Abstract
Mesenchymal stem cells (MSCs) are an attractive therapeutic cell source for treating renal diseases. MSC administration has been shown to improve renal function, although the underlying mechanisms are not completely understood. We recently showed that allogenic fetal membrane-derived MSCs (FM-MSCs), which are available noninvasively in large amounts, had a renoprotective effect in an experimental glomerulonephritis model. Here we investigated whether allogenic FM-MSC administration could protect kidneys from ischemia/ reperfusion (I/R) injury. Lewis rats were subjected to right nephrectomy and left renal I/R injury by clamping the left renal artery as an acute kidney injury (AKI) model. After declamping, FM-MSCs (5 × 105 cells) obtained from major histocompatibility complex (MHC)-mismatched ACI rats were intravenously administered. I/R-injured rats exhibited increased serum creatinine and BUN, whereas FM-MSC administration significantly ameliorated renal function. Histological analysis revealed that FM-MSC administration significantly suppressed tubular apoptosis and infiltration of macrophages and T-cells. Administration of FM-MSCs mainly homed into the lung, but increased serum IL-10 levels. Of interest is that renoprotective effects of FM-MSCs were abolished by using anti-IL-10 neutralization antibody, suggesting that IL-10 would be one of the candidate factors to protect rat kidney from I/R injury in this model. We concluded that allogenic FM-MSC transplantation is a potent therapeutic strategy for the treatment of AKI.
Introduction
Renal ischemia/reperfusion (I/R) injury is a major cause of acute kidney injury (AKI), which is associated with prolonged hospitalization and excess morbidity and mortality (38,42). As its prevalence has risen, it is increasingly recognized as a significant cause of end-stage renal diseases (6). Despite decades of laboratory and clinical investigations and the advent of renal replacement therapy, no effective new therapy has been introduced into clinical practice in decades (14,44). Therefore, due to the clinical importance of renal I/R injury, effective therapy for such injury should be considered.
Mesenchymal stem cells (MSCs) are multipotent stem cells present in bone marrow (BM), adipose tissue, and many other organs and are capable of differentiating into cells of different tissue lineages including adipocytes, osteocytes, and chondrocytes (9,10,34). Previous studies have shown that BM-MSCs have renoprotective effects and can ameliorate the renal damage due to AKI induced by I/R (25,41). Although BM-MSCs have promising renoprotective effects, BM aspiration is rather invasive and sometimes yields low numbers of MSCs after processing. Therefore, alternative sources of MSCs would be beneficial for both research and therapeutic purposes. Fetal membrane (FM), which is generally discarded as medical waste after delivery, has been shown to be a rich and easily expandable source of MSCs (21,35). Recently, we established a protocol to isolate MSCs from FM and reported that administration of allogenic FM-derived MSCs (FM-MSCs) (5 × 105 cells/rat) as well as autologous BM-MSCs (5 × 105 cells/rat) induced therapeutic angiogenesis in a rat hindlimb ischemia model (22). Moreover, we reported that administration of the same number of FM-MSCs (5 × 105cells/rat) has a therapeutic effect in anti-thymocyte antigen 1 (Thy1) nephritic rats (43). These results suggest that this dose is adequate to improve tissue damage, and therefore allogeneic FM-MSCs may be an attractive alternative to autologous BM-MSCs as a source of regenerative therapy.
We previously reported on the behavior of the transplanted FM-MSCs; a large number of FM-MSCs were observed in the lung, and a small number of FM-MSCs were detected in the liver, spleen, and kidney 24 h after transplantation, but FM-MSCs were rarely found in the lung, liver, or spleen at 7 days after transplantation, and no FM-MSCs were detected at 28 days (23,43). Similar observations were also reported using adipose-derived MSCs (16). However, the therapeutic effects of FM-MSC administration were shown on day 7 or 14 after transplantation. These results suggested that FM-MSCs contributed to tissue repair by producing humoral factors rather than by differentiating into injured cell types. Moreover, we showed that FM-MSC-secreted prostaglandin E2 (PGE2) was involved in renal repair of the anti-Thy1 nephritic rats (43). It was reported that PGE2-induced interleukin-10 (IL-10), in macrophages (29,39) and T-cells (11), could possess both anti-inflammatory and immunosuppressive properties. Our preliminary experiments revealed that the FM-MSC transplanted rats exhibited increased IL-10 production, and so we hypothesized that intravenously injected FM-MSCs could stimulate IL-10 secretion via PGE2 and investigated whether allogenic FM-MSC administration can be protective against renal structural and functional injury in I/R injury as a model of AKI. Furthermore, we also examined the role of FM-MSC-induced IL-10 by using an anti-IL-10 neutralizing antibody.
Materials and Methods
Animals
All experiments had received prior approval by the Animal Care Committee of the National Cerebral and Cardiovascular Center Research Institute. Different strains of rats were used according to their major histocompatibility complex (MHC) antigen disparity: 10-week-old male Lewis (MHC haplotype: RT-1l) and August Copenhagen Irish (ACI; MHC haplotype: RT-1a) rats (Japan SLC, Hamamatsu, Japan). Pregnant female green fluorescent protein (GFP)-transgenic Lewis rats (Institute of Laboratory Animals, Kyoto University, Japan) were used to study the distribution of transplanted FM-MSCs.
Isolation and Expansion of FM-MSCs
Isolation and expansion of FM-MSCs were performed as previously described (22). In brief, FM was obtained from two pregnant Lewis rats on day 15 postconception. Minced FM was digested with type II collagenase solution (300 U/ml; Worthington Biochemical, Lakewood, NJ, USA) for 1 h at 37°C. After filtration and centrifugation, FM-derived cells were suspended in α modification of minimum essential medium (α-MEM; Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Thermo Scientific, Woburn, MA, USA) and penicillin/streptomycin (Invitrogen) and cultured in standard plastic dishes (Iwaki, Tokyo, Japan). The adherent MSC populations appeared by days 5–7, and these FM-MSCs were used for the experiments at passages 3–6.
Experimental Model and Design
Lewis rats weighing 230 to 250 g were used throughout the experiments and randomly allocated into two groups: 1) control phosphate-buffered saline (PBS) injection alone (PBS group) and 2) FM-MSC injection (FM-MSC group). Rats were then subjected to I/R injury. All rats were anesthetized by intraperitoneal injection of pentobarbital sodium (50 mg/kg; Abbott Laboratories, North Chicago, IL, USA) and were subjected to 60 min of renal occlusion using Sugita Clip titanium mini temporary artery clips (Mizuho Medical Inc., Tokyo, Japan) to clamp the renal pedicles, and right nephrectomy was performed. Occlusion was visually confirmed by a change in kidney color to a paler shade. Reperfusion was initiated with the removal of artery clips and was visually confirmed by noting the subsequent blush. Prior to the removal of the artery clips, rats received PBS (WAKO Pure Chemical Industry Ltd., Osaka, Japan) or 5 × 105 FM-MSCs obtained from MHC mismatched ACI rats via the tail vein. Rats were sacrificed 6 h (n = 6 in each group), 12 h (n = 6 in each group), 24 h (n = 9 in each group), and 72 h (n = 8 in each group) after reperfusion. Sham-operated rats (sham group; n = 6 in each period) underwent the same procedure except that clamping was not done, and they received a PBS injection.
Blood was also collected at the time of sacrifice, and blood urea nitrogen (BUN) and serum creatinine levels were measured by SRL (Tokyo, Japan).
To assess the distribution of the injected cells, intravenous transplantation of the same number of FM-MSCs (5 × 105 cells) derived from GFP-transgenic Lewis rats was performed in I/R-injured ACI rats. Twenty-four hours after the injection, the rats were sacrificed, and sections of tissues were obtained from the kidney, lung, spleen, and liver and embedded in paraffin (n = 4 for each tissue).
To explore the role of IL-10 in this model, Lewis rats were induced I/R injury and received FM-MSC transplantation and anti-IL-10 neutralization antibody (0.05 μg/g body weight, which is more than sufficient a dose to neutralize serum IL-10 levels; R&D Systems, Minneapolis, MN, USA) at the time of reperfusions. The concentration of IL-10 in serum was determined using an enzyme-linked immunosorbent assay (ELISA) kit, according to the manufacturer's protocol (R&D Systems). The absorbance was measured by a microplate reader (Bio-Rad, Hercules, CA, USA) at 450 nm.
Morphology and Immunohistochemical Staining
Kidney tissues were fixed with 4% phosphate-buffered formalin solution (Wako Pure Chemical Industry Ltd.) and embedded in a paraffin block. Tissue sections (2 μm) were used for periodic acid-Schiff (PAS; Muto Pure Chemical Industry Co., Tokyo, Japan) or for immunohistochemical staining, which was performed with mouse anti-α-smooth muscle actin (α-SMA) antibody (clone 1A4; Dako, Glostrup, Denmark; undiluted), mouse anti-cluster of differentiation 68 (CD68) antibody (clone ED-1, Serotec, Oxford, UK; 1:100), mouse anti-CD3 antibody (BD Biosciences Pharmingen, San Jose, CA, USA; 1:100), and rabbit anti-GFP antibody (Invitrogen; 1:100). Following antigen retrieval, endogenous peroxidase activity was quenched with 1.5% H2O2 (WAKO Pure Chemical Industry Ltd.) for 10 min. The first antibodies were incubated for 1 h at room temperature, followed by incubation with DakoCytomation LASB2 System-HRP (Dako). The sections were visualized with 3,3′diaminobenzidine tetrahydrochloride (Dako) and counterstained with hematoxylin (Muto Pure Chemical Industry Co.). All histological slides were photographed using a digital microscope (BIOREVO BZ-9000; Keyence, Osaka, Japan). PAS-stained sections were scored by calculating the percentage of tubules in corticomedullary junction that displayed cell necrosis, loss of brush border, cast formation, and tubular dilation as follows: 0, none; 1, <10%; 2, 11–25%; 3, 26–45%; 4, 46–75%; and 5, >76%. At least 20 randomly selected fields per rat were scored. The number of CD68-and CD3-positive cells was evaluated by counting stained cells per areas in at least 20 randomly selected areas. The area of interstitial renal fibrosis in the outer medulla was calculated as a percentage using a computer-aided manipulator (Win Roof; Mitani, Fukui, Japan). The α-SMA staining percentage of total area was determined, and the mean value of 20 randomly selected interstitium was calculated. To evaluate the distribution of GFP-positive administered cells, we counted all the GFP-positive cells in one randomly selected section from each organ, and an overall average for all rats was calculated.
Terminal Deoxynucleotidyltransferase-Mediated dUTP Nick-End Labeling (TUNEL) Staining
TUNEL staining was performed using the in situ Apoptosis Detection Kit (Takara Bio, Otsu, Japan), according to the manufacturer's instructions. Briefly, sections were deparaffinized, subjected to antigen retrieval (proteinase K; WAKO Pure Chemical Industry Ltd. for 10 min), and were then incubated with 1.5% H2O2 for 10 min, followed by incubation with TdT enzyme solution for 90 min at 37°C. The reaction was terminated by incubation in a stop/wash buffer for 30 min at 37°C. The number of TUNEL-positive cell nuclei and the total numbers of cell nuclei stained with hematoxylin were counted in 20 random areas, and the percentage of TUNEL-positive nuclei against total cell nuclei was then calculated.
Quantitative Reverse-Transcription Polymerase Chain Reaction (qRT-PCR) Analysis
Total RNA was isolated from the kidney cortex using an RNeasy mini kit (Qiagen, Hilden, Germany). Obtained RNA was reverse transcribed into cDNA using a Quantitect Reverse Transcription kit (Qiagen). PCR amplification was performed using Power SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA). β-Actin transcript was used as an internal control. Primers were as follows: monocyte chemotactic protein-1 (MCP-1): 5′-atgcagttaatgccccactc-3′ (forward), 5′-ttccttat tggggtcagcac-3′ (back); IL-6: 5′-ccggagaggagacttcacag-3′ (forward), 5′-cagaattgccattgcacaac-3′ (back); transforming growth factor-β (TGF-β): 5′-ctactgcttcagctccacagaga-3′ (forward), 5′-accttgggcttgcgacc-3′ (back); type I collagen: 5′-aatggtgctcctggtattgc-3′ (forward), 5′-ggttcaccactgttgcct tt-3′ (back); β-actin: 5′-gccctagacttcgagc-3′ (forward), 5′-ct ttacggatgtcaacgt-3′ (back).
Statistical Analysis
All data were expressed as mean ± SEM. Comparisons of parameters among the three groups were made by one-way analysis of variance (ANOVA), followed by Tukey's test. A value of p < 0.05 was considered statistically significant.
Results
Protection of Renal Function by FM-MSC Transplantation
We examined the effect of FM-MSCs on renal function (Fig. 1). I/R injury induced significantly elevated serum creatinine level (1.51 ± 0.2 mg/dl; PBS group) 24 h later compared to the sham operation (0.31 ± 0.02 mg/dl). FM-MSC treatment significantly reduced this increase (0.77 ± 0.05 mg/dl, p < 0.05 vs. PBS group) (Fig. 1A). In addition, we observed an increase in BUN at 24 h after I/R injury (92 ± 10.3 mg/dl) in PBS group, while FM-MSC administration significantly inhibited the increase (56.7 ± 7.8 mg/dl, p < 0.05 vs. PBS group) (Fig. 1B). Similarly, FM-MSC transplantation also improved the renal function at 72 h after I/R injury (creatinine: 0.95 ± 0.13 mg/dl in PBS group vs. 0.53 ± 0.06 mg/dl in FM-MSC group, p < 0.05; BUN: 93.2 ± 13.9 mg/dl in PBS group vs. 48.1 ± 5.4 mg/dl in FM-MSC group, p < 0.05).
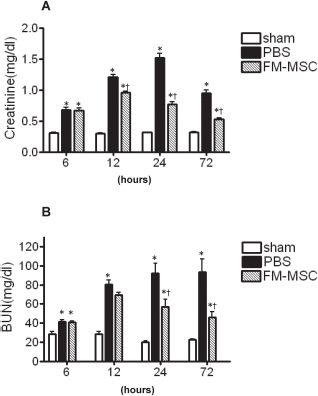
Effects of FM-MSC transplantation on renal function. Effect of fetal membrane-derived mesenchymal stem cell (FM-MSC) transplantation on renal function is summarized. Creatinine (A) and blood urea nitrogen (BUN) (B) were examined 6, 12, 24, and 72 h after sham operation (sham) or ischemia/ reperfusion injury treated with phosphate-buffered saline (PBS) or FM-MSC transplantation (FM-MSC). *p < 0.05 versus sham group, †p < 0.05 versus PBS group.
Effects on Histological Changes and Apoptosis of Tubular Epithelial Cells
We then tested the effect of FM-MSCs on histological changes (Fig. 2). PAS staining of the kidney sections obtained from PBS-treated I/R-injured rats at 24 h exhibited marked disruption of normal tubular morphology, including widespread degeneration of tubular architecture, tubular dilatation, swelling, and luminal congestion with loss of the brush border (PAS score: 2.53 ± 0.16 in PBS group vs. 0.07 ± 0.14 in sham group, p < 0.001) (Fig. 2A, B, and G). In contrast, FM-MSC treatment reduced severity with regard to the characteristic histological changes of I/R injury, including tubular atrophy (1.31 ± 0.06, p < 0.001 vs. PBS group) (Fig. 2C and G). At 72 h after reperfusion, PBS-treated rats exhibited more severe tubular injury than FM-MSC transplanted rats (3.52 ± 0.15 in PBS group vs. 1.71 ± 0.08 in FM-MSC group, p < 0.001) (Fig. 2G). Moreover, TUNEL-positive apoptotic cells were significantly increased in PBS-treated rats at 12 and 24 h after I/R injury (12 h: 12.78 ± 0.43%, 24 h: 11.34 ± 0.59%, p < 0.001 vs. sham group) compared with sham-operated rats (12 h: 0.16 ± 0.02%, 24 h: 0.08 ± 0.01%), while FM-MSC treatment significantly reduced the apoptotic cells (12 h: 4.98 ± 0.56%, 24 h: 2.14 ± 0.20%, p < 0.01 vs. PBS group) (Fig. 2D–F, H). Similarly, I/R-induced apoptosis (1.08 ± 0.10% in PBS group vs. 0.10 ± 0.02% in sham group, p < 0.01) was significantly attenuated by FM-MSC transplantation (0.74 ± 0.07%, p < 0.01) at 72 h after reperfusion.

Effects of FM-MSC transplantation on renal injury. Representative micrographs of periodic acid-Schiff (PAS) and terminal deoxynucleotidyltransferase-mediated dUTP nick-end labeling (TUNEL) staining in sham (A, D), PBS (B, E), and FM-MSC (C, F) groups at 24 h after reperfusion. Quantitative analysis of tubular damage (G) and the number of TUNEL-positive cells (H) are summarized. All stained sections were analyzed in 20 nonoverlapping low-power fields at 200× magnification. Scale bars: 50 μm. **p < 0.01, ***p < 0.001 versus sham group; ††p < 0.01, †††p < 0.001 versus PBS group.
Inhibition of T-Cell and Macrophage Infiltration by FM-MSC Transplantation
Next, we examined the effect of FM-MSCs on T-cell and macrophage infiltration into I/R-injured kidney (Fig. 3). At 6 h after reperfusion, infiltrating CD3-positive T-cells were significantly increased in PBS-treated kidneys (8.31 ± 0.41 cells/field, p < 0.001 vs. sham group) (Fig. 3B, G) compared with sham-operated kidneys (3.1 ± 0.2 cells/field) (Fig. 3A, G). On the contrary, FM-MSC treatment significantly suppressed the T-cell infiltration (5.8 ± 0.3 cells/field, p < 0.001 vs. PBS group) (Fig. 3C, G). A similar result was also observed at 12 h after reperfusion (PBS group 4.14 ± 0.30 cells/field vs. FM-MSC group 3.29 ± 0.31 cells/field, p < 0.001).

Effects of FM-MSC transplantation on T-cell and macrophage infiltration. Representative images of cluster of differentiation 3 (CD3) staining at 6 h and CD68 (antibody clone ED-1) staining at 72 h from kidney sections of sham (A, D), PBS (B, E), and FM-MSC (C, F) groups are shown. Quantification of CD3-positive cells (G) and ED-1-positive cells (H) was analyzed. Original magnification, 200×. Scale bars: 50 μm. Arrows indicate the positive staining for CD3. Real-time PCR showed monocyte chemotactic protein-1 (MCP-1; I) and interleukin (IL)-6 (J) mRNA levels at 6, 12, and 24 h. Results are expressed as relative expression against the expression in sham-operated rats. *p < 0.05, ***p < 0.001 versus sham group, †p < 0.05, †††p < 0.001 versus PBS group.
We then investigated the effect of FM-MSCs on interstitial macrophage infiltration, which is associated with interstitial damage and fibrosis. CD68 (antibody clone ED-1)-positive macrophages were increased in the PBS group at 12, 24, and 72 h after reperfusion (134.6 ± 3.8, 387.9 ± 17.0, and 812.7 ± 50.8 cells/field, respectively, p < 0.001 vs. sham group) compared to sham-operated rats (11.4 ± 1.1, 7.2 ± 0.7, and 20.8 ± 2.2 cells/field, respectively), while FM-MSC transplantation significantly suppressed interstitial ED-1-positive macrophage infiltration (105.6 ± 3.0, 310.6 ± 14.4, and 438.5 ± 33.2 cells/field, respectively, p < 0.001 vs. PBS group) (Fig. 3D–F, H). As we observed the inhibition of T-cell and macrophage infiltration, we examined the expression of various proinflammatory cytokines and chemokines in I/R-injured kidneys. qRT-PCR analysis showed that I/R injury induced increased MCP-1 expression at 6, 12, and 24 h after reperfusion (4.36 ± 0.97-, 3.35 ± 0.73-, and 7.62 ± 0.97-fold, respectively, p < 0.05 vs. sham group), and FM-MSC transplantation significantly suppressed this increased expression (2.02 ± 0.13-, 1.56 ± 0.35-, and 4.40 ± 0.54-fold, respectively, p < 0.05 vs. PBS group) (Fig. 3I). FM-MSC administration also significantly suppressed IL-6 mRNA expression at 6, 12, and 24 h after reperfusion (9.10 ± 1.43-, 4.69 ± 0.92-, and 1.82 ± 0.25-fold in FM-MSC group, respectively, p < 0.05 vs. 16.22 ± 2.45-, 7.61 ± 1.21-, and 3.34 ± 0.39-fold, respectively, in PBS group) (Fig. 3J).
Effects on Interstitial Phenotypic Changes in Kidney After I/R Injury
We next studied the effect of FM-MSCs on interstitial phenotypic alteration (Fig. 4), assessed by immunohistochemical staining using anti-α-SMA antibody, which is associated with interstitial damage and fibrosis. α-SMA expression was limited to vessels at 24 h after I/R injury in sham-operated (0.04 ± 0.01%), PBS-treated (0.06 ± 0.01%), and FM-MSC-treated kidney (0.06 ± 0.01%). However, interstitial α-SMA expression significantly increased in PBS-treated kidney at 72 h (10.09 ± 0.41%, p < 0.001 vs. sham group) compared to the sham-operated kidney (0.56 ± 0.04%), while FM-MSC transplantation suppressed the interstitial α-SMA expression (4.44 ± 0.38%, p < 0.001 vs. PBS group; Fig. 4A–D). Similarly, qRT-PCR analysis revealed that FM-MSC transplantation significantly decreased TGF-β and type I collagen mRNA expression, which are related with renal interstitial fibrosis, at 72 h after reperfusion (1.19 ± 0.14-fold and 1.61 ± 0.47-fold, respectively, p < 0.05 vs. PBS group) compared with the PBS group (2.32 ± 0.44-fold and 6.28 ± 1.87-fold, respectively, p < 0.05 vs. sham group) (Fig. 4E, F).

Effects of FM-MSC transplantation on phenotypic alteration. Representative images of α smooth muscle actin (α-SMA) staining are shown in kidney sections of sham (A), PBS (B), and FM-MSC (C) groups at 24 h and 72 h. Quantification of α-SMA-positive area (D) was analyzed. Original magnification: 200×. Scale bars: 50 μm. Real-time PCR showed transforming growth factor (TGF)-β mRNA level (E) and type I collagen at 72 h (F). Results are expressed as relative expression against the expression in sham-operated rats. *p < 0.05, ***p < 0.001 versus sham group, †p < 0.05, †††p < 0.001 versus PBS group.
Engraftment of Intravenously Injected FM-MSCs in Rats with I/R Injury
We previously reported that intravenously injected FM-MSCs were rarely observed in kidney at 7 days after transplantation. To investigate the distribution of intravenously administered FM-MSCs at the early period, we intravenously administrated FM-MSCs, which were derived from GFP-transgenic Lewis rats, into I/R-injured ACI rats at the time of reperfusion (Fig. 5). GFP-positive FM-MSCs were faintly detected in the kidney sections at 6 h (28.1 ± 1.1 cells/cm2) and 24 h (11.9 ± 1.4 cells/cm2) (Fig. 5A) as well as the liver (at 6 h: 32.1 ± 1.4 cells/cm2, at 24 h: 41.1 ± 3.2 cells/cm2) (Fig. 5B) and spleen (at 6 h: 26.2 ± 1.8 cells/cm2, at 24 h: 27.6 ± 3.3 cells/cm2) (Fig. 5C). On the contrary, we observed more GFP-positive FM-MSCs in lung (at 6 h: 335.9 ± 18.1 cells/cm2, at 24 h: 124.1 ± 8.5 cells/cm2) (Fig. 5D).

Distribution of intravenously transplanted FM-MSCs in I/R-injured rats. Representative micrographs showing localization of administrated green fluorescent protein (GFP)-positive FM-MSCs in kidney (A), liver (B), spleen (C), and lung (D) in a rat with I/R injury. Arrows indicate the positive staining for GFP. Original magnification: 400×. Scale bars: 50 μm.
Effects of IL-10 Neutralizing Antibody in FM-MSC Transplanted Rats with I/R Injury
As we observed rare FM-MSCs in kidney, we then examined the FM-MSC-derived humoral therapeutic factor (Fig. 6). In our previous study, we confirmed that FM-MSCs secreted a significant amount of PGE2 into FM-MSC supernatant (888.1 ± 123.3 pg/ml) (43). PGE2 enhanced the secretion of IL-10 from immune cells, and so we measured serum IL-10 levels and found a significant increase in IL-10 in the FM-MSC group at 24 h after reperfusion (113.0 ± 13.2 pg/ml), while serum IL-10 was not detected in PBS-treated group. Therefore, we attempted to confirm if IL-10 is a key mediator in this model. FM-MSC-treated rats that received anti-IL-10 neutralization antibody, exhibited undetectable serum IL-10 (<10 pg/ml) levels, and the renoprotective effect of FM-MSCs on I/R injury was cancelled (creatinine: 1.24 ± 0.2 mg/dl, p = 0.32, vs. PBS group; BUN: 83.3 ± 9.1 mg/dl, p = 0.54, vs. PBS group) (Fig. 6A, B).
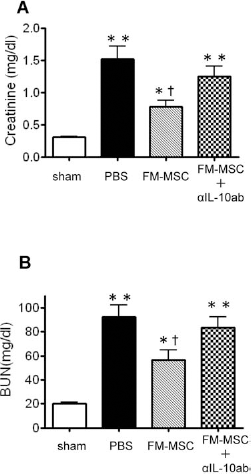
Effects of anti-IL-10 neutralizing antibody on FM-MSC transplantation treatment. Injection of anti-IL-10 neutralization antibody cancelled the effectiveness of FM-MSCs on creatinine (A) and BUN (B) levels. *p < 0.05, ** p < 0.01 versus sham group, †p < 0.05 versus PBS group.
Discussion
In this study, we investigated the renoprotective effects and the mechanism of FM-MSCs in AKI model; 1) intravenous injection of allogenic FM-MSCs improved renal function in rats with I/R injury, 2) allogenic FM-MSC administration inhibited tubular apoptosis, interstitial fibrosis, and infiltration of macrophages and T-cells in a rat model of I/R injury, 3) administered FM-MSCs mainly homed into the lung, but not into the kidney, 4) serum IL-10 levels were increased in FM-MSC-treated rats, but 5) renoprotective effects of FM-MSCs are abolished by anti-IL-10 neutralization antibody.
In this study, we employed FM-MSCs as a new type of MSCs. Several studies have shown beneficial effects of BM-MSC transplantation in AKI model rats (5,19,20,25– 28,41). However, clinical application of autologous BM-MSCs is limited because of several problems such as the invasiveness of extraction, inadequate cell numbers, and donor site morbidity (45). On the other hand, FM tissue possesses great advantages due to its abundance and easy accessibility and has been shown to be rich sources of MSCs (21,35). In our previous reports, we demonstrated that allogeneic FM-MSCs as well as autologous BM-MSCs are suitable cell sources for tissue regeneration using a rat hindlimb ischemia model. Therefore, we propose that allogeneic FM-MSCs might be a good alternative to autologous BM-MSCs.
Here we suggested that one of the therapeutic mechanisms of FM-MSCs is due to an anti-inflammatory effect via systemic IL-10 production. The underlying immunomodulatory mechanisms of MSCs remain largely unknown whether direct cell–cell contact or humoral factors are involved. Previously, we and others reported that PGE2 is one of the key modulators for the MSC-induced anti-inflammatory response (7,43). Nemeth et al. reported that BM-MSCs reprogrammed macrophages by releasing PGE2, and thereby IL-10, secreted from reprogrammed macrophages, attenuated septic conditions (28). In this study, we showed that FM-MSC-transplanted rats exhibited increased IL-10 in I/R-injured rats at 24 h after reperfusion. On the contrary, injecting anti-IL-10 neutralization antibody cancelled the renoprotective effect of FM-MSCs on I/R injury. These results indicated that FM-MSCs ameliorated renal I/R injury via IL-10 secretion. IL-10 is a pleiotropic cytokine with many immunosuppressive effects, and this potent anti-inflammatory cytokine could suppress the activation of neutrophils and monocytes and thereby prevent their production of chemokines and cytokines in animals and humans (12,13,31,32). However, the fact that IL-10 neutralizing antibody did not perfectly cancel the renoprotective effects suggests that IL-10 is not solely responsible for FM-MSC-derived therapeutic effects.
We demonstrated that FM-MSC transplantation inhibited macrophage infiltration in rat I/R-injured kidneys, compared with PBS treatment. Previous studies have focused on the anti-inflammatory effect of MSCs, which are able to inhibit systemic and local inflammatory responses in several models of inflammatory conditions (3,18,28). Inflammation also plays an important role in I/R-injured kidney, and macrophages are key participants in I/R injury (15). Monocyte/macrophage infiltration and monocyte/macrophage chemoattractants, such as MCP-1 (33,40,47) as well as macrophage-associated cytokines, such as IL-6 and TNF-α (40), were related in the I/R-injured kidney. In addition, inhibition of chemokine C-C motif receptor 2 (CCR2), receptor for MCP-1 (also known as chemokine C-C motif ligand 2; CCL2), markedly ameliorated disease manifestations in I/R-injured mice (17). In this rat I/R injury model, we demonstrated that administered FM-MSCs suppressed MCP-1 and IL-6 expression. Previously, we reported that allogeneic FM-MSC administration significantly attenuates macrophage infiltration in a rat model of acute glomerulonephritis (43). Therefore, FM-MSCs could modulate local inflammatory responses by inhibiting MCP-1 and IL-6 expression and contribute to the preservation of renal function and structure in renal I/R injury model.
In this study, T-cell infiltration occurred at 6 h and vanished by 24 h, and we demonstrated that FM-MSCs significantly reduced T-cell infiltration. In addition to a potent anti-inflammatory effect, many reports have highlighted the potential immunomodulatory effect of MSCs (1,30), such as suppression of T-cell activation and proliferation both in vitro and in vivo (4,23). Recent studies have also identified T-cells as important mediators (8,36,37,46), especially in so-called “hit-and-run” attacks; early T-cell infiltration at 4–6 h and resolution by 24 h (2,15,24). Our observation was consistent with this phenomenon. Therefore, we suggest that renoprotective effects of FM-MSCs, in part, may be due to the inhibition of systemic inflammatory responses, by controlling early T-cell infiltration after I/R and by attenuating the T-cell activation during the early reperfusion period.
In conclusion, this study showed that FM-MSC transplantation ameliorated renal function in a rat model of AKI. FM-MSCs are available noninvasively in large amounts, so we suggest that allogeneic FM-MSCs could provide a new therapeutic strategy for the treatment of AKI.
Footnotes
Acknowledgments
This work was financially supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology in Japan (grant No. 24790867 to H. Tsuda), a Health Labour Sciences Research Grant for Research on Allergic Disease and Immunology and a Grant-in-Aid for Progressive Renal Diseases Research, Research on Intractable Disease, from the Ministry of Health, Labour and Welfare of Japan. The authors declare no conflicts of interest.
