Abstract
Introduction
The risk of hepatocellular carcinoma persists in some patients despite achieving sustained virologic response with current interferon-free direct-acting antiviral therapy for hepatitis C. The subject of an even higher carcinoma risk in this context has been reported and is currently being debated. The quest for understanding this paradox relative to the dynamics of inflammatory biomarkers in cirrhosis patients receiving antiviral therapy thus remains a subject of importance.
Objective
Here, we aimed at evaluating the effects of direct-acting antiviral therapy-induced hepatitis C cure on plasmatic markers of systemic inflammation measured before, during and after treatment. Specifically, soluble immune mediator phenotype associations that impact the odds of hepatocellular carcinoma development and the related changes that arise upon direct-acting antiviral-mediated hepatitis C clearance in cirrhosis patients was investigated.
Methods
Employing multiplex technology that measured up to 91 circulating biomarker proteins, we profiled the plasma soluble immune mediator concentrations of cirrhosis patients who developed post-treatment hepatocellular carcinoma and their respective negative controls, before and after direct-acting antiviral treatment.
Results
Elevated pre-therapy concentrations of specific soluble immune mediators including MCP-3, GDNF, CDCP1, IL-17C, IL-17A, SLAMF1, CCL11, FGF-5, LIF-R, IL10, IL-10RA, IL-15RA, beta NGF, CCL28, CCL25 and NT-3 distinguished patients who developed post-treatment hepatocellular carcinoma relative to those that did not. Particularly, GDNF, FGF-5 and IL-15RA displayed independent predictive biomarker attributes for delineating carcinoma emergence regardless of
Conclusions
These results have considerable implications for delineating potential hepatocellular carcinoma emergence before initiating direct-acting antiviral therapy for hepatitis C in cirrhosis patients. They provide preliminary contribution to unravelling cases where the benefit of direct-acting antiviral therapies would be superior to the risk of developing carcinoma.
Keywords
Key summary
Established knowledge on this subject
Current interferon (IFN)-free direct-acting antivirals (DAAs) are effective at eliminating hepatitis C virus (HCV), but risks of residual liver disease and development of hepatocellular carcinoma persists. The hepatic inflammation that occurs during chronic hepatitis C causes systemic changes in blood soluble immune mediators (SIMs) that impact carcinogenetic processes involved in the growth, invasion and metastasis of hepatocellular carcinoma (HCC). DAA-induced HCV cure does not lead to a complete immunological restitution of the altered soluble inflammatory compartment in chronic hepatitis C.
Significant and/or new findings of this study
An elevated pre-therapy plasma profile of an extended repertoire of SIMs in cirrhosis was associated with HCC development post-DAA therapy. Successful DAA therapy did not alter the baseline elevated plasma SIM profile of cirrhosis patients that developed post-therapy HCC contrary to its effect in those that remained HCC-free.
Introduction
Hepatocellular carcinoma (HCC) still emerges in some patients despite clearance of hepatitis C virus (HCV) upon successful antiviral therapy. This has been shown not only for interferon (IFN)-based therapies 1 but also for therapies using IFN-free direct-acting antiviral (DAA) regimens. 2 Several studies have demonstrated that HCV cure by IFN-free DAA therapy does not seem to alter the short-term risk of HCC emergence, as it tends to remain still high, particularly in patients with established liver cirrhosis.3–6 There have been further concerns as to whether the risk of HCC recurrence may even be increased following IFN-free HCV therapy with DAAs. 7 A need thus exists to evaluate whether, and to what extent, HCC development is impacted by hepatic immune events in cirrhosis patients who receive IFN-free DAA therapy.
Preliminary reports provided an indication of a possible association between serum levels of distinct cytokines and the development of HCC.
8
,
9
Specifically, HCV clearance by IFN-free DAA therapy was reported to coincide with the induction of a rapid reduction in inflammation but increase in key HCC angiogenesis drivers such as the Vascular Endothelial Growth Factor (VEGF); an immune balance modification that may affect the anti-tumour surveillance machinery.
9
Further, a potential pre-treatment modification of a repertoire of soluble immune mediators in the serum of patients who eventually developed
Materials and methods
Patient population
In this study, we enrolled a total of 31 patients with baseline HCV-related liver cirrhosis who had undergone IFN-free hepatitis C therapy with DAAs at the liver outpatient clinic of Hannover Medical School (MHH). The criteria of cirrhosis diagnosis were based on liver histology (F5 and F6 according to ISHAK (modified Knodell score)), transient elastography (≥14.5 kPa), and definite morphological signs in ultrasound, magnetic resonance imaging (MRI) and computed tomography (CT) as detailed previously.
3
All patients had no co-existent chronic inflammatory disease(s) or any known cancers other than HCC. Of these patients, 15 were those who had developed post-treatment HCC at some point during treatment or within a 24-week follow-up period (herein referred to as Cirrh-to-HCC). Control groups including 16 age-matched patients who remained HCC-free upon therapy (herein referred to as Cirrh-to-NoHCC) and 10 healthy individuals were also recruited. All patients were HCC-free by ultrasound, CT or MRI technology before therapy start. In furtherance, an HCC emergence surveillance schedule was routinely done upon therapy initiation for all patients during antiviral therapy and during a six-month follow-up period. This surveillance schedule involved ultrasound imaging routinely performed at three-month intervals during antiviral therapy and 6 months after therapy. This surveillance schedule is borne out of routine clinical practice at our centre. The Cirrh-to-HCC patients included four recurrences and 11
Characteristics of patient cohorts and a description of the different DAA regimens for treatment according to international guidelines (HCV guidelines, European Association for the Study of the Liver (EASL), American Association for the Study of Liver Diseases (AASLD)) have been detailed in Table 1.
Individual patient characteristics.
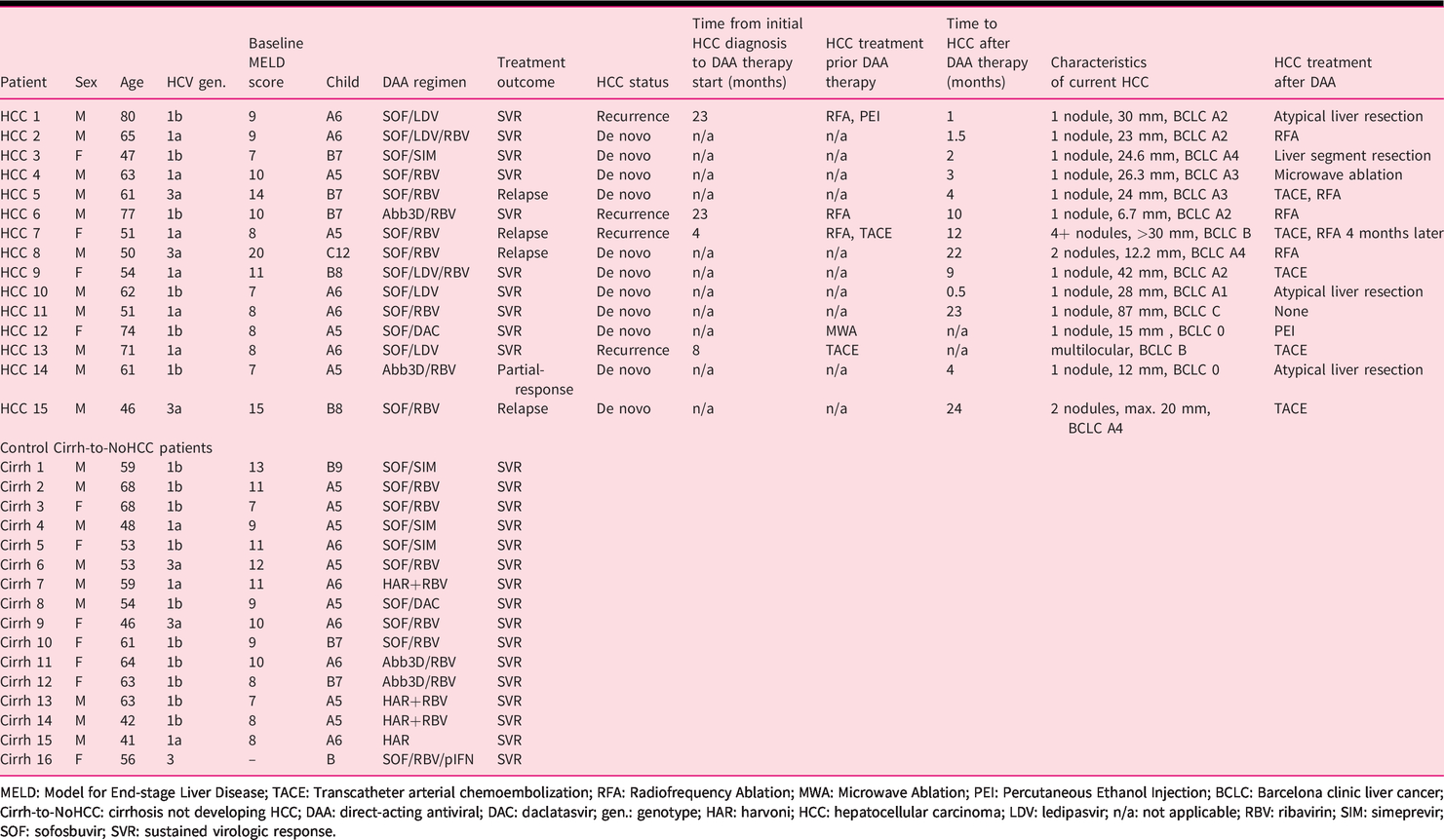
MELD: Model for End-stage Liver Disease; TACE: Transcatheter arterial chemoembolization; RFA: Radiofrequency Ablation; MWA: Microwave Ablation; PEI: Percutaneous Ethanol Injection; BCLC: Barcelona clinic liver cancer; Cirrh-to-NoHCC: cirrhosis not developing HCC; DAA: direct-acting antiviral; DAC: daclatasvir; gen.: genotype; HAR: harvoni; HCC: hepatocellular carcinoma; LDV: ledipasvir; n/a: not applicable; RBV: ribavirin; SIM: simeprevir; SOF: sofosbuvir; SVR: sustained virologic response.
Measurement of plasma SIM concentrations
We performed multianalyte plasma SIM profiling employing a multiplex technology (Olink’s Proseek Multiplex Inflammation), which simultaneously measured the expression of 91 multiple biomarker proteins (Supplementary Material Table S1), based on a proximity extension assay (PEA). The precision, reproducibility and scalability of this SIM measurement technology have been previously described.12–14 Plasma SIM concentrations of patients were analysed at baseline, and longitudinally at the end of therapy and follow-up of therapy as detailed in the Supplementary Material. The precise time-points at which plasma were sampled and used for SIM assessments is detailed in Supplementary Material Table 2. Based on a standardised limit of detection (LOD), a total of 17 proteins that had a missing data frequency of more than 45% were excluded from the analysis.
Statistical analyses
Data were analysed using GraphPad Prism software (GraphPad Software, La Jolla, California, USA) or Microsoft Excel (for spider graphs). Quantitative analyses were done using the student
Results
Pre-therapy elevated plasma SIM profiles characterised cirrhosis patients that developed HCC upon DAA therapy
Circumventing the low throughput of conventional methods, we here applied the innovative Proseek multiplex technology to profile multiple SIMs (
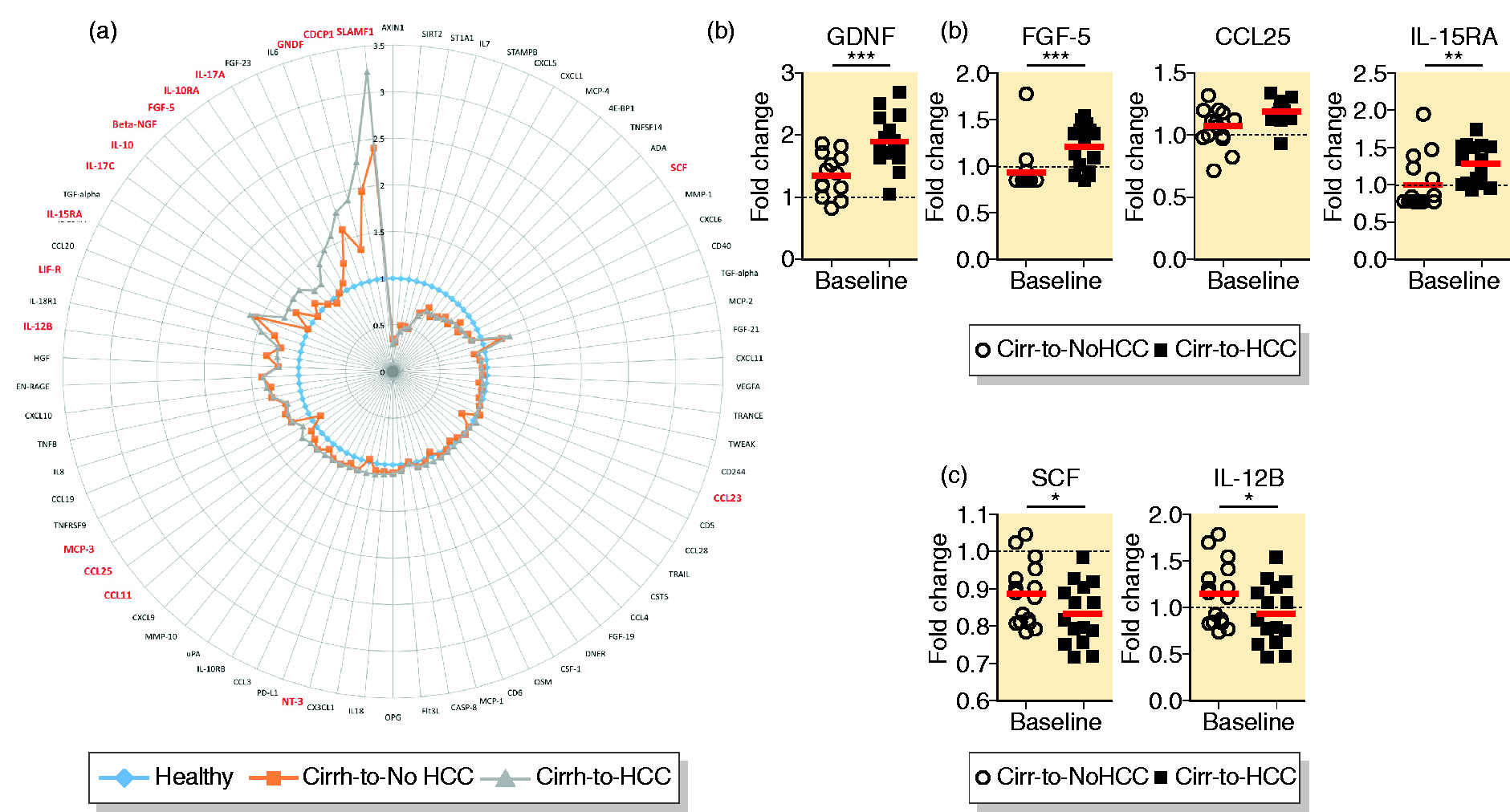
Plasma concentrations of soluble immune mediators (SIMs) at baseline. (a) Comparative pre-therapy SIM concentrations of cirrhosis developing hepatocellular carcinoma (Cirrh-to-HCC) and cirrhosis not developing hepatocellular carcinoma (Cirrh-to-NoHCC) patients relative to healthy controls. Significant variables are indicated in red. (b) Representative graphs of individual SIMs with increased concentrations at baseline in Cirrh-to-HCC patients with statistical
Further calculating the area under the receiver operating characteristic curves (AUROCs), three of the 16 elevated SIMs showed values above 0.80 (Figure 2(a)). We further evaluated the potential correlation between baseline plasma SIM levels and the emergence of recurrence and
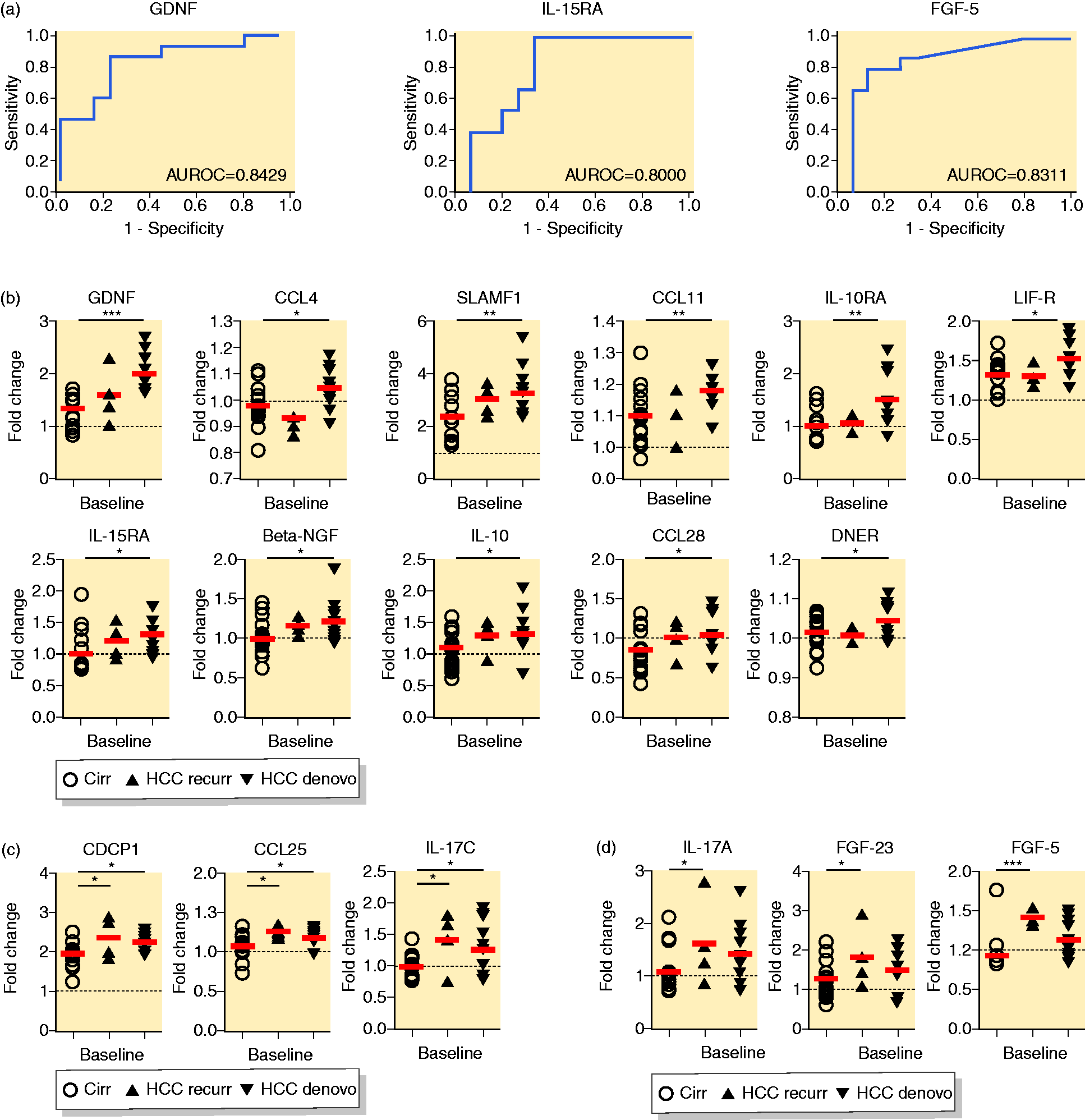
Association between baseline plasma soluble immune mediator (SIM) levels and the emergence of post-therapy hepatocellular carcinoma (HCC). (a) area under the receiver operating characteristic curve (AUROC) of markers of HCC development with values of ≥0.800 as compared to non-HCC control group. (b) Baseline plasma SIMs with concentrations higher exclusively in
Successful DAA therapy did not alter the baseline plasma SIM profiles in cirrhosis patients that developed post-therapy HCC
To further assess the possible existence of a differential regulation of distinct SIMs during IFN-free treatment of hepatitis C in relation to HCC emergence, we studied the plasma SIM kinetics in the two Cirrh-to-HCC and Cirrh-to-NoHCC patient cohorts (Figure 3). Plasma were longitudinally sampled at baseline (therapy start), end-of-therapy and follow-up (i.e. at least 12 weeks post sustained virologic response (SVR)) and the concentrations of the described SIMs measured. Patient Cirrh-16 who had received IFN-based therapy was exclude from the analysis. Patients Cirrh-15, HCC-1, HCC-9 and HCC-14 for whom no plasma samples were available at end-of-therapy and/or follow-up were also excluded from the analysis. We observed that while the HCC-free control cohort experienced significant therapy-mediated SIM reductions of some
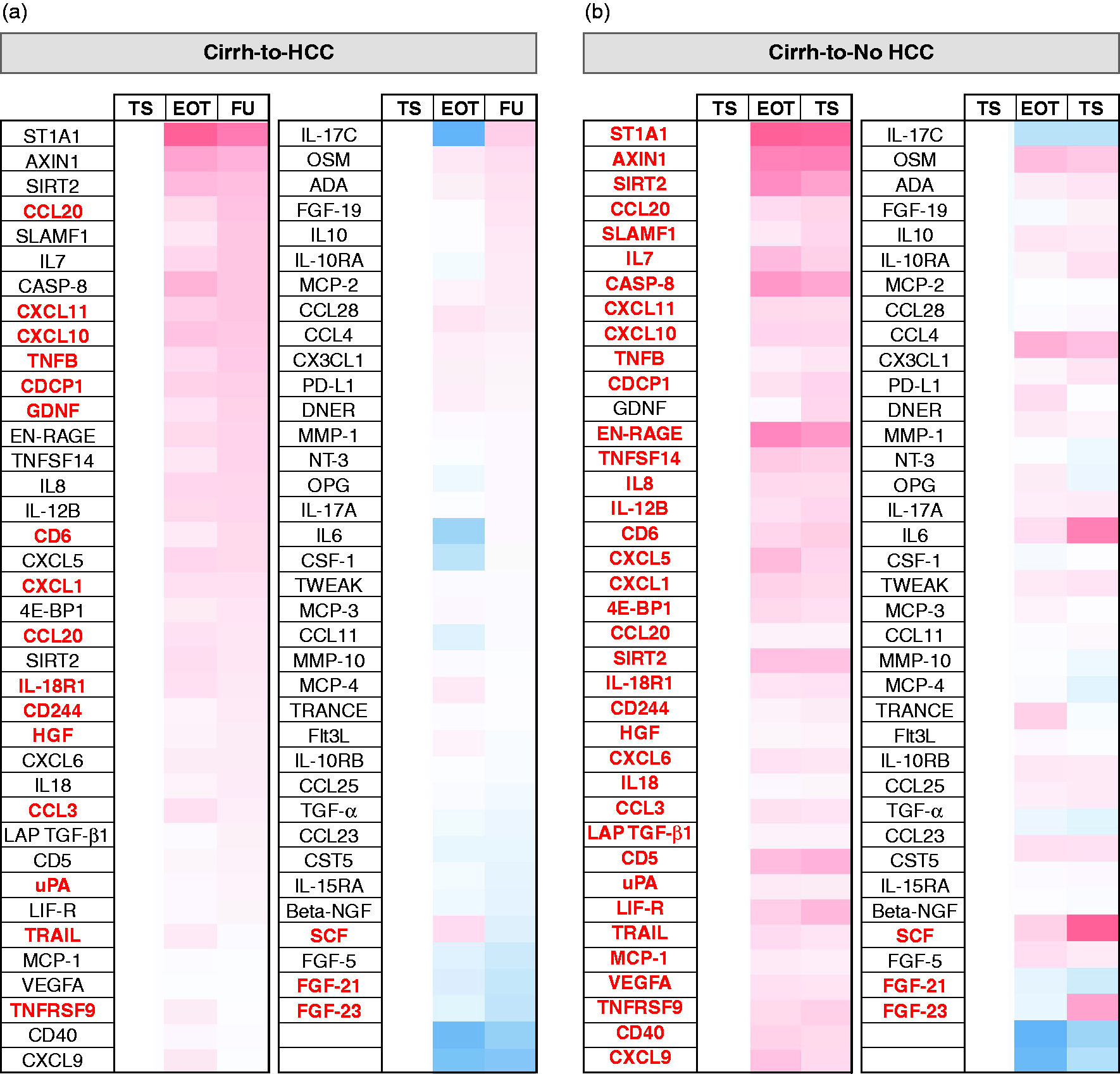
Effect of interferon (IFN)-free direct-acting antiviral (DAA) therapy on plasma soluble immune mediator (SIM) levels. Fold changes in plasma SIM concentrations of the cirrhosis patient cohorts that upon DAA therapy either developed HCC (Cirrh-to-HCC) or remained without any HCC (Cirrh-to-NoHCC) were calculated at end-of-therapy (EOT) and follow-up (FU) time-points in reference to baseline values. (a) Plasma SIM kinetics of Cirrh-to-HCC patients from therapy start (TS) through EOT to FU. (b) Plasma SIM kinetics of Cirrh-to-NoHCC patients from TS through EOT to FU. A one-way analysis of variance (ANOVA) with the recommended Geisser-Greenhouse correction was used for statistical evaluations. Significant variables are indicated in red.
The few SIMs that appeared normalised in the Cirrh-to-HCC cohort seemed to emanate from the
Discussion
In this plasma screening of a large repertoire of SIMs relative to HCC emergence upon IFN-free therapy for hepatitis C, we showed that a spectrum of pre-treatment SIM expressions were highly elevated in patients who later developed HCC and distinguished them from those who did not. Furthermore, pre-treatment SIM levels could differentiate
Chronic unresolved inflammation is known to promote and exacerbate malignancies. A clear example is HCC, which has over 90% of its cases arising in the context of liver injury and inflammation. Our study here suggests a pre-existing hyper-activated profile in Cirrh-to-HCC patients as evidenced by elevated baseline concentrations of a myriad of SIMs. To the best of our knowledge, these findings show for the first time that elevated pre-therapy SIM concentrations (such as those of GDNF, FGF-5 and IL-15RA) could serve as independent predictive biomarkers for HCC emergence, and distinguish Cirrh-to-HCC from Cirrh-to-NoHCC patients regardless of
Aside from IL-10, the signalling lymphocytic activation (SLAM) molecules are known for their roles in regulating immune responses, pathophysiology of neoplasm transformations and entry pathways of certain viruses.
21
Recently, SLAM molecules have come to the fore for their potential in diagnosis and therapy of various cancers. The signalling lymphocytic activation molecule family 1 (SLAMF1) in particular is the prototype member of the SLAM family of molecules that initiates signal transduction networks in many immune cells including T and B cells, dendritic cells, monocytes-macrophages, Natural Killer (NK) cells and Natural Killer T (NKT) cells that constitutively express them, thus modulating their activation and differentiation.22–24 In patients with colon cancer, for example, up-regulation or silencing of SLAMF1 expressed in lymphocytes was reported to be accompanied by increased or reduced lymphocytic cytotoxic activity respectively.
25
In our Cirrh-to-HCC patient cohort, the elevated SLAMF1 profile existent before DAA therapy initiation was an interesting observation in this regard. On the one hand, it fits very well into our assertion that the elevated SIM profile (as exemplified by SLAMF1) and characteristic systemic inflammation, may be a response to occult HCC in the liver. However, the characteristically higher SLAMF1 level in the patients who developed
Furthermore, assessing the potential immune correlates of IFN-free therapy-mediated HCV clearance with emergence of HCC by analysing longitudinal SIM kinetics, we confirmed our earlier report that innate SIM immunity may not necessarily normalise but experience strong reduction in the levels of specific SIMs in the cirrhosis control cohort that did not develop HCC.
26
Previous studies had reported a similar decline in the mean levels of the SIMs IP-10, MCP-1, MIP-1β, IL-18
19
and also of CCL-2, CCL-3, CCL-4, CXCL-8, CXCL-10, IL-1
On several scores, our findings in this study confirm and extend previous reports, especially by Debes et al. 8 and Villani et al. 9 However, one notable exception deserves a mention. The kinetics of plasma VEGF levels which were reported to be elevated until end-of-therapy before it normalised at SVR12 in chronic hepatitis C patients receiving IFN-free DAA therapy could not be confirmed. In our observation, Vascular Endothelial Growth Factor A (VEGFA) levels significantly declined in the cirrhosis patients that remained HCC-free post-therapy at both end-of-therapy and follow-up contrary to the unaltered levels in the patients who eventually developed HCC post-therapy, suggesting an association between VEGFA levels and HCC growth. On the contrary, other growth factors such as FGF-21, FGF-23 and stem cell factor (SCF) significantly increased in both cohorts in coincidence with HCV clearance whilst TGF-alpha, FGF-5, beta-NGF and other mediators such as IL-15RA, CCL25, CCL23 and CST5 all trended upwards exclusively in the Cirr-to-HCC cohort. The differences in observations could stem from the difference in sample size, peculiar cohort characteristics and the contributory role of the other members of the VEGF family that were non-discriminatorily measured in Villani et al. 9 compared to our assay which measured only VEGFA.
Our study has obvious strengths and limitations. The major strengths include the (a) prospective collection of samples (b) large panel of SIMs analysed (c) unbiased approach of SIM profiling and (d) homogeneous treatment cohort and follow-up for screening. Notwithstanding, the number of HCC cases which still remains small and the accompanying small fractions of recurrence and
In summary, we discovered that the pretreatment activation profile of the soluble immune compartment as measured by highly elevated SIM patterns correlates with post-therapy HCC development. We further demonstrate how the elevated SIM establishment existent before DAA therapy stays unperturbed upon DAA therapy in patients who develop post-therapy HCC contrary to those who remain HCC-free. These findings provide an important basis for a potential build-on to unravel cases where the benefit of DAA therapies would be unequivocally superior to the risk of developing HCC. Attainment of this feat would potentially contribute to improving the management of HCC and the quality of life of patients with chronic hepatitis C.
Ethics approval
The study protocol was approved in year 2014 by the ethics committee of Hannover Medical School (Study numbers: 2148-2014 and 2604-2014) and conducted as per the Helsinki declaration.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620976991 - Supplemental material for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620976991 for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients by Solomon Owusu Sekyere, Kerstin Port, Katja Deterding, Markus Cornberg and Heiner Wedemeyer in United European Gastroenterology Journal
Supplemental Material
sj-pdf-2-ueg-10.1177_2050640620976991 - Supplemental material for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients
Supplemental material, sj-pdf-2-ueg-10.1177_2050640620976991 for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients by Solomon Owusu Sekyere, Kerstin Port, Katja Deterding, Markus Cornberg and Heiner Wedemeyer in United European Gastroenterology Journal
Supplemental Material
sj-pdf-3-ueg-10.1177_2050640620976991 - Supplemental material for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients
Supplemental material, sj-pdf-3-ueg-10.1177_2050640620976991 for Inflammatory patterns in plasma associate with hepatocellular carcinoma development in cured hepatitis C cirrhotic patients by Solomon Owusu Sekyere, Kerstin Port, Katja Deterding, Markus Cornberg and Heiner Wedemeyer in United European Gastroenterology Journal
Footnotes
Acknowledgements
The authors wish to thank all of the participants who volunteered in this project. They are also very grateful to all physicians and study nurses at the liver outpatient clinic of Hannover Medical School. Finally, the authors appreciate the work of Olink Proteomics, Uppsala, Sweden in their innovative Proseek methodology for the plasma SIM measurements and for their data validation service. The authors of this study contributed as follows: conceptualization, SOS and HW; methodology, SOS and HW; Olink Proteomics, Uppsala, Sweden; software, SOS; KP; validation, SOS; HW; Olink Proteomics; formal analysis, SOS; HW; investigation, SOS; resources, HW; MC; KD; data curation, SOS, KP; writing – original draft preparation, SOS; writing – review and editing, SOS, KP, KD and HW; visualization, SOS and HW; supervision, HW; funding acquisition, HW.
Declaration of conflicting interests
SOS and KP have no conflicts of interest to declare. KD has received lecture fees from Gilead, AbbVie and Merck. MC has received lecture fees from AbbVie, Bristol-Myers Squibb, Boehringer Ingelheim Pharma, Gilead, Janssen-Cilag, MSD Sharp & Dohme/Merck, Roche Diagnostic, Roche Pharma and Siemens; advisory board fees from AbbVie, Bristol-Myers Squibb, Boehringer Ingelheim Pharma, Gilead, Roche Diagnostic and Roche Pharma; and data safety board fees from Janssen-Cilag. HW has received grants from AbbVie, Gilead, Roche, Roche Diagnostics, Abbott, Myr and Eiger, and consulting fees or honoraria from AbbVie, Abbott, BMS, Boehringer Ingelheim, Eiger, Gilead, Janssen, Novartis, MSD/Merck, Roche, Roche Diagnostics and Transgene. In addition, HW has received money for board memberships from AbbVie, Abbott, BMS, Boehringer, Eiger, Gilead, Myr, Novartis and Roche; honoraria for consultancy or speaking from Eiger, Janssen, Siemens, Abbott, AbbVie, Biolex, BMS, Boehringer Ingelheim, ITS, JJ/Janssen-Cilag, Medgenics, Merck/Schering-Plough, Novartis, Roche, Roche Diagnostics, Siemens, Transgene, ViiV; and for lectures, including service on speakers’ bureaus, from the Falk Foundation and OmnisMed.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Center for Research Grants SFB900 ‘Chronic Infections: Microbial Persistence and its Control’ and SFB 738 of the German Research Foundation (DFG).
Informed consent
All patients who participated in this study gave their written informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
