Abstract
Background
Recent reports from western countries have indicated an increased incidence and a decreased mortality in acute pancreatitis. However, the incidence assessment has often been hampered by the inclusion of both first-time and recurrent episodes of acute pancreatitis.
Methods
In this retrospective cohort study, all Swedish residents hospitalized with a first-time episode of acute pancreatitis between 1990 and 2013 were identified using national registers. Sex- and age-standardized incidence rates per 100,000 individuals and year were calculated, as were annual percent changes (APC) from joinpoint regression models.
Results
Overall, between 1990 and 2013, 66,131 individuals had a first-time episode of acute pancreatitis in Sweden. Comparing the first five years (1990–1994) to the last four years (2010–2013) of the study period, the overall incidence of acute pancreatitis increased from 25.2 (95% confidence interval (CI): 24.1, 26.3) to 38.3 (95% CI: 37.0, 39.5) cases per 100,000 individuals and year. An increase in incidence was observed irrespective of the subtypes of acute pancreatitis as well as the sex and age of the patients. Although the incidence of complicated acute pancreatitis declined in both men and women between 1990 and 2004, it started to increase in both sexes (APC 3.0; 95% CI: 0.5, 5.5 in men; APC 5.4; 95% CI: 2.6, 8.2 in women) from 2005 onwards.
Conclusion
Based on nationwide data, the incidence of first-time acute pancreatitis has increased in Sweden over a period of 24 years. The incidence of disease-related complications has also been on the rise during the past few years, after declining for more than 15 years before that.
Key summary
What is known
The incidence of acute pancreatitis has been increasing in many western countries, but many studies include first-time and recurrent acute pancreatitis, hampering the estimation of the true incidence of acute pancreatitis. Few studies have reported estimates of the incidence and trend of severe acute pancreatitis.
Key study findings
The incidence of first-time acute pancreatitis has increased during the last 24 years in Sweden, irrespective of sex, age or disease type. Severe acute pancreatitis has also been on the rise from 2005 onwards, after declining for more than 15 years before that.
Background
Over the past decades, based on either regional or national data, an increased incidence of acute pancreatitis has been reported from several western countries, including England,
1
Ireland,
2
Wales,
3
Sweden,4,5 Finland,
6
Denmark,
7
the Netherlands,
8
and the USA.
9
A more stable incidence has only been reported from a few countries, such as Germany
10
and Spain.
11
In Sweden, the national incidence of
While some of the aforementioned studies have shown a decreased mortality in acute pancreatitis,4,6,10 others have not shown such a change.3,9 Comparisons between studies are, however, difficult because of the variation in how mortality has been defined. For instance, many studies have only reported in-hospital mortality,6,9,10 which, compared to mortality that includes a certain time after hospital discharge (e.g. 30-day mortality), might not fully capture the mortality related to acute pancreatitis. 12
In this population-based study, using nationwide data between 1990 and 2013, we conducted an up-to-date analysis of the incidence and complications (including mortality) of first-time acute pancreatitis in Sweden.
Methods
Study design and setting
We identified all individuals hospitalized with a first-time episode of acute pancreatitis in Sweden between 1 January 1990 and 31 December 2013. Data on first-time acute pancreatitis and mortality were retrieved from validated national registers, as detailed below. The Swedish personal identity number, uniquely assigned to each Swedish resident, allowed for accurate linkage between registers. 13 The study was approved by the Central Ethical Review Board in Stockholm, Sweden.
Data sources
The
The
The
The
Ascertainment of first-time acute pancreatitis
Individuals discharged with a first-time diagnosis of acute pancreatitis (ICD-9: 577A; ICD-10: K85) from a Swedish hospital between 1990 and 2013 were identified via the Swedish Patient Register. None of these individuals had been diagnosed with pancreatitis (acute or chronic) or pancreatic cancer prior to 1990 (see supplementary Appendix for details and ICD codes). Gallstone-related acute pancreatitis was defined as episodes with gallstone-related disease (ICD-9: 574 and 575; ICD-10: K80 and K851) or gallstone-related surgery (the Swedish Classification of Operations and Major Procedures, sixth version: 5300, 5302, 5304, 5350–5359, 5388, 5394, and 9014; seventh version: JKA20, JKA21, JKB00, JKB01, JKE00, JKE02, JKE12, JKE18, JKE25, UJK02, and UJK05) 19 within three months of the hospitalization date, either during the initial hospital stay or after hospital discharge. All other episodes were defined as non-gallstone-related acute pancreatitis. As an additional subgroup, we also identified individuals with a probable alcohol-related etiology, defined as episodes with diagnosis codes for alcohol-related acute pancreatitis (ICD-10: K852, introduced in 2006) or alcohol abuse (see supplementary Appendix for details and ICD codes).
Complicated acute pancreatitis was defined as (a) in-hospital mortality or mortality within 30 days of hospital discharge or (b) admission to the intensive care unit or diagnosis or procedure codes indicating acute respiratory failure, renal failure (not combined with chronic renal failure (ICD-9: 586; ICD-10: N17)), surgical or percutaneous drainage of abdominal fluid collections, abdominal decompression or drainage of pleural fluid collections (see supplementary Appendix for details as well as ICD and procedure codes).
Exclusions
Exclusions were made for individuals with a reused or erroneous personal identity number as well as for those with a diagnosis of chronic pancreatitis or pancreatic cancer within the first three months or 12 months of follow-up, respectively, because of the high probability that they already had chronic pancreatitis or pancreatic cancer misdiagnosed as acute pancreatitis.
Statistical analyses
Sex- and age-standardized incidence rates of first-time acute pancreatitis (total, gallstone-related, and non-gallstone-related) per 100,000 individuals and year between 1990 and 2013 were calculated. The mid-year population for each sex, age, and year (retrieved from Statistics Sweden) was used as the denominator. 20 The sex and age (five-year groups; from 0–4 years up to >90 years) distribution of the Swedish population in 2013 was used as reference for standardization purposes.
The standardized incidence rates were calculated according to five categories of calendar period (1990–1994, 1995–1999, 2000–2004, 2005–2009, and 2010–2013). In subgroup analyses, the standardized incidence rates were calculated by sex, age at diagnosis (<50, 50—69, and ≥70 years), subgroups of non-gallstone-related acute pancreatitis (alcohol-related and non-alcohol-related), and development of complicated acute pancreatitis (no, yes).
We also performed log-linear joinpoint regression analysis over the study period, testing up to a maximum of three joinpoints, to investigate at which calendar year a significant change in the slope of the temporal trend occurred. The results were presented as annual percent change (APC) with 95% confidence intervals (CIs). 21
Statistical analyses were performed using the statistical software Stata (version 14, StataCorp, College Station, TX, USA) and SEER*Stat (version 4.3.1.0, Surveillance Research Program, National Cancer Institute SEER*Stat software (seer.cancer.gov/seerstat)).
Results
After exclusion of individuals with an incorrect personal identity number (
Characteristics of the Swedish population with first-time acute pancreatitis by calendar period (1990–2013).
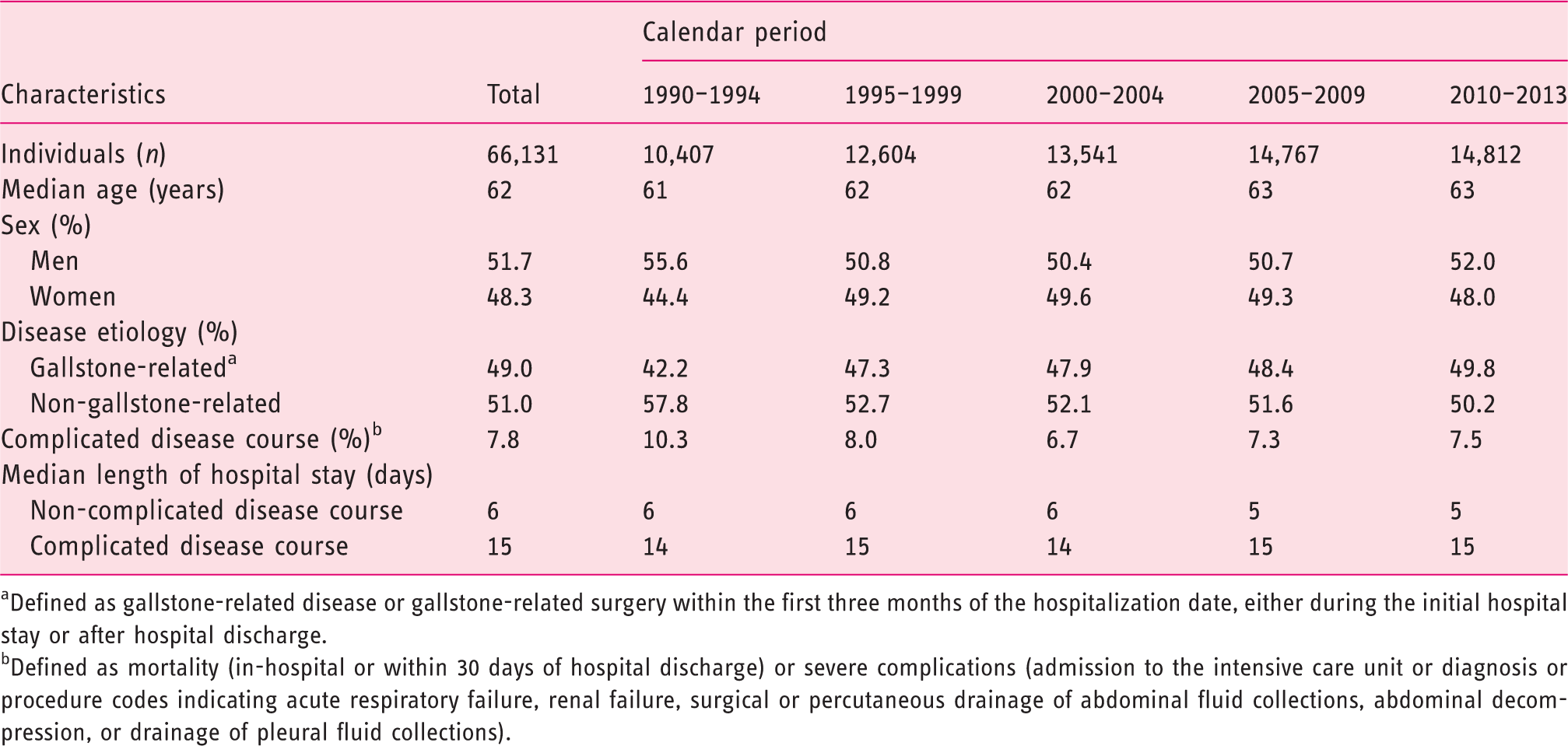
aDefined as gallstone-related disease or gallstone-related surgery within the first three months of the hospitalization date, either during the initial hospital stay or after hospital discharge.
bDefined as mortality (in-hospital or within 30 days of hospital discharge) or severe complications (admission to the intensive care unit or diagnosis or procedure codes indicating acute respiratory failure, renal failure, surgical or percutaneous drainage of abdominal fluid collections, abdominal decompression, or drainage of pleural fluid collections).
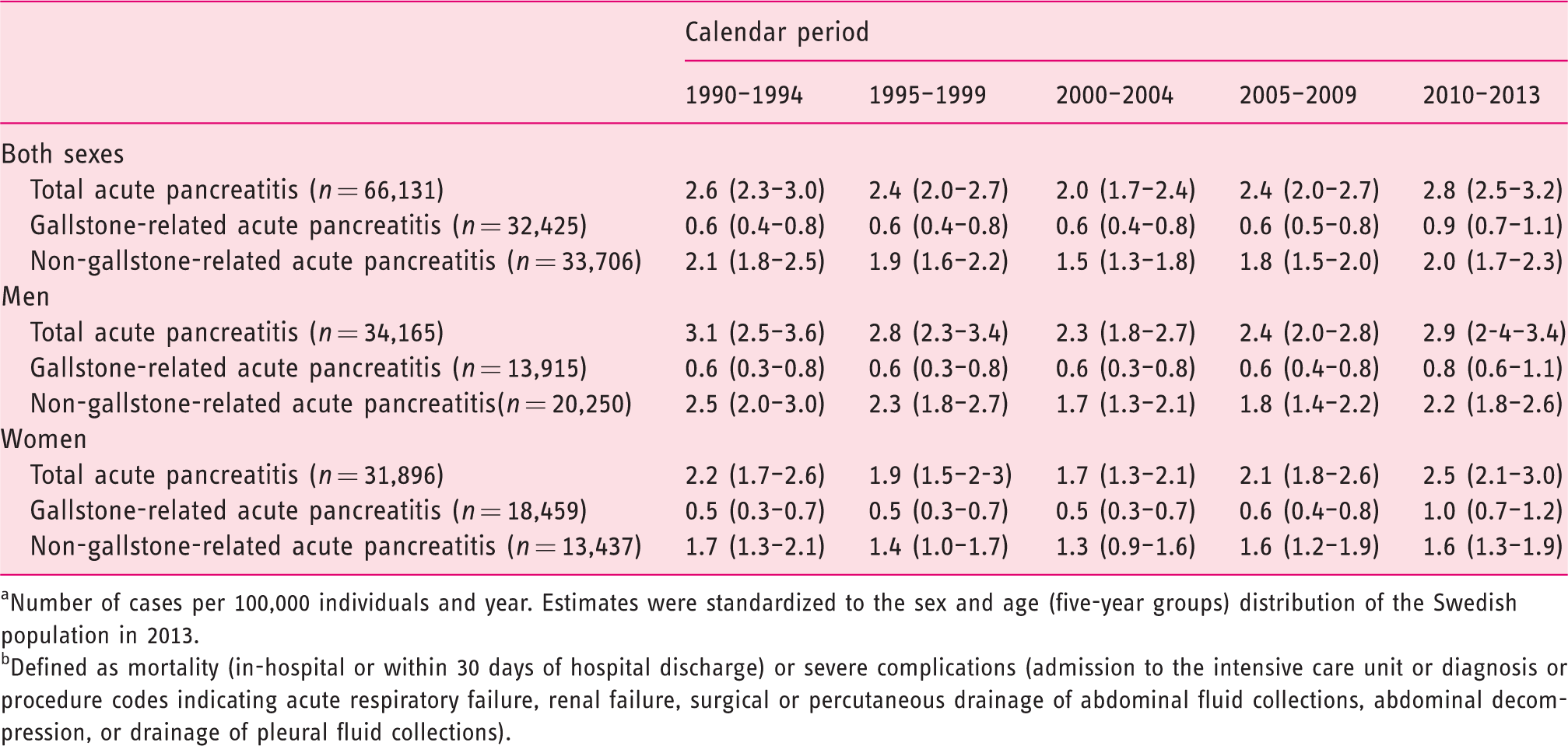
The sex- and age-standardized incidence rates of first-time acute pancreatitis between 1990 and 2013 are shown in Table 2. Comparing the first five years (1990–1994) to the last four years (2010–2013) of the study period, the overall incidence of acute pancreatitis increased from 25.2 (95% CI: 24.1, 26.3) to 38.3 (95% CI: 37.0, 39.5) cases per 100,000 individuals and year. An increased incidence was also seen for gallstone-related (from 10.8 to 19.1 cases per 100,000 individuals and year) and non-gallstone-related (from 14.5 to 19.2 cases per 100,000 individuals and year) acute pancreatitis. The findings were similar in men and women (Table 2) as well as in different age groups (Supplementary Tables 1 and 2), irrespective of the type of acute pancreatitis. In the analysis of alcohol-related acute pancreatitis, the incidence decreased from 1995 to 2004 and then started to increase from 2005 onwards (Supplementary Table 3).
Standardized incidence rates a (95% confidence intervals) of first-time acute pancreatitis in Sweden by calendar period and sex (1990–2013).
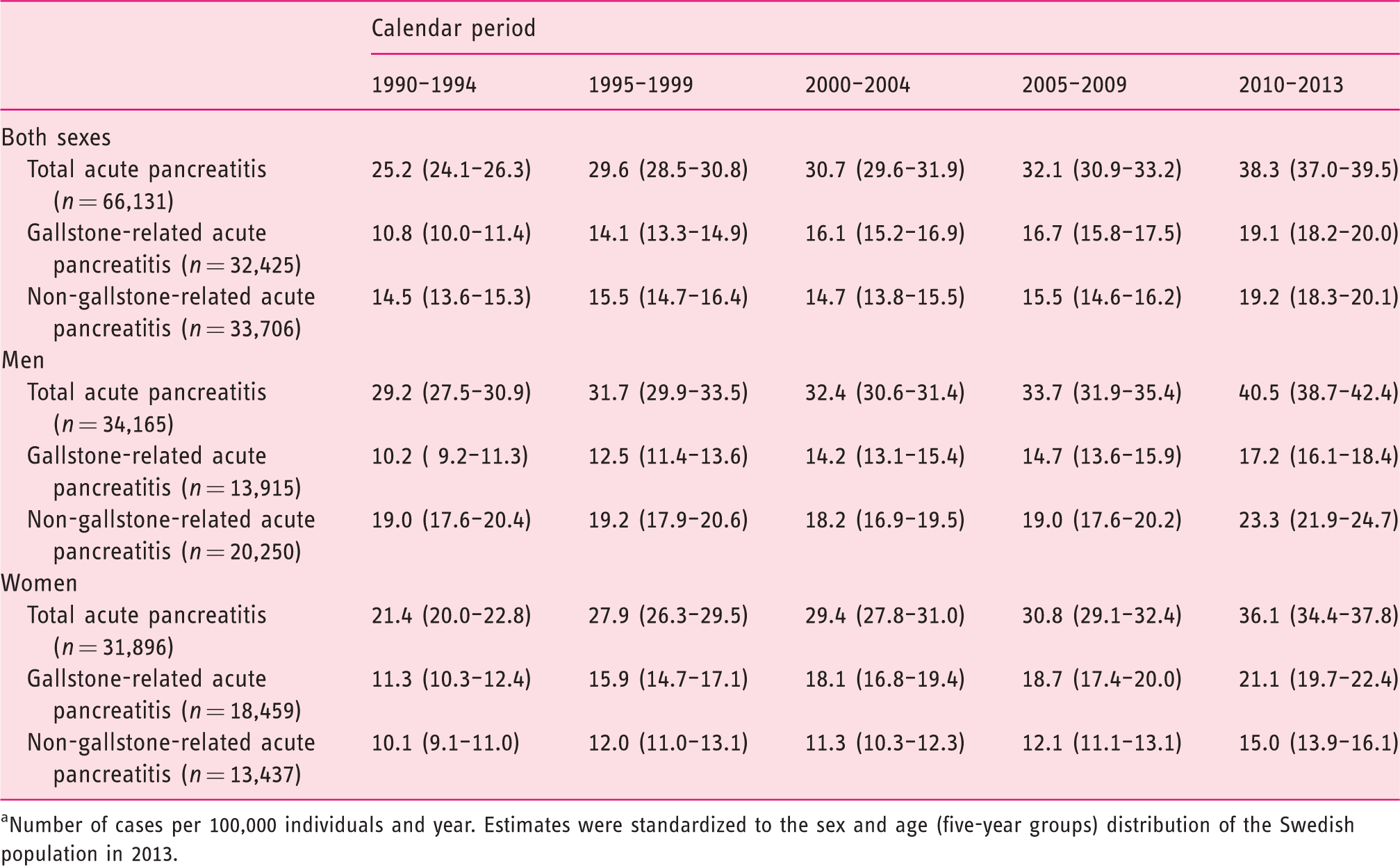
aNumber of cases per 100,000 individuals and year. Estimates were standardized to the sex and age (five-year groups) distribution of the Swedish population in 2013.
Although the incidence of gallstone-related and non-gallstone-related acute pancreatitis increased as a whole between 1990 and 2013, the shape of the sex-specific associations differed over the study period (Table 3, Figure 1). In men, the incidence of gallstone-related acute pancreatitis increased throughout the follow-up period (APC 2.5; 95% CI: 2.0, 2.9). For non-gallstone-related acute pancreatitis, after an initial increase in incidence (albeit non-statistically significant) in 1990 to 1994, the incidence declined in 1995 to 2005 (APC –1.6; 95% CI: –2.7, –0.4), before it started to increase during the rest of the follow-up period (APC 5.0; 95% CI: 3.4, 6.5). In women, the increase in the incidence of gallstone-related acute pancreatitis slowed down gradually over time (including a non-statistically significant decline in 2004–2006) (Table 3). The incidence of non-gallstone-related acute pancreatitis increased by more than 5% in 1990 to 1995 (APC 5.3; 95% CI: 3.2, 7.4) and in 2006 to 2013 (APC 5.1; 95% CI: 4.0, 6.2), but it declined by 1.4% (95 CI: –2.5, –0.2) in 1996 to 2005. Similar associations for men and women were seen in subgroups of non-gallstone-related acute pancreatitis (Supplementary Table 4).
Annual percent change (APC) a (95% confidence interval (CI)) of first-time acute pancreatitis in Sweden by sex (1990–2013).
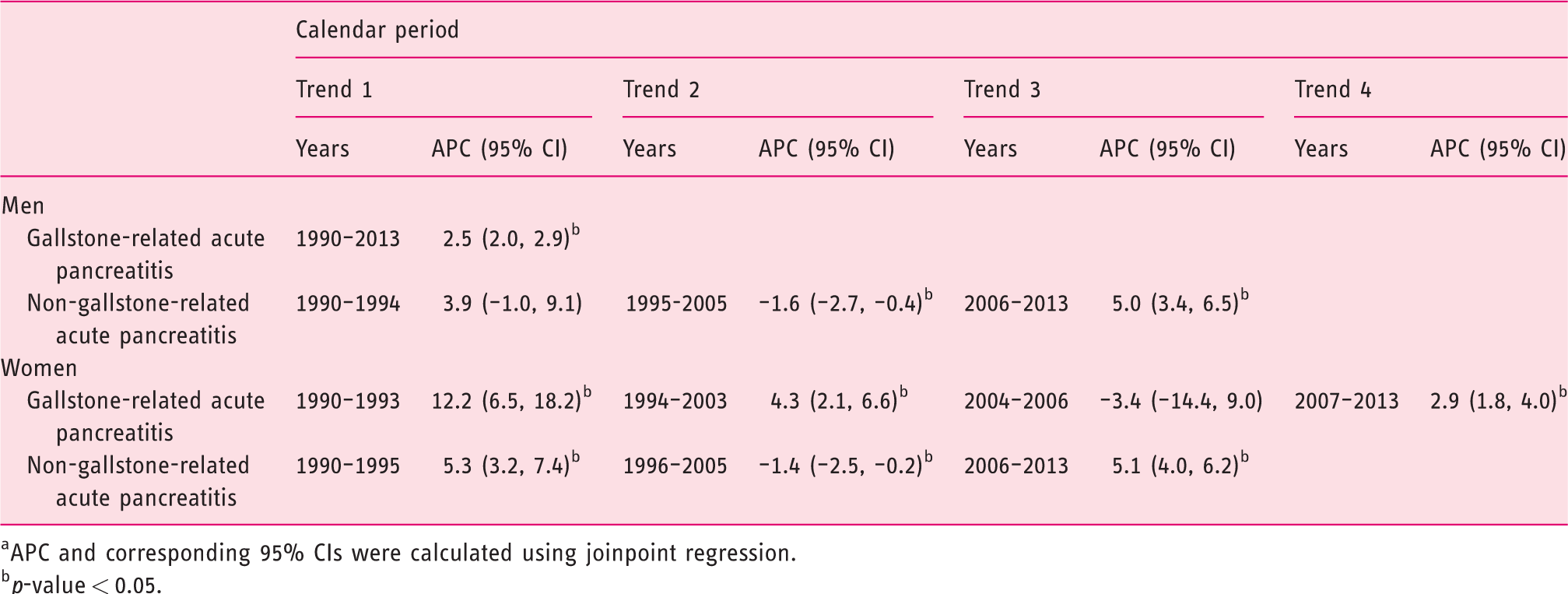
aAPC and corresponding 95% CIs were calculated using joinpoint regression.
b
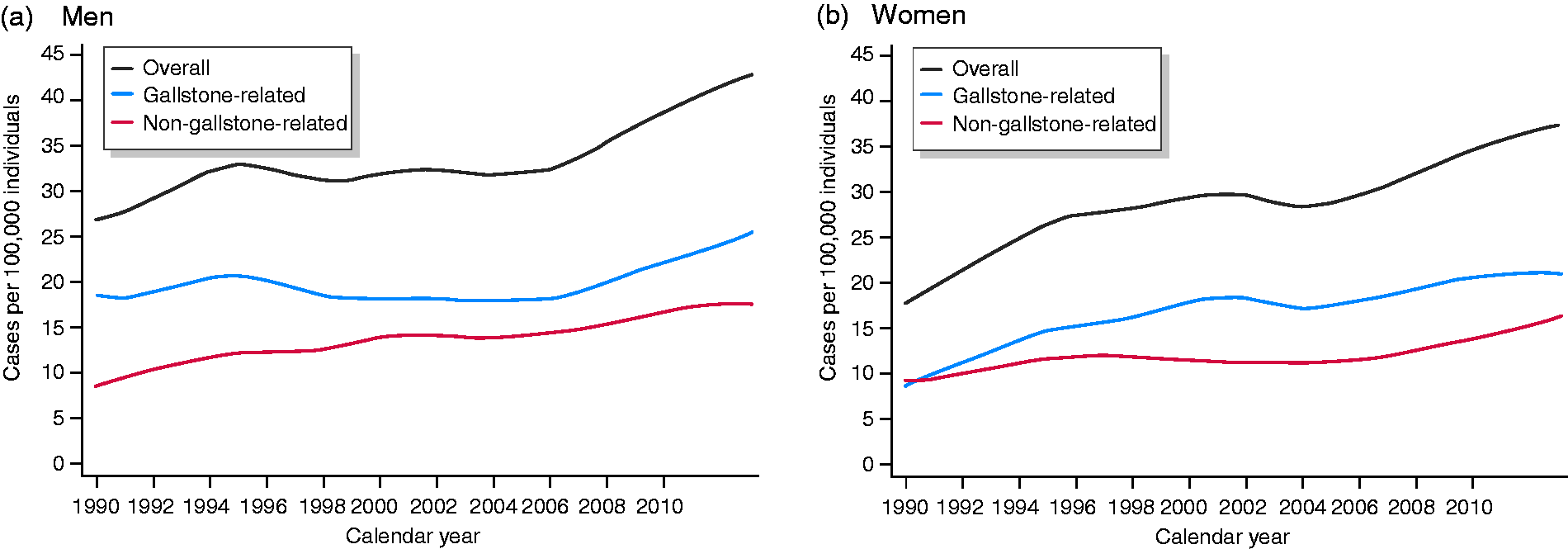
Age-adjusted incidence rates of first-time episode of acute pancreatitis in Swedish (a) men and (b) women, between 1990 and 2013. The mid-year population distribution in 2013 was used as a reference for standardization purposes.
The sex- and age-standardized incidence rate of first-time acute pancreatitis with a complicated disease course declined between 1990 and 2004, before it started to increase between 2005 and 2013 (Table 4). Similar trends were seen for gallstone-related and non-gallstone-related acute pancreatitis as well as in men and women. Using joinpoint regression, the incidence of complicated acute pancreatitis declined in men (APC –3.0; 95% CI: –4,7, –1.3) and women (APC –2.3; 95% CI: –4.3, –0.4) between 1990 and 2004, after which it started to increase in both sexes (APC 3.0; 95% CI: 0.5, 5.5 in men; APC 5.4; 95% CI: 2.6, 8.2 in women).
Standardized incidence rates a (95% confidence intervals) of first-time acute pancreatitis with complicated disease course b in Sweden by calendar period and sex (1990–2013).
aNumber of cases per 100,000 individuals and year. Estimates were standardized to the sex and age (five-year groups) distribution of the Swedish population in 2013.
bDefined as mortality (in-hospital or within 30 days of hospital discharge) or severe complications (admission to the intensive care unit or diagnosis or procedure codes indicating acute respiratory failure, renal failure, surgical or percutaneous drainage of abdominal fluid collections, abdominal decompression, or drainage of pleural fluid collections).
Discussion
In this nationwide population-based study, we observed that the incidence of first-time acute pancreatitis increased over the past 24 years in Sweden, although the shape of the association differed over the study period. An increase in incidence was present irrespective of the type of acute pancreatitis as well as the sex and age of the patients. In addition, after an initial decline up until the mid-2000s, the incidence of complicated acute pancreatitis (defined as mortality in-hospital or within 30 days of hospital discharge and/or certain organ dysfunctions) increased during the last years of the study period up to a level comparable to that of the early 1990s.
Several studies have reported an increased incidence of acute pancreatitis in western countries (e.g. in the UK, Scandinavia, and the USA),1–4,6–9 with a more stable incidence reported from only a few countries (e.g. in Germany and Spain).10,11 A previous study based on nationwide Swedish data reported that the incidence of first-time acute pancreatitis increased between 1988 and 1997, after which the incidence was stable until the end of follow-up in 2003. 4 In another Swedish study, based on regional data, the incidence of acute pancreatitis increased by 3.9% per year between 1985 and 1999. 5 In our study, the overall incidence of acute pancreatitis in Sweden increased by more than 50% between 1990 and 2013. This increase was more pronounced for gallstone-related acute pancreatitis (87% increase in women and 69% in men) than for non-gallstone-related acute pancreatitis (49% increase in women and 23% in men). While the incidence of gallstone-related acute pancreatitis increased rather gradually over time, it should be noted that the incidence of non-gallstone-related acute pancreatitis seemed to vary over time, with increased incidence in the early 1990s and the late 2000s and decreased incidence in the mid-1990s to mid-2000s. In subgroup analyses, this variability seemed to be explained by a sharp decline of alcohol-related acute pancreatitis in the late 1990s. A similar finding was seen in the previously mentioned study based on regional Swedish data, in which the incidence of alcohol-related acute pancreatitis decreased by 5.1% per year between 1985 and 1999. 5
During the past decades in Sweden, there have been substantial changes in the prevalence of several common lifestyle risk factors for acute pancreatitis, such as abdominal obesity, cigarette smoking, and alcohol abuse. Obesity, an acknowledged risk factor for gallstone disease,22,23 is a growing problem worldwide, and Sweden is no exception.24,25 A role of obesity, particularly abdominal adiposity, has also been suggested in the development of acute pancreatitis, both gallstone-related and non-gallstone-related.26–29 It is, therefore, likely that the increased prevalence of obesity has contributed to the increased incidence of acute pancreatitis in our study. In contrast, the prevalence of cigarette smoking has declined steadily in Sweden over the past decades, 30 and only 11% of the Swedish population were estimated to be daily smokers in 2013. 25 As such, it is very unlikely that cigarette smoking had an impact on the observed increase in incidence of acute pancreatitis. Finally, with respect to alcohol abuse, the average yearly alcohol consumption per person in Sweden (based on the total amount of purchased alcohol beverages) increased steadily from 1990 to 2004 (from 7.8 to 10.5 liters of alcohol). 31 Between 2004 and 2013, the average yearly alcohol consumption was fairly stable around 10 liters per person. 32 During the same time period, the estimated overall risk consumption of alcohol (>14 and >9 standard drinks of alcohol per week in men and women, respectively) declined, owing to a decline among individuals aged 16 to 29 years. 25 However, the alcohol consumption was practically unchanged or even increased among older age groups. 25 Our data for alcohol-related acute pancreatitis did not fully follow the trends in alcohol consumption in Sweden, as the incidence decreased from 1995 to 2004 and then started to increase from 2005 onwards. Whether altered drinking behaviors, especially among older age groups, might have affected the observed incidence rates beyond 2005 is speculative and out of scope for the current study. It is also important to stress that a specific ICD code for alcohol-related acute pancreatitis was introduced in 2006, leading to a relative underdiagnosis of alcohol-related acute pancreatitis in the years prior to 2006. Thus, our incidence estimates for alcohol-related acute pancreatitis should be interpreted with caution.
Apart from lifestyle risk factors, it should also be noted that the rates of cholecystectomies have been declining in Sweden over the past years. 33 Reasonably, this decline is due to a reduced number of cholecystectomies in individuals with biliary colic pain, among whom a future gallstone-related acute pancreatitis would have been prevented by early cholecystectomy. 34 Thus, some of the increased incidence rates of gallstone-related acute pancreatitis might be explained by this issue.
In the previous Swedish study based on nationwide data, the authors reported that the disease-related mortality (defined as death within 90 days of admission) declined throughout the entire follow-up period (1988 to 2003). 4 In our study, the incidence of complicated acute pancreatitis declined between 1990 and 2004, which may indicate improved clinical management of acute pancreatitis and/or improved and earlier diagnostics of acute pancreatitis (due to a more frequent use of serum amylase and radiologic imaging in patients with abdominal pain). 35 The latter could also, in theory, explain a proportion of the overall increase in incidence, particularly for non-complicated acute pancreatitis, which might have gone undetected with the less sensitive diagnostics of years past. However, the incidence of complicated acute pancreatitis increased from 2005 onwards, alongside an increase in the incidence of non-complicated acute pancreatitis. The complicated form of acute pancreatitis requires careful in-hospital management and will eventually be detected. Thus, our findings for complicated acute pancreatitis might indicate that there has been an actual increase in the incidence of acute pancreatitis in Sweden.
An interesting finding in our study, especially from a clinical perspective, was that the median hospital stay for patients with acute pancreatitis did not substantially change during the study period – if anything, it actually increased for those with complicated disease course (14 days in 1990 to 1994 versus 15 days in 2010 to 2013). While the reason for this finding is unclear, it does indicate that physicians – despite advances in intensive and surgical care – have not been able to find sufficient clinical treatments to improve the disease course of complicated acute pancreatitis.
The strengths of this study included its population-based design, the complete nationwide coverage of Swedish national registries, and the accessibility of the Swedish health care system to all Swedish residents. These features enabled identification of all individuals with a first-time episode of acute pancreatitis, ensured accurate estimation of mortality and complications related to acute pancreatitis, and reduced selection bias. The high accuracy of a recorded diagnosis of acute pancreatitis in the Swedish Patient Register should also have led to a low misclassification of the diagnosis. 15 With that said, some study limitations must be mentioned. We cannot rule out that some individuals with gallstone-related acute pancreatitis were misclassified as having non-gallstone-related acute pancreatitis, and vice versa (especially since the specific ICD codes for gallstone-related and alcohol-related acute pancreatitis have not been validated). While such misclassification might have affected the incidence estimates for subtypes of acute pancreatitis, it should not have had any impact on our findings for the total incidence of acute pancreatitis. Furthermore, it is possible that some individuals with a history of acute pancreatitis prior to 1987 – when the Swedish Patient Register did not have nationwide coverage – might have been misclassified as having a first-time episode of acute pancreatitis. However, the median time to a recurrent episode of acute pancreatitis has been reported to be less than a year in Sweden. 19 Such misclassification should, therefore, most likely have led to an overestimation of the incidence estimates during the first years of the study period.
In conclusion, the incidence of first-time acute pancreatitis has increased in Sweden over a period of 24 years. The mortality and complications of the disease have also been on the rise during the past few years, after declining for more than 15 years before that. The increasing incidence of acute pancreatitis, particularly gallstone-related disease, will continue to comprise a health care challenge in western countries.
Supplemental Material
UEG913737 Supplemental Material - Supplemental material for Rising incidence of acute pancreatitis in Sweden: National estimates and trends between 1990 and 2013
Supplemental material, UEG913737 Supplemental Material for Rising incidence of acute pancreatitis in Sweden: National estimates and trends between 1990 and 2013 by Viktor Oskarsson, Servah Hosseini, Andrea Discacciati, Per Videhult, Anders Jans, Anders Ekbom and Omid Sadr-Azodi in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics approval
The study was approved by the Central Ethical Review Board in Stockholm, Sweden.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the Swedish Society of Medicine; the Centre for Clinical Research, Sörmland, Uppsala University; and the Uppsala and Örebro Research Council, Sweden. Grant number for funding from Swedish Society of Medicine – SLS-684471 Grant number for funding from Centre for Clinical Research Sörmland DLL-746261 Grant number for funding from Uppsala and Örebro Research Council RFR-736251.
Informed consent
Informed consent is not required for register-based research in Sweden.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
