Abstract
Background:
Ischemia/reperfusion injury (IRI) causes cellular dysfunction and death in organs like the kidney, heart, and brain. It involves energy depletion during ischemia and oxidative stress, inflammation, and apoptosis during reperfusion. Kidney IRI often leads to acute kidney injury (AKI) in various clinical scenarios. The omentum, an adipose tissue with healing properties, has been used to treat injuries in different organs.
Objective:
This study aimed to assess the omentum’s healing effects on reducing IRI’s adverse effects after renal ischemia in Wistar rats.
Method:
A total number of 36 male Wistar rats were used in a study on IRI-induced AKI. Rats were divided into 6 groups of normal kidneys wrapped with omentum “Sham-1” and “Sham-2,” ischemic kidney wrapped with omentum as “OMT-1” and “OMT-2,” and ischemic kidney without omentum as “Control-1” and “Control-2.” Ischemia was induced by clamping the left renal artery for 45 minutes. The omentum was transposed onto the injured kidney in “OMT” group. After sacrifice at weeks 4 and 8, kidney histology and blood samples were analyzed for kidney function markers.
Results:
On the first day after surgery, there was an immediate increase in creatinine and blood urea nitrogen (BUN) levels, which then decreased by day 28. Both OMT groups showed significantly lower levels of creatinine and BUN compared to Control groups on day 1, but after 28 days differences were not statistically significant. Histological analysis using H&E and Masson’s trichrome staining revealed significantly higher levels of inflammatory cell infiltration and hyperemia in the OMT groups. However, fibrosis and glomerular shrinkage were higher in the Control groups.
Conclusion:
Using an omental flap significantly prevented fibrosis within the renal parenchyma, slow down the AKI progression, and potentially serving as a promising therapeutic strategy for kidney dysfunction.
Introduction
Ischemia/reperfusion injury (IRI) is defined as the paradoxical exacerbation of cellular dysfunction and death, following restoration of blood flow to ischemic organs. 1 Ischemia/reperfusion starts a cascade of deleterious cellular responses leading to cell death via necrosis or apoptosis. 2 Ischemia/reperfusion injury is a common mode of injury that affected multiple organs such as kidney, heart, and brain. 3 The injury process occurs within 2 stages: during ischemia, the main factor is cell energy depletion, secondly during reperfusion, the role of oxidative and microcirculatory stress, besides inflammation and apoptosis. 4 Multiple studies show the role of oxidative stress, inflammation, and apoptosis in the initiation and development of IRI. 5
Kidney IRI is a common cause of acute kidney injury (AKI), which occurs in various clinical settings such as administration of vasoconstrictive drugs or radio-contrast agents, sepsis, trauma, transplantation, myocardial infarction, and cardiac surgery, that all lead to circulatory shock and hypotension. There is a mismatch between oxygen supply and demand, causes a reduction in oxidative metabolism, with progressive injury and death of tubular epithelial cells. 6 Acute kidney injury is a complex pathological situation mostly involving hemodynamic, inflammatory, and direct toxic effects at the cellular level, leading to morbidity and mortality.7,8 Ischemia/reperfusion injury–induced AKI is associated with changes in hemodynamics and dysfunction of endothelial cells owing to high levels of reactive oxygen species (ROS) and reactive nitrogen species that lead to decreased production of nitrogen oxide and intracellular energy store exhaustion. 9
The omentum is an adipose tissue attached to the greater curvature of the stomach, containing immune cells called “milky spots.” The omentum has the innate characteristic of adhering to the injured sites in the abdomen.10,11 Additionally, the omentum is a mobile organ that covers the peritoneal cavity and deals with infections and contaminants. It also has a role in controlling inflammation, inducing revascularization and tissue regeneration. Commonly, the omentum is wrapped around infection sites to avoid spreading to healthy sites. Furthermore, the omentum has a characteristic feature to pursue and contain the injury site. This phenomenon produces many immunomodulatory cells alongside the proliferating cells having stem cell properties, in a process called omentum activation.12,13 Omentum is an available source of adult stem cells that could be used to repair and regenerate injured tissue. 14 Omentum has been known for its various functions in promoting revascularization, reconstruction, and tissue regeneration. The activated omentum secretes angiogenic and various growth factors that promote the formation of new blood and lymphatic vessels, which subsequently connect with the vascular structures of the injured organ. This process aids in the removal of tissue debris and coagulated blood, while also ensuring an adequate blood supply that mitigates ischemic damage to the affected tissue. 15 Many studies show its potential revascularization effect on ischemic tissues such as the brain, myocardium, and extremities. Omentum has been also utilized in supporting neural regeneration on the transected spinal cord in both animal and human research.16-21 After the activation, the omentum size increases enormously (up to 20-fold) by producing new tissue consisting of stromal cells, interstitial cells, and blood vessels. The stromal cells of expanded omentum express markers of stem cell activity such as fibroblast growth factor (FGF) and vascular endothelial growth factor (VEGF) and express adult stem cell markers including SDF-1α, CXC motif chemokine receptor 4 (CXCR4), WT-1, as well as pluripotent embryonic stem cell markers, such as Nanog, Oct-4, and SSEA-1. 22
In this study, we try to evaluate the healing effect of omentum on diminishing adverse effects of renal IRI in Wistar rat model.
Materials and Methods
This research was conducted in the Animal Laboratory of Urology Research Center of Tehran University of Medical Science, Tehran, Iran, between November 2023 and March 2024. The study was approved by the Ethics Research Committee of Tehran University of Medical Sciences (IR.TUMS.AEC.1401.191). Ethical principles in animal studies were considered in all phases of the research. Thirty-six male Wistar rats (Pasteur Institute, Tehran, Iran), aged 8 to 10 weeks and weighing 210 ± 20 g, were used in the rat model of IRI. The rats were randomly divided into 6 groups, “ischemic kidney wrapped with omentum, euthanized after 4 weeks” namely OMT-1 group (n = 6), “ischemic kidney wrapped with omentum, euthanized after 8 weeks” namely OMT-2 group (n = 6), “ischemic kidney without omentum, euthanized after 4 weeks” namely Control-1 group (n = 6), “ischemic kidney without omentum, euthanized after 8 weeks” namely Control-2 group (n = 6), “normal kidney wrapped with omentum, euthanized after 4 weeks” namely Sham-1 group (n = 6), and “normal kidney wrapped with omentum, euthanized after 8 weeks” namely Sham-2 group (n = 6). All rats were acclimatized for 7 days. After acclimatization, rats were anesthetized with injection of ketamine 10% (70 mg/kg) and xylazine 2% (10 mg/kg) intraperitoneally. In “Control” and “OMT” groups, rats underwent laparotomies with the midline abdominal incision. After dissecting renal pedicles, non-traumatic vascular clamps (Micro-serrefine curved 6 mm, Fine Science Tools Inc, Foster, California, USA) were applied to left renal artery for 45 minutes to achieve ischemia of kidney. The right kidney in all rats was preserved. Rats were maintained under anesthesia and placed on heating pads during ischemia. The color change of the kidney from red to dark purple indicates the ischemia. Reperfusion started after removing the clamps and was observed by the return of color to red. In the “OMT” group, transposition of the omentum was performed without primary sutures on the injured kidney. To mobilize the omentum, 5 ml of polydextran particle slurry (1:1 in sodium chloride solution) was added into the abdominal cavity. 23 Transection of the omental flap started from the hepatic flexure of the colon with preservation of blood supply from the base of the flap. Considering the kidney size, an appropriate length of omentum was transected to reach the kidney without any tension. The incision was closed in 2 layers by 3-0 polyglycolic acid interrupted sutures. In the “Control” group, the omentum was completely resected to prevent the omentum from fusing to the injured kidney. Sham operation was done by laparotomy midline incision (previously described) and transposition of the omentum to the normal kidney. All rats were kept in standardized laboratory conditions, controlled light (day/night cycle of 12 h/12 h), fed standardized rodent food, and given filtered water. The average temperature of the room was 25 ± 2°C with 55 ± 5% humidity. Rats were euthanized at weeks 4 and 8 after inducing IRI. At euthanasia, kidney was resected for histologic examination.
Renal Function Measurement
Creatinine and blood urea nitrogen (BUN) were measured before the procedure, 1st day, 14th day, and 28th day after the procedure. Colorimetric Jaffe reaction (Sigma Diagnostics, Inc, St Louis, Missouri, USA) was used to measure the creatinine, and improved colorimetric Jung method (BioChain Institute, Inc, Hayward, California, USA) calculated the BUN from the measurement of urea. Serum samples were collected and stored at −80°C until the analysis.
Tissue Sampling and Histopathological Examination
After sacrifice, the kidney tissue was fixed in 10% phosphate-buffered formalin. Several grades of ethanol were used for dehydration, the tissues were embedded in paraffin, and serial sections were obtained from the blocks with a thickness of 5 µm. Then, the tissue sections were mounted on slides, and staining (H&E) was performed. An expert pathologist scores each section in several random fields blindly using a light microscope. The histopathological parameters and quantifying of tissue damages included: “hyperemia (congestion in medullary blood vessels), enlargement of Bowman’s space, shrinkage of glomerular tuft, and infiltration of inflammatory cells.” Scoring was performed using a 0 to 5 scale: normal (score 0); up to 20% (score 1), 21% to 40% (score 2), 41% to 60% (score 3), 61% to 80% (score 4), and more than 80% (score 5). Each group’s average of all sections indicates a “total histopathological score.” 24
For Masson’s trichrome staining, the tissues were fixed in 10% neutral-buffered formalin for 24 to 48 hours, then dehydrated through a series of graded alcohols. The tissues were embedded in a paraffin block. Sections were taken with the 5-µm slices mounted on the slides, and deparaffinized with xylene. Staining was done with Weigert’s iron hematoxylin, Biebrich scarlet-acid fuchsin, and aniline blue solution. Finally, sections were dehydrated through a series of graded alcohols and cleared in xylene. Scoring was performed with the same method used for H&E staining. 25
Statistical Analysis
Analysis was conducted using GraphPad Prism 9 (GraphPad Software, CA, USA). The descriptive variables were summarized using frequencies and percentages, while continuous variables were described with either medians and interquartile ranges for non-normal distributions or means and standard deviations for normal distributions. Statistical comparisons were made using the Kruskal-Wallis and Mann-Whitney U tests for skewed data (after multi-comparison analysis significance level was considered as .017). Analysis of variance test was used for normally distributed data, with post hoc tests conducted using Tukey’s range test.
Results
Renal Function Parameters
The rats that were euthanized on days 28 and 56 were analyzed separately. Considering the rats euthanized on day 28, no significant differences in creatinine levels were observed between the groups on the preoperative day. However, on postoperative day 1, there was a significant increase in creatinine levels in the Control-1 and OMT-1 groups compared to the preoperative day as shown in Table 1 (P values <.0001). Additionally, the creatinine level in Control-1 was significantly higher than in OMT-1 (P value <.0001). Subsequently, on day 28, there was a significant decrease in creatinine levels in both Control-1 and OMT-1 groups compared to day 1 (P values <.0001). No differences were found between Control-1 and OMT-1 group in terms of creatinine levels on day 28 (P value = .982), with creatinine levels on day 28 not statistically different from preoperative levels in the Control-1 and OMT-1 groups (P values = .360 and .999, respectively).
Comparison of Creatinine and BUN Level Between Time Points and Groups in Rats That Were Euthanized After 4 Weeks.

Sham-1 vs Control-1.
Sham-1 vs OMT-1.
Control-1 vs OMT-1.
P value <.05 compared to preoperative.
P value <.05 compared to day 1.
While comparing BUN level, no statistically significant differences were observed between the groups on the preoperative day (Table 1). However, on the first day after surgery, there was a notable increase in BUN levels in both the Control-1 and OMT-1 groups compared to the preoperative day (P values <.0001), with Control-1 showing significantly higher BUN levels than OMT-1 (P-value <.0001). Subsequently, by day 28, there was a significant decrease in BUN levels in both Control-1 and OMT-1 compared to day 1 (P value <.0001). There were no discernible differences between Control-1 and OMT-1 in terms of BUN levels on day 28 (P value = .346), and BUN levels on day 28 were not statistically different from preoperative levels in either group (P values = .988 and .967, respectively). Creatinine and BUN levels trend during the study period have been shown in Figure 1.
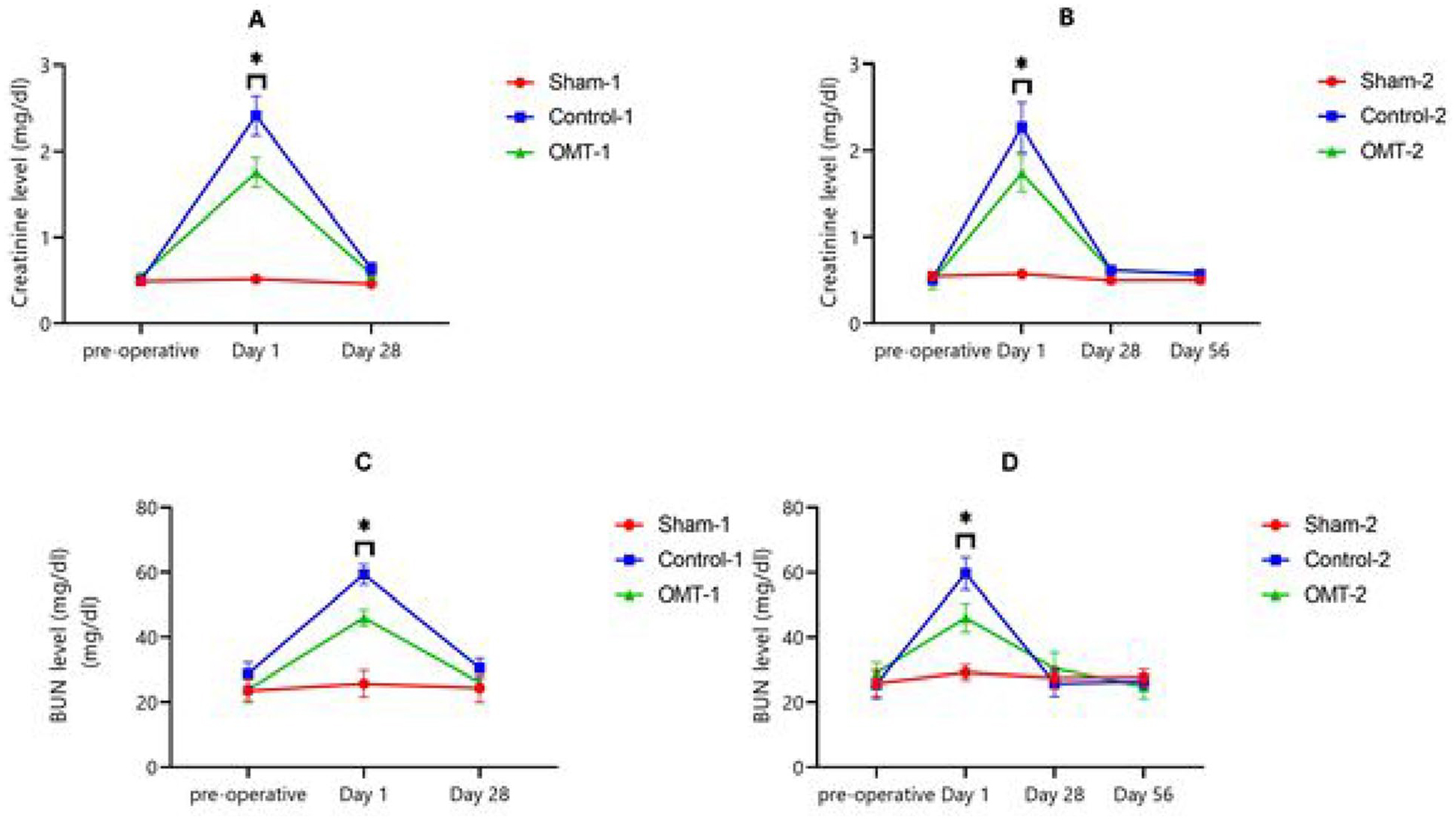
Creatinine and BUN levels during the study period. (A) Creatinine trend in 4 weeks. (B) Creatinine trend in 8 weeks. (C) BUN trend in 4 weeks. (D) BUN trend in 8 weeks.
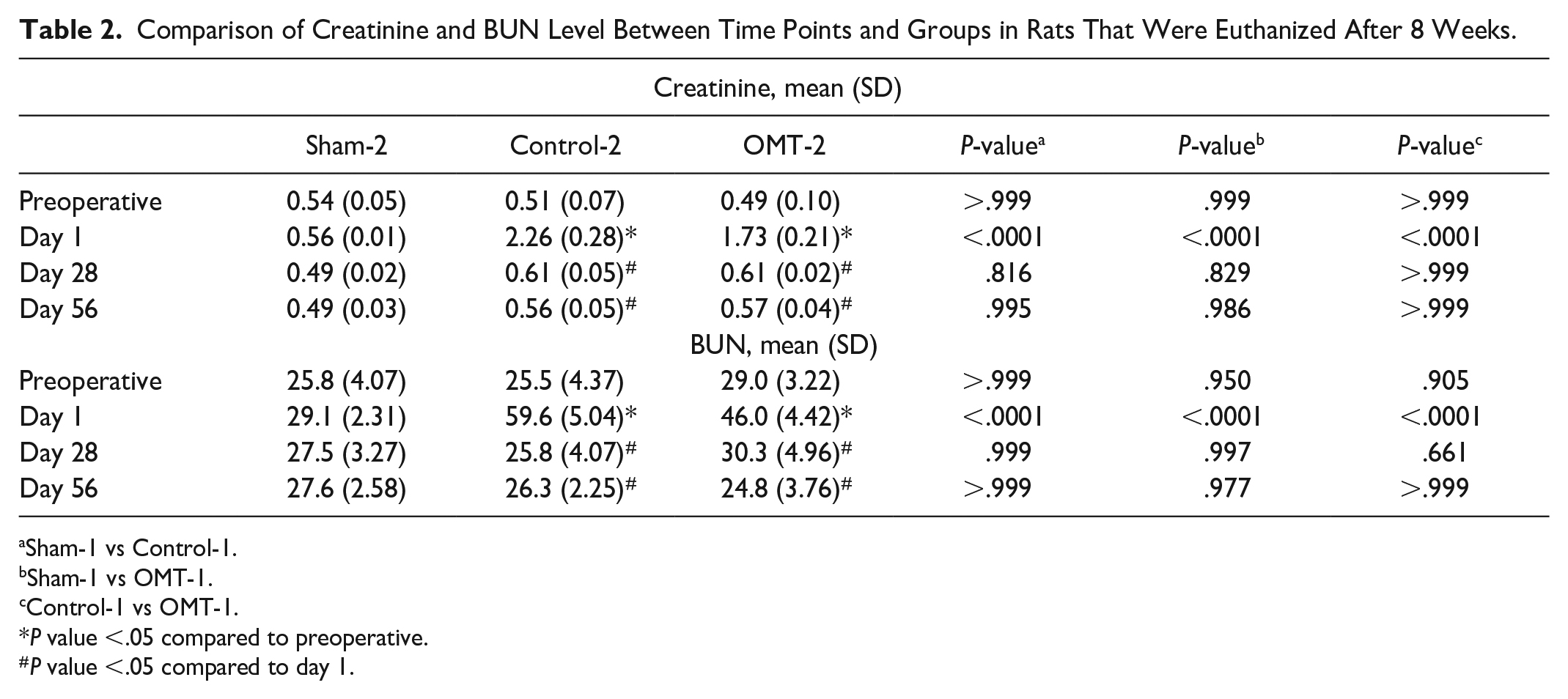
Similar results were observed in the rats euthanized on day 56. On postoperative day 1, there was a significant increase in creatinine levels in the Control-2 and OMT-2 groups compared to the preoperative day (P value <.0001), with Control-2 showing significantly higher creatinine levels than OMT-2 (P value <.0001). Subsequently, on day 28, there was a significant decrease in creatinine levels in both Control-2 and OMT-2 groups compared to day 1 (P value <.0001). No differences were observed between Control-2 and OMT-2 group in terms of creatinine levels on day 28 (P value >.999), with creatinine levels on day 28 not statistically different from preoperative levels in the Control-2 and OMT-2 groups (P values = .938 and .816, respectively). Furthermore, the results on day 56 mirrored those on day 28, with no significant differences in creatinine levels between the 2 time points as demonstrated in Table 2.
Comparison of Creatinine and BUN Level Between Time Points and Groups in Rats That Were Euthanized After 8 Weeks.
Sham-1 vs Control-1.
Sham-1 vs OMT-1.
Control-1 vs OMT-1.
P value <.05 compared to preoperative.
P value <.05 compared to day 1.
Similarly, on the first day after surgery, there was a significant increase in BUN levels in both the Control-2 and OMT-2 groups compared to the preoperative day (P value <.0001), with Control-2 exhibiting significantly higher BUN levels than OMT-2 (P value <.0001). Subsequently, by day 28, there was a significant decrease in BUN levels in both Control-2 and OMT-2 groups compared to day 1 (P value <.0001). No differences were observed between Control-2 and OMT-2 group in terms of BUN levels on day 28 (P value >.999), and BUN levels on day 28 were not statistically different from preoperative levels in either group (P value >.999). Furthermore, the results on day 56 were consistent with those on day 28, with no significant differences in BUN levels between the 2 time points as indicated in Table 2.
Histopathological Findings
Hyperemia, enlargement of Bowman’s space, shrinkage of glomerular tuft, and infiltration of inflammatory cells were assessed by H&E staining (Figure 2). Glomerular shrinkage was significantly higher in Control-1 versus OMT-1 on day 28 (Table 3). Although the shrinkage was higher in Control-2, it was not statistically significant on day 56 (Table 4). While hyperemia was higher in OMT-1 on day 28 and in OMT-2 on day 56, the difference was significant only on day 28. Infiltration of inflammatory cells was higher in both OMT-1 and OMT-2 groups with a significant difference only in the OMT-2 group. Bowman’s space was increased in both Control-1 and OMT-1 groups on day 28. Although the increase was slightly more in OMT-1, the differences were insignificant. Considering the rats that were euthanized after 8 weeks, Bowman’s space increased significantly in OMT-2. Masson’s trichrome staining was performed to assess renal fibrosis (Figure 3). Fibrosis was significantly higher in Control-1 versus OMT-1 on day 28. The same result was observed in the rats that were euthanized after 8 weeks.

Histopathological sections of rat kidney, H&E staining. The thin arrow shows the glomerular shrinkage of the tuft. A thick arrow indicates hyperemia. The black star demonstrates the inflammatory cell infiltration and the red one shows the increase of Bowman’s space.
Comparison of the Histopathological Findings in Rats That Were Euthanized After 4 Weeks (H&E and Masson’s Trichrome).

Kruskal-Wallis test.
P value <.017 comparing Sham-1 vs Control-1.
P value <.017 comparing Sham-1 vs OMT-1.
P value <.017 comparing Control-1 vs OMT-1.
Comparison of the Histopathological Findings in Rats That Were Euthanized After 8 Weeks (H&E and Masson’s Trichrome).

Kruskal-Wallis test.
P value <.017 comparing Sham-2 vs Control-2.
P value <.017 comparing Sham-2 vs OMT-2.
P value <.017 comparing Control-2 vs OMT-2.
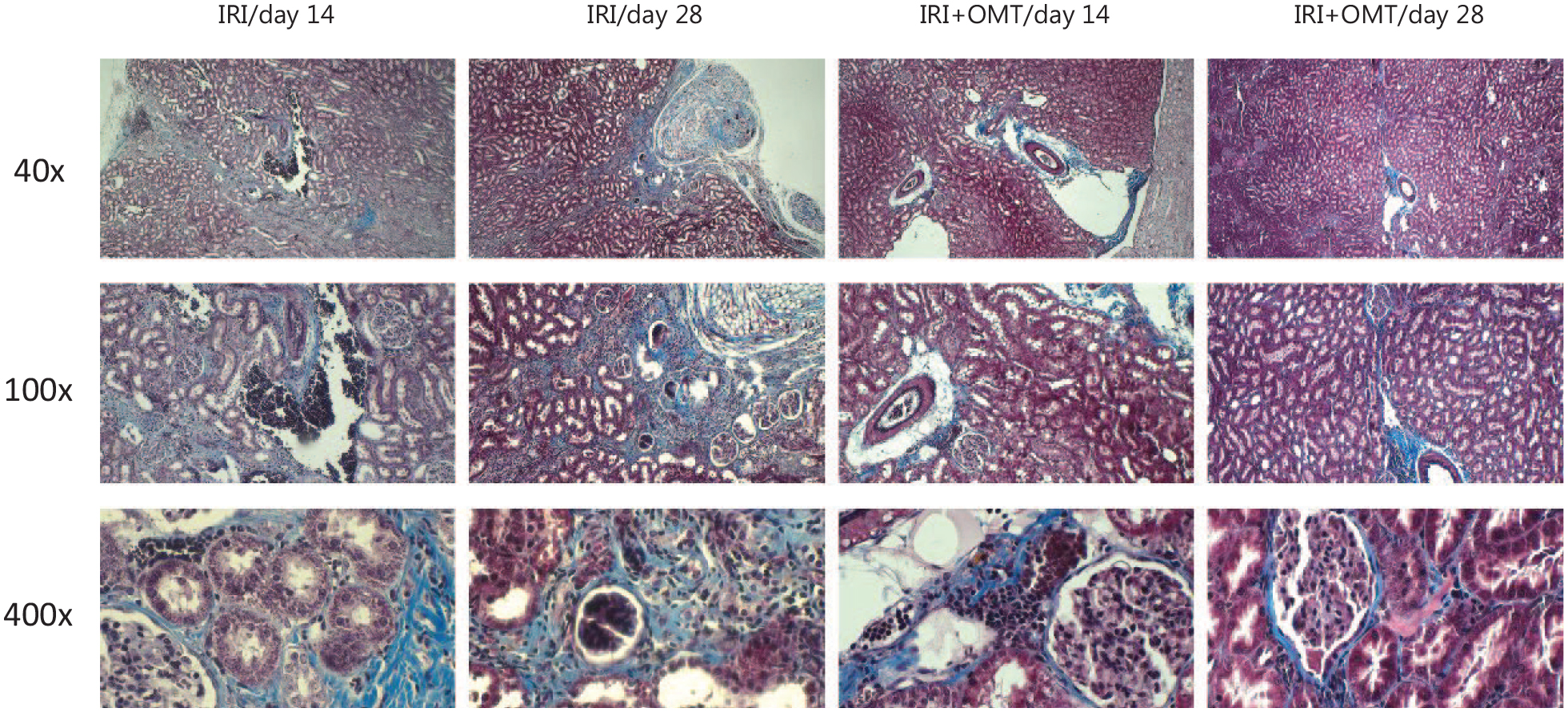
Histopathological sections of rat kidney, Masson’s trichrome staining. Blue stains show renal sclerosis.
Discussion
The etiology of AKI is primarily associated with a reduction in renal perfusion, leading to renal hypoxia. Subsequent elevation of creatinine, BUN rise, and decline in glomerular filtration rate are significant complications of this situation. Reversing the ischemia may result in adverse outcomes such as necrosis and cellular swelling, known as IRI. Renal dysfunction due to IRI has been observed in various clinical scenarios, including cardiac and aortic surgeries, trauma, shock, burns, and organ transplantation. Clinical management necessitates dealing with reperfusion when tissue ischemia occurs. Despite the established correlation between the severity of damage and ischemia duration, prompt reperfusion is not always feasible. Extensive research efforts have been dedicated to elucidating the mechanisms underlying IRI; however, consensus on the precise pathophysiology remains elusive.4,26,27 Several pharmacological agents have been employed to mitigate IRI complications. Urocortin, a peptide from the corticotropin-releasing factor family, exhibits notable cardioprotective effects post-myocardial IRI by modulating various signaling pathways. Polyethylene glycols and exogenous adenosine also demonstrate therapeutic potential in ameliorating IRI by protecting mitochondria, cytoskeleton integrity, and enhancing Akt/eNOs engagement, respectively.28-30 Excessive production of ROS post-IRI induces oxidative stress, leading to cellular damage through diverse mechanisms. Antioxidants and free radical scavengers have shown promise in mitigating IRI-induced oxidative stress. Agents like ulinastatin, propofol, and certain herbal products with antioxidative properties, such as Picrorhiza kurroa, naringin, and aqueous garlic extract, have been implicated in reducing IRI complications. 31 Activation of the renin-angiotensin system is identified as a risk factor exacerbating IRI, with angiotensin 2 exerting adverse effects through renal vasoconstriction, increased vascular sensitivity to sympathetic stimulation, and apoptosis induction. Prior studies suggest that angiotensin-converting enzyme inhibitors and angiotensin receptor blockers can confer renoprotective effects against IRI-related complications. 32
Currently, mesenchymal stem cells (MSCs) have garnered considerable attention in the field of tissue repair and regenerative medicine due to their inherent self-renewal capabilities. The therapeutic potential of MSCs has prompted researchers to investigate the efficacy of MSC-based interventions in IRI. Homing capacity of MSCs in damaged tissues is attributed to its therapeutic effects. Homing ability, beyond anti-inflammatory and vascular-supportive roles, is crucial for MSCs to effectively localize within damaged organs and enhances the paracrine functions and contributes to the preservation of microvessel density. Signals from injured renal cells are detected by transplanted MSCs, which leads to their chemoattraction to the affected area. The CXCR4 serves as a specific receptor for the chemokine stromal cell–derived factor-1 (CXCL12), with CXCR4-expressing cells playing a pivotal role in renal repair during AKI. The localization of damaged tissues by MSCs is mediated by stromal cell–derived factor-1, which is significantly upregulated during AKI and promotes the homing of CXCR4-expressing cells to the injured renal tissue.33-36 In AKI, there are notable alterations in the levels of VEGF, hepatocyte growth factor (HGF), epidermal growth factor, insulin-like growth factor-1 (IGF-1), and transforming growth factor-beta (TGF-β), all of which contribute to the apoptosis of endothelial cells. These growth factors are also recognized as critical elements in cellular regeneration and renal repair processes. MSCs release various bioactive molecules directly or through extracellular vesicles which include VEGF, HGF, IGF-1, interleukin-10, FGF, and TGF-α. Additionally, MSCs downregulate the expression of pro-inflammatory cytokines such as interleukin-1 beta and tumor necrosis factor-alpha, thereby exerting anti-inflammatory and anti-apoptotic effects that facilitate renal repair.37-39 In a notable study by Mathew et al, extracellular vesicles derived from MSCs were utilized to mitigate retinal IRI. These extracellular vesicles were isolated from human MSCs and administered into the vitreous humor of a rat model 24 hours post-retinal ischemia. Results indicated enhanced functional recovery and a significant reduction in apoptosis. 40 Another study by Kim et al evaluated post-IRI renal function following the administration of adipose-derived stem cell–derived conditioned medium (ADSC-CM) in a rat model. By forming 3-dimensional spheroids from ADSCs and collecting conditioned medium after 48 hours of incubation, the researchers observed significantly lower levels of BUN and creatinine in the ADSC-CM group, along with reduced collagen content, apoptosis, increased proliferation, and higher antioxidant levels in both the medulla and cortex. 41
Mesenchymal stem cells are present in various tissues such as adipose tissue, peripheral blood, and bone marrow. The omentum, a vascular structure rich in growth factors and MSCs, has been recognized for its healing capabilities attributed to its migratory properties toward sites of injury. 42 In this study, male Wistar rats were utilized to assess the impact of omental flap on IRI. It is noteworthy to mention that female rats are often excluded from clinical trials due to the complexities of the menstrual cycle, which leads to variable hormone levels and complicates data interpretation. While male sexes have similar sex hormones, female hormones fluctuate cyclically. Females generally have a protective advantage in renal diseases, attributed to estrogen and progesterone. Additionally, outcomes in AKI are usually more favorable in females. To reduce hormonal variability and create a consistent model of renal injury, this study used male rats. 43 Moreover, a unilateral renal ischemia approach was performed in this study. Ischemia/reperfusion injury can be induced through unilateral or bilateral clamping of the renal arteries. Shorter ischemic durations associated with bilateral clamping do not allow strict control of renal damage that may be too mild or too severe. In contrast, unilateral clamping allows for extended ischemic periods that allow us to study a larger range of severity of renal injury and facilitate the investigation of AKI following a prolonged post-IRI phase. Furthermore, unilateral clamping preserves an intact kidney post-surgery, significantly diminishing the risk of mortality due to renal failure. Consequently, unilateral IRI is often preferred in experimental settings.44-46
In a study by Fay et al, 47 autologous omentum was transposed as a flap in a rat model of spinal cord injury, resulting in significant preservation of nerve fibers after 16 weeks. Di Taranto et al 48 utilized omental flap (harvested via laparoscopy) reconstruction to treat lower limb ulcers in lymphedema patients, leading to reduced ulcer size and improved lymphatic drainage (confirmed by lymphoscintigraphy). Additionally, Paglione et al investigated the impact of omental MSC infusion on IRI in a rat model. The MSC was injected intraparenchymal at reperfusion stage. Findings showed significantly lower levels of creatinine and BUN in the omental MSC group. 49 In a study by Garcia-Gomez et al, rats underwent right total nephrectomy and left 5/6 nephrectomy in order to induce chronic kidney disease (CKD). Transpositioning of the omentum was performed on the remaining left kidney to assess its potential role on slowing the progression of CKD. Creatinine and BUN were significantly lower in group receiving omentum. Finding also showed that the differences were higher in week 12 versus week 6. In H&E staining, hyperemia (seen in experimental kidneys) demonstrated that the injured kidney is well perfused while the lack of red blood cells in the Control group suggested poor perfusion. Inflammatory cells were not infiltrated in both experimental and control tissues. Masson’s trichrome staining also was carried out to distinguish renal fibrosis. This test showed fibrosis 2 weeks after 5/6 nephrectomy and experimental kidneys had significantly less fibrosis. 50
In this study, creatinine and BUN levels rose immediately after ischemia/reperfusion in both Control and both OMT groups on postoperative day 1 and gradually declined until postoperative day 28, almost near the normal range. Creatinine and BUN levels were significantly higher in both Control groups comparing to OMT groups in the first postoperative day. Creatinine and BUN levels in day 28 were not statistically different, and the same findings were observed in day 56. These findings revealed that omentum flap can slow the progression of AKI. In our study, renal function parameters came back to the normal range after 28 days. Our damage to the kidney with ischemia/reperfusion might be not as severe (as 5/6 nephrectomy) to distinguish the potential effect of omentum. This finding also can be associated to the impact of the other normal kidney that compensated ischemic kidney. For better evaluation of omentum ability, more severe damage with bilateral ischemia/reperfusion may be helpful. Another option for the measurement of accurate renal parameters without the interference of the healthy kidney is ureteral catheterization in the experimental kidney.
Fibrosis is a pathological phenomenon characterized by connective tissue deposition, and affected soft tissue in different organs including the heart, liver, and lungs due to many conditions. Accumulation of fibroblasts and collagens plays a key role in the fibrosis process. 51 Renal fibrosis is a progressive process resulting in end-stage renal disease, a critical condition that necessitates dialysis or kidney transplantation. Renal fibrosis is a consequence of the wound-healing process after renal trauma. 24 In a study by Jo et al, a 3-dimensional bio-printed omentum patch was used in unilateral ureteral obstruction in rat model. After 14 days, histopathological staining demonstrated significant alteration in fibrosis. The omentum showed antifibrosis characteristic in unilateral ureteral obstruction. 52 Endothelial dysfunction is one of many factors responsible for renal fibrosis. In a cross-sectional study by Noishiki et al, reactive hyperemia index (RHI) was evaluated in keloid (the result of excessive fibrosis) and non-keloid patients. This study aimed to assess the effect of endothelial dysfunction by using RHI on excessive fibrosis occurring in the keloid formation process. On logistic regression, they found poor RHI values associated with keloid development. 53
In our study, fibrosis was observed in both groups after 28 days. The severity of fibrosis was not changed in day 56. However, our findings demonstrated that the application of omental flap exhibited a significant preventive effect on fibrosis within the renal parenchyma. Histological analysis using H&E staining indicated a notable increase in hyperemia (especially in the first month) and infiltration of inflammatory cells (significantly on the second month) in OMT groups. As literature review showed, adequate blood flow is necessary for prevention from fibrosis and can be associated with hyperemia.
Some degree of inflammation was always presented in damaged tissue. However, inflammation is a double-edged sword, sometimes resulting in tissue regeneration and sometimes leading to fibrosis. 54 Our study findings show that infiltration of inflammatory cell is significantly higher in OMT groups (especially after 8 weeks). Further animal studies should perform to understanding the exact inflammatory cells infiltrated in renal tissue after transpositioning the omentum and finding the molecular pathways involved in preventing the fibrosis.
To evaluate the effect of omentum in AKI progression after IRI more precisely, it would be beneficial to assess creatinine and BUN levels during the first post-IRI week. However, this was one of our study limitations. Further studies should be carried out to closely monitor creatinine and BUN levels in the first week after IRI.
Conclusion
Overall, the incorporation of an omental patch stands as a promising and innovative therapeutic strategy for preserving renal function. In light of these findings, numerous studies and clinical trials are underway to explore novel treatment modalities for kidney diseases.
Footnotes
Acknowledgements
The authors specially thank the Sina hospital, Tehran University of Medical Sciences, Tehran, Iran.
Ethics Approval
This research was conducted in the Animal Laboratory of Urology Research Center of Tehran University of Medical Science, Tehran, Iran. The study was approved by the Ethics Research Committee of Tehran University of Medical Sciences (IR.TUMS.AEC.1401.191). Ethical principles in animal studies were considered in all phases of the research.
Consent to Participate
Not applicable.
Consent for Publication
All authors agree to publication.
Availability of Data and Materials
Data will be provided on request.
Author Contributions
Amirreza Shamshirgaran: Writing original draft; Abdolreza Mohammadi, Leonardo Oliveira Reis, and Fateme Guitynavard: Editing and reviewing; Parisa Zahmatkesh and Milad Akbarzadeh Moallem Kolaei: Ruining animal lab; Gholamreza Mesbah: Pathology; Alireza Khajavi: Data analysis; Seyed Mohammad Kazem Aghamir: Conceptualization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
