Abstract
Background
The use of interferon-free direct-acting antiviral agents (DAAs) is associated with a rapid short-term decrease in liver stiffness in chronic hepatitis C-infected patients with sustained virologic response (SVR).
Objective
The objective of this article is to evaluate long-term changes in liver elasticity in hepatitis C patients with SVR using transient elastography (TE), FIB-4 and APRI.
Methods
A total of 143 patients were treated with DAAs and reached SVR. Patients received TE measurement (median (range)) at treatment start (baseline), follow-up week 24 (FU24) and follow-up week 96 (FU96). Laboratory data were examined at each date and FIB-4 and APRI were calculated.
Results
Liver elasticity showed a significant decrease from baseline to FU24 (13.1 (3.1–75) kPa to 9.3 (2.9–69.1) kPa;
Conclusion
During long-term follow-up, the majority of patients with SVR had further improved liver stiffness values. Still, a significant proportion of patients may show long-term liver stiffness progression and thus continued TE follow-up is recommended.
Keywords
Key summary
1. Summarise the established knowledge on this subject: - Liver stiffness measurements (LSMs) improve rapidly over a short-term follow-up after successful treatment with direct-acting antiviral agents (DAAs) (follow-up week 24 (FU24)), which is mainly affected by an improvement of hepatic inflammation and by a decrease in portal hypertension. 2. What are the significant and/or new findings of this study? - Short-term reduction in LS values was observed until FU24 in almost every patient. - Further LSM regression was seen in patients with early cirrhosis only but not in individuals with more advanced cirrhosis, which implies that patients with advanced cirrhosis do not fully recover despite hepatitis C virus eradication. - Progression of LSM occurred despite viral clearance in a significant proportion of all patients. - This finding has implications for clinical practice as LSM measurements both at baseline and at FU24 provide relevant information for long-term management of patients after sustained virologic response.
Introduction
Hepatitis C virus (HCV) infection affects approximately 170 million people worldwide, 1 causing liver cirrhosis and hepatocellular carcinoma (HCC) and leading to liver transplantation and ultimately death.2–4 Since the introduction of direct-acting antivirals (DAAs), sustained virologic response (SVR) rates have improved substantially in patients at all fibrosis stages.5–10
Liver biopsy remains the gold standard for the assessment of liver fibrosis in patients with chronic liver disease even though it is an invasive procedure, has limitations regarding the sample volume and is influenced by significant intra- and inter-observer variability. 11 However, liver biopsy in HCV patients is of only minor importance nowadays, finding its use only in special cases. 12
National and international guidelines recommend a standard assessment of fibrosis in HCV patients be performed by transient elastography (TE),12,13 which is a non-invasive method to measure liver stiffness (LS). TE allows a rapid follow-up of a patient’s liver elasticity and development of fibrosis with excellent diagnostic accuracy.14–16 However, fibrosis assessment by TE has been evaluated only in patients with active HCV infection and therefore diagnostic accuracy does not apply to patients with SVR. Recently published short-term studies revealed that patients with an SVR from HCV show a significant improvement in liver stiffness measurements (LSMs).17–20 Likewise, liver inflammation and portal hypertension decrease rapidly in the same observation period.21,22 The influence of these parameters on LSMs might be the explanation for the rapid initial decrease in TE values. In patients with SVR, this influence no longer exists. Therefore, cut-off values generated in patients with active HCV infection cannot be valid in SVR patients. 23
Additionally to TE measurements, non-invasive fibrosis scores such as the Fibrosis-4 Index (FIB-4) and aspartate aminotransferase to platelet ratio index (APRI) have been established in international guidelines. 12 These scores show overall a good diagnostic performance.24,25
So far, long-term fibrosis assessment has been possible only in SVR patients treated with interferon-based therapies. Reaching SVR has shown beneficial effects with a halt or even regression of fibrosis. 26 This study aimed to evaluate long-term changes in liver elasticity in HCV patients with SVR, until 96 weeks after end of treatment, using TE and non-invasive fibrosis scores FIB-4 and APRI. We also intended to analyse different patient groups to identify patients at risk according to the collected data.
Patients and methods
In a prospective, single-centre cohort study at Hannover Medical School, we examined 290 HCV-infected patients between January 2014 and December 2017. The study was approved by the local ethics committee of Hannover Medical School on 31 October 2011 for the evaluation of short-term follow-up and on 11 August 2017 for the evaluation of long-term follow-up. Written informed consent was obtained from all patients. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
All patients included had a chronic HCV infection at treatment start, which was confirmed by anti-HCV immunoassay (RealTime HCV assay, Abbott Molecular, Des Plaines, IL, USA) or by polymerase chain reaction (COBAS AmpliPrep/COBAS TaqMan, Roche, Pleasanton, CA, USA) for quantifiable HCV RNA. Patients received therapy with an interferon-free direct-acting antiviral agent (DAA) for eight to 24 weeks.
Clinical and laboratory assessments were performed at treatment start, which was defined as baseline (BL), at 24 weeks after the end of treatment (FU24) and 96 weeks after the end of treatment (FU96) (Figure 1(a)).
APRI: aspartate aminotransferase to platelet ratio index; BL: baseline; DAA: direct-acting antiviral agent; EOT: end of treatment; FIB-4: Fibrosis-4 Index; FU: follow-up; FU24: follow-up at week 24; FU96: follow-up at week 96; HCC: hepatocellular carcinoma; HIV: human immunodeficiency virus; LTX: liver transplantation; TE: transient elastography.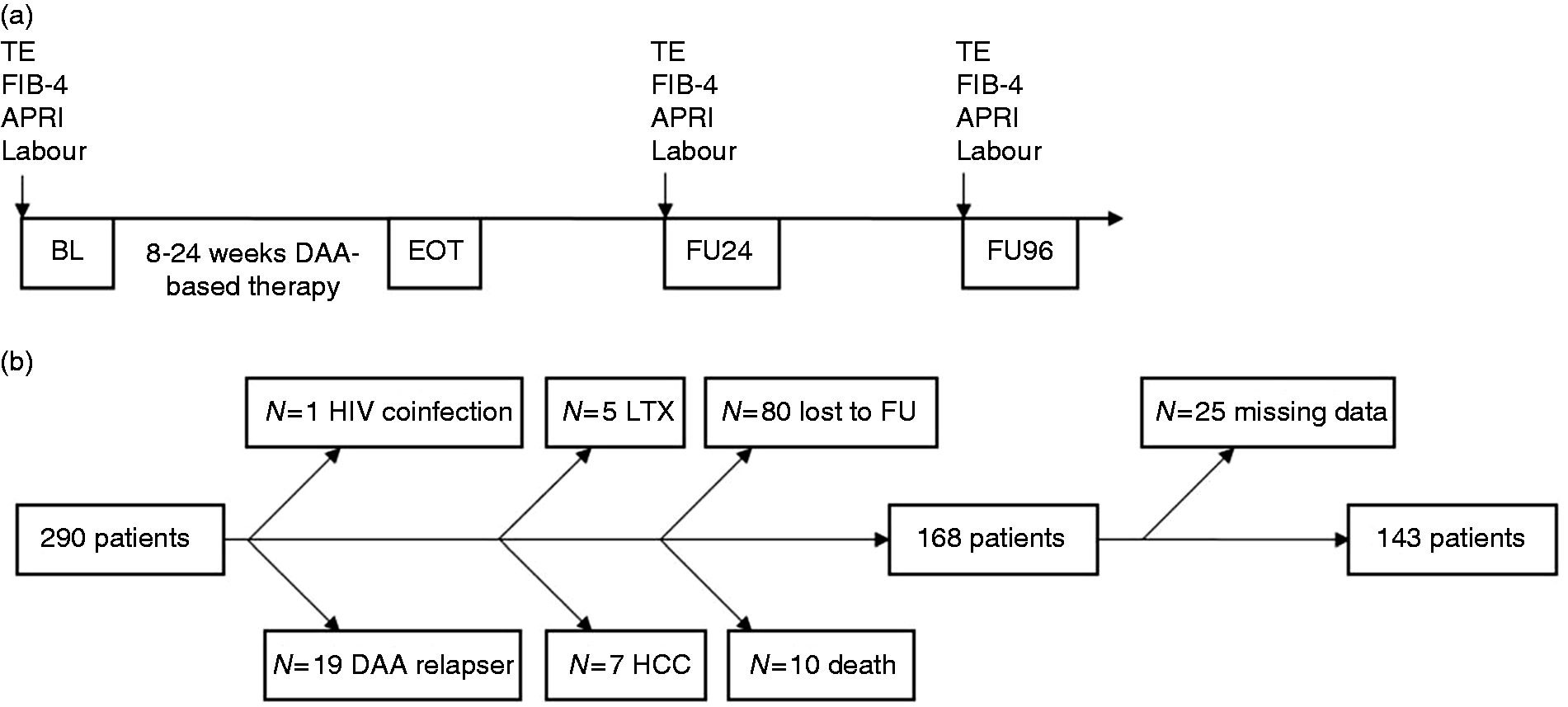
The main exclusion criteria were human immunodeficiency virus (HIV) coinfection, development of HCC or liver transplantation during follow-up and relapsing from a former DAA therapy. All patients included reached SVR defined as no detectable HCV RNA at FU24. After applying our inclusion and exclusion criteria, 143 patients were included for further analysis (Figure 1(b)). The baseline characteristics of the study cohort compared to the patients who were excluded from the study are presented in Supplementary Table 1. Excluded patients more often suffered from liver cirrhosis, and had higher LS values and lower platelet counts but no differences in aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels at baseline in comparison to the study cohort.
Laboratory assessment
At each observation period, patients received a complete blood count – including a platelet count – as well as measurements of AST, ALT, bilirubin, albumin, cholinesterase (CHE) and international normalised ratio (INR).
TE
We performed an LSM by TE (FibroScan®, Echosens, Paris) at BL, FU24 and FU96. LS is expressed in kilopascal (kPa).14,16 Fibrosis staging was performed by TE once at baseline according to the cut-off values for the METAVIR-scoring system: F0/F1 ≤ 7.2 kPa; F2 > 7.2 kPa; F3 > 9.5 kPa; F4 > 14.5 kPa.
17
Depending on the skin-capsule distance, either a medium (
FIB-4 and APRI calculations
At BL, FU24 and FU96, non-invasive fibrosis scores FIB-4 [age (years) × AST (IU/l)/(platelet count (109/l)× √ALT (IU/l))] 28 and APRI [(AST (IU/L)/AST upper limit of normal (IU/l)/platelet count (109/l))× 100], 29 the model of end-stage liver disease (MELD) score and Child-Pugh-Turcotte (CPT) score were calculated.
Subgroup analysis
We analysed patients with cirrhosis and impaired liver function defined as bilirubin >35 µmol/l and/or albumin <35 g/l and/or platelets < 60,000/µl. Assessing the delta (Δ) TE during follow-up, we classified an increase of ≥1.5 kPa as progression and a decrease of ≤1.5 kPa as regression.
Statistical analysis
Continuous variables are given as median and range and categorical data as frequency and percentage. We applied the Wilcoxon test to compare two paired groups, used the Mann-Whitney
Results
Patient characteristics of the study cohort at baseline.

Values are given as median and range.
ALT: alanine aminotransferase; APRI: AST to platelet ratio index; AST: aspartate aminotransferase; BMI: body mass index; CHE: cholinesterase; CPT: Child-Pugh-Turcotte score; FIB-4 Index: Fibrosis-4 Index; INR: international normalised ratio; IU/l: international units per litre; µmol/l: micromole per litre; g/l gram per litre; µl: microlitre; kU/l: kilo units per litre; kPa: kilopascal; MELD: model of end-stage liver disease; TE: transient elastography.
Long-term changes in LS
Patients presented at BL with a median TE value of 13.1 (3.1–75) kPa and decreased significantly until FU24 (9.3 (2.9–69.1) kPa, APRI: aspartate aminotransferase to platelet ratio index; FIB-4: Fibrosis-4 Index; FU: follow-up; FU24: follow-up at week 24; FU96: follow-up at week 96; kPa: kilopascal; TE: transient elastography.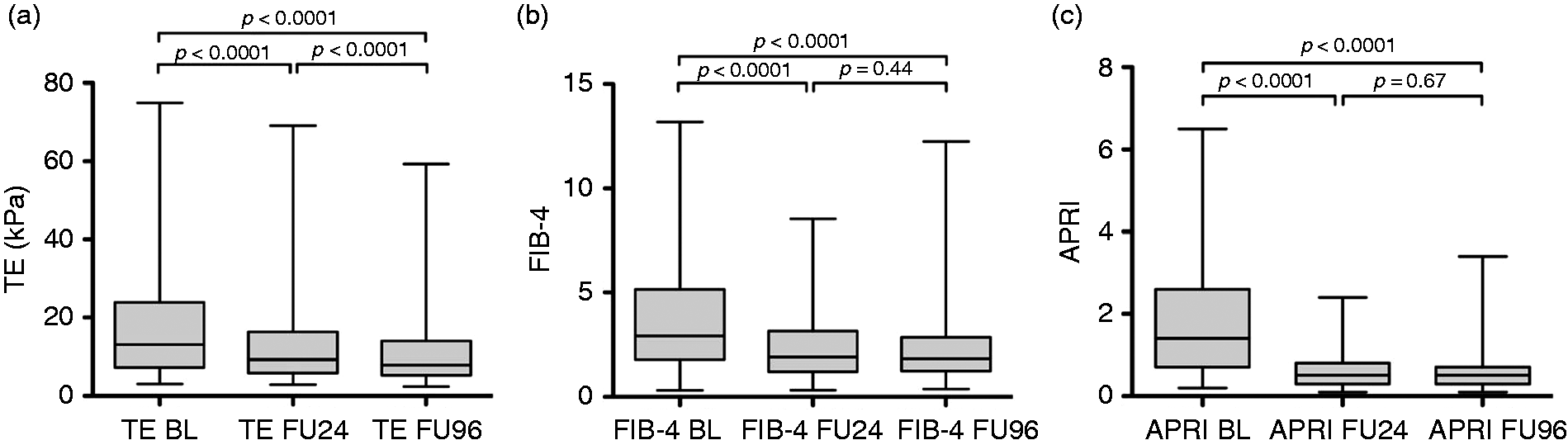
Long-term changes in laboratory parameters
We observed a significant improvement in the aminotransferases AST (
Long-term changes in patients with and without cirrhosis
Long-term changes of patients with and without cirrhosis.
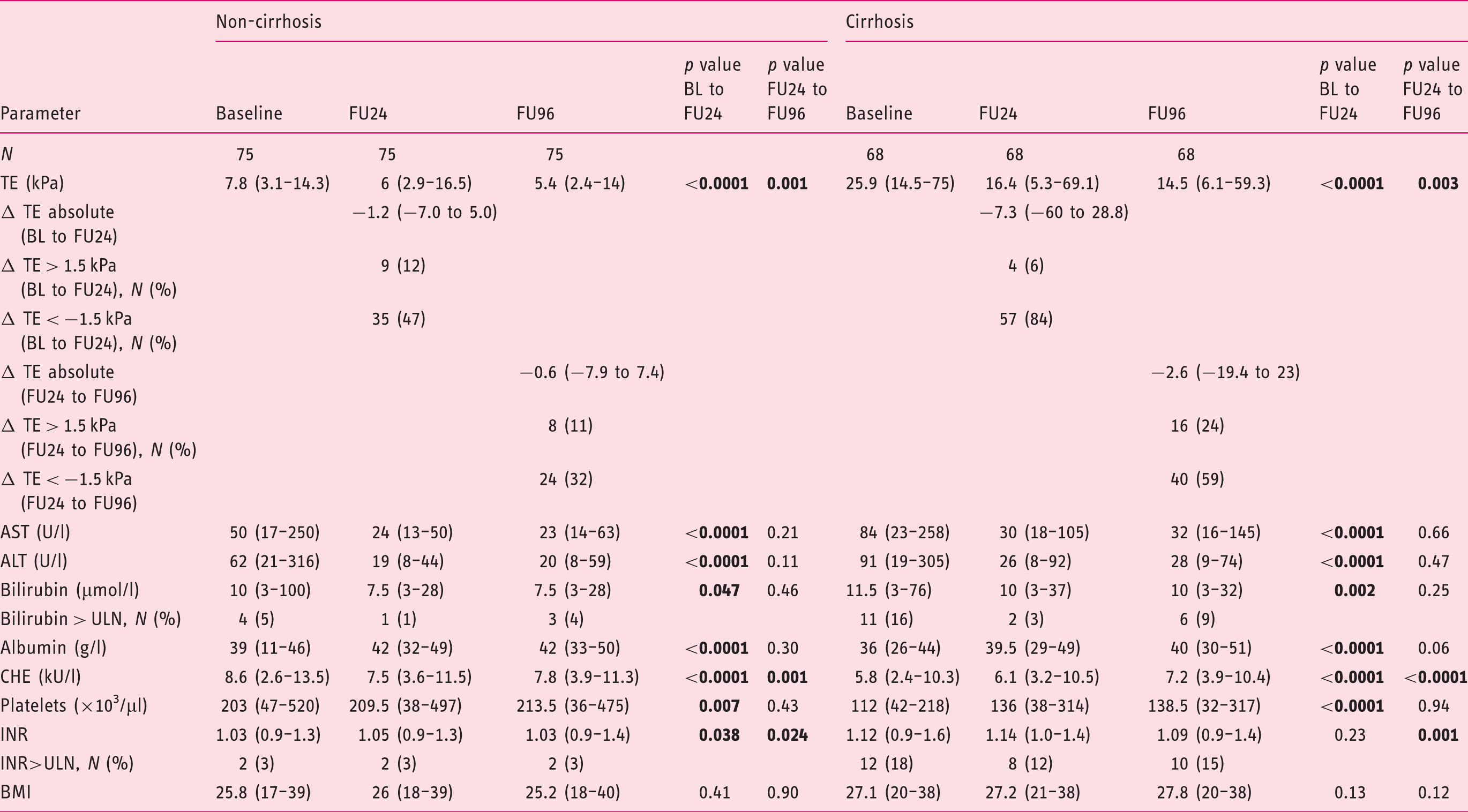
Values are given as median and range;
ALT: alanine aminotransferase; APRI: AST to platelet ratio index; AST: aspartate aminotransferase; BL: baseline; BMI, body mass index; CHE: cholinesterase; FIB-4: Fibrosis-4 Index; FU24: follow-up at week 24; FU96: follow-up at week 96; INR: international normalised ratio; IU/l: international units per litre; µmol/l: micromole per litre; g/l: gram per litre; µl: microlitre; kU/l: kilo units per litre; TE: transient elastography; ULN: upper limit of normal.
Differences between cirrhotic patients with and without impaired liver function
Long-term changes of cirrhotic patients with and without impaired liver function.
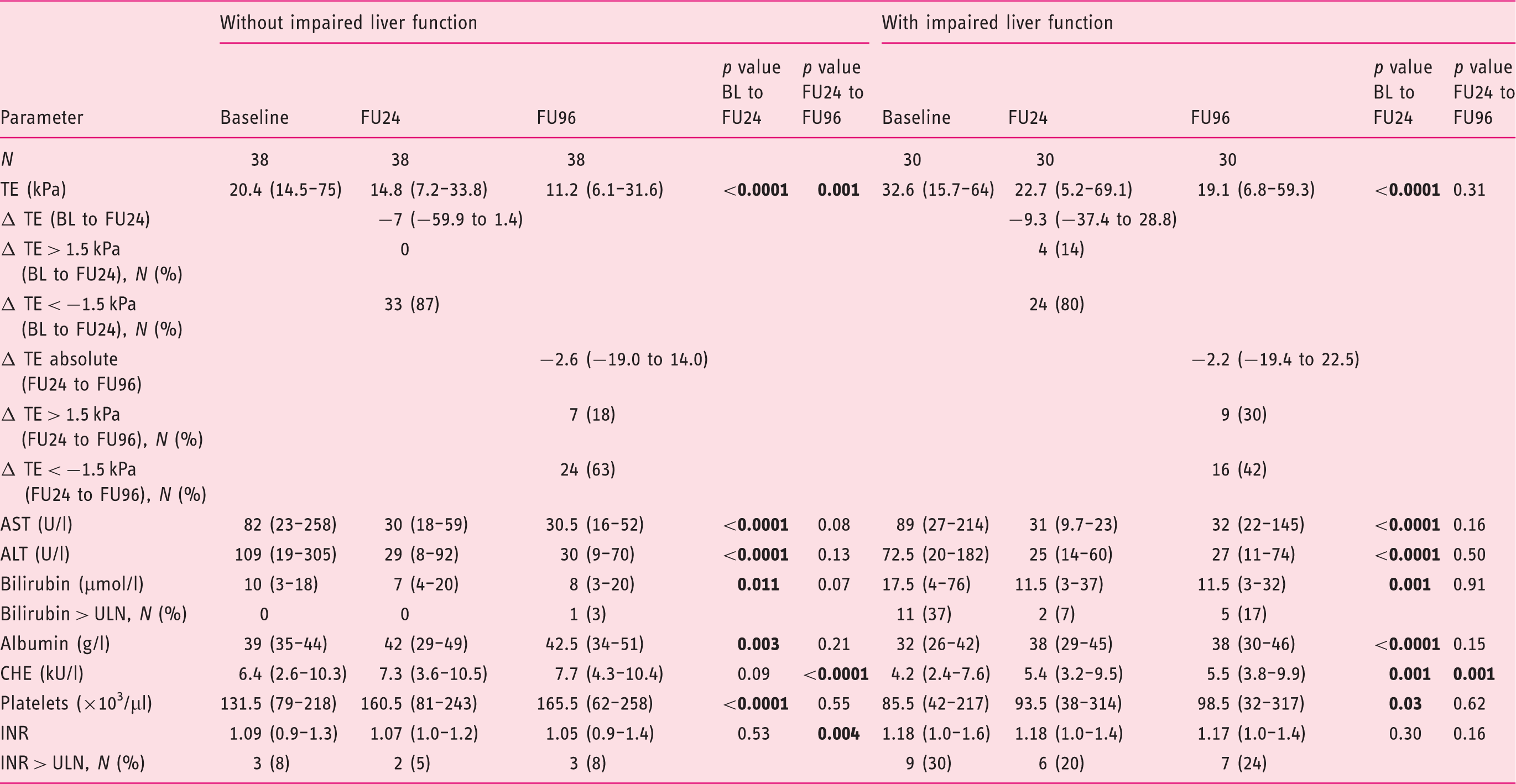
Values are given as median and range;
APRI: AST to platelet ratio index; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BL: baseline; CHE, cholinesterase; FU24: follow-up at week 24; FU96: follow-up at week 96; FIB-4, Fibrosis-4 Index; INR: international normalised ratio; IU/l: international units per litre; µmol/l: micromole per litre; g/l: gram per litre; kU/l: kilo units per litre; µl: microlitre; TE: transient elastography; ULN: upper limit of normal.
Long-term progression of LS
In 24 patients (17%), LSM increased significantly from FU24 to FU96 with a median ΔTE of +3.1 (1.7–22.5) kPa ( Individual course of patients progressing (n=24) with their median TE values between FU24 and FU96. BL: baseline; FU: follow-up; FU24: follow-up at week 24; FU96: follow-up at week 96; kPa: kilopascal; TE: transient elastography.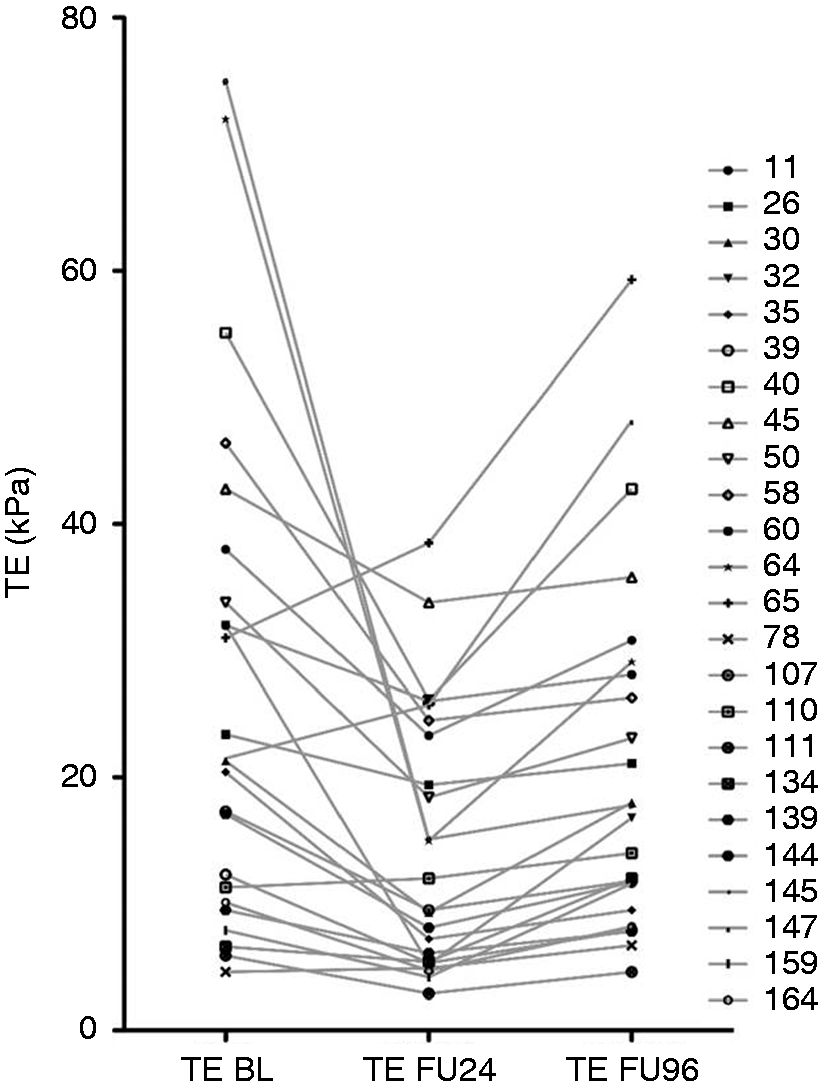
(a) Univariate analysis of factors predicting a progression of TE values from FU24 to FU96.
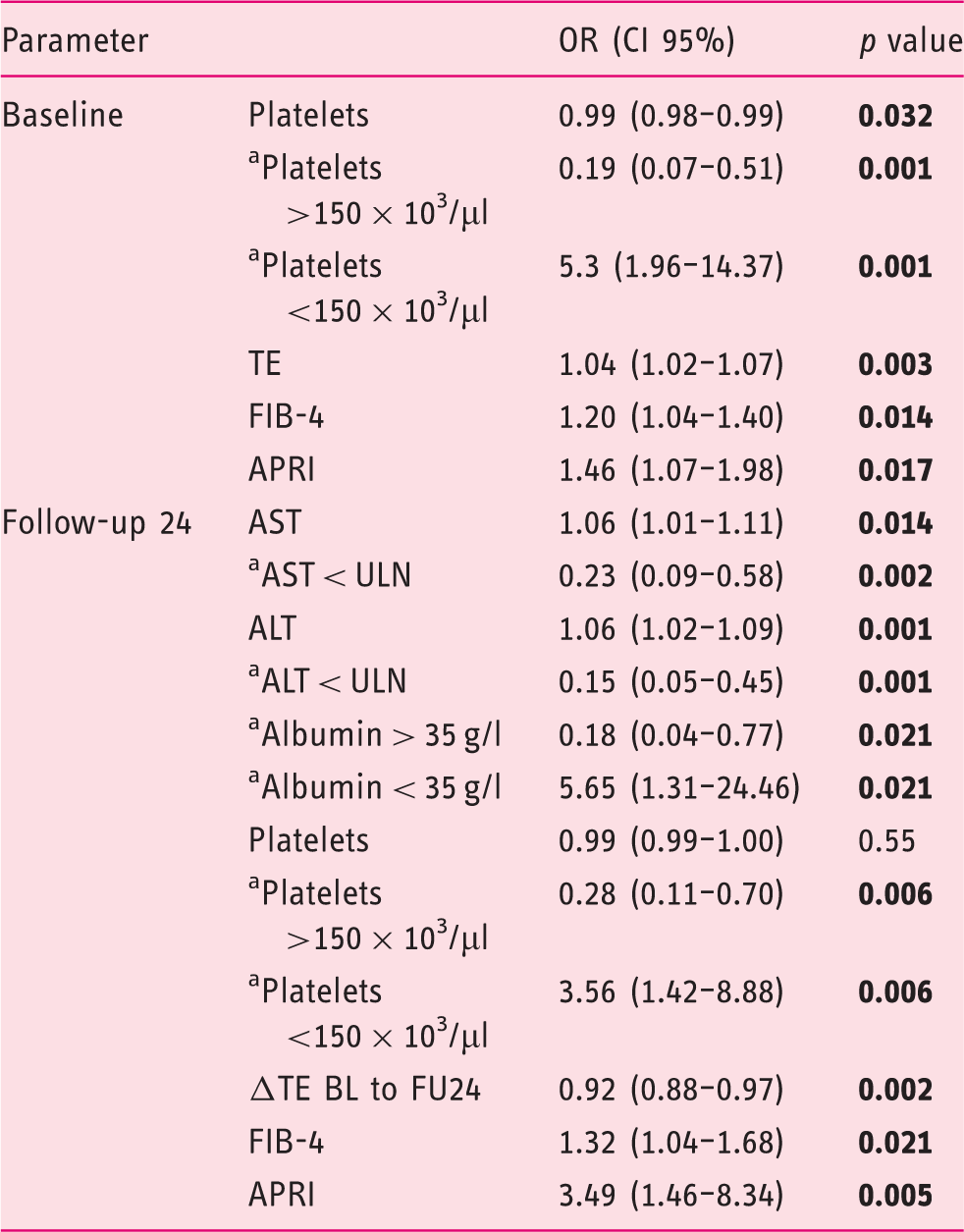
Values are given as odds ratio and 95% confidence interval;
Binary endpoints.
ALT: alanine aminotransferase; APRI: AST to platelet ratio index; AST: aspartate aminotransferase; BL: baseline; CI 95%: 95% confidence interval; FIB-4: Fibrosis-4 Index; FU24: follow-up at week 24; g/l, gram per litre; µl: microliter; OR: odds ratio; TE: transient elastography; ULN: upper limit of normal.
(b) Multivariate analysis of factors predicting progression of TE values from FU24 to FU96.

Values are given as odds ratio and 95% confidence interval;
AST: aspartate aminotransferase; BL: baseline; CI 95%: 95% confidence interval; FU24: follow-up at week 24; FU96: follow-up at week 96; µl: microlitre; OR: odds ratio; TE: transient elastography.
Discussion
The current study investigates, for the first time, the long-term course of LS following successful interferon-free antiviral treatment. Key findings of this study were that (i) a short-term reduction in LS until FU24 was seen in almost every patient regardless of stage of liver disease, (ii) further regression was seen in patients with early cirrhosis but not in individuals with cirrhosis and impaired liver function and (iii) progression of LS values occurred despite viral clearance in about one-sixth of the patients.
In this study a rapid fall in LS was seen over the short-term course until FU24, which is most likely driven by declines in inflammation as already described in recently published studies.18,20,29,30 Early improvements in liver elasticity reflect a reduction in hepatic inflammation, an improvement in hepatic function and/or an improvement in portal hypertension.17,20,21 The early TE decline in this study also paralleled improvements in biochemical and haematological parameters supporting this hypothesis.
We demonstrated that LS continues to improve over the long-term course of two years after the end of therapy. TE improvements paralleled an increase in serum albumin, CHE and INR, indicating a continued improvement in liver function beyond FU24. In contrast to LSM by TE, FIB-4 and APRI did not show significant decreases between FU24 and FU96, which is not really surprising considering that liver transaminases normalised in the majority of patients already until FU24. Therefore, liver elasticity seems to be more suitable to assess the long-term course following HCV-SVR. To what extent improvements in LSMs at FU96 reflect regression of liver fibrosis remains speculative as we obtained no sequential liver biopsies. However, it is tempting to speculate that this was indeed the case in some patients since previous studies following patients after interferon-based regimens demonstrated that fibrosis regression is possible.26,31,32
The LS kinetics differed between patients with and without liver cirrhosis. Patients known to have cirrhosis demonstrated until FU24 a stronger decline in LSM compared to patients without liver cirrhosis, which can be explained by the more elevated aminotransferases at the commencement of treatment. Portal hypertension that is known to improve over the short-term course21,33 seems to remain unchanged over the long-term course, as the study revealed no significant difference in platelet count between FU24 and FU96.
Interestingly, we observed a difference in long-term LSMs between patients with and without impaired liver function. While the LSM values decreased strongly in both groups until FU24, it was no longer possible to identify any significant decline in patients with impaired liver function between FU24 and FU96. This observation implies that patients with impaired liver function do not fully recover despite HCV eradication. Instead, these patients may continue to deteriorate and, consequently, should be closely monitored by laboratory chemical follow-up as well as ultrasound follow-up for potential complications. One other important aspect that can be concluded is that patients with an HCV infection should be treated with DAAs at an early stage.
Interestingly, we observed an increase in LS over the long-term course in about one-sixth of patients despite viral clearance. A large proportion of these patients already had rather high LS values before antiviral treatment. From a clinical perspective, it is important to evaluate possible factors explaining LS progression after SVR. Firstly, a change in lifestyle following eradication of HCV, potentially with weight gain, may play a role. 34 However, in our cohort body mass index did not reveal any significant differences over the course and did not explain TE changes. Other causes might include an increase in alcohol consumption or other hepatic comorbidities. As we did not have detailed information on follow-up alcohol intake or sequential liver biopsies, these assumptions remain speculation. Future studies will need to address these factors as possible causes for LS increases in more detail.
In the current study, it was possible to identify factors associated with the increase in LS between FU24 and FU96. Multivariate analysis revealed that a low ΔTE between BL and FU24 and an elevated AST level at FU24 are associated with LS progression at FU96.
Our study has limitations. The lack of a sequential biopsy makes it impossible to state the changes of fibrosis degree following HCV-SVR. Indirect ultrasound criteria and laboratory chemical parameters were used to assess portal hypertension rather than using the gold standard hepatic venous pressure gradient measurement. This study has a selection bias as patients with HCC or significant ascites were excluded from the study. Finally, our study did not collect personal data from the patients such as diabetes, alcohol intake and lifestyle modification.
In summary, we observed a significant drop in LS over the long-term course in a large portion of patients, which is most likely to be indicative of a persistent improvement in hepatic function and portal hypertension. Patients with an increase in LS during and after successful antiviral treatment should be examined for potentially harmful cofactors and possibly should be supervised more closely in follow-up care as there seems to be a high risk of disease progression. Furthermore, patients should be treated at an early stage. As previously recommended, new cut-off values for hepatic fibrosis staging after HCV healing are needed for this specific patient group. 23
Supplemental Material
Supplementary table -Supplemental material for Long-term changes in liver elasticity in hepatitis C virus-infected patients with sustained virologic response after treatment with direct-acting antivirals
Supplemental material, Supplementary table for Long-term changes in liver elasticity in hepatitis C virus-infected patients with sustained virologic response after treatment with direct-acting antivirals by Veronika Pietsch, Katja Deterding, Dina Attia, Kristina Imeen Ringe, Benjamin Heidrich, Markus Cornberg, Michael Gebel, Michael Peter Manns, Heiner Wedemeyer and Andrej Potthoff in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
None declared.
Funding
This work was supported in part by Else-Kröner-Fresenius-Stiftung in the context of the dissertation programme
Ethics approval
The study was approved by the local ethics committee of Hannover Medical School on 31 October 2011 for the evaluation of short-term follow-up and on 11 August 2017 for the evaluation of long-term follow-up. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
Informed consent
Written informed consent was obtained from all patients.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
