Abstract
Background
Interferon-free, direct-acting antiviral treatments can result in a sustained virologic response in nearly 100% of patients with chronic hepatitis C virus infection.
Aims
The purpose of this study was to evaluate histological improvement after achieving a sustained virologic response to direct-acting antiviral treatments in patients with chronic hepatitis C.
Methods
Among 691 patients with chronic hepatitis C who achieved a sustained virologic response to direct-acting antivirals, 51 underwent liver biopsy 41 ± 20 weeks after the end of treatment despite normal transaminase levels. In 20 patients, liver biopsy specimens obtained a median of 1.2 years before the start of treatment were available.
Results
Among the 51 patients who underwent post-sustained virologic response biopsies, the grade of inflammation was A0 in 18 patients, A1 in 24, A2 in eight, and A3 in one; the stage of fibrosis was F0 in three patients, F1 in 20, F2 in 15, F3 in nine, and F4 in four. Among the nine post-sustained virologic response biopsy specimens with moderate-to-severe inflammation (≥A2), four showed S1-to-S3 steatosis (>5% of hepatocytes affected). In the 20 paired biopsy specimens, the inflammation grade significantly regressed (
Conclusions
Even if transaminases become normal after obtaining a sustained virologic response, significant histological inflammation of unknown cause was found in some patients. Additionally, improvement in liver fibrosis was not evident in the short term.
Key summary
Summarize the established knowledge on this subject
A sustained virologic response (SVR) to interferon-based therapy resulted in histological improvements in the grade of inflammation and stage of fibrosis in patients with chronic hepatitis C virus (HCV) infection.
What are the significant and/or new findings of this study?
Histological improvement, defined as a significant decrease in inflammatory score with no worsening of fibrosis, was observed also in patients with a SVR to direct-acting antiviral (DAA) treatment. The residual inflammation after obtaining a SVR was attributable to steatohepatitis at least in some patients, but was of unknown cause in others. Although fibrous bundles/septa bridging between portal tracts became thin after DAA therapy, improvement in liver fibrosis was not evident in this short-term study. Hepatic iron overload commonly observed in patients with chronic HCV infection could be improved after achieving a SVR to DAAs.
Introduction
An estimated 71 million people are chronically infected with hepatitis C virus (HCV) worldwide, and approximately 399,000 people die annually from hepatitis C-related causes, including mainly cirrhosis and hepatocellular carcinoma (HCC). 1 A sustained virologic response (SVR) is merely a surrogate marker for the virologic cure of HCV infection, and the ultimate goal of anti-HCV treatments is to prevent the development of cirrhosis and its complications. Until recently, peginterferon-ribavirin regimens had been the standard of care for patients with chronic hepatitis C. In the era of interferon therapy, a number of studies showed histologically that a SVR resulted in improvements in the grade of inflammation and stage of fibrosis in the long term.2–4
Nowadays, interferon-free, direct-acting antiviral (DAA) treatments can result in a SVR in nearly 100% of patients with chronic HCV infection. Yet, long-term outcomes of patients treated with DAAs remain unknown. An increasing number of studies have shown that a DAA-induced SVR can lead to improvement in non-invasive, clinical variables. 5 However, histological improvement after achieving a SVR to DAA treatment remains to be evaluated in patients with chronic hepatitis C.
Liver biopsy is indeed invasive, but still considered the “gold standard” for diagnosing chronic liver diseases, grading inflammatory activity, and staging liver fibrosis. 6 In chronic hepatitis C, it is also important to identify co-morbidities, such as autoimmune hepatitis, steatohepatitis, and hemochromatosis. In addition, liver biopsy provides other valuable information. For example, previous studies showed that a majority of chronically HCV-infected patients with persistently normal alanine aminotransferase (ALT) levels have some degree of histological liver damage. 7 In patients with persistently elevated ALT levels after obtaining a SVR, occult HCV infection has sometimes been described. 8 We previously reported that occult HCV infection is extremely rare (if any) in patients who have had a SVR to DAAs. 9
The aim of this study was to evaluate the short-term histological characteristics after achieving a SVR to DAA treatment in patients with chronic hepatitis C. First, we assessed the post-SVR liver biopsy specimens obtained from 51 patients with chronic hepatitis C treated with interferon-free DAA regimens. The presence of occult HCV infection was also determined by sensitive polymerase chain reaction assays. Second, in the 20 patients for whom pre-treatment biopsy specimens were available, histological improvement was also elucidated by comparing the paired biopsy specimens.
Methods
Patients
During the period from September 2014–June 2017, a total of 706 non-transplant patients with chronic hepatitis C started to receive interferon-free DAA treatments in our hospital. Patients were excluded if they had used immunomodulatory drugs, decompensated cirrhosis, viable HCC, hepatitis B surface antigen, or had other likely causes of chronic liver disease. None of the patients showed evidence of coinfection with human immunodeficiency virus. The procedures in this study were in accordance with the Helsinki Declaration of 1964 (2013 revision) and were approved by the Ethics Committee of Osaka City University Graduate School of Medicine on 1 September 2014 (No. 2905). Written informed consent was obtained from each patient.
Treatment
Patients with HCV genotype 1a or 1b infection were treated with ledipasvir 90 mg/sofosbuvir 400 mg for 12 weeks (
Clinical evaluations
Blood cell counts and routine biochemical tests were performed with the use of standard procedures. Hepatitis B surface antigen and core antibody were detected by chemiluminescence enzyme immunoassay. Genotypes of HCV were identified using an HCV genotype primer kit (Institute of Immunology Co. Ltd, Tokyo, Japan). Serum HCV RNA was measured with the use of the COBAS TaqMan assay (Pleasanton, California, USA) 10 (with a lower limit of quantification of 15 IU/ml) and Abbott RealTime assay (Des Plaines, Illinois, USA) 11 (with a lower limit of quantification of 12 IU/ml). Serum α-fetoprotein levels were determined by chemiluminescence enzyme immunoassay. Serum concentrations of type IV collagen 7S domain were measured by radioimmunoassay. The genetic polymorphisms rs8099917 near the interleukin 28B gene were examined in patients who consented to genetic analyses. 12 Liver stiffness and controlled attenuation parameter (CAP) values were measured by transient elastography (FibroScan; EchoSens, Paris, France).13,14
Liver biopsy
The flow of patients is shown in Figure 1. A total of 691 (97.9%) of the 706 patients achieved a SVR (331/335 (98.8%) to ledipasvir/sofosbuvir, 145 of 152 patients (95.8%) to daclatasvir plus asunaprevir, 24 of 25 patients (96.0%) to ombitasvir/paritaprevir/ritonavir, 22 of 22 patients (100%) to elbasvir plus grazoprevir, and 169 of 172 patients (98.3%) to sofosbuvir plus ribavirin.) Among the 691 patients, 57 consented to undergo liver biopsy after having SVR. After excluding six patients who had abnormal ALT levels (>42 U/l for men and >27 U/l for women), 51 patients with normal ALT levels after having a SVR were included in analyses (referred to as “patients with post-SVR biopsy”). In 20 patients, liver biopsy specimens taken a median (interquartile range) of 1.2 (0.4 − 2.4) years before the start of DAA treatment were available (referred to as “patients with paired biopsy”).
Study flowchart. Fifty-one patients with normal alanine aminotransferase (ALT) levels after having sustained virologic responses (SVRs) were referred to as “patients with post-SVR biopsy”. Twenty patients in whom liver biopsy specimens were taken a median of 1.2 years before the start of direct-acting antiviral (DAA) treatment were referred to as “patients with paired biopsy”.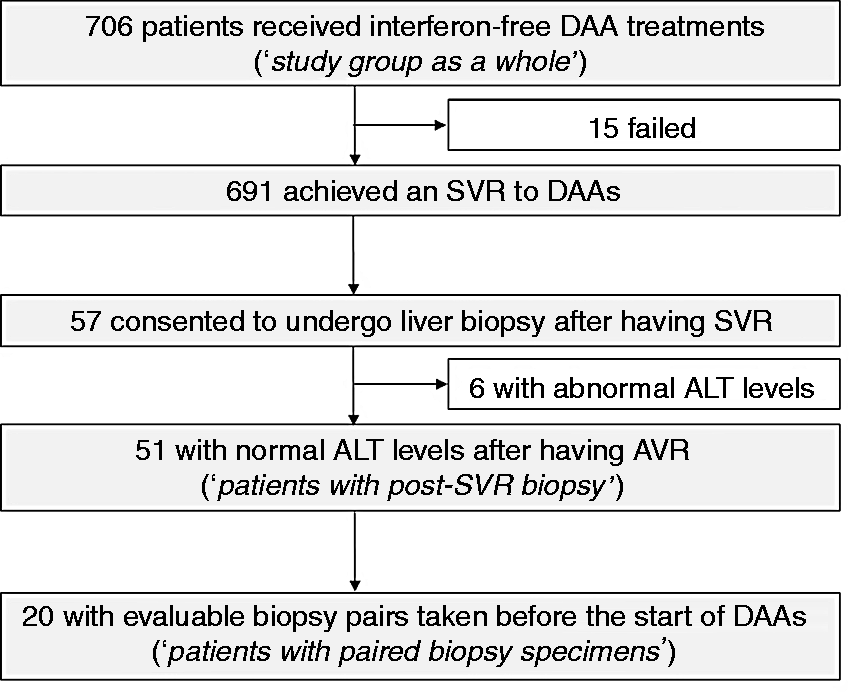
Liver biopsy specimens were obtained from each patient with a 15-gauge Tru-Cut needle under ultrasound guidance 41 ± 20 weeks after the end of treatment. Liver tissues were fixed in formalin immediately after biopsy and embedded in paraffin. A portion of each biopsy specimen was also immediately placed in RNAlater (Qiagen, Valencia, California, USA) and temporarily stored at −20℃. We attempted to amplify the HCV RNA from 25 ng of the extracted RNA from the liver needle biopsy specimen by the sensitive Abbott RealTime assay. Four-micrometer-thick sections were stained with hematoxylin-eosin, Azan-Mallory, orcein, and Prussian blue stains. All liver biopsy specimens were evaluated by an experienced histopathologist (Y.I.) blinded to the clinical data. The histological findings were assessed by grading the inflammatory activity and staging fibrosis according to the Knodell scoring system 15 and the METAVIR scoring system. 16 Iron deposition was evaluated on a scoring system proposed by Rowe et al., 17 based on Prussian blue staining as follows: grade 0 (granules absent/barely discernible at ×400 magnification), grade 1+ (granules barely discernible at ×250); grade 2+ (discrete granules resolved at ×100); grade 3+ (discrete granules resolved at ×25); and grade 4+ (masses visible at ×10 or grossly visible). Steatosis was graded in accordance with the nonalcoholic fatty liver disease (NAFLD) activity score as follows: S1 (5–33% of hepatocytes affected), S2 (34–66% affected), and S3 (>66% affected). 18 A liver sample was considered adequate if it was longer than 15 mm and contained six or more portal tracts.
Statistical analysis
Statistical analysis was performed with R software (The R Foundation for Statistical Computing, Vienna, Austria). Distributions of continuous variables were analyzed by the Mann–Whitney
Results
Changes in clinical parameters in the study group as a whole
As expected, serum ALT, α-fetoprotein, and type IV collagen 7S levels decreased significantly and platelet count increased significantly in the study group as a whole at the end of treatment and 12 weeks after treatment (Supplementary Material Figure 1). In the 141 patients for whom valid elastography data were available before treatment and 12–48 weeks after treatment, liver stiffness significantly decreased, and the CAP marginally increased (Supplementary Material Figure 2).
Characteristics of patients with post-SVR biopsy
Characteristics of the 51 patients with post-sustained virologic response (SVR) biopsy.
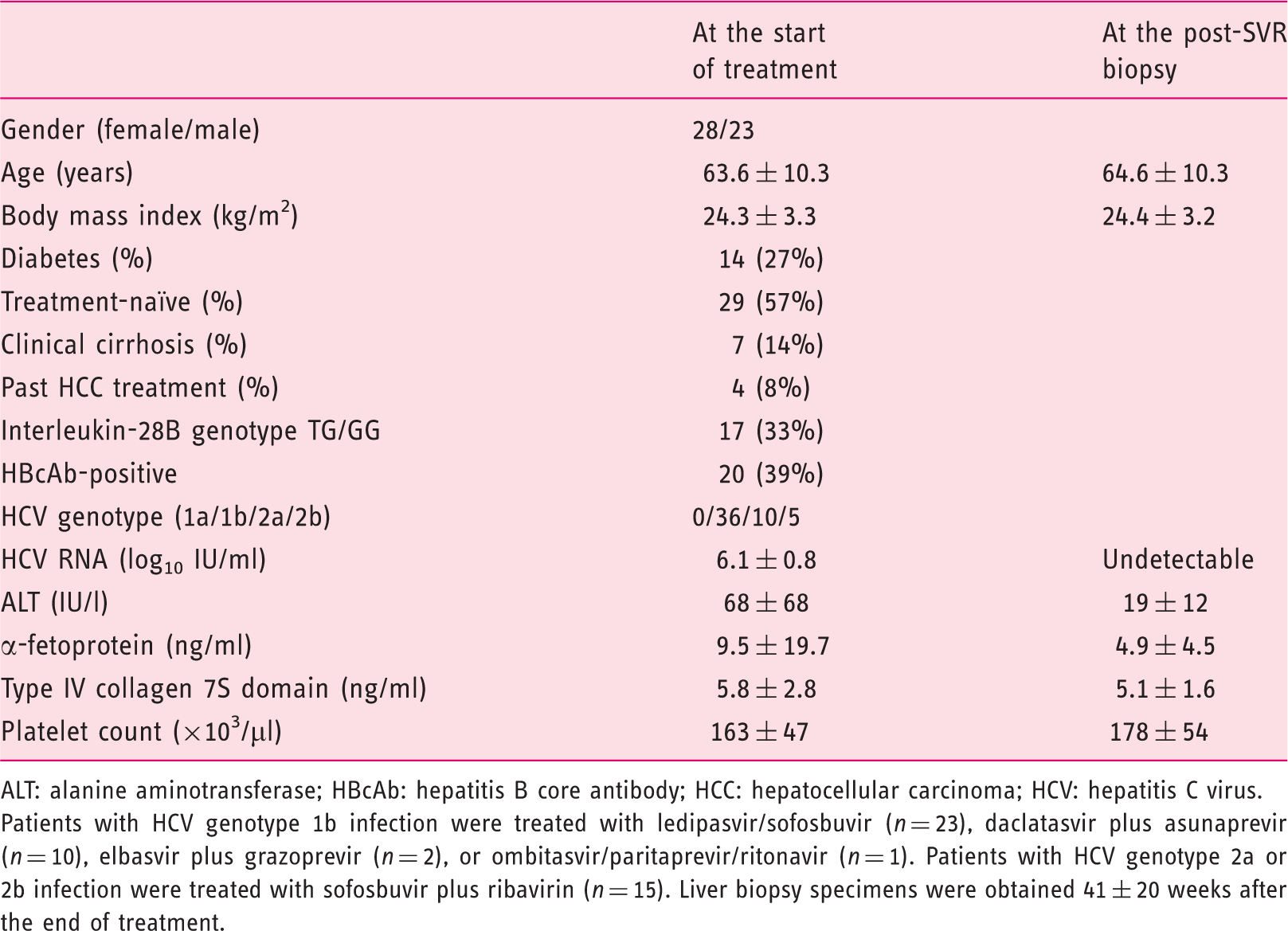
ALT: alanine aminotransferase; HBcAb: hepatitis B core antibody; HCC: hepatocellular carcinoma; HCV: hepatitis C virus.
Patients with HCV genotype 1b infection were treated with ledipasvir/sofosbuvir (
HCV RNA detection in serum and in liver of patients with post-SVR biopsy
Rates of undetectable HCV RNA in serum during and after treatment are shown in Figure 2. The rates were lower with the Abbott RealTime assay than with the COBAS TaqMan assay during treatment. Of interest, even patients in whom the serum HCV-RNA remained positive on the Abbott RealTime assay at the end of treatment could achieve a SVR after treatment. HCV RNA was not detected in the post-SVR liver biopsy specimens of the 51 patients, including the four patients who remained positive for serum HCV-RNA at the end of treatment.
Rates of undetectable hepatitis C virus (HCV) RNA in serum of the 51 patients with post-sustained virologic response (SVR) biopsy during and after treatment. Serum HCV RNA was not detected at the end of treatment or at 12 weeks post-treatment in any SVR patient with use of the TaqMan assay. In contrast, with use of the RealTime assay, serum HCV RNA remained positive at the end of treatment in four (8%) patients, but became negative at 4–12 weeks post-treatment.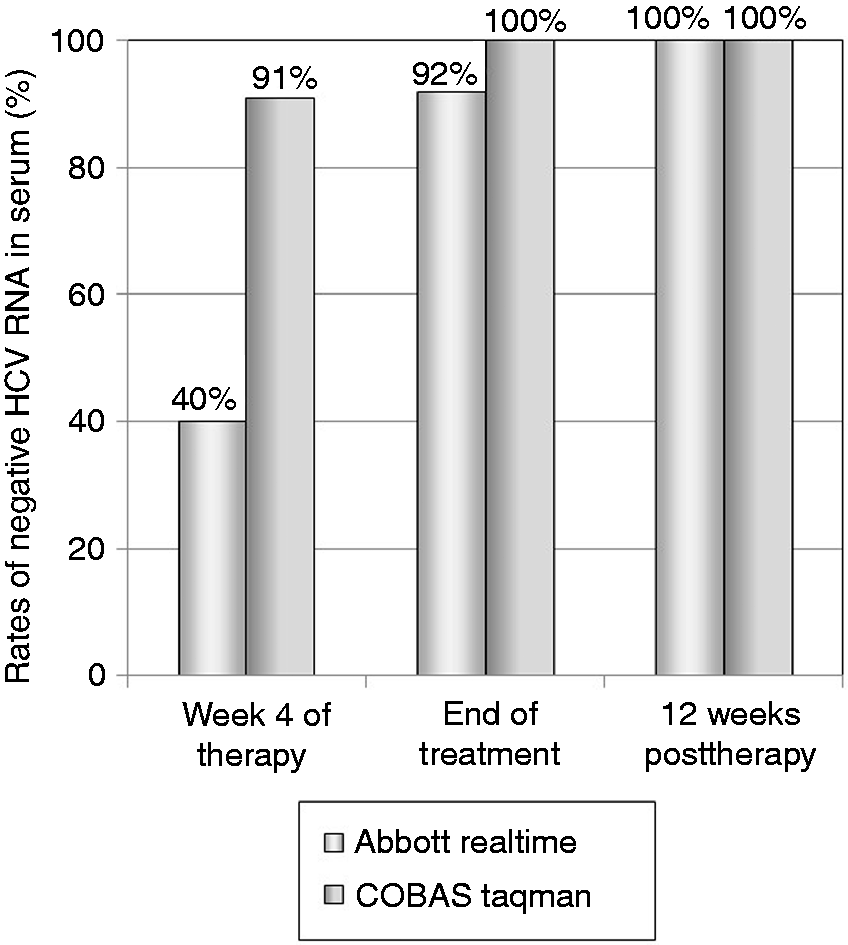
Histological evaluations of post-SVR biopsy
The results of the histological evaluations of hepatic inflammation, fibrosis, iron deposition, and steatosis in the 51 post-SVR biopsy specimens are shown in Figure 3. The only one biopsy specimen with severe inflammation (A3) post-SVR showed active lymphocyte and plasma cell infiltrates in the portal area, suggesting autoimmune hepatitis (Supplementary Material Figure 3). Among the eight post-SVR biopsy specimens with moderate inflammation (A2), four showed S1 to S3 steatosis (>5% of hepatocytes affected), three of which also showed hepatocellular ballooning, characterizing steatohepatitis (Supplementary Material Figure 4).
Results of the histological evaluations of hepatic inflammation (a), fibrosis (b), iron deposition (c) and steatosis (d) in the 51 post-sustained virologic response (SVR) biopsy specimens. The grade of inflammation was A0 in 18 patients, A1 in 24, A2 in eight, and A3 in one; the stage of fibrosis was F0 in three patients, F1 in 20, F2 in 15, F3 in nine, and F4 in four; the grade of iron deposition was zero in 29 patients, 1+ in 10, 2+ in six, 3+ in five, and 4+ in one; and the grade of steatosis was S0 in 29 patients, S1 in 15, S2 in six, and S3 in one.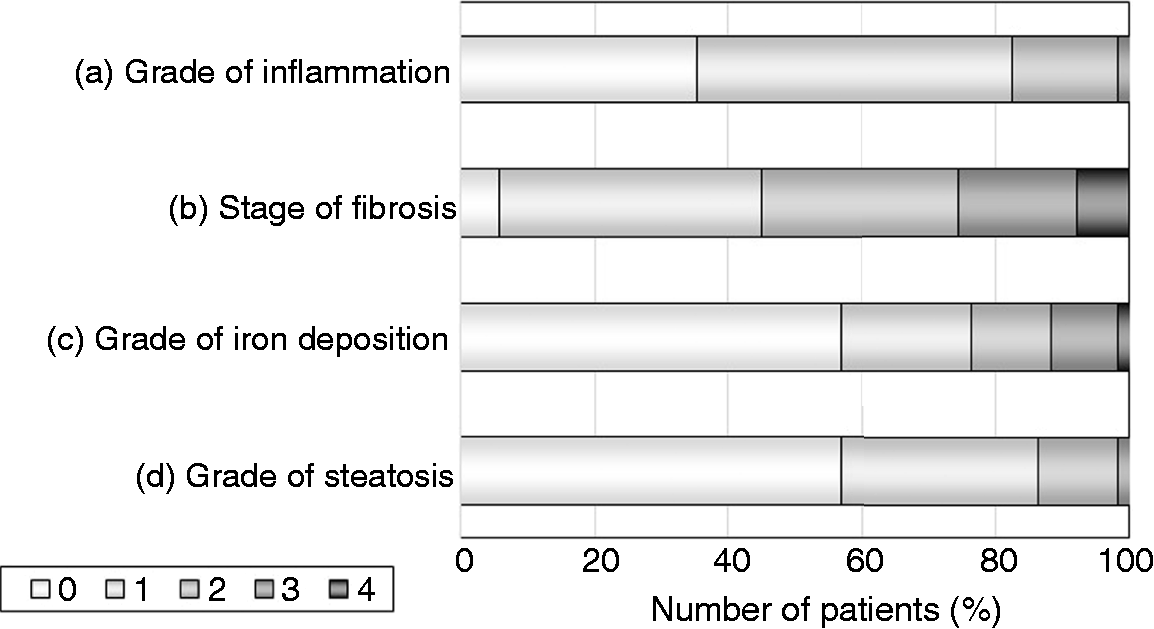
Changes in histology in patients with paired biopsy
Characteristics of the 20 patients with paired biopsy.

ALT: alanine aminotransferase; HBcAb: hepatitis B core antibody; HCC: hepatocellular carcinoma; HCV: hepatitis C virus; SVR: sustained virologic response.
Patients with HCV genotype 1b infection were treated with ledipasvir/sofosbuvir (
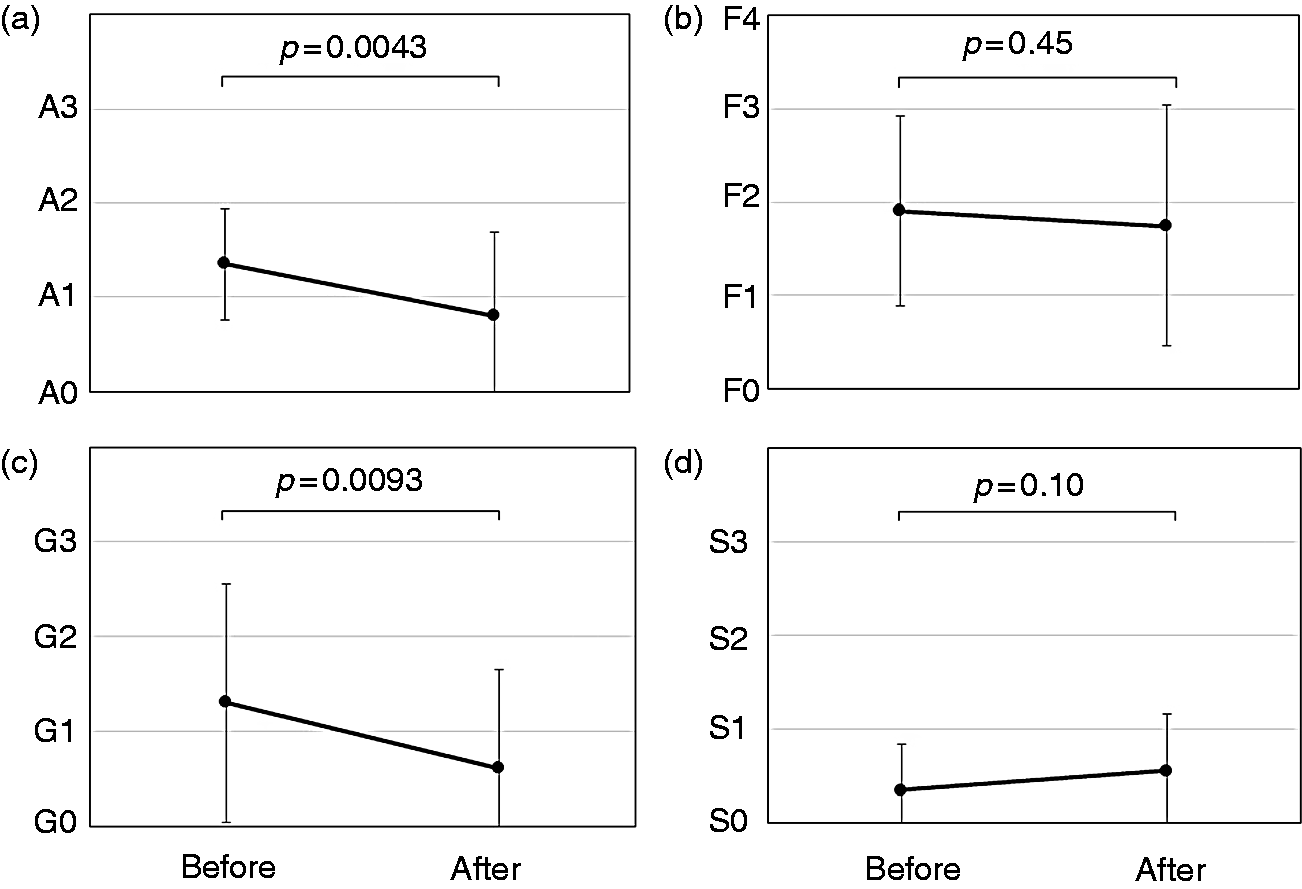
Changes in histological findings in the 20 patients with evaluable pairs of biopsy specimens. The grade of inflammation (a) had significantly regressed (
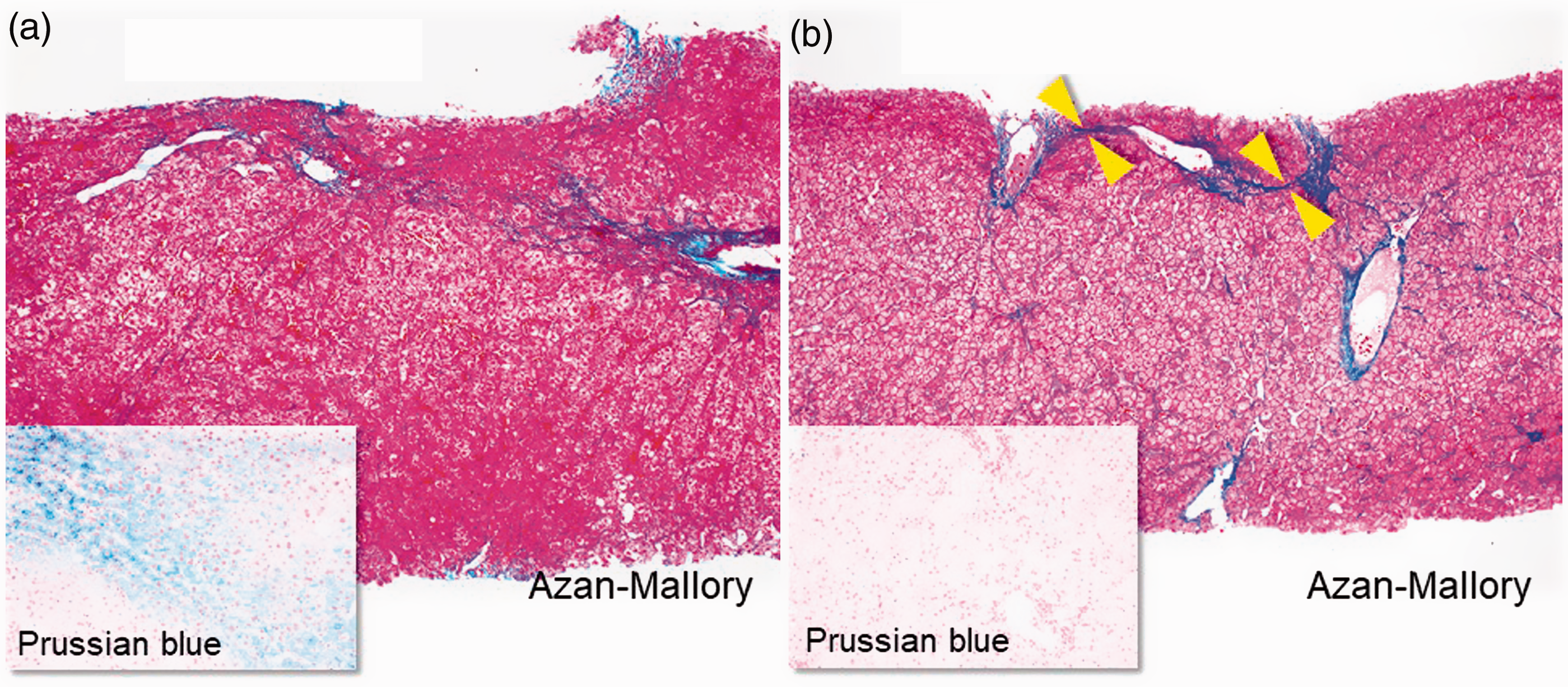
Liver biopsy specimens obtained from a 74-year-old woman with genotype 1b infection who relapsed after peginterferon-ribavirin plus faldaprevir treatment previously and then achieved an sustained virologic response (SVR) to ledipasvir/sofosbuvir treatment (Azan-Mallory and Prussian blue stains; original magnification, all ×100): METAVIR score of A2F2 before therapy (a) and A1F2 after therapy (b). Fibrous bundles/septa bridging between portal tracts are thinner in post-SVR (arrowheads). As aminotransferases decreased aspartate aminotransferase (AST) from 35 to 33 U/l and alanine aminotransferase (ALT) from 31 to 26 U/l), the inflammatory score also decreased after obtaining a SVR, as did the grade of iron accumulation evaluated with Prussian blue stain.
Discussion
To our knowledge, this is the first study to evaluate histological outcomes in patients with chronic hepatitis C who achieved a SVR to interferon-free DAA treatments. We examined 51 liver biopsy specimens obtained with informed consent after achieving a SVR. In the first part of this study, 18% of the post-SVR liver biopsy specimens showed clinically significant inflammation (≥A2) despite normal ALT values. In the second part of this study, regression in fibrosis was not evident in the 20 paired biopsies in the short term, although histological improvement, defined as a significant decrease in inflammatory score with no worsening of fibrosis, was found in the majority of patients who achieved a SVR to DAAs.
Steatohepatitis was one of possible causes of the residual inflammation in the post-SVR liver biopsy specimens. In this study, both the histological grade of steatosis and CAP (as determined by transient elastography) increased slightly but not significantly after the treatment. Schlevogt et al. reported that DAA-induced SVR is associated with weight gain during long-term follow-up. 19 Body weight did not change significantly in our patients during the short term (Tables 1 and 2). In a previous study of 214 patients who received daclatasvir plus asunaprevir therapy, 20 the CAP was elevated significantly after treatment. In the era of interferon-based therapies, intrahepatic steatosis was reported to be one of the independent predictors of the development of HCC after treatment. 21 Patients with steatohepatitis should be carefully followed up with periodical liver function testing and abdominal imaging even after achieving a SVR.
In the remaining post-SVR biopsies, the cause of the residual inflammation remains unknown. Whitcomb et al.
22
reported that among the 36 allograft liver biopsy specimens from patients who achieved a SVR after receiving a liver transplant for chronic HCV infection, 69% had histologic features of active inflammation. Nearly all patients had been treated with interferon-based regimens. They raised the possibility that chronic HCV infection might induce a dysfunctional immune response, which remains active despite viral eradication, and suggested that the inflammatory reaction to HCV persists in the liver tissue long after serologic clearance. In the present study, the grade of residual inflammation did not correlate with the interval from the end-of-treatment to the post-SVR biopsy (
We believe that the occult HCV infection could not be the cause. Elmasry et al. 23 reported that positive- and negative-strand HCV RNA was detected in four liver tissue specimens from nine patients in whom serum ALT levels did not normalize despite a SVR to DAAs for post-transplant recurrent HCV infection. Responding to their report, we previously demonstrated that HCV RNA was not found in any liver samples obtained from 25 non-transplant patients with sustained normal ALT levels after having a SVR. 9 In the present study, we confirmed the same results in an additional 26 patients, including some in whom serum HCV RNA remained positive on the Abbott RealTime HCV assay at the end of treatment, and then achieved a SVR.24,25
Previous studies 5 reported that liver stiffness regressed after attaining a SVR, but it remains unclear whether such improvement in the results of non-invasive tests might really reflect the reversal of liver fibrosis or reduced inflammation (or both). Knop et al. 26 showed that the dynamics of the improvement in liver stiffness were more pronounced between baseline and end of treatment than between baseline and 24 weeks after treatment. Taken together with our paired biopsy results, the short-term improvement in non-invasive test results are mainly attributable to reduced inflammation.
In patients with chronic hepatitis B, large clinical trials of entacavir or tenofovir disoproxil fumarate showed both short- and long-term histological improvements at one year and 3–7 years, respectively.27,28 Sun et al. 29 recently proposed a classification to evaluate the dynamic changes in the quality of fibrosis, namely predominantly progressive or predominately regressive and indeterminate, which displayed an overall balance between progressive and regressive scarring. As shown in Figure 5, predominately regressive scarring (defined as most (>50%) fibro-septal stroma in the liver biopsy specimens showing features of scars with thin, densely compacted stroma and largely dark staining on the connective tissue stain) was found in four post-SVR biopsies, but none of the pre-treatment biopsies (data not shown). Because previous studies (including ours)30,31 have shown that progressive fibrosis in liver despite a SVR is closely associated with development of HCC, longer observation is required.
Hepatic iron overload is commonly observed in patients with chronic HCV infection, and can cause liver damage by increasing oxidative stress. The mechanisms involve mainly impaired hepcidin expression and the altered expression of other iron-metabolism-related genes. Previous studies in the interferon era32,33 showed that successful HCV eradication restored the relative impairment of hepcidin production. In this study, we showed in liver biopsy specimens for the first time that SVR to DAAs also can improve the iron overload condition.
Prati et al.
34
proposed updated upper limits of normal for ALT (30 U/l for men and 19 U/l for women), which were more sensitive for identifying persons with HCV infection. We further divided our subjects with “normal” ALT into two groups according to the updated upper limits of normal (≤30 U/l or 31–42 U/l for men and ≤19 U/l or 20–27 U/l for women) (Supplementary Material Table 2). The grade of inflammation was higher in patients whose ALT was normal but 31–42 U/l in men or 20–27 U/l in women than in those whose ALT was ≤30 U/l in men or ≤19 U/l in women (
Our study had several limitations. First, because this was a retrospective study of a real-world cohort, the timing of liver biopsy was not uniform. In particular, the first paired biopsy specimens were taken a median (interquartile range) of 1.2 (0.4 − 2.4) years before the start of DAA treatment. However, previous studies showed that the rate of fibrosis progression was only 0.10–0.133 fibrosis unit per year in the natural history of untreated patients with chronic hepatitis C.2,35 Therefore, the stage of liver fibrosis was probably unchanged during the lag time in the majority of patients. Second, there may be a selection bias, since the difference in age was significant between patients with post-SVR biopsy and patients without post-SVR biopsy. However, the proportions of patients with cirrhosis and other baseline characteristics did not differ between patients with post-SVR biopsy and those without biopsy. Lastly, because of the limited number of patients and short duration of observation, it was difficult to evaluate the impact of a SVR to DAAs on the regression of liver fibrosis in our study. Large, long-term studies are needed to draw final conclusions.
In summary, histological improvement, defined as a significant decrease in inflammatory score with no worsening of fibrosis, was observed in 55% of the patients with a SVR to DAA treatment. However, improvement in liver fibrosis was not evident in this short-term study. Even if ALT normalized after obtaining a SVR, clinically significant histological inflammation remained in 16% of the patients. The residual inflammation was attributable to steatohepatitis at least in some patients, but was of unknown cause in others.
Supplemental Material
Supplementary Tables -Supplemental material for Short-term histological evaluations after achieving a sustained virologic response to direct-acting antiviral treatment for chronic hepatitis C
Supplemental material, Supplementary Tables for Short-term histological evaluations after achieving a sustained virologic response to direct-acting antiviral treatment for chronic hepatitis C by Masaru Enomoto, Yoshihiro Ikura, Akihiro Tamori, Ritsuzo Kozuka, Hiroyuki Motoyama, Etsushi Kawamura, Atsushi Hagihara, Hideki Fujii, Sawako Uchida-Kobayashi, Hiroyasu Morikawa, Yoshiki Murakami and Norifumi Kawada in United European Gastroenterology Journal
Supplemental Material
Supplementary Figures -Supplemental material for Short-term histological evaluations after achieving a sustained virologic response to direct-acting antiviral treatment for chronic hepatitis C
Supplemental material, Supplementary Figures for Short-term histological evaluations after achieving a sustained virologic response to direct-acting antiviral treatment for chronic hepatitis C by Masaru Enomoto, Yoshihiro Ikura, Akihiro Tamori, Ritsuzo Kozuka, Hiroyuki Motoyama, Etsushi Kawamura, Atsushi Hagihara, Hideki Fujii, Sawako Uchida-Kobayashi, Hiroyasu Morikawa, Yoshiki Murakami and Norifumi Kawada in United European Gastroenterology Journal
Footnotes
Acknowledgments
The authors are grateful to Yoko Yasuhara, Sanae Deguchi, and Ayano Fujikawa for their assistance in collecting the data, to Saori Itami for her technical assistance, and to Masaaki Korenaga and Misako Sato-Matsubara for their valuable comments on this study.
Declaration of conflicting interests
Norifumi Kawada has received lecture fees from Gilead Sciences Inc., AbbVie Inc., and MSD K.K., and research grants from Gilead and AbbVie.
Funding
This work was supported in part by JSPS KAKENHI (Grant Number 16K09369).
Ethics approval
The procedures in this study were in accordance with the Helsinki Declaration of 1964 (2013 revision) and approved by the Ethics Committee of Osaka City University Graduate School of Medicine on 1 September 2014 (No. 2905).
Informed consent
Written informed consent was obtained from each patient.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
