Abstract
While for many years investigators had worked on highly effective direct-acting antiviral agent (DAA) therapy, we are now encountering challenges on the appropriate timing of DAA therapy in patients with decompensated cirrhosis. Improvement in hepatic function and quality of life can be achieved following successful therapy but not in all patients. Predictors of improvement or failure to improve have been noted but these are currently not robust enough to ubiquitously apply them to clinical practice. The lowest probability of improvement in hepatic function and avoidance of Model for End-stage Liver Disease (MELD) “purgatory” appears to be in those with MELD >20 while the more likely scenario of improvements is in those with MELD <15. Ideally, patients with a MELD score >20 should be transplanted first and treated for hepatitis C virus (HCV) infection after liver transplantation (LT). Those with MELD score <15 should be considered readily for treatment while in those with MELD of 15–20, treatment has to be individualized with full discussion of the pros and cons of treating them pre- or post-LT. However, it is to be appreciated that the majority of patients with decompensated cirrhosis across the world may not be eligible for liver transplant or may not have access to LT; thus, these patients should be considered for HCV therapy with the hope and expectation that they still gain variable degrees of benefit from successful DAA therapy.
Keywords
Introduction
The estimated global prevalence of hepatitis C virus (HCV) infection is 1.0%. The Eastern Mediterranean Region had the highest prevalence (2.3%) followed by the European Region (1.5%) and African Regions (1.0%). 1 The estimated prevalence of HCV viremia in sub-Saharan Africa has varied from 0.5-2.1% according to subregion, and in North Africa has been estimated at 1.7% where Egypt represents the most HCV prevalent country. 2 The infection causes progressive hepatic inflammation which can lead to cirrhosis in 10–20% of individuals over 20–30 years, with a 3–6% annual risk of hepatic decompensation. 3 Once hepatic decompensation develops, it can result in markedly decreased survival and impaired quality of life.3,4 Liver transplantation (LT) remains the ultimate, definitive, and life-saving procedure; however, donor organ shortage is still a major limitation. The introduction of highly effective direct-acting antiviral agents (DAAs) has led to a paradigm shift in HCV treatment with high sustained virologic response (SVR) rates and safety profiles which can then result in lowering Model for End-stage Liver Disease (MELD) scores,5,6 improved quality of life, and lowered morbidity from cirrhosis. 4 However, some DAAs such as protease inhibitors are contraindicated in patients with decompensated cirrhosis and there might be a few tolerability issues, particularly with ribavirin. Further, some patients who experienced SVR resulting in lower MELD scores after successful DAA therapy may continue to have poor quality of life, so-called “MELD purgatory,” which decreases their chances of achieving a liver transplant. 7 Predictors associated with improvements in decompensated cirrhosis after DAA therapy are being more extensively evaluated in order to determine the appropriate candidates for HCV treatment in this specific subgroup of patients.
Natural history of chronic HCV infection
Chronic HCV infection is a slowly progressive disease which can lead to cirrhosis in 20–30 years. Once cirrhosis has developed, the risk of hepatic decompensation is approximately 3–6%/year. 3 Patients with decompensated cirrhosis (Child–Turcotte–Pugh, CTP class B or C) may variably develop portal hypertensive-related complications such as ascites, jaundice, variceal bleeding, or hepatic encephalopathy. Successful HCV treatment results in lower risk of hepatic failure, hepatocellular carcinoma, liver-related deaths, and all-cause mortality while the definitive treatment for patients with decompensated cirrhosis is LT.8,9 In those who undergo LT and have untreated hepatitis C infection or have failed therapy, there can be a significantly accelerated course of disease, compared to non-LT setting, where cirrhosis can develop in up to 30% at 5 years after LT. 8 A small proportion of patients (2–9%) may develop allograft failure within 1–2 years after LT due to the development of cholestatic hepatitis, a condition associated often with high hepatitis C viremia. 9 However, with the advent of DAAs, SVR has been achieved in >85% of LT-recipients, favorably altering the natural history of post-transplant hepatitis C–associated liver disease. 10 Due to the effectiveness of DAA therapy in this era, the prevalence of HCV infection is expected to decline, but still, hepatocellular carcinoma and liver-related deaths are expected to continue rising worldwide through the year of 2030. 11
Outcome of HCV treatment in decompensated cirrhosis
Summary of clinical trials on hepatitis C virus treatment in patients with decompensated cirrhosis.4,5,4
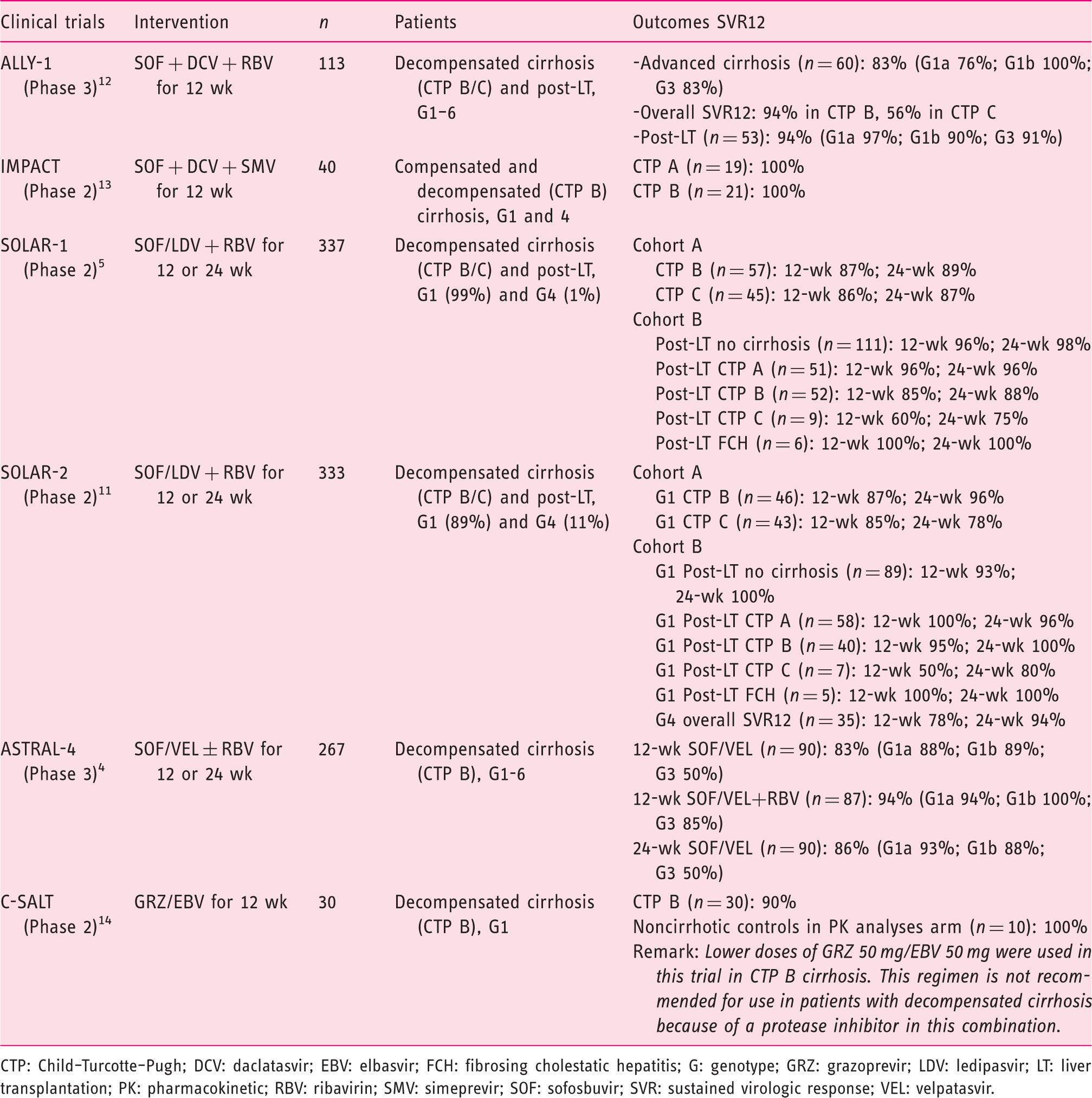
CTP: Child–Turcotte–Pugh; DCV: daclatasvir; EBV: elbasvir; FCH: fibrosing cholestatic hepatitis; G: genotype; GRZ: grazoprevir; LDV: ledipasvir; LT: liver transplantation; PK: pharmacokinetic; RBV: ribavirin; SMV: simeprevir; SOF: sofosbuvir; SVR: sustained virologic response; VEL: velpatasvir.
Sofosbuvir/ledipasvir
In Cohort A of the SOLAR-1 study, adults with HCV genotype 1 or 4 infection with decompensated liver disease (CTP B or C) and post-LT were randomized to receive either a 12- or 24-week course of sofosbuvir/ledipasvir and ribavirin. Patients receiving the 12-week regimen had an SVR12 rate of 86–87%, which was similar to the 24-week regimen with SVR12 rate of 87–89%.
6
In cohort B, post-LT decompensated liver disease (CTP B and C) patients had a lower SVR12 rate of 60–85% with the 12-week regimen and 75–88% with the 24-week regimen.
6
Likewise, in the SOLAR-2 study, which included more of genotype 4 patients (
Sofosbuvir/velpatasvir
In the ASTRAL-4 trial in adults with HCV genotype 1–6 infection with decompensated cirrhosis who were randomized to three arms (sofosbuvir/velpatasvir 12 weeks; sofosbuvir/velpatasvir + ribavirin 12 weeks; sofosbuvir/velpatasvir 24 weeks), the overall SVR12 rate was 83%, 94%, and 86%, respectively. Notably, among HCV genotype 3 infection, the treatment groups without ribavirin had lower SVR12 rate of 50%. 5
Sofosbuvir plus daclatasvir
In the ALLY-1 trial, 60 patients with advanced cirrhosis with genotype 1, 2, 3, 4, and 6 were enrolled and the CTP classifications were 20% A, 53% B, and 27% C. In patients with cirrhosis, 82% (95% confidence interval (CI) 67.9–92.0%) of patients with genotype 1 infection achieved SVR12, while SVR rates in those with genotypes 2, 3, and 4 were 80%, 83%, and 100%, respectively; SVR12 rate was higher in patients with CTP class B (94%), versus class C (56%). 13
Real-world data on hepatitis C virus treatment in patients with decompensated cirrhosis. 3
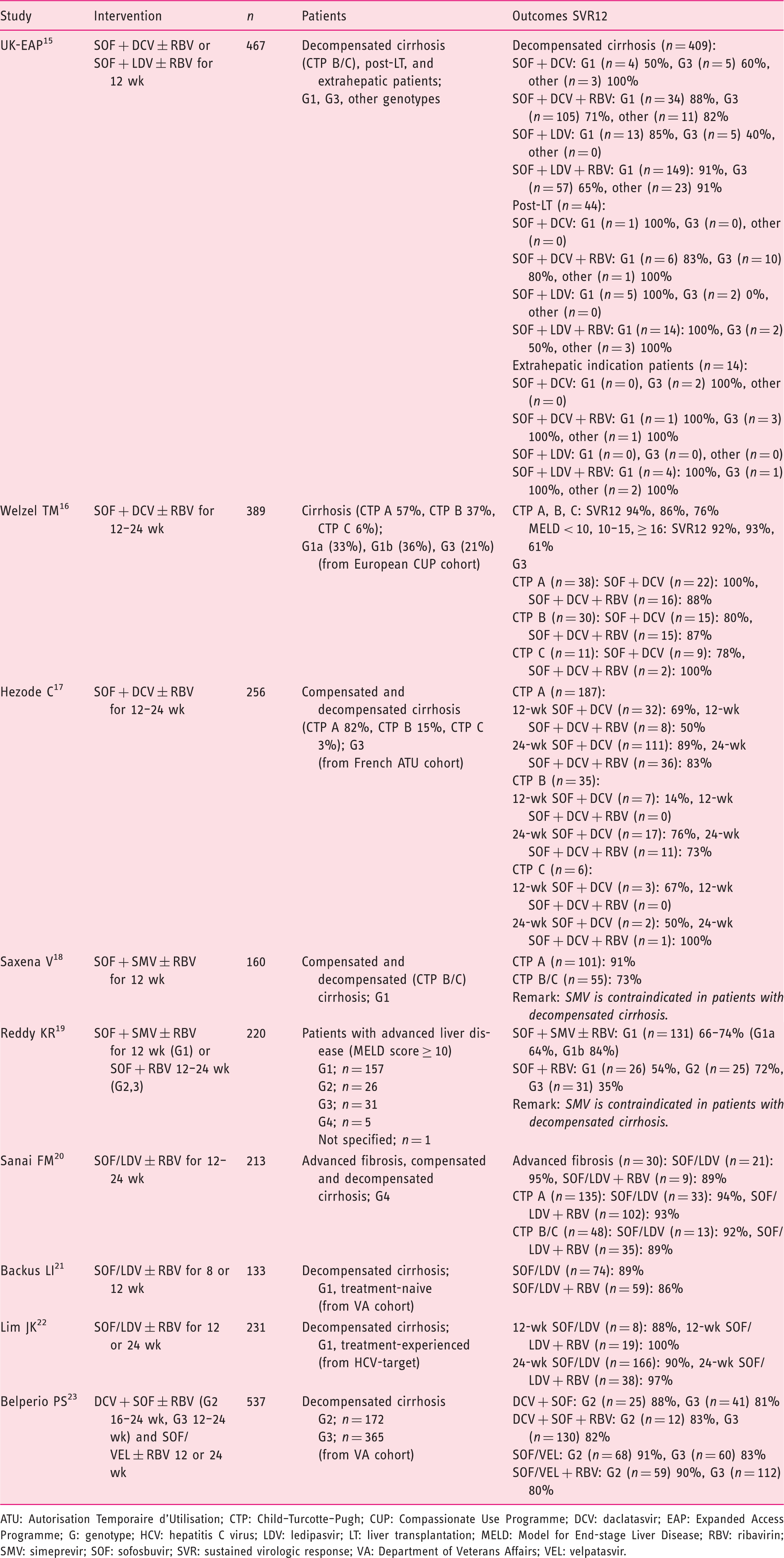
ATU: Autorisation Temporaire d'Utilisation; CTP: Child–Turcotte–Pugh; CUP: Compassionate Use Programme; DCV: daclatasvir; EAP: Expanded Access Programme; G: genotype; HCV: hepatitis C virus; LDV: ledipasvir; LT: liver transplantation; MELD: Model for End-stage Liver Disease; RBV: ribavirin; SMV: simeprevir; SOF: sofosbuvir; SVR: sustained virologic response; VA: Department of Veterans Affairs; VEL: velpatasvir.
Challenges for DAA treatment in patients with decompensated cirrhosis
Effects of DAA treatment on CTP and MELD score
Successful HCV treatment can improve hepatic function in the majority of patients over the short term with 60% of patients achieving improvement in MELD score, while 17% had no change, and 23% had worsening of MELD score.
10
Improvement of MELD scores following HCV treatment in decompensated cirrhosis varied with a median change of –2 points (
Predictors associated with improvements in decompensated cirrhosis after DAA therapy
Various studies have made attempts at identifying predictors associated with clinical improvement after DAA therapy, and also overcome the MELD purgatory state in patients with decompensated cirrhosis. According to the ELITA study ( Suggested hepatitis C virus treatment algorithm in the direct-acting antiviral agent era in patients with cirrhosis (modified from 26,31,35,37) Abbreviations: HCV, hepatitis C virus; CTP, Child-Turcotte-Pugh; LT, liver transplantation; MELD, Model for End-stage Liver Disease; LDLT, living donor LT; GFR, glomerular filtration rate; SVR, sustained virologic response; DAA, direct-acting antiviral agents.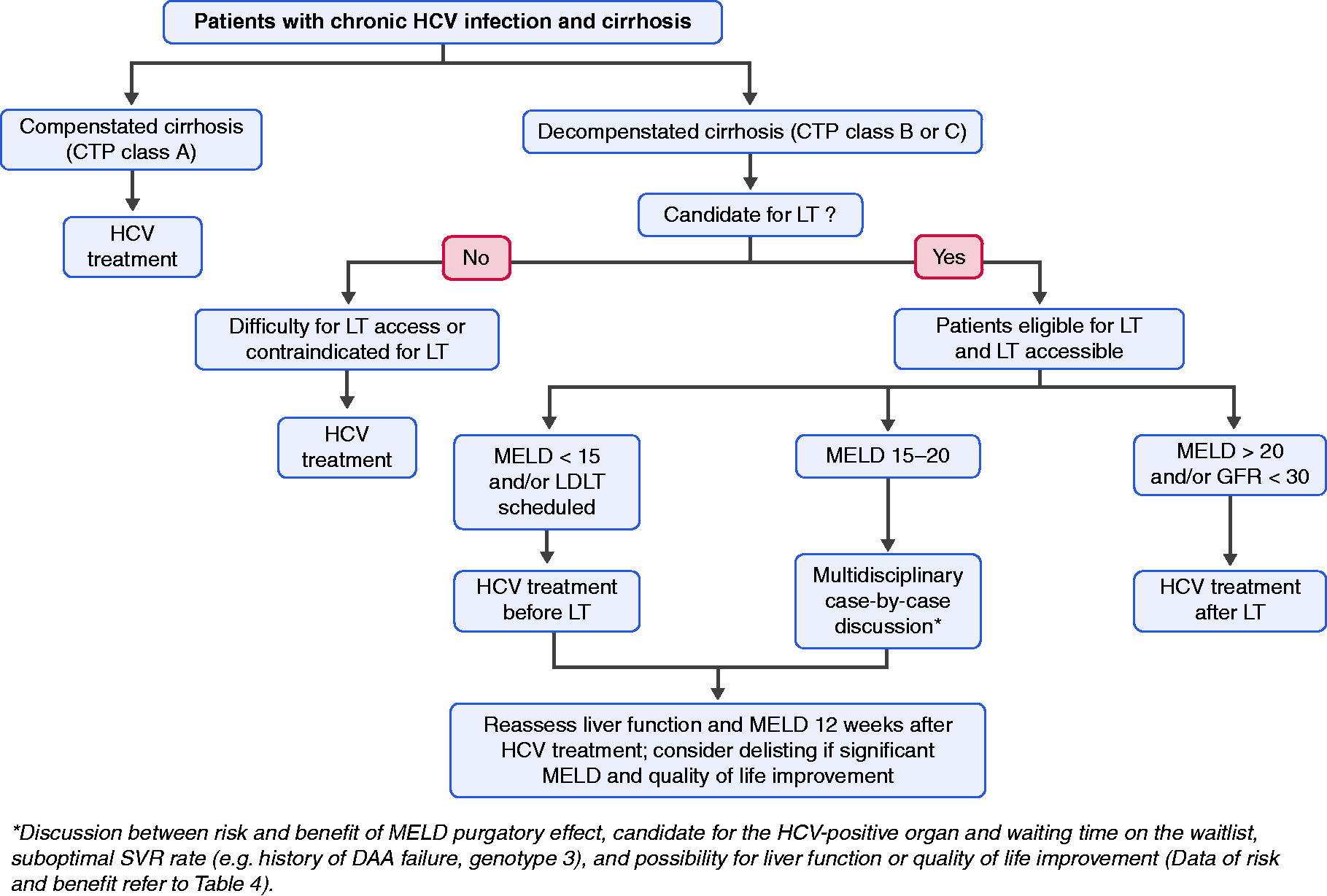
Adverse effects and drug-drug interactions of DAA in decompensated cirrhosis
Current guideline recommendations accept the use of sofosbuvir-based DAA regimens with the combination of ledipasvir, velpatasvir, daclatasvir, with or without ribavirin, for HCV treatment in patients with decompensated cirrhosis. NS3/4 protease inhibitors are contraindicated because of their potential to worsen the severity of their liver disease. Apart from sofosbuvir which is mainly excreted by the kidneys, most of the DAAs are metabolized by the liver with biliary excretion as a major route. Pharmacokinetic studies of these drugs showed no clinically relevant effect on the drug exposure in patients with hepatic impairment, and thus, no dose adjustment is required.29–31 However, in patients with severe renal impairment (glomerular filtration rate < 30 mL/min), sofosbuvir use is not better recommended, and HCV treatment is suggested to be deferred until after transplantation. 32 Recent data, however, suggest that sofosbuvir-based therapy can be used safely and with efficacy in those with chronic kidney disease stages 4 and 5, although the studies have been done in those with well-compensated liver disease.33,34 With ribavirin, the incidence and severity of ribavirin-induced anemia has been noted to be increased in cirrhosis. 10 Thus, in patients with decompensated cirrhosis, it is suggested ribavirin be started with an initial dose of 600 mg/day and uptitrated according to patients' tolerability.
Guideline recommendation for HCV therapy in patients with decompensated cirrhosis
Treatment recommendations for hepatitis C virus–infected patients with decompensated cirrhosis.34,35
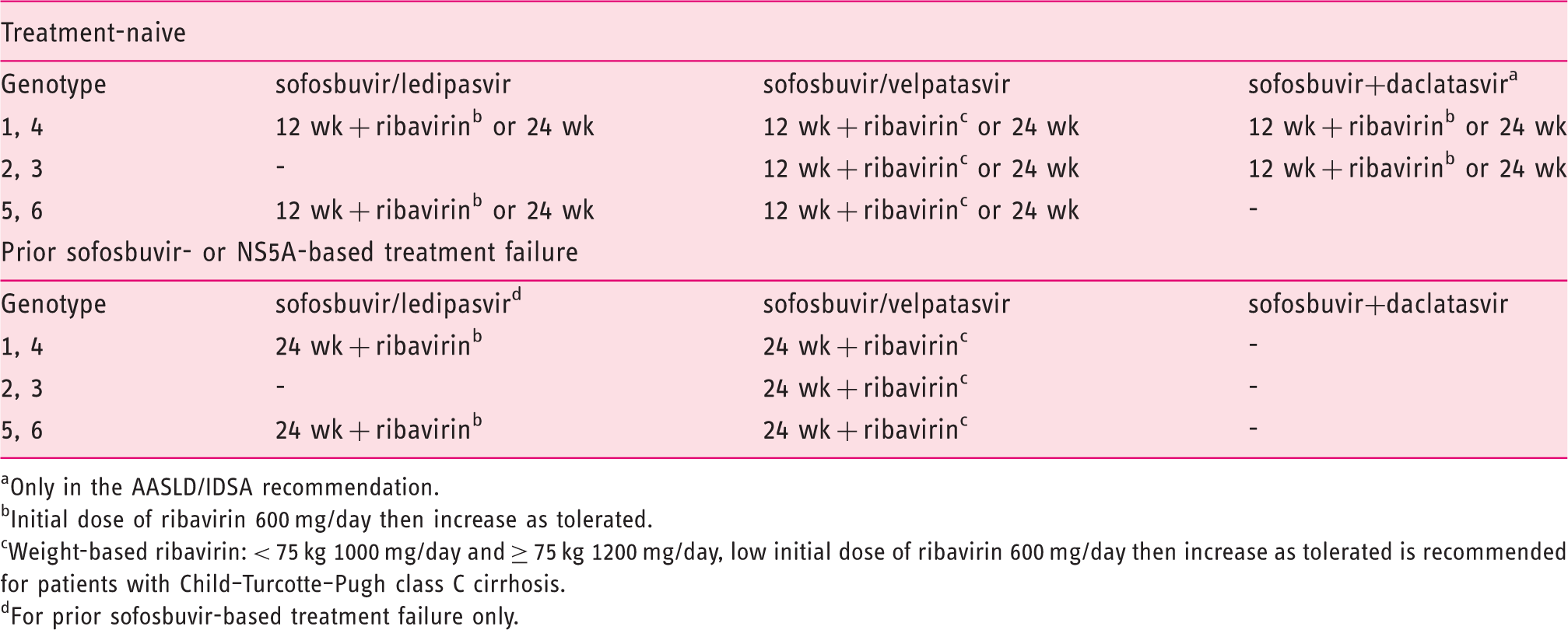
Only in the AASLD/IDSA recommendation.
Initial dose of ribavirin 600 mg/day then increase as tolerated.
Weight-based ribavirin: < 75 kg 1000 mg/day and ≥ 75 kg 1200 mg/day, low initial dose of ribavirin 600 mg/day then increase as tolerated is recommended for patients with Child–Turcotte–Pugh class C cirrhosis.
For prior sofosbuvir-based treatment failure only.
Controversial issues on HCV therapy in patients with decompensated cirrhosis versus post-LT
HCV therapy in patients with decompensated cirrhosis should be individualized particularly if LT is an option and is available; in such cases the challenge is to decide on therapy before or after LT. Existing data suggest that successful HCV therapy is associated with improvement of hepatic function in 20–60% which may lead to delisting in some patients (MELD < 16, 16–20, > 20 with the delisting probability of 35, 12, 5% respectively).10,26,27 However, the risk of progressive liver disease, MELD purgatory state, and hepatocellular carcinoma still remain. After successful HCV eradication, while some patients have MELD score reduction and are delisted, they may still have complications from cirrhosis and poor quality of life, so-called “MELD purgatory” or a MELD “limbo” state where these patients are in a “no person's” land in that they are neither fully functional and normal nor are they on the “radar scene” for achieving a transplant since they do not have a transplantable MELD. Such a state has been noted to evolve mostly in those successfully treated for their HCV while they had CTP class C liver disease and high MELD scores (>20). In patients with a MELD score >20, SVR rate has been reported to be as low as 25% and with limited safety profiles for DAAs. 37 Further, a modeling study, based on data on efficacy of DAAs and the expected improvement in hepatic function, suggests that it may not be cost-effective to treat HCV prior to LT in those with MELD score of >20 and that therapy be pursued after LT. Thus, a shift towards treating HCV in those on the liver transplant waiting list and with a MELD score >20 to after LT is evolving as a strategy. This argument for post-transplant treatment is further augmented in regions of the world where HCV-positive organs are becoming increasingly available due to the unfortunate epidemic of deaths in those with opioid abuse in persons with injection drug use.32,38 The suggested HCV treatment algorithm in the DAA era in patients with decompensated cirrhosis is shown in Figure 1.
Risks versus benefits in treating hepatitis C virus–infected patients with decompensated cirrhosis in the context of liver transplantation (modified from 37).

CTP: Child–Turcotte–Pugh; GFR: glomerular filtration rate; HCC: hepatocellular carcinoma; HCV: hepatitis C virus; LT: liver transplantation; MELD: Model for End-stage Liver Disease; SVR: sustained virologic response.
In summary, in the era of safe and effective DAA therapy, in patients with compensated cirrhosis, HCV treatment is intuitively indicated in order to improve hepatic function and avoid transplantation. 32 However, not all patients are able to achieve significant improvement in hepatic function and avoid LT. The data thus far suggests that improvement is more often achieved in those with MELD score of < 1539 and less likely in those with MELD > 20, while those in between are in a “ gray zone” for improvement. Thus, the timing of HCV treatment either in pre-LT or post-LT period should be individualized in liver transplant candidates, while universal treatment should be in those who are transplant ineligible or when LT is not available (Figure 1). For patients with decompensated cirrhosis with a MELD score < 15 on the LT waiting list, DAA therapy should ideally be considered because the probability of hepatic function improvement and delisting are high. 27 However, in patients with a MELD score > 20 and CTP class C, studies had shown lesser degree of improvement in hepatic function after HCV therapy27,38 and lower SVR rates; 37 thus, a recommendation of treating HCV after LT is reasonable and this particularly is more relevant in areas where there is access to HCV-positive organs. The decision to treat HCV pre-LT or post-LT in those with MELD between 15 and 20 has to be individualized. However, it is to be appreciated that the majority of patients with decompensated cirrhosis across the world may not be eligible for liver transplant or may not have access to LT; thus, these patients should be considered for HCV therapy with the hope and expectation that they still gain variable degrees of benefit from successful DAA therapy.
Footnotes
Declaration of conflicting interests
Sirina Ekpanyapong declares no conflict of interest. K. Rajender Reddy has served on the Advisory Board of Gilead, Abbvie, Merck, Spark Therapeutics, Shionogi, and Dova, has received Research Support (paid to the University of Pennsylvania) from Abbvie, Gilead, Merck, Conatus, Intercept, Mallinckrodt, Exact Sciences and serves on a Data and Safety Monitoring Board for Novartis.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not‐for‐profit sectors.
