Abstract
Case series summary
A retrospective, single-center case series describes five cats with primary portal vein hypoplasia (PVH). This case series outlines clinical signs, diagnostics, treatments and prognosis in cats diagnosed with primary PVH based on histopathology. Pathology records from cats that underwent liver biopsies were retrospectively searched to identify cats with histologic features of portal vein hypoperfusion. Patients with concurrent hepatic conditions such as portosystemic shunts, portal vein obstruction, arteriovenous fistulas and other hepatic conditions that complicate vasculature were excluded, leaving five cases for inclusion. In this case series, 3/5 cats were asymptomatic, with abnormalities detected on biochemistry or abdominal ultrasound, one cat presented with lethargy and one cat presented with gastrointestinal signs associated with a foreign body. Of the five cats in this series, four underwent treatment with follow-up biochemistry profiles. One cat in this study was a well-controlled diabetic and hyperthyroid, while one cat was newly diagnosed with hyperthyroidism after biopsies, which complicated liver enzyme interpretation. Treatment protocols were variable and encompassed management of concurrent metabolic disorders, use of hepatoprotective medications, hepatic encephalopathy therapy, antimicrobial treatment, copper chelation and immunosuppressive therapy. A larger case series or a prospective study is necessary to establish the optimal treatment plan, as there were no established standards for managing this condition for this cohort. Four cats with confirmed follow-up had an average survival time of 878.5 days after diagnosis, indicating a favorable prognosis.
Relevance and novel information
PVH is a stereotypical microscopic finding in liver portal tracts, most often linked to portosystemic shunts. It can occur with any condition that reduces portal blood flow. In the absence of a shunt, arteriovenous fistula or portal vein obstruction, it is termed primary portal vein hypoplasia, which is poorly characterized in cats.
Introduction
Primary portal vein hypoplasia (PVH) is a congenital anomaly reported in dogs, but rarely in cats, with exact prevalence unknown. 1 PVH is microscopically characterized by portal vein hypoperfusion (PVHp) due to abnormal development of the vitelline veins during embryogenesis. 2 The vitelline veins give rise to the portal vein, and disruption at any stage can impair the formation and growth of the smallest intrahepatic portal branches, resulting in PVH. 2 Histological abnormalities in portal tracts with PVHp include decreased portal vein diameter or absence of portal veins, increased number of arteriolar profiles, periportal fibrosis, hepatocyte atrophy, increased ductular profiles, lipogranulomas and sinusoidal dilatation in the periportal regions.1,3 –5 However, these features are not exclusive to primary PVH and can be seen with intra- or extrahepatic portosystemic shunts (PSSs), intrahepatic arteriovenous fistulas, portal vein obstruction, cirrhosis and congenital hepatic fibrosis. 1 PVH is diagnosed after excluding other causes of PVHp, including PSS, portal vein obstruction or arteriovenous fistula through imaging, along with histopathologic findings in multiple liver lobes.
In dogs, clinical severity of PVH is diverse and depends on the degree of hypoplasia and loss of hepatocellular function. 1 Clinical signs range from subclinical to variable degrees of gastrointestinal upset, hepatic encephalopathy (HE) and stunted growth.6,7 Clinicopathological abnormalities in dog cases include persistently increased liver enzymes including alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT) and total bilirubin (TBili). 3 Other clinicopathological changes include increased blood ammonia levels and decreased blood urea nitrogen. 3 Abdominal ultrasound (AUS) is utilized initially to evaluate hepatic abnormalities and assess portal blood flow for the identification of PSSs. 7 In one study, dogs with PVH had no detectable abnormalities of the hepatic parenchyma or vasculature on AUS. 3 CT angiography may be considered in cases with a higher suspicion for PSS. A previous study reported a sensitivity of 96% and specificity of 89% for CT angiography in detecting PSS compared with 68% and 84%, respectively, for AUS. 8 Based on these findings, CT angiography and histopathology of multiple liver lobes are considered the gold standard diagnostic for dogs.
Most dogs with primary PVH are asymptomatic and managed with hepatoprotectants, such as s-adenosylmethionine with silybin-phosphatidylcholine complex (SAMe-SPC), to support hepatic health and regeneration. 7 Dogs with progressive liver dysfunction may require additional therapy, including low-protein diet modification and management of HE with lactulose, which reduces ammonia absorption, and metronidazole, which decreases ammonia-producing gut bacteria.6,7 Portal hypertension, another potential consequence, can be managed with spironolactone, an aldosterone antagonist that helps reduce fluid accumulation.6,7 This case series aims to outline the clinical signs, diagnostics, treatments and prognosis in cats diagnosed with primary PVH.
Case series description
Medical records from Angell Animal Medical Center in Boston, MA, were reviewed for cats with liver biopsies between October 2014 and August 2023. Cases were identified using terms related to primary PVH, including ‘hypoperfusion’, ‘portal vein atrophy’, ‘arteriolar hyperplasia’ and ‘biliary hyperplasia’. Inclusion criteria included liver biopsies, baseline biochemistry and AUS to evaluate liver architecture and exclude other portal vascular abnormalities. Exclusion criteria included other hepatic vascular disease or the absence of imaging. For included cases, the following data were collected: signalment, clinical history, therapies trialed, relevant examination findings, preoperative biochemistry, ultrasound results performed by a board-certified radiologist, surgical reports detailing biopsy techniques, histopathology, heavy metal screening, cultures and follow-up information.
Outcomes were assessed using recheck biochemistries, therapeutics initiated, additional diagnoses and survival time from discharge. As long-term follow-up was often performed by referring veterinarians, a questionnaire was distributed to owners via email or telephone. This questionnaire evaluated survival status, current medications, concurrent conditions, recheck biochemistries, biopsies or imaging, and quality of life after the diagnosis. Survival time was evaluated from time of biopsy to study completion.
Of the 29 initial cases, 24 were excluded because of other diagnosed hepatic pathologies (n = 23) or absence of ultrasound (n = 1). Five cats met the inclusion criteria. Their signalments, presenting complaints and physical examination findings are summarized in Table 1, biochemistry results in Table 2 and gross hepatic appearance, copper quantification, and aerobic and anaerobic culture results in Table 3. Biochemistry and complete blood cell count results were available in all cats to exclude prehepatic causes of hyperbilirubinemia. Liver enzymes and bilirubin are highlighted in Table 2. The most common elevation in all five cats was ALT, with a mean of 749.8 U/l (range 27–211), followed by AST (mean 192.4 U/l, range 18–465), ALP (mean 204.5 U/l, range 15–579), GGT (mean 6.6 U/l, range 1–10) and TBili (mean 0.82 mg/dl, range 0.1–2.3).
Signalment, presenting complaint and initial findings for five cats with primary portal vein hypoplasia

ALT = alanine aminotransferase; DSH = domestic shorthair; FS = female spayed; MC = male castrated; NSF = no significant findings
Liver biochemistry results at the time of presentation for five cats with primary portal vein hypoplasia

Values in bold are outside the reference interval
ALT = alanine aminotransferase; ALP = alkaline phosphatase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase; N/A = test was not submitted or value not available; TBili = total bilirubin
Biopsy method, liver gross appearance, histopathology and ancillary testing in five cats with primary portal vein hypoplasia

dw = dry weight; N/A = test was not submitted or value not available; ppm = parts per million; RI = reference interval
Cat 1 was asymptomatic at presentation, with incidental increases in liver enzyme activity and an unremarkable AUS. Histopathology and ancillary tests are outlined in Table 3. This cat was treated with SAMe (22 mg/kg PO q24h) with SPC (22 mg/kg PO q24h) and ursodeoxycholic acid (11 mg/kg PO q24h) for 9 months. Follow-up liver enzymes (Figure 1) demonstrated improvement after initiating therapy, with continued improvement after discontinuation. The greatest improvement was ALT, followed by AST. At the time of writing, cat 1 maintained a good quality of life with no concurrent disease or ongoing treatments.

(a) Highlights cat 1’s liver enzyme values from the time of diagnosis, which is noted as day 0. Day 160 highlights recheck liver enzymes while cat 1 was still on treatments, including s-adenosylmethionine with silybin-phosphatidylcholine complex (SAMe-SPC) and ursodeoxycholic acid. During subsequent follow-up of liver enzyme values, cat 1 was no longer under active treatment. (b) Highlights cat 1’s total bilirubin (TBili) values from the time of diagnosis, which is noted as day 0. Day 160 highlights recheck liver enzymes while cat 1 was still on treatments, including SAMe-SPC and ursodeoxycholic acid. During subsequent follow-up of TBili values, cat 1 was no longer under active treatment. ALT = alanine aminotransferase; ALP = alkaline phosphatase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase
Cat 2 exhibited mild lethargy, accompanied by increased liver enzymes. A mild extrahepatic bile duct dilation was observed on AUS, but other findings were unremarkable. Histopathology and ancillary tests are outlined in Table 3. Cat 2 was treated with lactulose (150 mg/kg PO q12h) for 1 month and metronidazole (7 mg/kg PO q12h) for 3 months to address possible HE, given reported lethargy. This cat was also a well-controlled diabetic on glargine (1 unit SC q12h) and hyperthyroid on methimazole (0.9 mg/kg PO q12h). Initial follow-up after discontinuing lactulose showed mild improvement of liver enzymes but an increase in TBili (Figure 2). Subsequent follow-up after discontinuing metronidazole demonstrated further improvement in all values. One year after biopsy, ALT remained the highest enzyme at 1040 U/l. The follow-up questionnaire noted that cat 2 had a good quality of life with PVH and concurrent metabolic diseases.
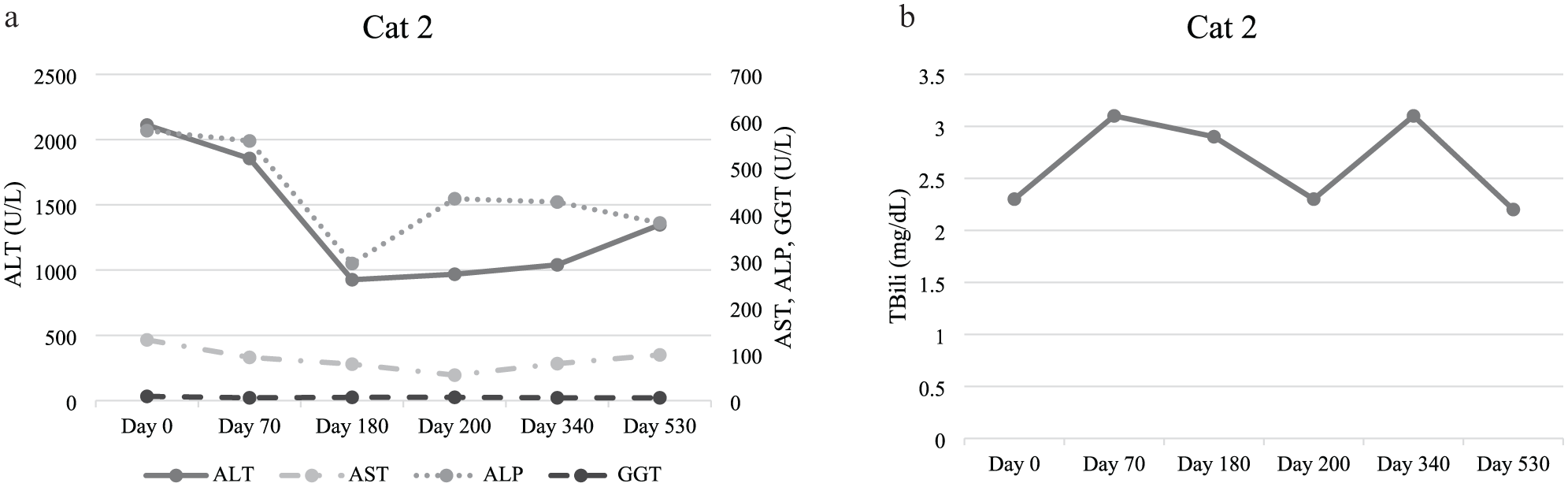
(a) Highlights cat 2’s liver enzyme values from the time of diagnosis, which is noted as day 0. At the day 70 follow-up, liver enzymes results reflected that cat 2 was receiving metronidazole, with lactulose having been discontinued after 30 days. During subsequent follow-up of liver enzyme values, cat 2 was no longer under active treatment aside from diabetic and hyperthyroid management. (b) Highlights cat 2’s total bilirubin (TBili) from the time of diagnosis, which is noted as day 0. At the day 70 follow-up, liver enzyme results reflected that cat 2 was receiving metronidazole, with lactulose having been discontinued after 30 days. During subsequent follow-up of liver enzyme values, cat 2 was no longer under active treatment aside from diabetic and hyperthyroid management. ALT = alanine aminotransferase; ALP = alkaline phosphatase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase
Cat 3 presented febrile, vomiting and anorexic with increased liver enzymes. A linear foreign body was identified on AUS, likely accounting for the presenting signs. Histopathology and ancillary tests are outlined in Table 3. Cat 3 was not started on treatment and was lost to follow-up.
Cat 4 was asymptomatic with normal liver enzyme activity but was included because of a hyperechoic hepatic parenchyma on AUS during a hyperthyroidism work-up. Histopathology and ancillary tests are outlined in Table 3. Based on bile culture and sensitivity results, enrofloxacin (5 mg/kg PO q24h) was initiated. Follow-up biochemistry after 2 weeks demonstrated that liver enzymes remained within the reference interval (RI). This cat was an outlier in the series, as liver enzymes were consistently normal. Cat 4 was euthanized because of pulmonary nodules and pleural effusion observed on thoracic ultrasound; no additional diagnostics or necropsy were performed. Survival time from biopsy was 288 days.
Cat 5 was asymptomatic at presentation, with incidental increases in liver enzymes on routine biochemistry. A mottled hepatic parenchyma was seen on AUS. Histopathology and ancillary tests are outlined in Table 3. Aerobic and anaerobic bile cultures grew Staphylococcus pseudintermedius, initially considered a potential contaminant but treated prophylactically. Therapeutic management included penicillamine (5.7 mg/kg PO q12h) for increased copper, 1 week of amoxicillin-clavulanic acid (13.75 mg/kg PO q12h) based on culture results, SAMe (22.6 mg/kg PO q24h) with SPC (22.3 mg/kg PO q24h) and ursodeoxycholic acid (7.5 mg/kg PO q24h). Penicillamine was not consistently administered. Follow-up biochemistry showed progressive liver enzyme elevations (Figure 3). Concurrently, this cat was diagnosed with hyperthyroidism and treated with methimazole (0.25 mg/kg PO q24h). At the same time, cat 5 was started on chlorambucil (5.26 mg/m2) for 8 months because of persistently increased liver enzymes. Chlorambucil was initiated because of the persistent enzyme elevation and re-evaluation of the biopsy, which showed periportal hepatitis interpreted as reactive or non-specific. More extensive lymphocytic inflammation would have raised concern for lymphocytic portal hepatitis and was the rationale behind initiating chlorambucil. Follow-up testing showed initial improvement that plateaued after discontinuing chlorambucil. Cat 5 was alive with a good quality of life according to available records at the time of writing.
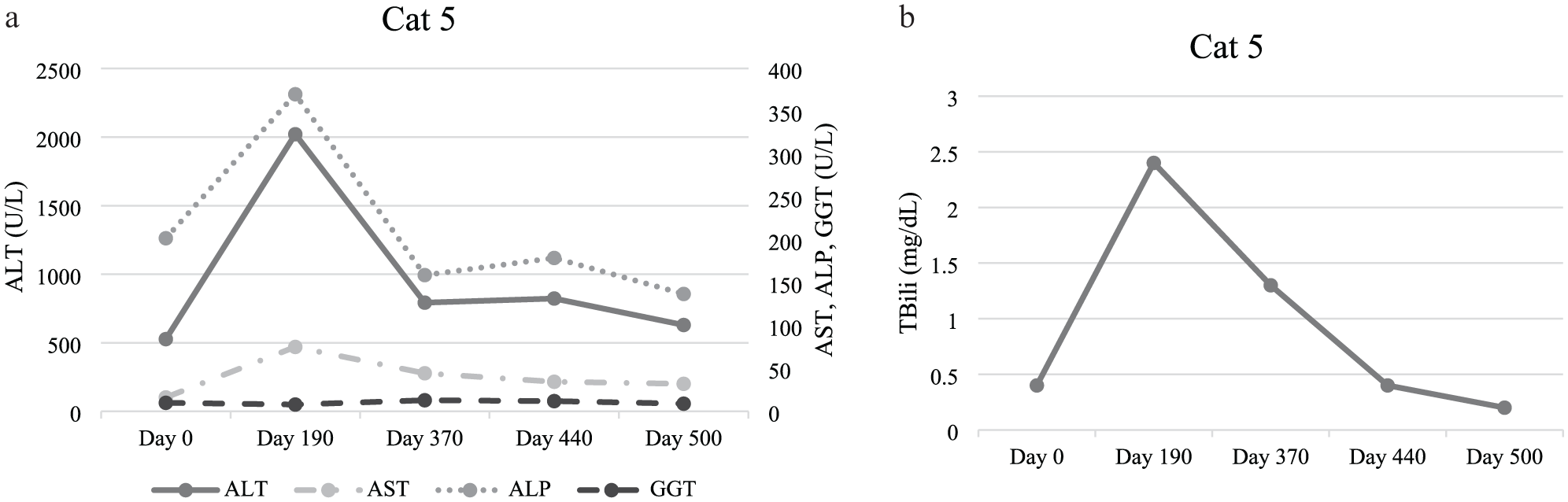
(a) Highlights cat 5’s liver enzyme values from the time of diagnosis, which is noted as day 0. Cat 5 received penicillamine therapy for copper accumulation, but administration was inconsistent because of gastrointestinal upset. Liver enzyme values were not re-evaluated while cat 5 was on penicillamine. At the day 190 evaluation, cat 5 was receiving s-adenosylmethionine with silybin-phosphatidylcholine complex (SAMe-SPC) and ursodeoxycholic acid. A diagnosis of hyperthyroidism was documented at this time and methimazole was initiated. Chlorambucil was subsequently added to the treatment regimen at this time. Day 370 reflects recheck liver enzymes, while cat 5 was a well-controlled hyperthyroid as well as receiving SAMe-SPC, ursodeoxycholic acid and chlorambucil. At day 440, liver enzymes were stable, prompting discontinuation of chlorambucil while maintaining all other treatments. By day 500, liver enzyme values continued to remain stable with well-controlled hyperthyroidism and continuing SAMe-SPC and ursodeoxycholic acid. (b) Highlights cat 5’s total bilirubin (TBili) values from the time of diagnosis, which is noted as day 0. Cat 5 received penicillamine therapy for copper accumulation, but administration was inconsistent because of gastrointestinal upset. TBili was not re-evaluated while cat 5 was on penicillamine. At the day 190 evaluation, cat 5 was receiving SAMe-SPC and ursodeoxycholic acid. A diagnosis of hyperthyroidism was documented at this time and methimazole was initiated. Chlorambucil was subsequently added to the treatment regimen at this time. Day 370 reflects recheck TBili, while cat 5 was a well-controlled hyperthyroid as well as receiving SAMe-SPC, ursodeoxycholic acid and chlorambucil. At day 440, TBili demonstrated overall stability, prompting discontinuation of chlorambucil while maintaining all other treatments. By day 500, TBili continued to reflect stability with well-controlled hyperthyroidism and continuing SAMe-SPC and ursodeoxycholic acid. ALT = alanine aminotransferase; ALP = alkaline phosphatase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase
At the time of writing, 3/5 cats were still alive, one was euthanized for unrelated causes and one was lost to follow-up. The mean survival time after liver biopsy was 878.5 days (range 288–1827), indicating PVH generally carried a favorable prognosis with appropriate medical management.
Discussion
This case series outlined clinical signs, clinicopathological changes, histopathology, treatment, follow-up and survival time in five cats with primary PVH, a condition not widely reported in cats. In dogs, PVH often presents asymptomatically; however, gastrointestinal upset, HE or stunted growth can occur.6,7 In this case series of five cats, three were asymptomatic, with abnormalities detected on biochemistry or AUS, one cat presented with lethargy and one cat presented with gastrointestinal signs associated with a foreign body.
As observed in dogs, elevation of ALT ranging from mild to severe was the most consistent laboratory abnormality in the cats of this series. 3 Three out of the five cats were referred for asymptomatic liver enzyme elevation, prompting further diagnostics, including imaging and biopsies. Ultrasound was an important diagnostic screening factor for PSS and vascular anomalies. The presence of a PSS cannot be definitively excluded without a CT scan, which remains the gold standard for vascular evaluation. However, in this series, CT was not pursued as all cats lacked clinical evidence consistent with a PSS, and AUS failed to identify anomalous vasculature. Given these findings, the likelihood of a PSS was considered low, and the diagnosis was supported by biochemical and histopathologic changes and absence of ultrasonographic evidence of PSS. Histopathology features most commonly associated with PVH include decreased portal vein diameter, arteriolar and biliary hyperplasia, lobular atrophy and hepatocellular vacuolation, which were all common findings in these cats.1,3–5 Cat 3 had an abnormal presentation as the clinical signs were attributed to a foreign body rather than to PVH. Liver biopsies from cat 3 had minimal hepatic lipidosis, also associated with an intestinal foreign body, as hepatic lipidosis typically occurs with prolonged anorexia, making it an uncommon finding with PVH. 9
Follow-up was available for 4/5 cats in this series, and there was no standardized treatment for primary PVH. Hepatoprotectants are the main therapies used in dog cases, and other therapies can be added based on the development of signs compatible with liver dysfunction.
HE is described as a neuropsychiatric syndrome occurring when over 70% of liver function is lost. 10 Cat 2 received treatment for suspected HE due to concurrent lethargy. Improvement in all liver enzymes were noted in this cat, ultimately resulting in discontinuation of medications. Continued improvement of liver enzymes was noted despite discontinuation, and it was unclear if medications had a significant benefit. None of the cats in this series showed evidence of ascites secondary to PH, which is caused by increased resistance or blood flow through portal circulation. 11
A subset of cats in this series had well-managed conditions: hyperthyroidism and diabetes mellitus (DM). Hyperthyroidism was pre-existing in cat 2, previously treated in cat 4 and diagnosed after biopsy in cat 5, while DM was pre-existing in cat 2. In another study, approximately 75% of hyperthyroid cats have increased ALP and ALT, and over 90% showed elevation in at least one enzyme. 12 Histopathologic changes were typically non-specific and unrelated to PVH, and the etiology of enzyme elevation remains unclear. 12 In DM, ALT or ALP elevations were reported in 40–50% of patients, often reaching several times the upper RI, with mild hyperbilirubinemia sometimes reflecting hepatic lipidosis. 13 In this case series, the cat with concurrent DM showed no histologic evidence of hepatic lipidosis, making it unlikely that the increased enzymes were secondary to diabetes.
The impact of hepatoprotective supplements on lifespan is not well understood. SPC is an antioxidant that helps to reduce ongoing oxidative damage, while SAMe protects the liver cells. 7 Ursodeoxycholic acid is an off-license synthetic hydrophilic bile acid that has a role in hepatic inflammation and promotes bile flow. 7 Cat 1 showed liver enzyme improvement after 9 months of treatment with both medications; however, further improvement occurred after discontinuing, leaving it unclear what role these therapies played in the improvement.
Cat 4 had normal liver enzymes before biopsy and was treated with enrofloxacin based on bile culture and sensitivity results. Antibiotic selection should be guided by culture and sensitivity findings while adhering to the best antimicrobial stewardship practices, although some variation between clinicians is expected. Liver enzymes remained normal after discontinuation, making this cat an outlier for being the only cat with normal enzymes.
Cat 5 received SAMe-SPC, ursodeoxycholic acid and penicillamine. Penicillamine, an off-license copper-chelator, forms water-soluble complexes with excess hepatic copper for urinary excretion. 14 Copper elevation can occur primarily and secondarily in various liver diseases, requiring appropriate management for favorable outcomes. 15 Penicillamine can cause adverse effects, including gastrointestinal upset, which led to discontinuation in cat 5. 14 A rare side effect of penicillamine is hemolytic anemia, which typically occurs secondary to severe hepatic necrosis and systemic copper release; however, this was not observed in this cat. 15 The progressive increase in ALT in this cat was likely related to hyperthyroidism, as enzyme values improved after methimazole. The concurrent use of chlorambucil for periportal hepatitis as an immunosuppressant and anti-inflammatory agent had an unclear effect on enzyme improvement, as the initial biopsy changes were considered reactive or non-specific rather than indicative of lymphocytic portal hepatitis.
Follow-up was available in 4/5 cats in this series and showed an overall favorable prognosis, with a mean survival time of 878.5 days from the time of biopsy. Canine studies also report a favorable prognosis, with a survival time of over 5 years. 3 However, as PVH is a congenital condition, progression can occur, making follow-up crucial. The cats’ ages should be considered, as two were seniors at the time of diagnosis. However, their advanced age also suggests that this condition may remain non-progressive for many years. Further studies should be considered to best determine the prognosis of PVH with regulated treatments.
This study has limitations, including the series size, inconsistent follow-up and incomplete medical records. Other limitations include the lack of CT, which is the gold standard to exclude PSS, lack of ammonia levels, and the potential influence of concurrent conditions and treatments affecting serial liver enzyme elevation. Continued monitoring with follow-up biochemistry, imaging and, when indicated, repeat biopsies may assist in assessing disease progression. Future prospective studies with a larger series size, standardized treatments and consistent follow-up are needed to evaluate therapeutic response, long-term outcomes and frequency of hepatic dysfunction in asymptomatic cats.
Conclusions
Primary PVH in cats is an uncommon condition but should be considered in cats with asymptomatic increased liver enzymes. Management with a combination of therapies shows success and a favorable prognosis. However, because of a lack of standardized treatment protocols, further prospective studies are needed to identify the most effective therapeutic protocols.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
