Abstract
A 77-year-old man was diagnosed with an infrarenal abdominal aortic aneurysm coexisting with a horseshoe kidney detected on a computed tomography (CT) scan. Since the presence of three accessory renal arteries was confirmed, reconstruction for renal protection was done by performing transperitoneal laparotomy instead of endovascular treatment. After performing anastomosis of a Y-shaped artificial graft, the major accessory renal artery was reconstructed under intraoperative renal perfusion. We report a successful surgical repair without renal isthmus division, where only autologous blood transfusion was performed. Although a postoperative CT scan showed partial renal infarction, creatinine levels, indicative of renal function, were comparable to preoperative levels. Horseshoe kidney is the most common fusion defect of the kidneys with aberrant accessory renal arteries. Thus, clearly constructed images are key to establish an effective operative strategy. Furthermore, comparing preoperative and postoperative CT images could also provide important feedback on the accuracy of treatment strategy.
Keywords
Introduction
Horseshoe kidney (HSK) is the most common congenital kidney anomaly, with a prevalence of 0.2%. 1 It is characterized by medial fusion of the kidneys anterior to the aorta and beneath the inferior mesenteric artery. The concomitant presence of HSK in patients with abdominal aortic aneurysm (AAA) is rare. Planning an appropriate surgical strategy, such as the transperitoneal or retroperitoneal approach or endovascular surgery, is challenging for surgeons due to the complexity of various accessory renal arteries. Clearly constructed images play a vital role in developing an operative strategy. Visualizing the arteries, neighboring renal vein, and ureter on a preoperative CT scan is important. The CT scan was instrumental in developing an operative strategy: reconstructing the accessory renal arteries, controlling lumbar artery bleeding that was not clearly visible due to the bifurcation behind the isthmus, and avoiding injury to the neighboring tissue. Comparing preoperative and postoperative CT images could provide feedback on the accuracy of operative strategy.
Case
During a workup for mitral valve regurgitation and atrial fibrillation surgery 3 years prior, a 41 × 43 mm infrarenal AAA and HSK were detected on CT in a 77-year-old man with a history of hypertension. Despite the absence of abdominal pain on physical examination, the maximum diameter of the AAA expanded to 48 × 54 mm, with a right common iliac artery aneurysm measuring 25 mm, warranting surgical treatment. The renal isthmus was positioned over the AAA and had accessory renal arteries (ARAs): two on the right side and one on the left (Figure 1). The Crawford classification was Type Ⅲ. 1
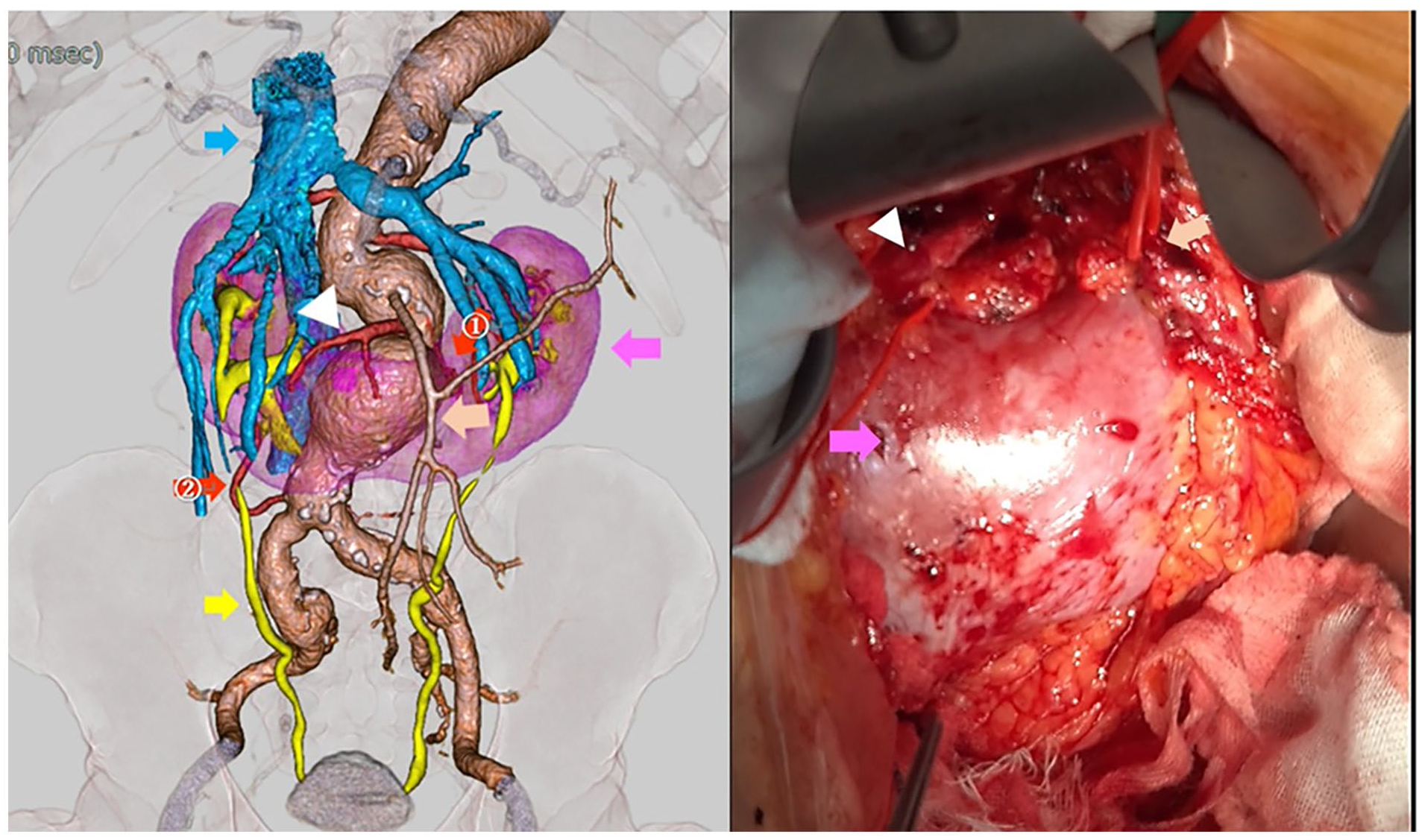
Preoperative enhanced computed tomography and intraoperative findings. The abdominal aortic aneurysm, horseshoe kidney (pink arrow), accessory renal arteries (the diameters are white arrowhead: 6.1 mm, red arrow ①: 2.4 mm, and red arrow ②: 3.3 mm), renal veins (blue arrow), inferior mesenteric artery (beige arrow), and ureter (yellow arrow).
Owing to the need for ARA reconstruction, the patient underwent open surgery. The aneurysm was approached with a transperitoneal midline laparotomy, not a peritoneal approach, because a major part of the major accessory renal artery was located on the right side and the isthmus slightly adhered to the aneurysm.
The bilateral iliac arteries and abdominal aorta were clamped after the dissection of the proximal neck of the aneurysm and heparin (100 unit/kg) administration. An aortotomy was then performed, during which the lumbar arteries were ligated. A 10-Fr cannula was inserted into the major ARA stump on the right, measuring 6.1 mm in diameter. In addition, a cold crystalloid solution was manually delivered (500 mL), with an additional 100 mL administered every hour. The perfusate consisted of 1000 mL of lactated Ringer’s solution with mannitol (4 gm/L), sodium carbonate (1.68 g/L), heparin sodium (2000 unit/L), and methylprednisolone (125 mg/L), cooled to 4℃. 2 Owing to the strong enhancement of the isthmus observed in the CT scan, symphysiotomy was not performed. After the Y-shaped artificial graft (a 16 mm × 8 mm Dacron graft) was anastomosed, the major ARA on the right was reconstructed to the left leg of the graft due to the natural positioning of the renal artery orifice (Figure 2).

Intraoperative findings and postoperative enhanced computed tomography. The renal isthmus (yellow arrow) is positioned over the abdominal aorta and reconstructed accessory renal artery (white arrowhead) on the left leg of the graft and a partial renal infarction (white arrow).
Subsequently, the pair of bilateral small ARAs and the inferior mesenteric artery were ligated.
The ischemic time for the ARA was 60 min; only autologous blood transfusion was performed.
The postoperative CT scan clearly showed partial renal ischemia and the reconstructed accessory renal artery. No postoperative complications occurred; postoperative serum creatinine level was maintained at 0.91–1.10 mg/dL, similar to preoperative levels. The CT scan also indicated that the ligation of ARAs was not harmed. The patient was discharged and was healthy during outpatient follow-up till now.
Discussion
HSK is the most common congenital kidney anomaly, with a prevalence of 0.2%. It is characterized by medial fusion of the kidneys anterior to the aorta, beneath the inferior mesenteric artery, and anomalous rotation of the urinary tract. The concomitant presence of HSK in patients with AAA is rare, observed in only 0.12% of cases. 3 The pathophysiology underlying the interaction between HSK and abdominal aortic aneurysm is unknown; it usually involves arteriosclerosis or congenital abnormality of the cell component of the aortic wall. Since the patient’s aortic wall dilation was the largest behind the isthmus, friction between the pulsative aortic wall and isthmus might have induced aortic wall dilation. Planning an appropriate surgical strategy for managing these patients, such as a transperitoneal or retroperitoneal approach or endovascular surgery, is challenging for surgeons. Clearly constructed images play a vital role in the operative strategy, and reconstruction of an ARA > 2 mm in diameter is recommended to avoid renal ischemia. 1 We ligated the small-sized renal artery (number ① and ②, Figure 1). Although the renal ischemia shown on the postoperative CT scan (Figure 2) was greater in area than expected, postoperative serum creatinine levels were similar to its preoperative levels. Furthermore, although aneurysm repair in the modern era is mostly achieved endovascularly, 4 open surgeries may have a crucial role in cases of complex renal artery perfusion. If our case was managed via an endovascular approach, complex procedures such as fenestration configuration for the renal artery and long-term durability such as endoleak would have been required, posing concerns about adverse effects on renal function. Concomitant AAA with HSK involving major ARAs should be treated with open surgery considering the patient’s operation tolerance. HSK has clinical relations to renal trauma and tumors, and artificial graft infection is a concern about surgical re-laparotomy of the kidney in the future. 5
To conclude, surgical repair of AAA complicated by HSK might be challenging for surgeons due to the complexity of various ARAs. Clearly constructed images play a vital role in establishing an operative strategy. Furthermore, comparing the preoperative and postoperative CT images could provide important feedback on the accuracy of the treatment strategy.
