Abstract
Recently, a new low-density lipoprotein apheresis device (Rheocarna®; Kaneka Corporation, Osaka, Japan), a blood purification therapy, was approved in Japan for managing chronic limb-threatening ischemia with refractory ulcers. Here, we describe a case of chronic limb-threatening ischemia that was treated with the Rheocarna. A 65-year-old Asian man with an ulcer on the right heel was admitted to our hospital. Angiography revealed chronic total occlusion with severe calcification of the anterior tibial, peroneal, and posterior tibial arteries. The patient underwent distal bypass of the saphenous vein; however, the bypass was occluded in the early postoperative period. The Rheocarna was used, and the ulcers improved significantly postoperatively. Although endovascular treatment was eventually performed on the occluded bypass graft to completely heal the ulcer, the Rheocarna could be an alternative treatment option in challenging cases of chronic limb-threatening ischemia.
Introduction
Chronic limb-threatening ischemia (CLTI) is a clinical syndrome defined by the presence of peripheral artery disease (PAD) combined with rest pain, gangrene, or lower-limb ulceration for over 2 weeks, and CLTI is the most advanced form of PAD.1,2 The optimal treatment for CLTI is revascularization, such as bypass surgery or endovascular treatment (EVT).3 –5 However, challenging cases with insufficient peripheral circulation or microcirculation after revascularization are often encountered, 6 and adjuvant therapy becomes important. Recently, a new low-density lipoprotein apheresis (LDL-A) device (Rheocarna®; Kaneka Corporation, Osaka, Japan) (Figure 1), a blood purification therapy that selectively removes the causative agents of CLTI, such as low-density lipoprotein cholesterol (LDL-C) and fibrinogen, using porous cellulose beads containing negatively charged dextran sulfate and hydrophobic tryptophan, has received approval in Japan for the management of CLTI with refractory ulcers. LDL-A has been traditionally used for PAD and its efficacy has been reported, 7 but the Rheocarna usage has no prerequisite for the presence of refractory hyperlipidemia compared to conventional therapy. And its use twice a week over a 3-month treatment period is approved for insurance reimbursement (24 times maximum). 8 However, real-world data for this technique are limited. Here, we describe a case of CLTI treated with Rheocarna.

Circuit diagram of the Rheocarna® apheresis device.
Case presentation
A 65-year-old Asian man who underwent hemodialysis for chronic glomerular nephritis at 39 years of age was referred to our hospital for an ulcer on his right heel (Figure 2(a)). His medical history included hypertension and dyslipidemia. The patient underwent multiple percutaneous coronary interventions for angina pectoris. His medications included aspirin (100 mg/day), clopidogrel (75 mg/day), bisoprolol (5 mg/day), pitavastatin (4 mg/day), and olmesartan (20 mg/day).

(a) The wound before treatment (30 × 30 mm). (b) Angiography images before treatment. (c) Angiography images presenting occlusion after surgery (white arrow). (d) The wound after Rheocarna® therapy (20 × 5 mm). (e) Angiography images after endovascular treatment. (f) The wound after endovascular treatment.
The right ankle-brachial index (ABI) 9 value was 0.71. The right-sided skin perfusion pressure (SPP) was 23 mmHg in the dorsal area and 21 mmHg in the plantar area. Routine laboratory tests revealed decreased hemoglobin (10.1 g/dL) and serum albumin (3.1 mg/dL) levels. The inflammatory marker levels (C-reactive protein, 0.3 mg/dL) were normal, and chest radiography revealed no abnormalities. Electrocardiography at rest demonstrated a normal sinus rhythm. Transthoracic echocardiography revealed diffuse left ventricular dysfunction (left ventricular ejection fraction, 45%). Pre-treatment right lower extremity angiography showed chronic total occlusions with severe calcification in the anterior tibial, peroneal (PA), and posterior tibial arteries (Figure 2(b)). Based on ABI and angiographic findings, the patient was diagnosed with CLTI. We attempted EVT of the PA, but the guidewire did not pass through the lesion because of severe calcification. The patient underwent distal bypass of the saphenous vein at another hospital. It was a popliteal-posterior tibial bypass with a detached saphenous vein. However, the bypass was occluded during the early postoperative period (Figure 2(c)). Therefore, we decided to treat the patient with Rheocarna. The LDL-C and fibrinogen levels immediately before the start of the Rheocarna regimen were 78 and 550 mg/dL, respectively. The Rheocarna was administered 15 times, and the quantity of blood flow was measured in the range of 100–120 mL/min for 2 h for each session. The apheresis was carried out through the arteriovenous fistula that the patient used for hemodialysis. Heparin was used as an anticoagulant, and no vasodilators were used during treatment. The patient’s ulcer gradually and significantly improved over 2 months (Figure 2(d)), the ulcer was reduced from 30 × 30 mm to 20 × 5 mm, and plantar SPP improved from 21 mmHg to 31 mmHg. Moreover, angiographical findings showed improved peripheral microcirculation. But ABI value remained essentially unchanged. After the Rheocarna administration, LDL-C levels decreased from 78 to 42 mg/dL, and fibrinogen levels decreased from 550 to 353 mg/dL. Eventually, EVT with plain balloon angioplasty was successfully performed on the occluded bypass graft (Figure 2(e)), and the ulcer resolved 1 month later (Figure 2(f)). However, the patient died of heart failure 7 months after the EVT.
Discussion
Here, we reported a case of CLTI treatment using Rheocarna, which decreased LDL-C and fibrinogen levels, improved microcirculation, and significantly improved refractory ulcers. CLTI comprises both macrocirculatory and microcirculatory diseases involving impaired blood flow rheology. Blood and plasma viscosities, important factors in blood flow rheology, are major threats associated with cardiovascular disease and mortality. 10 Viscosity causes endothelial dysfunction and atherosclerosis, disturbing blood flow. Although the detailed mechanism by which viscosity causes endothelial dysfunction and atherosclerosis remains unclear, few studies have reported that viscosity is strongly associated with endothelial shear stress in PAD. 11 LDL-A can improve blood and plasma viscosity in terms of shear rates, not just merely reduce LDL-C levels. 12 In addition, LDL-A reduces levels of fibrinogen, 13 a major determinant of blood and plasma viscosity. Decreased fibrinogen levels improve endothelial function, improving the microcirculation. 14 Thus, modulating LDL-C and fibrinogen levels, which affect blood and plasma viscosity, is a very important therapeutic target. In this case, LDL-C and fibrinogen levels improved after Rheocarna administration, resulting in partial healing of the ulcer via improved microcirculation.
Moreover, in this case, Rheocarna treatment preceded EVT despite the bypass being occluded in the early postoperative period because the use of EVT for infrainguinal venous bypass grafts in patients with PAD remains controversial. 15 In addition, hemodialysis is a known risk factor for lower extremity bypass failure in CLTI patients. 16 Furthermore, the patient was recently reported to have concomitant triglyceride deposit cardiomyovasculopathy (TGCV) based on a 123I-β-methyl-p-iodophenyl-pentadecanoic acid (BMIPP) wash-out rate of 4.3% and diffuse coronary atherosclerosis on coronary angiography (Figure 3(a)–(d)). TGCV is a metabolic disease characterized by excessive triglyceride deposit accumulation in the heart or vessels and has a high target vessel revascularization rate and poor prognosis.17,18 It has been reported that the factors relevant to coronary stent restenosis, including vascular endothelial growth factor-alpha, transforming growth factor-beta, and interleukin-6, exhibit distinct expression levels in mouse models of TGCV compared with those in the wild-type mice, which may influence the pathophysiology and treatment of CLTI, but the details are unknown. 19 Therefore, we considered that enforcing EVT against the occluded bypass graft would reocclude it; we used the Rheocarna first. Rheocarna therapy decreases LDL-C and fibrinogen levels and improves the microcirculation, which might contribute to the improvement of refractory ulcers even in cases of occluded bypass. The Rheocarna recently received approval and limited reports detail its use, and its detailed mechanism remains unclear. In addition, although we selected Rheocarna in this case, we need to figure out medical therapy with prostanoid, which may improve outcomes after revascularization in patients with a poor runoff like this, 20 as a potential therapeutic option. Furthermore, studies are required to evaluate its real-world effects.
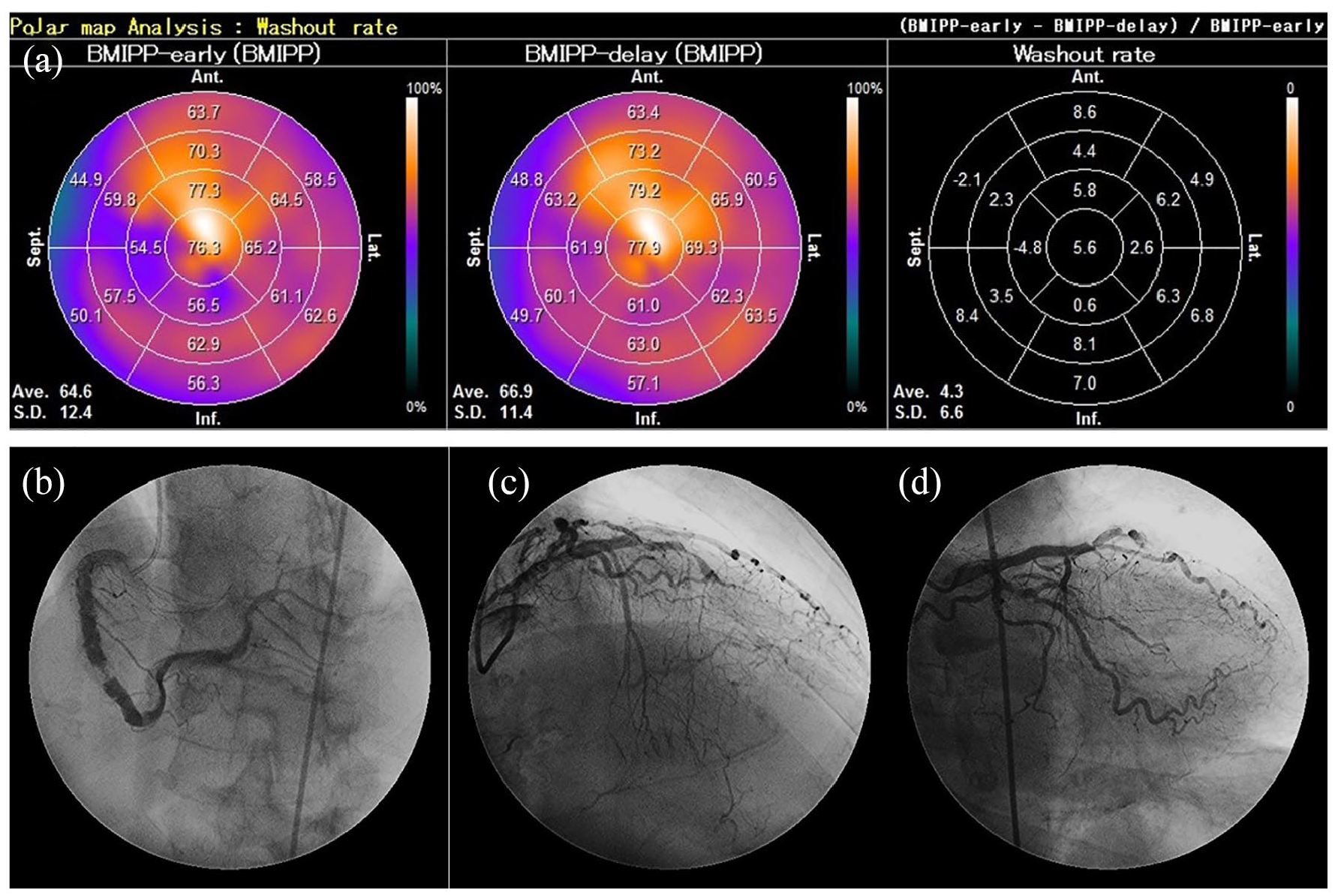
Iodine-123(123I)-β-methyl-p-iodophenyl-pentadecanoic acid (BMIPP) myocardial single-photon emission computed tomography and diffuse atherosclerosis on coronary angiography. 123I-BMIPP wash-out rate of <10% is a diagnostic criterion for triglyceride deposit cardiomyovasculopathy. (a) 123I-BMIPP washout rate. (b) Right coronary artery. (c) Left anterior descending coronary artery. (d) Left circumflex coronary artery.
Conclusion
The Rheocarna is a potential treatment option for CLTI in cases in which the indications for EVT are unclear or in challenging cases of refractory ulcers in daily clinical practice.
Footnotes
Acknowledgements
The authors thank all staff who contributed to the management of this case, including doctors, nurses, clinical engineers, pharmacists, laboratory technicians, and medical clerks.
Author contributions
The contributions of respective joint authors are below
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
