire and catheter passage was felt when attempts to advance these instruments were made. Due to concern that the vessel had been perforated, the catheter and guidewire were removed. Catheter placement was then attempted in the left external jugular vein. Again, after successful venipuncture, the guidewire was unable to be advanced further than approximately 4 cm. Ultrasonography of the cervical neck was performed, and the external jugular veins were unable to be traced beyond the level at which the guidewires terminated. The 18-gauge over-the-needle catheter was sutured in place to allow for an angiographic study. Both CT angiography and fluoroscopic angiography were considered to evaluate the vascular anatomy in the neck; due to the desire for a shorter anesthesia, the injection of a lower dose of contrast medium and the ability to generate a dynamic study, fluoroscopic angiography was selected over CT angiography.
The cat was transported to the fluoroscopy suite (OEC 9900 Elite; GE Healthcare) and placed in dorsal recumbency. Iopamidol contrast medium (1.5 ml, Isovue-370; Bracco Diagnostics) was mixed with 1.5 ml saline (0.9% NaCl), and an injection into the 18-gauge catheter in the left external jugular vein was performed with the thoracic inlet centered in the fluoroscopy beam (Figure 1). The fluoroscopic angiogram revealed that the left external jugular vein extended only two-thirds of the cranial-caudal length of the neck, never reaching the thoracic inlet. The contrast medium then traveled cranially from the left external jugular vein into the transverse jugular vein and further into the contralateral external jugular vein, which was only noted to extend two-thirds of the cranial-caudal length of the neck (Figure 1). From the external jugular veins, contrast medium flowed into the anterior and posterior facial veins bilaterally, which then drained into the vertebral veins bilaterally. Once in the vertebral veins, blood flow traveled caudally through the thoracic inlet, until the vertebral veins connected to the vena cava, and blood then continued to the heart (Figure 1). No internal jugular veins or external jugular veins caudal to the thoracic inlet or brachiocephalic veins were noted.
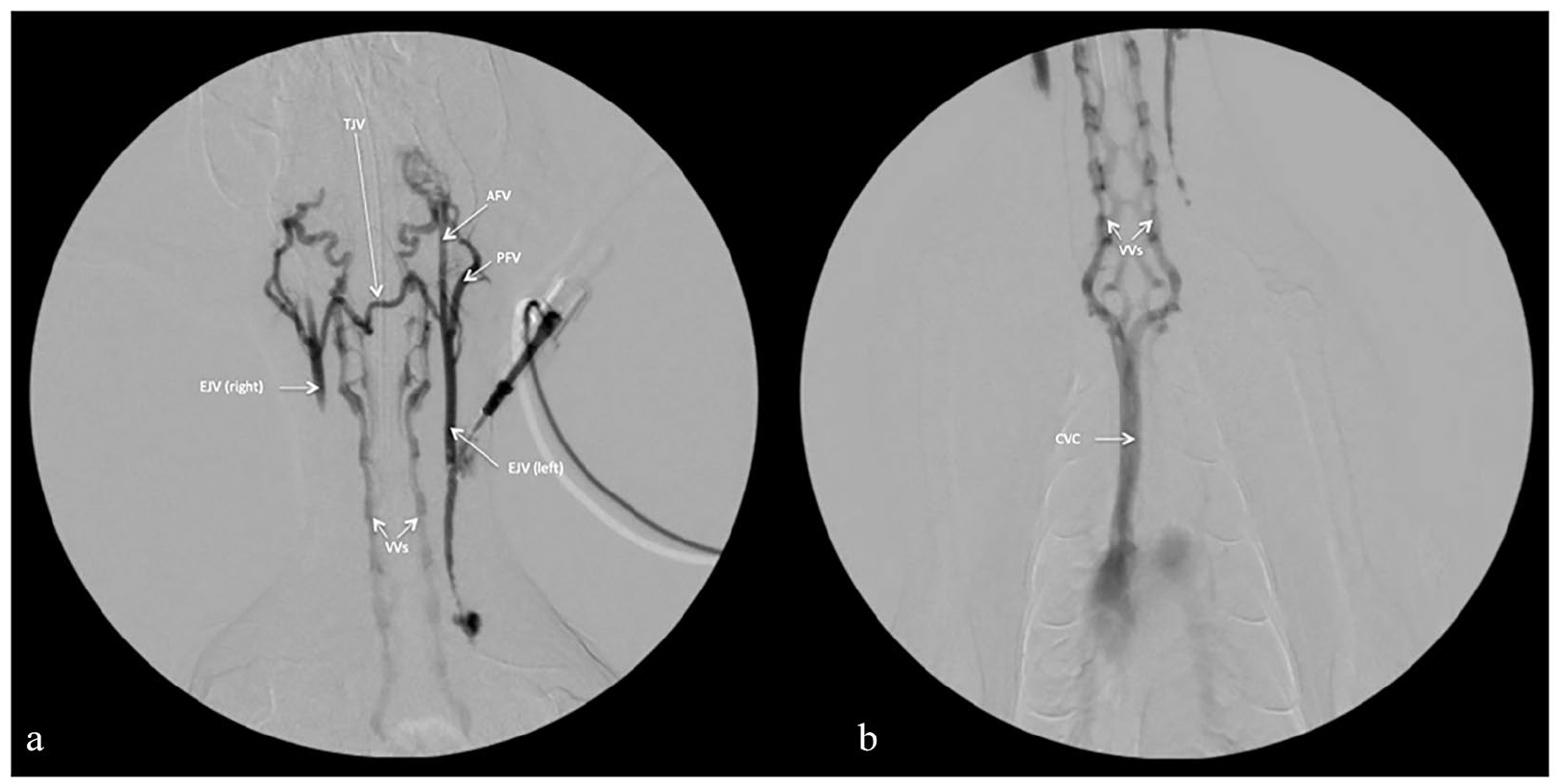
Fluoroscopic images after the contrast medium injection with (a) the caudal skull centered and (b) thoracic inlet centered. (a) After the injection of contrast medium into the left external jugular vein (EJV left), the contrast medium travels cranially into the anterior facial vein (AFV), posterior facial vein (PFV) and transverse jugular vein (TJV). After traveling through the TJV, the contrast medium can be seen in the right EJV (EJV right), and then finally draining into the vertebral veins (VVs). (b) The contrast medium can be seen traveling caudally from the VVs to the cranial vena cava (CVC)
Unfortunately, owing to the anomalous venous anatomy in the cervical region, a hemodialysis catheter was unable to be placed. Discussion regarding the use of peritoneal dialysis or placement of a double-lumen catheter into another vein (eg, femoral vein) was had with the owner; however, humane euthanasia was elected at that time. No necropsy was performed.
Discussion
When severe anuric acute renal failure that cannot be medically managed develops, renal replacement therapies such as intermittent hemodialysis or continuous renal replacement therapy are often the only effective treatment modalities. In veterinary medicine, renal replacement therapies, such as intermittent hemodialysis, are almost exclusively performed by the placement of a large bore double-lumen catheter in one of the jugular veins. Other vessels may be considered for access; however, vessel size and the mobility of the site limits the ability to successfully maintain a longer-term catheter in alternative sites, especially in cats and small- to medium-sized dogs.
Cats ingesting Lilium species can develop varying degrees of kidney injury, and while reports of cats with anuric renal failure secondary to intoxication are infrequent in veterinary medicine, the prognosis is often guarded.1,2 Unfortunately, in the cat of this report, ingestion of Lilium species led to severe acute kidney injury with metabolic derangements and anuria, and by the time of presentation to our hospital, renal replacement therapy was required. The inability to obtain venous access in the jugular veins of this cat led the owner to elect euthanasia due to the guarded prognosis, even with other modalities being available, such as peritoneal dialysis.
In cats, the major venous drainage from the head and cerebral sinuses are the vertebral veins, but the external jugular veins also contribute to drainage. 3 It is common for the external jugular veins and vertebral veins to communicate extensively in cats during venous drainage. 3 The internal jugular veins contribute minimally to drainage of the head as several studies have shown the internal jugular veins in cats are often not present or small.3,4
In one study, CT examinations of the head and neck in cats showed no variation in the presence and appearance of the external jugular veins and concluded that an alteration in the presence or conformation of the exter-nal jugular vein would be considered abnormal. 4 The absence of a unilateral jugular vein has been documented in a single case when a CT angiographic study was performed in a cat that had experienced trauma and was demonstrating amaurosis. 5 It is possible that the anatomical variation noted in the cat of this report occurs regularly, but this phenomenon may not be appreciated commonly due to the lack of indication for placement of a catheter of significant length in the external jugular vein. In this cat, when a standard over-the-needle catheter was placed (as opposed to the long hemodialysis catheter), bleeding was noted from the over-the-needle catheter, and this catheter was able to be aspirated masking the presence of a venous anomaly.
When it was determined that hemodialysis via an external jugular vein catheter was not going to be possible in the cat of this report, alternatives were considered, including double-lumen catheter placement in a different vein and peritoneal dialysis.6–8 Maintenance of a femoral catheter can be challenging in an active and mobile feline patient. In addition, the small size and tortuosity of the femoral vein as it enters into the abdomen and the constant movement of the hind legs would likely lead to catheter kinking and difficulty maintaining adequate blood flow rates to successfully perform hemodialysis.
In this case, limited characterization of the anomalous vessels was achieved due to the clinical status of the patient and decision to pursue humane euthanasia. Better characterization of the anomalies would have been possible if advanced imaging (eg, CT or MRI) had been performed. In addition, a necropsy could have allowed for more detailed investigation of the anatomy of this cat.
Conclusion
To the authors’ knowledge, this is the first report of a cat with bilateral external jugular vein anomalies resulting in blind endings that did not communicate with the cranial vena cava. This anomaly was discovered due to a poisoning leading to acute kidney injury and the subsequent need for hemodialysis. While this anatomical variation is likely uncommon, it should be an important differential to consider when faced with challenging external jugular vein catheterizations in feline patients.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
