Abstract
Objective:
Renal fusion anomalies are rare and usually present as horseshoe kidneys or crossed fusion ectopia. The complex renal anatomy seen in patients with these anomalies can present a challenge. Pre-operative planning is therefore paramount in the surgical management of these cases. Herein we report the use of interactive virtual three-dimensional (3D) reconstruction to aid renal surgery in patients with fusion anomalies of the kidney.
Materials and Methods:
A total of seven cases were performed between May 2016 and October 2020. 3D reconstruction was rendered by Innersight Labs using pre-operative computed tomography (CT) scans.
Results:
Five patients had malignant disease and two patients had benign pathology. Robotic and open operations were performed in four and three patients, respectively.
Conclusion:
The use of 3D reconstruction in the cases reported in this series allowed for the identification of variations in renal vasculature, and this informed the choice of operative approach.
Oxford Centre for Evidence-Based Medicine Evidence Level:
4
Keywords
Introduction
Fusion anomalies of the kidney are rare but most often present as horseshoe kidneys, complex crossed fused renal ectopia or fused pelvic kidneys (cake kidneys) in about 1 in 400 or 1 in 1000 people, respectively.1,2 The surgical management of benign and malignant diseases involving horseshoe kidneys and crossed ectopic fusion anomalies can be challenging, owing largely to the complex anatomy and the difficulty of mobilisation. Advancements in surgery have enabled the use of minimally invasive approaches in this population and optimal pre-operative planning using three-dimensional (3D) reconstructed images from computed tomography (CT) scans, can aid surgery. We report a case series of partial nephrectomies and hemi-nephrectomies carried out with the benefit of pre-operative 3D reconstruction in patients with horseshoe kidney, crossed fused renal ectopia and fused pelvic kidneys.
Materials and methods
We retrospectively reviewed the records of seven patients with horseshoe kidneys, crossed fused renal ectopia and fused pelvic kidneys who underwent a partial nephrectomy or heminephrectomy between May 2016 and October 2020 and had signed appropriate consent for data collection. 3D reconstruction was carried out by Innersight Labs based on routine pre-operative CT scans using an observer-independent automated algorithm as described previously. 3 The automated segmentation was quality-edited by biomedical engineers (Innersight Labs) and verified by a clinical expert in the clinical setting. The cases are summarised in Table 1, a detailed report for each case is provided online (Supplemental Material).
Summary of cases.
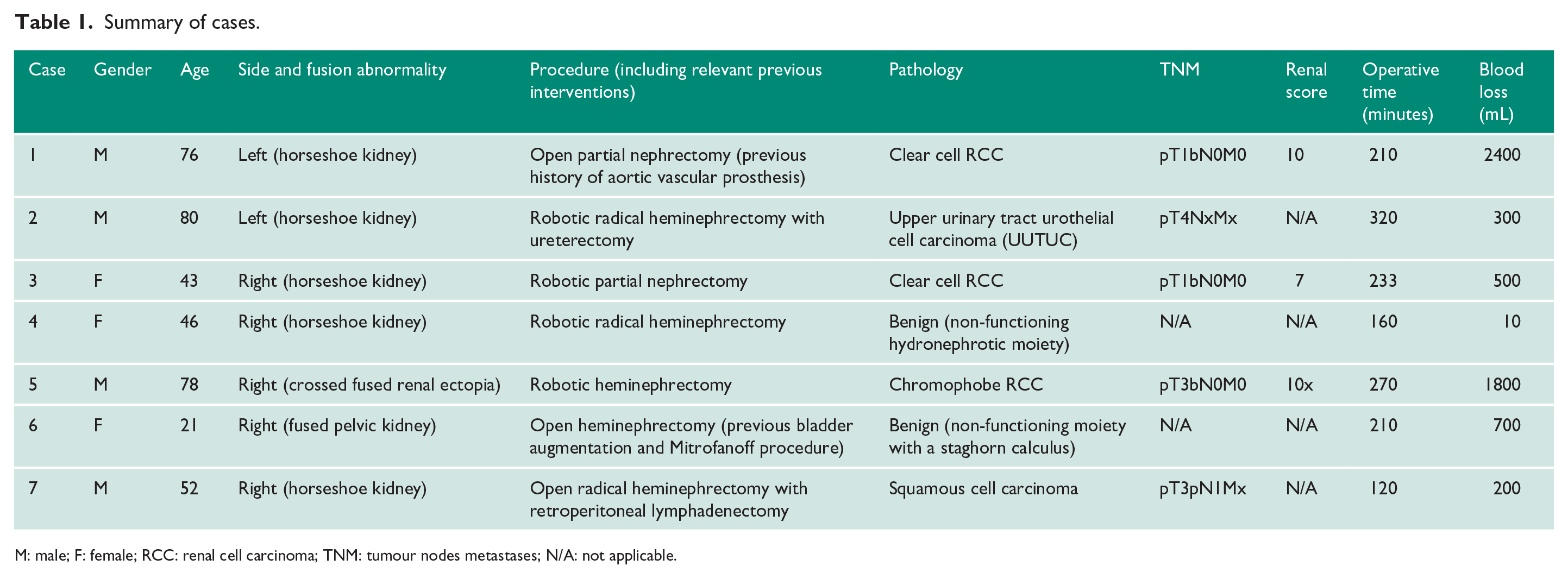
M: male; F: female; RCC: renal cell carcinoma; TNM: tumour nodes metastases; N/A: not applicable.
For the open-surgical approach, patients were in the supine position and access was created through a subcostal or midline incision. In patients who underwent robotic surgery, Veress needle insufflation was carried out after which an 8 mm camera port was inserted near the border of the lateral rectus muscle, all other ports were placed under direct vision, and included three further 8-mm ports and a 12-mm periumbilical assistant port.
Results
The case details and perioperative outcomes are summarised in Table 1.
Figure 1 shows the 3D reconstruction for each case together with a corresponding representative CT section of the renal pathology. Links to online 3D reconstructions are provided in supplementary material. Figure 2 demonstrates the complex anatomy in a patient with a history of previous open infrarenal aortic vascular prosthesis insertion for which a partial nephrectomy in the left moiety was planned (Case 1).
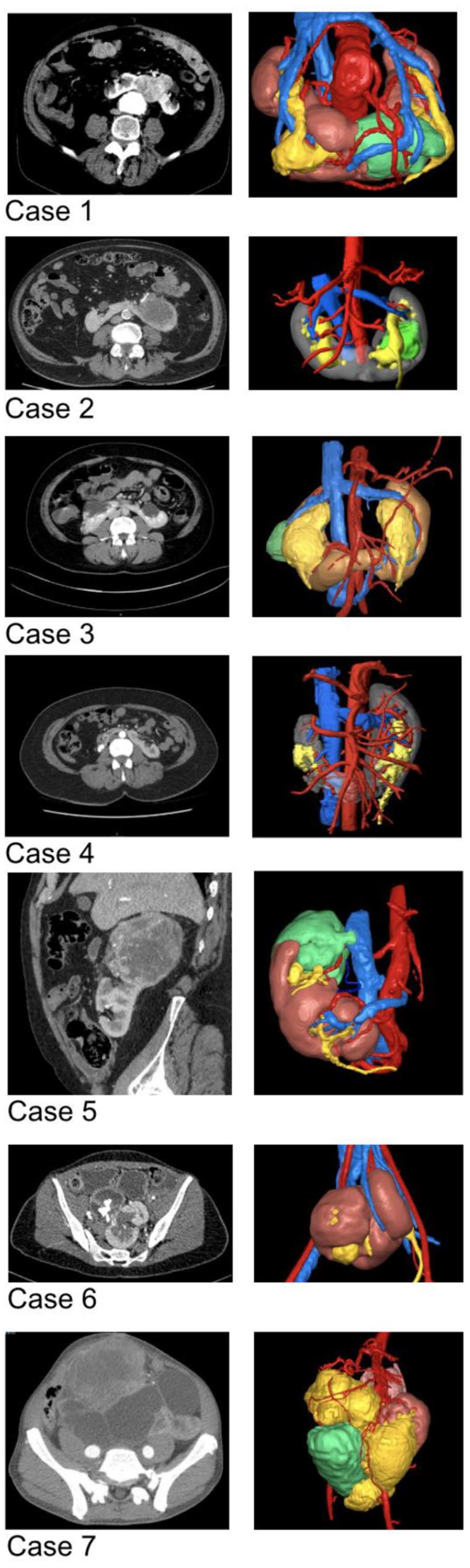
Abdominal CT scans (left) and the corresponding 3D reconstructions (right) for the patients in this series.
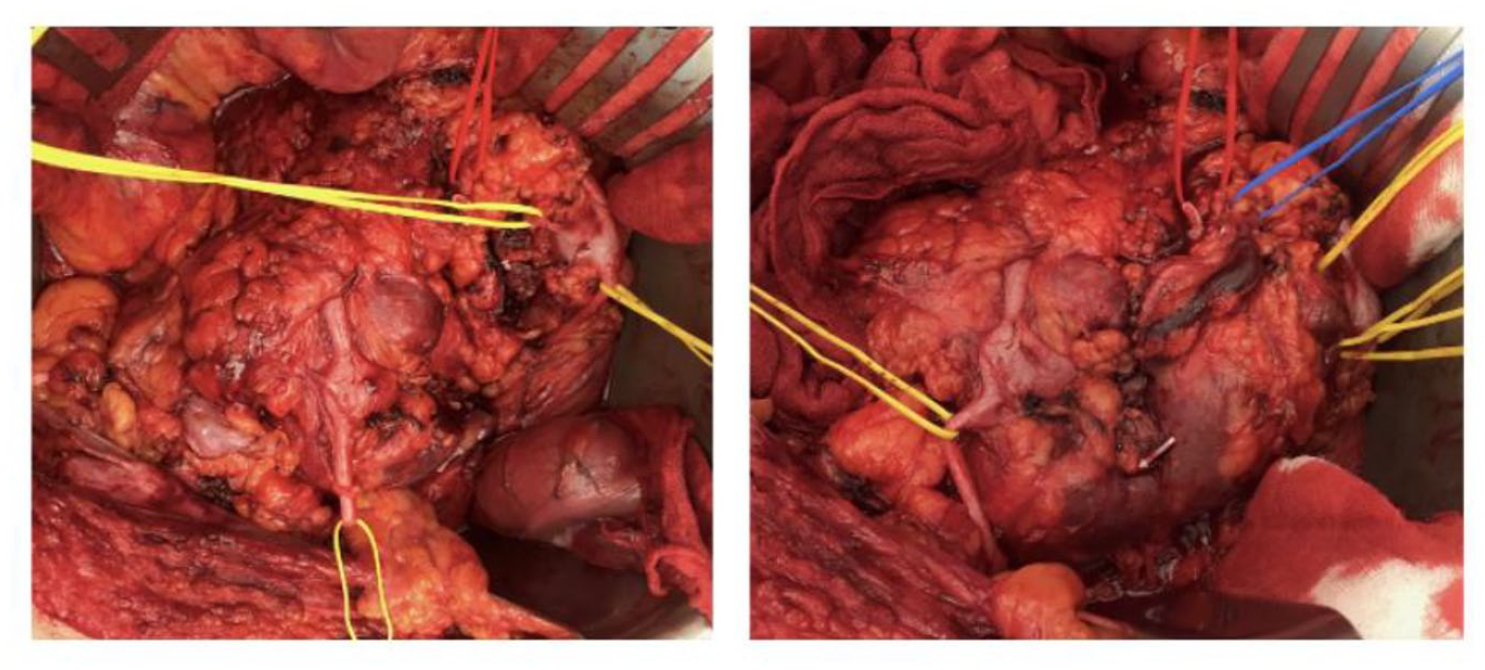
Intraoperative images from Case 1 with blood vessels (red, blue sloops) as well as ureters and renal pelvis (yellow sloops) identified.
Discussion
Patients with horseshoe kidneys and other renal fusion anomalies are more susceptible to certain urological conditions. There is a higher risk of developing urinary tract infections, nephrolithiasis, nephroblastomas, and urothelial cancers, but the incidence rate of renal cell carcinoma is comparable to the general population. 4 The surgical management of renal pathology in patients with renal fusion anomalies is challenging, largely due to the unusual anatomy which impacts on the mobilisation of the kidney and requires more comprehensive pre-operative planning.
As can be seen from our experience, the vascular anatomy of horseshoe kidneys and other renal fusion anomalies is quite variable. Although Graves 5 described distinct vascular presentations of horseshoe kidneys, there have been shown to be exceptions to this, with up to 64% showing a deviation from his classification in one study. 6 The complexity of the vasculature and the lack of a collateral blood supply means that there is an increased risk of complications from the procedure. 6 Knowledge of the patient’s vasculature will aid the surgeon in the choice of operative approach.
Despite the potential difficulties of operating on renal fusion anomalies, 7 minimally invasive approaches have been shown to be feasible and safe. Laparoscopic surgery in this population is becoming increasingly common, and a recent case series with 19 patients who underwent laparoscopic surgery for tumours in their horseshoe kidney reported no complications greater than Clavien-Dindo grade II. 8 In high-volume centres, robotic surgery is likely to offer many potential benefits, such as shorter post-operative stay and less intraoperative blood loss.
3D reconstruction is a useful tool for pre-operative planning of complex renal cases. Glybochko et al. 9 noted that 3D reconstruction is beneficial compared to 2D imaging because it allows for greater accuracy and precision in determining the location and measurements of anatomical structures. This is of particular importance when performing a partial nephrectomy or heminephrectomy in renal fusion anomalies with their unpredictable vascular supply. 10 In our series, 3D reconstruction assisted in the identification of complex anatomy such as in Case 1 with secure identification of the vasculature following previous open aortic prosthesis surgery, or for example in Cases 5 and 6. In Case 5, 3D reconstruction enabled us to access the main renal artery early on and to recognise the independent blood supply to the lower moiety. This allowed a clampless heminephrectomy and a relatively safer access to the caval tumour thrombus. In Case 6, in which an augmented bladder with a Mitrofanoff was located anterior to the fused pelvic kidney the course of the right and left blood supply was clearly identified along with the site of fusion. A limitation of our case series is that the utility of 3D reconstruction to facilitate renal surgery and improve patient outcomes and costs has not been investigated in a randomised setting in comparison to standard 2D cross-sectional imaging.
Conclusion
In our experience, partial nephrectomies and hemi-nephrectomies are feasible in patients with renal fusion anomalies, using both open and minimally invasive approaches. Pre-surgical planning with interactive virtual 3D reconstruction demonstrates the complex anatomy and informs the choice of procedure, surgical approach, and patients selected for surgery.
Supplemental Material
sj-docx-1-uro-10.1177_20514158211068310 – Supplemental material for Interactive virtual 3D image reconstruction to assist renal surgery in patients with fusion anomalies of the kidney
Supplemental material, sj-docx-1-uro-10.1177_20514158211068310 for Interactive virtual 3D image reconstruction to assist renal surgery in patients with fusion anomalies of the kidney by Naomi Morka, Lorenz Berger, Eoin Hyde, Faiz Mumtaz, Ravi Barod, Prasad Patki, Niels Graafland, Kees Hendricksen, Maxine Tran and Axel Bex in Journal of Clinical Urology
Footnotes
Conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.B. and E.H. are employed by Innersight Labs. All other authors report no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Contributorship
N.M. and A.B. were involved in the conception, design, data acquisition, and drafting of the original manuscript. F.M., R.B., P.P., N.G., K.H. and M.T. provided resources. A.B., F.M., R.B., P.P., N.G., K.H., M.T., E.H. and L.B. critically revised the manuscript. E.H. and L.B. assisted in data interpretation and the use of software. A.B. supervised the work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
