Abstract
Purpose of program:
Different models exist to guide successful implementation of electronic health tools into clinical practice. The Contrast Reducing Injury Sustained by Kidneys (Contrast RISK) initiative introduced an electronic decision support tool with physician audit and feedback into all of the cardiac catheterization facilities in Alberta, Canada, with the goal of preventing contrast-associated acute kidney injury (CA-AKI) following coronary angiography and intervention. This report describes the change management approaches used by the initiative and end-user’s feedback on these processes.
Sources of information and methods:
The Canada Health Infoway Change Management model was used to address 6 activities relevant to project implementation: governance and leadership, stakeholder engagement, communications, workflow analysis and integration, training and education, and monitoring and evaluation. Health care providers and invasive cardiologists from all sites completed preimplementation, usability, and postimplementation surveys to assess integration and change success.
Key findings:
Prior to implementation, 67% of health providers were less than satisfied with processes to determine appropriate contrast dye volumes, 47% were less than satisfied with processes for administering adequate intravenous fluids, and 68% were less than satisfied with processes to ensure follow-up of high-risk patients. 48% of invasive cardiologists were less than satisfied with preprocedural identification of patients at risk of acute kidney injury (AKI). Following implementation, there were significant increases among health providers in the odds of satisfaction with processes for identifying those at high risk of AKI (odds ratio [OR] 3.01, 95% confidence interval [CI] 1.36-6.66, P = .007), quantifying the appropriate level of contrast dye for each patient (OR 6.98, 95% CI 3.06-15.91, P < .001), determining the optimal amount of IV fluid for each patient (OR 1.86, 95% CI 0.88-3.91, P = .102), and following up of kidney function of high risk patients (OR 5.49, 95%CI 2.45-12.30, P < .001). There were also significant increases among physicians in the odds of satisfaction with processes for identifying those at high risk of AKI (OR 19.53, 95% CI 3.21-118.76, P = .001), quantifying the appropriate level of contrast dye for each patient (OR 26.35, 95% CI 4.28-162.27, P < .001), and for following-up kidney function of high-risk patients (OR 7.72, 95% CI 1.62-36.84.30, P = .010). Eighty-nine percent of staff perceived the initiative as being successful in changing clinical practices to reduce the risk of CA-AKI. Physicians uniformly agreed that the system was well-integrated into existing workflows, while 42% of health providers also agreed.
Implications:
The Canada Health Infoway Change Management model was an effective framework for guiding implementation of an electronic decision support tool and audit and feedback intervention to improve processes for AKI prevention within cardiac catheterization units.
Purpose
More than 14 000 people in Alberta, Canada, undergo coronary angiography or percutaneous coronary angiography (PCI) each year to diagnose or treat coronary artery disease. Historically, almost one in ten develop contrast-associated acute kidney injury (CA-AKI) as a procedural complication.1-4 However, CA-AKI is preventable: institutions that have implemented evidence-based preventative strategies have achieved substantial reductions in CA-AKI incidence with rates that approach 4%. Inconsistent uptake of risk mitigation measures, such as preprocedural identification of at-risk patients,5-8 minimization of iodinated radiocontrast media exposure,9-12 and periprocedural intravenous hydration,13,14 is well documented.1,15,16
The
This article describes the implementation of the Contrast RISK decision-support tool within Alberta, guided by the Canada Health Infoway Change Management model, and highlights relevant factors governing successful health technology implementation and evaluation.
Methods
Health Infoway Change Management Model
We considered the following in selecting a model: (1) the purpose of the framework; (2) the levels included within the framework (eg, provider, organization, and system); (3) the degree of inclusion, depth of analysis, and operationalization of implementation concepts (eg, process, determinants [barriers and facilitators], strategies, and evaluation); and (4) the framework’s orientation, which includes the setting and type of intervention. Based on these factors, we chose to utilize the Health Infoway Change Management Framework. Canada Health Infoway is an independent, non-profit corporation founded by the Government of Canada that aims to improve Canadian health care systems and delivery of electronic health projects.18-20 Through Canada Health Infoway, the Pan-Canadian Change Management Network identified 6 key elements in the process of adopting electronic health tools in clinical practice: governance and leadership, stakeholder engagement, communications, workflow analysis and integration, training and education, and monitoring and evaluation. Our intent was to use this framework to manage anticipated barriers to implementation, assess change readiness, ensure meaningful engagement, create conditions that foster change, and target behavior change in the way prevention approaches for CA-AKI were used in the catheterization units.
Governance and Leadership
Strong governance and leadership were identified as crucial in establishing project priorities and providing direction to support consistent and appropriate project progress. 21 In this respect, we addressed leadership on 2 levels.
First, prior to project initiation, a research team was created, as well as a formal project charter that outlined key project elements and established predetermined objectives. Then, a steering committee was formed, which included: the principal investigators; physician site-leaders recruited from each catheterization unit; technology/platform experts to support the development and integration of the electronic decision-support tool; and designated research coordinators. The committee was tasked with implementing the initiative and encouraging collective engagement and ownership. Quality improvement, project skills, and prior experience were not prerequisites in selecting physician leaders, and participants received no funding to undertake leadership roles or attend project meetings.
Second, physician site-leaders, unit managers, and unit educators who already provided relevant day-to-day leadership in the catheterization laboratories designated resources from their teams to assist with the implementation of the initiative. In addition, these leaders reviewed and revised site-specific protocols for implementation, and interacted with colleagues to answer questions, offer immediate guidance, and further generate support for the research initiative. This collective guidance and leadership were essential to meeting the research goals and effecting change. The 3-physician site-leads, unit managers, and unit educators remained committed to the project for more than 3 years. We did not provide additional funding to support the physician leaders though we did work with physicians who had leadership roles and nursing staff with quality improvement roles in each catheterization unit.
Stakeholder Engagement
Stakeholders were defined as the population included in the research initiative and were empowered to make decisions that were in line with that of research project priorities. 21 Alberta has 3 cardiac catheterization units (2 in the city of Edmonton and 1 in the city of Calgary). Stakeholders relevant to the project across these 3 sites included cardiologists, registered nurses, radiology technologists, physiological technicians, unit educators, and managers working within cardiac catheterization units. 22 However, the combination and composition of these providers varied depending on the specific catheterization unit. The decision support tool was designed for use by providers caring for inpatients or outpatients, undergoing procedures for acute coronary syndrome or stable coronary disease.
To better understand the intricacies of each site’s workflow and care provider dynamics, in-person meetings at all 3 cardiac catheterization sites were held prior to implementation. This informed appropriate mapping of implementation strategies for each site and established rapport with common leadership positions across all locations, who then disseminated information and guidance to the individual staff members who perform the daily operation objectives throughout project deployment. All stakeholders were consulted on updates to project progress and research decisions that would affect their workflow procedures, giving them an opportunity to voice concerns and provide suggestions. User advice was incorporated to ensure that stakeholders felt supported by the research team, approved of research project-related changes to work duties, and were comfortable utilizing new tools and processes.
Communications
Effective communication between the research team and all stakeholders ensured that health care staff were equipped with the information necessary to incorporate the most appropriate information related to Contrast RISK research protocol within clinical practice. 21
Both in-person and digital communication strategies were utilized. The project team met weekly throughout, but meetings with the steering committee were less frequent—closer to monthly (with more frequent meetings added as needed at the start of the project when implementation activities were most active) and every 3 months later in the project to coordinate preparation for scheduled audit and feedback sessions. These meetings provided a forum to solicit feedback from staff, relay relevant project information, and report on project progress to support continued behavior change.
A dedicated research coordinator was on site in the catheterization lab each Monday to Friday for 3 months at the start of implementation, then made weekly site visits to each site and these were ultimately reduced to visits every 3 months for the remainder of the project. During this time, the coordinator facilitated staff training and personalized the site workflow as well as fielding questions and trouble shooting. This engagement supported all stakeholders to feel a connection to the research. Digital communications were designed to reinforce information from the site-specific protocols and were made available via a project web site (https://cumming.ucalgary.ca/research/icdc/research/contrast-risk-project) including site-specific protocol (Supplemental Material 1), frequently asked questions document (Supplemental Material 2), video presentation, quarterly newsletters, and e-mail communiques, which ensured that stakeholders were capable of contacting research team members with more urgent matters outside of scheduled meeting hours and provided references that stakeholders could refer to at any time for relevant project information. 23
Workflow Analysis and Integration
Workflow analysis and integration assessed how day-to-day operations were conducted in each unit to identify areas of improvement and forecast the effects of change initiatives. 21 In doing so, the initiative was better able to integrate into existing work cultures and become sustainable fixtures in patient care.
All three cardiac catheterization units in Alberta utilized the Alberta Provincial Project for Outcomes Assessment in Coronary Heart Disease (APPROACH) clinical information system in routine patient care at the time of the project. 22 Therefore, the electronic decision-support tool was integrated into this existing clinical information system to eliminate the disruption associated with staff learning to utilize new programs, changing data entry workflows, and troubleshooting technology issues. We chose a model for the decision support tool that used variables that were already being input into the APPROACH system for data collection, which eased incorporation into existing data collection processes. The laboratory data used in the model were automatically populated in the fields, to minimize additional manual data entry. Details about how the tool could be accessed, used, and its incorporation into workflow are provided in Supplemental Material 1.
To support the human factor changes required to utilize the new risk model and decision support tools, the project team collaborated in-person with the site cardiologist lead, unit managers, clinical nursing educators, and other health providers to better understand the unique staff composition, responsibilities, and workflow processes at each cardiac catheterization laboratory. Site-specific procedural protocols that most seamlessly incorporated the new workflow elements to collect and enter the data required to execute risk models for the decision support tools, communicate information on safe contrast levels and recommended amounts of intravenous fluid, and ensure the follow-up creatinine testing required by the Contrast RISK project were then presented for approval by each location (Supplemental Material 1).
Training and Education
Training and education imparted relevant knowledge and skills to end-users that allowed them to adopt implemented research changes and utilize them appropriately in a clinical setting. 21
Education to generate buy-in for the change initiative was achieved through presentation of evidence reviews to stakeholders of the inconsistent uptake of CA-AKI risk-mitigation strategies in Alberta, highlighting the opportunity that the Contrast RISK initiative offered to improve patient outcomes. To guide staff on the new behaviors required to use the decision-support tool correctly, site-specific workflow protocols that explained which staff member was to perform what task and when to do so were developed, reviewed by stakeholders, and then revised as per their recommendations (Supplemental Material 1). Final verification of the protocols was completed in person with staff at each catheterization unit. Training was delivered to all staff prior to deployment of the electronic tool to review new workflow procedures. To maximize engagement, we provided several meetings scheduled at different times and different days in each catheterization unit to reach the most users as possible, as each individual’s availability differed based on the shift they were working, and a given day’s workload. Following completion of the introductory training sessions, research coordinators remained available on site initially daily for the first 3 months after introduction to guide staff in real time while catheterization procedures were occurring. This enabled users to ask questions, practice completing action prompts, and better understand the processes required to use all components of the Contrast RISK decision-support tool in routine clinical practice. As health care providers began to understand how to best use the decision-support tool, research team members were no longer required to be on-site for immediate assistance.
The focus of the education delivered to staff was to build the new processes to determine, communicate, and document risk into their workflow, as well as provide actionable information to physicians at the appropriate time. The physicians’ education focused on motivating behavior change to consider contrast volume and fluid strategies to reduce risk of AKI. Educational content was embedded in the audit and feedback reports provided to all physicians on a continuous basis to reinforce knowledge about the study initiative, functions of the decision support tools, and strategies to improve their clinical practice. We tailored the content of education sessions and resources based on the needs of the users and their role.
Monitoring and Evaluation
Monitoring and evaluation occurred throughout the course of the project to determine whether change initiatives were being implemented appropriately and effectively. 21
Routine, informal monitoring was conducted via in-person and digital communications with stakeholders to anecdotally determine the impact of research decisions and protocols on end-users. Procedural data measures relevant to the workflow changes introduced by the Contrast RISK initiative, including the number of patients with a model-calculated preprocedural risk estimation and documentation of contrast dye and IV fluids, were pulled monthly from existing APPROACH data servers for rapid assessment and feedback on the fidelity of implementation during the initial period of implementation at each site. We aimed for 90% of eligible patients to have these steps completed and documented in the APPROACH system before moving forward with the next steps of the project and were able to achieve and sustain these targets.
Once these initial implementation metrics were achieved, anonymized, aggregate data summaries were made available to stakeholders in bimonthly reports that highlighted improvements in practice and areas of opportunity to encourage sustained behavior changes. Each physician also received individualized audit and feedback reports quarterly, with summarized clinical measures of performance, including contrast dye volume, hydration rates, and CA-AKI outcomes. These measures were compared against those of colleagues at the same site and across the province. The physician audit and feedback system has been described in detail elsewhere. 24 Both formal and informal monitoring helped guide adjustments to work protocols, updates to the decision-support tool, and behavior recommendations.
Formative evaluations targeting all stakeholders were conducted at different points across the continuum of change to confirm if project goals were being achieved. The Conjoint Health Research Ethics Board (CHREB) in the University of Calgary provided ethics approval. All participants provided written informed consent. Prior to project commencement, health providers and invasive cardiologists completed surveys to assess their baseline satisfaction with CA-AKI prevention processes in their center and their perceptions about the potential benefits and challenges associated with implementing an electronic decision support tool for CA-AKI prevention. These results were used to inform project delivery strategies to try and preemptively address barriers to change. These surveys were then disseminated again upon project completion to assess changes in health care provider perceptions. Surveys to assess tool usability were also distributed 3 months after each round of physicians was stepped in and the decision support tool was implemented. The survey assessed tool usability, satisfaction with different aspects of the Contrast RISK initiative, and perceived impact on their behavior. General feedback was also collected with open-ended questions. Preimplementation and postimplementation responses to the survey questions were measured using 5-point Likert scales.
The patient experience related to the initiative was also evaluated with structured telephone interviews to measure satisfaction with information provided, knowledge of postprocedural instructions, and the transition of care to the community, which have been reported elsewhere. 25
The initiative’s impact on processes of care measures (adherence to preventative measures related to contrast volume and intravenous fluid administration), and clinical outcomes (CA-AKI incidence, and downstream kidney and cardiovascular events) was evaluated at the completion of the project, as has been reported elsewhere. 17
Statistical Analyses
Responses to preimplementation and postimplementation surveys were compared using ordinal logistic regression models that included an independent variable to determine preimplementation versus postimplementation phase collection. The parallel regression assumption was tested and met for all models, which indicated no violation of the proportional odds assumption (Brant test P > .05). Common odds ratios could thus be reported for responses to each of the questions on the basis that they could be considered generalizable to all possible cut points where ordinal responses (from the 5-point Likert scale) could be dichotomized (eg, 1 vs 2, 3, 4, or 5; 1 or 2 vs 3, 4, or 5; 1, 2, or 3 vs 4 or 5; 1, 2, 3, or 4 vs 5). Statistical analyses were performed using Stata Statistical Software: Release 13 (College Station, TX: StataCorp LP).
Key Findings
Preimplementation Surveys
Characteristics of participants
Fifty-five allied health professionals completed the preimplementation survey. The majority (67%) were registered nurses, 80% were female, 66% had worked in their profession for over 10 years, and 65% had more than 5 years of work experience in the cardiac catheterization unit (Figure 1).
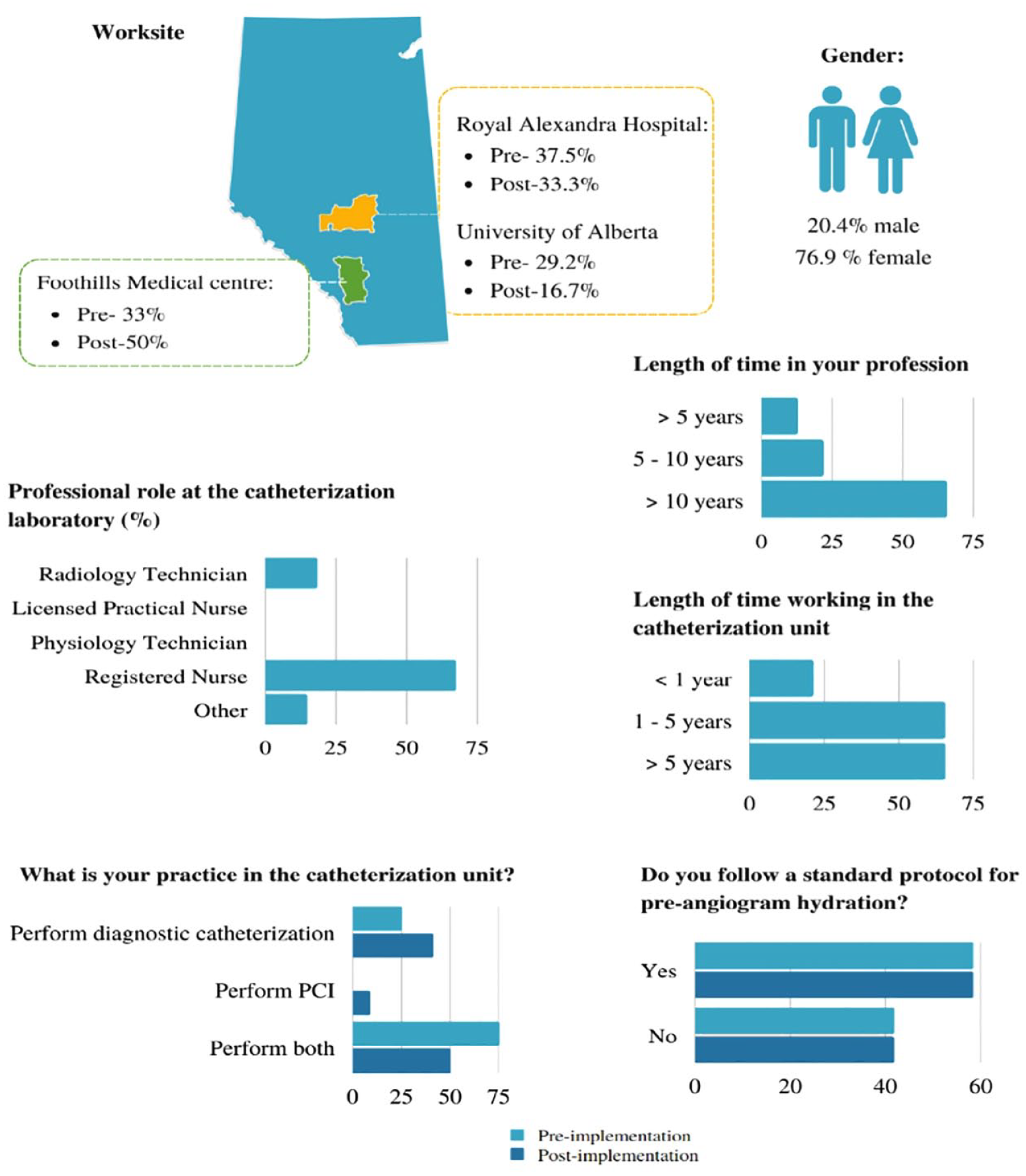
Health care provider survey respondent characteristics.
Of a total of 34 invasive cardiologists practicing in the province of Alberta at the start of the study, 29 (85%) completed the preimplementation survey (Figure 2). Physician respondents were equally distributed across Alberta’s 3 cardiac catheterization sites. Most physicians (75%) performed both catheterization and interventional procedures, but 6 (25%) performed only diagnostic catheterization (Table 1). All physicians indicated that CA-AKI risk was an important consideration when planning angiography. However, only 10 (42%) indicated that they used a formal risk scoring method to assess patient CA-AKI risk and 9 (38%) rarely used a method to determine a threshold safe contrast dye volume. Nine (38%) agreed that information on contrast volume would always influence their practice and 8 (33%) indicated they always ordered serum creatinine 48 to 72 hours postangiography in high-risk patients.

Health professionals’ and physicians’ satisfaction with catheterization unit care processes for prevention of acute kidney injury preimplementation and postimplementation of the initiative.
Physician Characteristics and Self-Reported Strategies Used for Prevention of Acute Kidney Injury Preimplementation and Postimplementation of the Initiative.

Note. All values represent n (%). PCI = percutaneous coronary angiography; AKI = acute kidney injury.
Not assessed in postimplementation survey since this was systematically implemented for all patients at increased risk of AKI following implementation of the initiative.
Preimplementation perspectives
Health provider and physician satisfaction with existing CA-AKI prevention processes prior to implementation of the Contrast RISK initiative are reported in Figure 2. Most health providers (67%) were satisfied with their ability to identify patients at high-risk of CA-AKI, but 67% felt neutral or dissatisfied with processes to quantify appropriate contrast dye volumes, 47% were neutral or dissatisfied with processes for appropriate intravenous fluid administration, and 68% were neutral or dissatisfied with postcatheterization follow-up protocols for high-risk patients. Unlike other health care professionals, only 11 (48%) physicians were satisfied with processes to identify patients at high-risk of CA-AKI and 7 (30%) were satisfied with processes to quantify the appropriate level of contrast media. Most physicians (61%) were satisfied with processes for IV fluid administration and 52% were satisfied with processes for follow-up of high-risk patients.
Most health providers (77%) believed that the project’s greatest benefit would be a reduction in CA-AKI incidence or an improvement in patient outcomes, whereas 42% anticipated that the greatest challenges would be physician adherence with recommendations and 33% identified the increased time required to input data to execute risk models could be a challenge.
Evaluation of education sessions
Thirty-eight health providers completed a written evaluation of the project education sessions. The majority of participants (63%) were registered nurses, 79% were female, 71% had worked in their profession for over 10 years, and 68% had more than 5 years of work experience specifically in the cardiac catheterization unit.
Written responses grouped by primary feedback theme indicated that the sessions were perceived to be informative with the appropriate format, length, and content. Some participants would have preferred a longer question and answer period along with distribution of the educational materials used during the session to participants immediately afterwards. Two respondents noted that the training may have been more useful if the cardiologists were also present at the same sessions. Nine participants thought that the greatest challenge with implementation of the initiative would be related to the increased workload and time required to complete data entry prior to each procedure.
Assessment of tool usability
Fifteen (63%) health professionals agreed or strongly agreed that the computerized decision support tool was easy to use compared with 2 (8%) who found the tool to be unnecessarily complex. However, only 10 (42%) allied health professionals agreed it was well integrated into the existing APPROACH system. Notable written feedback themes included preexisting issues with the background user interface that the decision-support tool did not appropriately circumnavigate, challenges with integration into workflows (due to both a lack of computers and handover between staff members), and excess information provided during training.
Despite the preimplementation and usability concerns that were reported by staff, there was increasing uptake in use of the decision support tool over the initial months following its implementation, and after 5 months the decision support tool was completed prior to 90% of eligible procedures (Supplemental Material 3); this was sustained over the subsequent 2 years of the project. Physicians uniformly (100%) agreed that the system was easy to use and well-integrated, although 1 respondent indicated that the tool was unnecessarily complex, and 1 felt that they would require assistance to properly use the decision support (17%).
Postimplementation evaluation
Fifty-one eligible health professionals completed the postimplementation evaluation (Figures 2 and 3). All four core aspects of the Contrast RISK initiative were well received, with the majority (82%) satisfied or very satisfied with processes to identify those at high risk of CA-AKI, 78% satisfied or very satisfied with processes to determine a safe contrast dye limit, 65% satisfied or very satisfied with processes to deliver optimized intravenous fluid, and 59% satisfied or very satisfied with processes for follow-up of those at high risk. Written comments were positive and showed that 59% of staff perceived that the initiative was beneficial in improving patient outcomes: with 29% perceiving this occurring via a reduction in the volume of contrast dye being administered and 29% perceiving this occurring via increasing awareness about the importance of preventative measures for CA-AKI. Health providers perceived that the greatest challenge in implementation was adhering to recommendations for hemodynamically optimized IV fluid administration.

Physicians’ self-reported strategies used for prevention of acute kidney injury preimplementation and postimplementation of the initiative.
Similarly, all physician respondents were satisfied or very satisfied with the core aspects of the initiative. Of the 12 respondents (40% of preimplementation participants), 8 (74%) always used the risk scoring system to estimate their patients CA-AKI risk and 7 (64%) reported always ordering a serum creatinine 48 to 72 hours postangiography in high-risk patients. Five (46%) physicians agreed that the recommended safe contrast volume always influenced their practice. Written comments indicated that 8 (89%) respondents perceived that the initiative had changed their practice to reduce contrast dye exposure or increase fluid administration.
Comparison of preimplementation versus postimplementation perspectives
In ordinal logistic regression models comparing preimplementation versus postimplementation perspectives of health care providers, implementation of the initiative led to a 3-fold increase in the odds of satisfaction with processes for identifying those at high risk of AKI (odds ratio [OR] 3.01, 95% CI 1.36-6.66, P = .007), an almost 7-fold increase in the odds of satisfaction with processes for quantifying the appropriate level of contrast dye for each patient (OR 6.98, 95% CI 3.06-15.91, P < .001), no difference in the odds of satisfaction with processes for determining the optimal amount of IV fluid for each patient (OR 1.86, 95% CI 0.88-3.91, P = .102), and an over 5-fold increase in the odds of satisfaction with processes for follow-up of kidney function of high risk patients (OR 5.49, 95% CI 2.45-12.30, P < .001) among health care staff working in the catheterization units. The effects of the initiative were stronger among physicians in several areas, with an almost 20-fold increase in the odds of satisfaction with processes for identifying those at high risk of AKI (OR 19.53, 95% CI 3.21-118.76, P = .001), an over 20-fold increase in the odds of satisfaction with processes for quantifying the appropriate level of contrast dye for each patient (OR 26.35, 95% CI 4.28-162.27, P < .001), no significant difference in the odds of satisfaction with processes for delivery of the optimal amount of IV fluid for each patient (OR 3.24, 95% CI 0.80-13.21, P = .100), and an over 7-fold increase in the odds of satisfaction with processes for follow-up of kidney function of high risk patients (OR 7.72, 95% CI 1.62-36.84.30, P = .010) (Supplemental Material 4). The implementation of the initiative also led to significant increases in physician self-reported use of a calculation to identify the threshold amount of contrast for a patient at risk of AKI (OR 42.68, 95% CI 6.46-282.06, P < .001) and in routinely checking serum creatinine 48 to 72 hours postangiography (OR 5.20, 95% CI 1.23-22.00, P = .251) (Figure 3; Supplemental Material 5). Recognition of the risk of AKI as important for planning angiography or intervention and the influence of information on contrast volume on practice, which were reported by most physicians as being important or very important prior to implementation of the initiative, did not significantly change with implementation. Many physicians also reported that they used a standard protocol to guide intravenous hydration both before and after implementation of the initiative. Consultation with a nephrologist before procedures in high-risk patients, which was not promoted by the Contrast RISK initiative, was reported to be infrequent both before and after implementation (Figure 3).
Implications
In this report, we have outlined the implementation of a computerized clinical decision support and audit and feedback intervention for prevention of CA-AKI across all cardiac catheterization units in Alberta, incorporating 6 activities within the Health Infoway Change Management Framework. Deployment of this initiative across all of Alberta’s cardiac catheterization units according to this framework led to successful uptake of the decision support tools into practice and the usability of the system was generally well received by physicians and allied health providers. Comparisons between preimplementation and postimplementation survey responses suggest that the initiative improved care providers’ satisfaction with processes for identifying patients at high risk of CA-AKI, using appropriate levels of contrast media and IV fluids, and promoting follow-up of kidney function for high-risk patients. These findings are important because the acceptance by end-users of these changes to structures and processes of care in cardiac catheterization units were a critical prerequisite to the goals of the Contrast RISK project.
Our report adds to previous literature on how systematic efforts can be implemented to reduce the incidence of CA-AKI with coronary procedures. Brown et al collected qualitative and quantitative data from 10 PCI centers in the United States and reported that clinical leadership and institution-focused efforts to standardize preventive practices were associated with lower rates of CA-AKI. 18 Our study provides more specific knowledge related to the process of incorporating an electronic decision-support tool for this purpose. Our findings identify several aspects that likely underlie the effectiveness of its implementation. First, resistance to change was minimized by aligning care provider priorities with project goals.26,27 Preimplementation data suggested that providers were not fully satisfied with the state of preventative steps for CA-AKI, including identification of appropriate contrast dye volume and IV fluid use, identifying an opportunity and motivation for change. Training and education activities leveraged care providers’ desire to provide better quality patient care in these two areas by outlining the existing state of CA-AKI care in Alberta and identifying how the initiative provided a unique opportunity to improve use of evidence-based preventative approaches to AKI.
Second, regular updates from the research team to all stakeholders in the form of online communications and in-person project update presentations helped provide users with the capacity to make changes that aligned with the project objectives and maintain enthusiasm for efforts to continue improving practice. Physician comments reflected satisfaction with the regular data that they received in the form of audit and feedback reporting. 24 Postimplementation surveys from allied health providers and physicians further suggested there was increased satisfaction with institutional processes for CA-AKI prevention following implementation of the initiative. The perceived strengths of the initiative in specific domains varied for health professionals and physicians, which was likely due to differences in roles and greater exposure to some aspects than others for health professionals versus physicians.
There were several areas where implementation of the project could have been improved. First, workflow analysis and integration could have been more effective had the project team coordinators also been integrated earlier at each site prior to beginning the project rather than introducing new personnel for the purpose of the project, since a consistent resource person on-site regularly may improve behavior change. However, this was limited by both human resources and funding availability. Interestingly, there was some discrepancy in opinions on the success of integration of the decision support tool between physicians, who all agreed that the project was well integrated, and other health providers, where just over 40% agreed. This may be due to differences in the extent of interaction with the clinical information system and its impact on workflow between physicians and health providers. For instance, data entry to support the decision support tool was largely the responsibility of health providers before and during the procedure, whereas physicians received the information without having to complete additional tasks in the clinical information system. Therefore, interpretation of participant feedback should recognize the context of the feedback provider’s clinical role, available resources, and impact on their own work processes.
We found that despite comprehensive in-person and personalized education sessions delivered to the cardiac catheterization units at the start of the project, staff required additional training and support to use the clinical decision support tool in the initial weeks following implementation. This illustrates that training and education is required not only prior to project initiation but also during early phases after implementation to help properly transition knowledge from didactic training settings into regular clinical practice. An additional complexity to the implementation of our project resulted from the stepped-wedge cluster randomized design of the accompanying trial, which required additional training for staff to recognize changes over time in the physicians who were versus were not receiving the intervention. Our experience demonstrated that providing end-users with education on research trial design and rationale was important when it influenced working requirements. Ensuring consistent messaging from clinical leadership was critical to strengthen end-user prioritization of the project’s implementation. Unit managers and nurse educator inclusion offered immediate, valuable perspectives on important research decisions and assistance in problem solving. Incorporating these stakeholders into the project steering committee may have streamlined communication and improved commitment to change. Providing financial compensation to physician and quality improvement leaders for such initiatives may be another way to strengthen motivation, adherence, and sustainability of a clinical decision support initiative of this nature.
Our experience highlights successful use of an existing framework to guide implementation of a computerized decision support initiative at a system level across a provincial health system, addresses capacity building and motivational factors that encouraged desired behavior changes, and identifies opportunities where improvements could be achieved in similar work in the future. This information is relevant for researchers and health system decision makers seeking to incorporate electronic decision support tools into clinical practice in the most time and cost-efficient manner possible. Despite the generalizability of these lessons learned, it is crucial that future researchers implementing similar computer decision support initiatives thoroughly understand their local clinical research environment to best adapt recommendations and shape implementation strategies to fit the unique needs of their setting.
Conclusions
The process of implementing this computerized clinical decision support and audit and feedback system illustrates the challenging, multi-step process involved with change management. The Canada Health Infoway Change Management Framework was a suitable model that underpinned the integration of new roles and responsibilities of health care providers for AKI prevention in cardiac catheterization units. Shared project goals and strong teamwork best supported deployment of the initiative. Future health technology implementation projects may consider the experience and lessons learned from our change management experience, incorporating effective training, communication, and governance to further encourage desired behavior change.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581231206127 – Supplemental material for Change Management Accompanying Implementation of Decision Support for Prevention of Acute Kidney Injury in Cardiac Catheterization Units: Program Report
Supplemental material, sj-docx-1-cjk-10.1177_20543581231206127 for Change Management Accompanying Implementation of Decision Support for Prevention of Acute Kidney Injury in Cardiac Catheterization Units: Program Report by Bryan Ma, Matthew T. James, Pantea A. Javaheri, Denise Kruger, Michelle M. Graham, Bryan J. Har, Benjamin D. Tyrrell, Shane Heavener, Clare Puzey and Eleanor Benterud in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The Alberta Provincial Project for Outcomes Assessment in Coronary Heart Disease (APPROACH) has been made possible by support from Alberta Health Services (Cardiac Sciences), the Libin Cardiovascular Institute, and Mazankowski Alberta Heart Institute. The funders were not involved in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The authors thank all the health staff and physicians working in Alberta’s cardiac catheterization units for their participation in the project.
Ethics Approval and Consent to Participate
The Health Research Ethics Board of the University of Alberta and Conjoint Health Research Ethics Board (CHREB) of the University of Calgary provided approval (REB17-0039).
Consent for Publication
All authors consent to the publication of this report.
Availability of Data and Materials
Data and materials from this program may be requested from the corresponding author.
Author Contributions
BM, MTJ, and EB drafted the report. All authors contributed to the initiative and provided critical input and contributed to revisions of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by a Partnership for Research and Innovation in the Health System (PRIHS) grant (#201600425) from Alberta Innovates and Alberta Health Services. MTJ was supported by a Canadian Institutes of Health Research (CIHR) Foundation Award.
Sharing
Research materials related to this study may be requested from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
