Abstract
Tumor lysis syndrome (TLS) consists of a rapid tumor cell breakdown that ends in multiple metabolic derangements and can occur spontaneously or be triggered by oncological therapies. It is a serious oncological emergency, primarily seen in hematological malignancies like non-Hodgkin lymphomas and particularly Burkitt’s lymphoma, as well as in certain bulky solid tumors like neuroblastoma. This report discusses a case of spontaneous TLS in a patient with metastatic cholangiocarcinoma. We present a case of a 72-year-old Hispanic male with a history of hypertension, tobacco use disorder, and alcoholism who was admitted for 20 days of worsening abdominal distention, edema, dyspnea, cough, and hyporexia. Physical examination revealed jaundice and signs of ascites. Laboratory studies showed mildly elevated liver enzymes and a resolved hepatitis B infection. A multiphase computerized tomography (CT) scan of the abdomen revealed a large heterogeneous hepatic mass, ascites, lymphadenopathy, and portal vein thrombosis. Chest CT showed nodules with perilymphatic distribution. A paracentesis yielded 3000 cc of ascitic fluid, showing a serum-ascites albumin gradient of 1.81 g/dL and neutrophil predominance. Tumor markers indicated elevated CA 19-9 levels. Subsequent liver biopsy confirmed moderately differentiated adenocarcinoma, and the immunohistochemistry supported a diagnosis of cholangiocarcinoma. On day 5 of hospitalization, the patient’s renal function worsened, leading to spontaneous TLS. Overall, TLS is a life-threatening emergency in cancer patients, necessitating regular lab tests for early diagnosis, especially in those on chemotherapy or with known solid tumors. There is no consensus on diagnosing spontaneous TLS.
Introduction
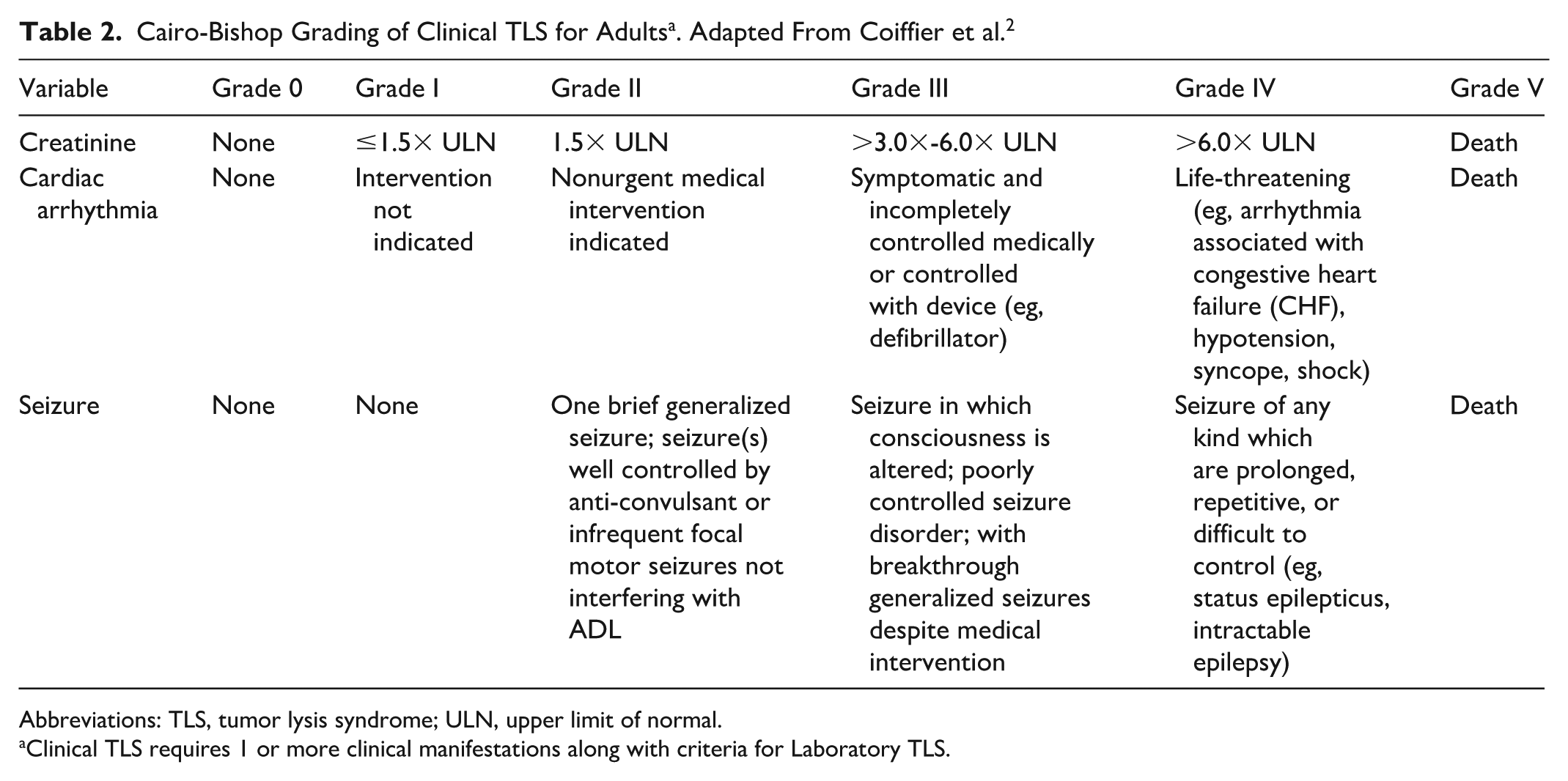
Tumor lysis syndrome (TLS) is the result of a rapid tumor cell breakdown that ends in multiple metabolic derangements. The concept of TLS emerged soon after the integration of cytotoxic chemotherapy into oncological care. 1 The syndrome is characterized by a massive and abrupt release of cellular components, such as cations, anions, protein products, and nucleic acids, into the bloodstream.1,2 This metabolic disruption can occur spontaneously or more commonly be triggered by the initiation of oncological treatment. It is frequently associated with cytotoxic chemotherapy but can also result from endocrine therapies, targeted agents, radiotherapy, and glucocorticoid therapy. 3 TLS can be diagnosed using the well-known Cairo and Bishop definition, which allows for clinical and laboratory-based diagnosis and grading of the disease (see Tables 1 and 2). It ranks among the most common life-threatening oncological emergencies, typically observed in hematological malignancies, especially non-Hodgkin lymphomas, particularly Burkitt’s lymphoma, acute lymphoid leukemia, and acute myeloid leukemia. 2 Although solid tumors only represent about 1% of TLS cases in adults, certain bulky solid tumors that are responsive to chemotherapy, such as neuroblastoma, germ-cell tumors, and small-cell lung cancer, are classified as intermediate risk disease. The risk stratification of TLS is based on the type of tumor, tumor burden, grade, and patient factors.2,4 Here, we present a case of spontaneous TLS in a patient diagnosed with metastatic cholangiocarcinoma.
Abbreviation: TLS, tumor lysis syndrome.
Laboratory TLS is defined as either a 25% change from baseline or any 2 or more serum values within 3 days before or 7 days after the initiation of chemotherapy (assuming the patient has received adequate hydration and hypouricemic agent).
Abbreviations: TLS, tumor lysis syndrome; ULN, upper limit of normal.
Clinical TLS requires 1 or more clinical manifestations along with criteria for Laboratory TLS.
Case Presentation
A 72-year-old Hispanic male with a past medical history of hypertension, tobacco use disorder (22 pack/year), and alcoholism presented to the hospital for a 20-day history of progressing abdominal distention, lower extremity edema, dyspnea, dry cough, and hyporexia. The physical exam was remarkable for jaundice, dullness at the right flank, and positive signs of shifting dullness and fluid wave. On arrival, laboratory studies showed mildly elevated liver enzymes, serum creatinine within the normal limits (unclear baseline) per the laboratory report, normal coagulation tests, normal potassium, and low ionized calcium (Table 3). The viral hepatitis panel was unremarkable except for a resolved hepatitis B infection profile.
Laboratory Results.

Abbreviation: AST, Aspartate Aminotransferase, ALT, Alanine Aminotransferase, PT, Prothrombine Time, INR, International Normalized Ratio, PTT, Partial Thromboplastin Time, LDH, Lactate Dehydrogenase, EMA, Epithelial Membrane Antigen, not available.
Multiphase contrast-enhanced CT scan of the abdomen was done and demonstrated a large hepatic mass of heterogeneous appearance that retracted the capsule, irregular enhancement in the arterial phase, which progressed in the portal phase, thickening and striation of the greater omentum with soft tissue nodules, ascites, multiple lymphadenopathies, and chronic portal vein thrombosis (Figure 1). CT of the chest revealed nodules with a perilymphatic distribution.

(A) Abdominal CT on arterial phase and (B) portal phase showing a large hepatic mass of heterogeneous appearance.
The patient underwent diagnostic and therapeutic paracentesis, yielding 3000 cc of ascitic fluid. Fluid studies reported albumin 1.19 g/dL, serum-ascites albumin gradient 1.81 g/dL, pH 7.6, glucose 94 mg/dL, total proteins 1.99 g/dL, LDH 73 U/L, erythrocytes 646 cell/mm3, leucocytes 248 cell/mm3 (neutrophiles 20%, lymphocytes 77%). Cytology and cell block showed reactive mesothelial hyperplasia. Furthermore, tumor markers were measured, revealing elevated CA 19-9 levels (1693.6 U/mL) and a normal level of carcinoembryonic antigen (CEA; 2.1 ng/mL) and alpha-fetoprotein (2.0 ng/mL).
Subsequently, a percutaneous CT-guided liver biopsy was performed. Pathology reported moderately differentiated adenocarcinoma. Immunohistochemistry stains were positive for cytokeratin 19, cytokeratin 20, cytokeratin 5.2, cytokeratin AE1/AE3, and negative for WT-1, NKX3.1, CDX-2, CA19-9, PSA, synaptophysin, TTF-1, HepPar-1, glypican 3, MOC-31, CEA, EMA, and cytokeratin 7. With a Ki67 of 40% to 50% in neoplastic cells. Although cytokeratin 20 is not commonly found in biliary tract adenocarcinomas, the positivity for cytokeratin 19, along with microscopy images highly suggestive of cholangiocarcinoma (Figure 2), clinical presentation, multiple imaging studies, negative bronchoalveolar lavage cytology, normal colonoscopy, and normal upper endoscopy, collectively support a diagnosis of cholangiocarcinoma as the primary tumor.
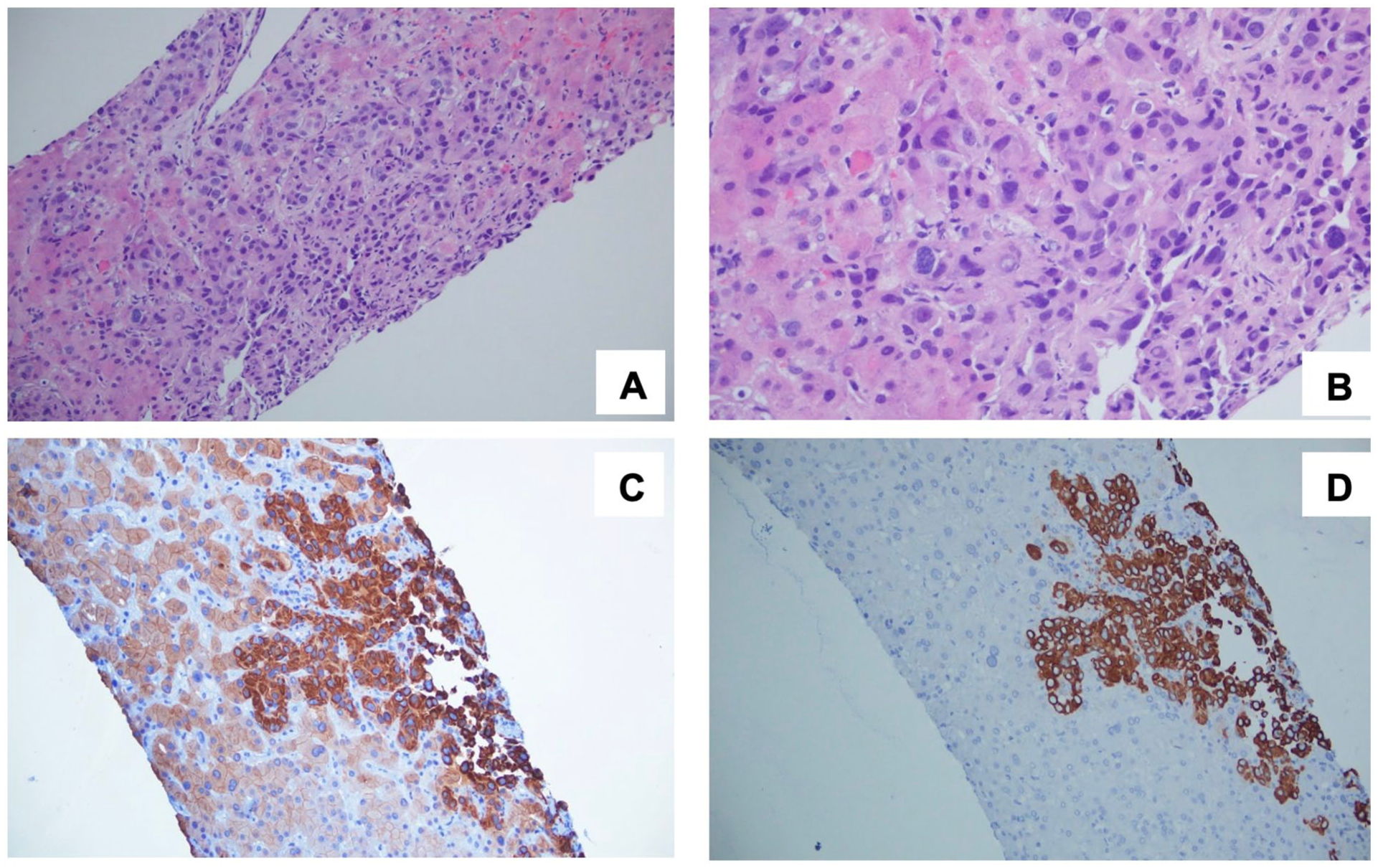
(A) H&E 200× neoplastic epithelial cells that form nests and a few poorly defined tubules. (B) H&E 400× poorly defined tubules with marked nuclear pleomorphism, hyperchromatism, and atypical mitoses. IHC positive (C) cytokeratin 5.2 and (D) positive cytokeratin 20 on neoplastic cells. H&E, hematoxylin and eosin; IHC, immunohistochemistry.
On the fifth day of hospitalization, the patient’s renal function began to deteriorate, reflected in an elevated creatinine level. Additionally, moderate hyperkalemia, hypocalcemia, hyperuricemia, and normal phosphorus levels were observed. The electrocardiogram was unremarkable, and the patient exhibited no neurological deficits. Based on these findings, a diagnosis of spontaneous TLS was established.
The patient was started on IV fluids (normal saline 0.9%) at a rate of 999 mL/h, allopurinol 300 mg orally twice daily, and hyperkalemic treatment per the hospital algorithm, which included calcium gluconate 2 g IV followed by insulin regular 10 UI and glucose IV bolus, achieving a potassium <5.5 in the next potassium check. Over the subsequent days, the patient’s renal function improved, as did the electrolyte abnormalities. However, despite these positive developments, the progression of the disease significantly impacted the patient’s overall condition and quality of life, prompting a shared decision to focus solely on comfort measures, in line with the patient’s wishes and goals of care.
Discussion
TLS represents a significant oncological emergency characterized by metabolic disturbances that lead to hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia, disrupting normal physiological balance. These imbalances can lead to severe complications such as lethal arrhythmias, renal impairment, seizures, and even death. TLS is divided into spontaneous and therapy-induced forms.
The Cairo and Bishop definition applies only to patients who have started chemotherapy either 3 days before or within 7 days after the initiation of treatment. The patient mentioned above had recently been evaluated for malignancy when he began to experience a 2.6-fold increase in his creatinine levels and met 3 of the 4 criteria for electrolyte abnormalities. However, the patient was not undergoing any cancer therapy. Therefore, based on the significant tumor burden and metastatic behavior of the disease, a diagnosis of spontaneous TLS was made, a rare condition that occurs when tumor cells lyse without current oncological treatment.
Spontaneous occurrences of TLS in solid tumors are rare. Risk factors such as bulky disease (>10 cm), advanced disease (multiple metastases), medication concomitant use (such as thiazide diuretics and aspirin), prior reduced renal function, and alcohol abuse should be considered for risk stratification in these cases.2,3
Cholangiocarcinoma is a rare form of cancer, and spontaneous TLS has only been documented in 2 other cases, both involving males of a similar age and comparable advanced metastatic disease burden. The first case was reported by Ali et al as the first known instance of spontaneous TLS in a patient with metastatic cholangiocarcinoma. They presented a 66-year-old man who presented with right upper quadrant pain, hepatomegaly, and jaundice. Further imaging studies revealed multiple liver metastases and the presence of ascites. Patient’s initial laboratory results indicated elevated levels of uric acid, LDH, and liver enzymes, while renal function and electrolytes remained normal. Despite not receiving any treatment, the patient developed acute renal failure, hyperkalemia, hyperphosphatemia, hypocalcemia, and increasing uric acid levels, fulfilling the Cairo–Bishop criteria. 5
The second case, described by Dong et al, involved a 59-year-old man with newly diagnosed metastatic cholangiocarcinoma who presented with weakness, fatigue, and lightheadedness. Imaging revealed a significant hepatic mass along with extensive pulmonary and hepatic metastases. Initially misdiagnosed with sepsis with multiorgan dysfunction, subsequent laboratory tests revealed hyperuricemia, hyperkalemia, and renal failure, meeting the Cairo–Bishop criteria for TLS. Unfortunately, the patient passed away shortly thereafter due to multiorgan failure. 6
It remains unclear whether cholangiocarcinoma is associated with a higher risk of TLS. However, it is worth mentioning that liver and lung metastases, as seen in our patient, may increase the likelihood of spontaneous TLS. 7 Additionally, some reports suggest that significant hyperuricemia (defined as levels exceeding 8 mg/dL) is more commonly associated with spontaneous TLS rather than other conditions, such as acute kidney injury due to acute tubular necrosis. 8
Conclusion
TLS is a life-threatening oncologic emergency that can arise in various types of cancer. Effective risk stratification and routine laboratory tests are crucial for patients undergoing chemotherapy to ensure early diagnosis of therapy-induced TLS. Unfortunately, while spontaneous TLS is less common than therapy-induced TLS, it is often underdiagnosed due to its atypical presentation and the lack of routine screening in untreated patients. The diagnosis of spontaneous TLS can be easily overlooked due to nonspecific laboratory findings and the tendency to attribute metabolic disturbances to other causes.
Currently, there is no established consensus or criteria for diagnosing spontaneous TLS, making it challenging to identify patients at immediate risk. Further research and efforts are needed in this area. Nonetheless, it is essential to conduct routine tests to monitor and rule out the development of spontaneous TLS in patients with known solid tumors, even if they are not scheduled for treatment in the future.
This approach can help mitigate the potential underdiagnosis of spontaneous TLS, which poses a significant risk of morbidity and mortality in patients with solid tumors, regardless of therapy. Additionally, it may be necessary to adapt or revise the Cairo and Bishop criteria to facilitate and include the diagnosis of spontaneous TLS.
Footnotes
Acknowledgements
The Ethics and Investigations Committee of Hospital Pablo Tobón Uribe for authorizing the review of clinical records. Juan Pérez, MD, for his contribution of pathology reports and histology images and Aaron Bisig, MD, for his radiology consultation and assistance with image selection.
Ethical Considerations
Ethical approval to report this case series was obtained from “Comité de Investigaciones y Ética en Investigaciones—Hospital Pablo Tobón Uribe (HC 1547191).”
Consent to Participate
Verbal informed consent to participate was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.