Abstract
Background
The use of 19-gauge (G) stainless steel needles for endoscopic ultrasound-guided fine-needle biopsy of a pancreatic mass often results in technical difficulties due to an inability to advance the relatively rigid needle out of the endoscope. More flexible nitinol-based needles might decrease such technical difficulties and thus increase diagnostic accuracy.
Objective
In this prospective multicenter randomized single-blinded study we compared the diagnostic value of those two needle types in patients with a solid pancreatic lesion.
Methods
Patients with a solid pancreatic mass were diagnosed with endoscopic ultrasound-guided fine-needle biopsy using one puncture with each needle in a randomized fashion. The primary endpoint was the diagnostic accuracy of each needle. Secondary endpoints included time for puncture, amount of tumour tissue obtained, and technical failure. Histological specimens were centrally reviewed by a pathologist blinded to the final needle type and final diagnosis (ClinicalTrials.gov Identifier: NCT02909530).
Results
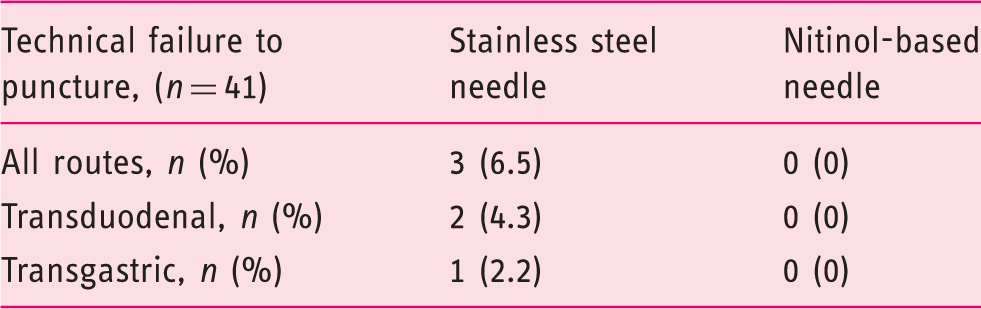
Out of 46 prospectively recruited patients, central pathological examination was available for 41. Diagnostic accuracy for the two needles combined was 87.8%. Diagnostic accuracy was 66% and 68% using the stainless steel- and nitinol-based needle respectively. Time spent for puncturing was 137 ± 61 s (mean ± standard deviation) for the stainless steel and 111 ± 53 s for the nitinol-based needle (p = 0.037). Technical failure occurred in three (6.5%) cases using the stainless steel- and in none using the nitinol-based needle.
Conclusions
Usage of a nitinol-based 19-G needle failed to present a significant superior accuracy compared with a stainless steel needle in endoscopic ultrasound-guided fine-needle biopsy of solid pancreatic lesions.
Key summary
19-gauge (G) endoscopic ultrasound fine-needle biopsy (EUS-FNB) stainless steel needles are able to obtain more cellular material than 22-G needles. Due to the greater needle diameter, technical difficulties often occur, especially during the advancement of the needle out of the endoscope. Nitinol-based 19-G EUS-FNB needles are more flexible and may therefore potentially overcome current limitation of stainless steel. Use of a nitinol-based 19-G needle failed to present significant superior accuracy compared with a stainless steel needle in EUS-FNB of solid pancreatic lesions. Use of a nitinol-based 19-G needle reduced the time required to perform the puncture significantly.
Introduction
Today, endoscopic ultrasound (EUS)-guided fine-needle aspiration (FNA) or -biopsy (FNB) is the standard for obtaining a histological specimen of a suspicious pancreatic mass. 1 The principle of this technique was first described in 1930 2 and since then has evolved to include the variety of needle tip types and diameters we use today in our endoscopy units. 3 Despite the progress that has been made to improve the technique, a survey involving endosonographers from 29 countries revealed a level of sensitivity that was considerably lower than that reported in the literature. 4 Two of the main reasons were the lack of rapid on-site cytopathological evaluation (ROSE) and the absence of microcore isolation. Changing the needle diameter from 22-gauge (G) to 19-G increased the cellular material obtained from the puncture site in a comparative study. 5 However, due to the greater needle diameter, 5 out of 26 punctures (19.2%) resulted in technical failure. Obtaining histological material from the pancreatic head is especially important because most of the pancreatic ductal adenocarcinomas (PDAC) are located in this region. 6 However, choosing the transduodenal route often results in significant bending of the endoscope with the inability to advance the rigid 19-G needle. 7 The reported success rate of 33 out of 35 (94%) EUS-FNB for acquiring histological material via the transduodenal route with a 19-G needle is often difficult to reproduce in a regular setting. 8 In the latter study the authors reported that in many cases the needle needed to be pushed out of the endoscope in the stomach before advancing the endoscope into the duodenum.
The use of nitinol instead of stainless steel promised to result in more flexible needles for EUS-FNB, and reports of their clinical application have been encouraging. However, to the best of our knowledge all published studies are of a single-arm design and thus did not present a direct comparison between the two needle types.8–12
In this prospective, multicenter, single-blinded, randomized trial we aimed to compare the diagnostic yield of a 19-G stainless steel EUS-FNB needle and a 19-G nitinol needle in regard of the histological diagnosis of a solid pancreatic mass as a primary endpoint. Secondary endpoints included diagnostic yield subdivided by puncture route as well as diagnostic accuracy depending on the order of the needles, time spent on puncture, technical failure rate and histological amount obtained.
Methods
Patients
After obtaining written informed consent, consecutive patients with a solid pancreatic mass and indicated for EUS-FNB were prospectively recruited in four German centres between September 2016 and December 2017. Incapacity to give informed consent, haemorrhagic disease, disorders of hemostasis and coagulation (PT < 60%, PTT > 42 s and platelets < 60000/µL), oral anticoagulants and dual platelet inhibitor therapy were exclusion criteria. The study, the protocol and patient informed consent forms were approved by the ethics committee of each participating centre before recruitment. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki, as reflected in a priori approval by each institution’s human research committee. The ethics committee of the University of Ulm approved the study on the 1 September 2016. The trial has been registered in a publicly accessible registry (ClinicalTrials.gov identifier: NCT02909530).
EUS-FNB procedure
The Olympus EZ Shot 2 19-G was used for the stainless steel needles and the EZ Shot 3 19-G Plus for the nitinol-based needles. After initiation of sedation and visualization of the solid pancreatic target lesion, the puncture was performed with both needles: only one puncture per needle in a predefined randomized order. Additional puncture attempts were allowed according to protocol and were made at the endosonographer’s discretion. The results of those additional punctures were not analyzed by needle type for the study endpoints. Due to the different handle shapes, needle assignment was not blinded to the examiners who performed EUS-FNB. The predefined order was identified during the procedure by opening a sealed envelope. The puncture technique (i.e. with suction) was recorded for each patient, but was not predefined by the study in order to allow the examiners to use the technique they were most experienced with. The time from insertion of the needle into the endoscope to extraction was recorded per needle in seconds. Technical failure was defined as the inability to advance the needle out of the endoscope in order to perform the puncture. The examiners were instructed to use histological instead of cytological analysis to evaluate the puncture material. Thus, after the puncture, the obtained material was collected for each needle and placed separately in buffered 4% formaldehyde solution for histological examination. The specimens were labelled either ‘1’ or ‘2’ in order to blind the pathologist to the needle type used.
Follow-up and adverse events
Mainly in order to identify the correct final diagnosis, each centre performed follow-up by outpatient visits and telephone calls. Adverse events related to the procedure that occurred during follow-up were prospectively reported. In addition, further examinations like CT scans, percutaneous biopsies and surgical interventions with histological examination of the pancreatic lesions were prospectively recorded up to 1.5 years after the puncture in order to rule out malignancy.
Central histological examination
After inclusion of the final patient, histological slides from all centres were collected and centrally analyzed by an experienced pathologist who was blinded regarding the needle type used. The pathologist reviewed both slides with the knowledge they were from the same patient. Instead of an initially planned subjective comparison of histological quality using a visual analogue scale and overall satisfaction, the slides were scanned and the total surface area and the tumour proportion were measured manually by the pathologist to an accuracy of square millimetres. Measurement was performed by manual delineation of normal and tumorous tissue without automatic delineation algorithms in order to avoid false positive results. All measurements were performed using NDP.serve 3.1.85 (Hamamatsu Photonics K.K., Japan).
Final diagnosis
In cases of the presence of malignant disease on the slides acquired through the intervention, final diagnosis was made by the reference pathologist. In cases that were unclear or of benign disease, final diagnosis was based on the surgically resected specimen, or on further biopsies that were obtained. If the reference pathologist did not diagnose a malignant disease and no surgical specimen could be obtained, the final diagnosis was made based on a minimum clinical follow-up time of 1 month up to 1.5 years for cases that were unclear in the first follow-up. Spontaneous resolution or no change in the radiological finding indicated a benign disease. A rapid progression of the lesion or deterioration of the clinical course indicated a malignant disease.
Statistical analysis
Sample size was calculated with an estimated diagnostic accuracy of 80% and 60% for nitinol-based and stainless steel needles, respectively. Sample size needed to test 1 proportion: 1-sample, 2-sided equality, type I error rate, α of 5% and power, 1−β of 0.8. The calculation indicated that 32 patients were required to detect a significant difference of 20% in diagnostic accuracy between the two needles. We anticipated a loss to follow-up of 25%, thus we planned to recruit 40 patients.
The primary endpoint was the difference in diagnostic accuracy of each needle, defined by comparison of histological diagnoses of the specimens obtained and the final diagnosis of the pancreatic lesion. Secondary endpoints included diagnostic accuracy differentiated by puncture route (transduodenal or transgastric), and diagnostic accuracy depending on the order of the needles, time spent on the puncture, technical failure and histological quality. All analyses were performed using IBM SPSS Statistics 24 (SPSS Inc. Chicago Illinois, USA). Normally distributed continuous variables are reported as mean and standard deviation (SD), non-normally distributed variables are reported as median and interquartile range (IQR). Significance was evaluated using the Student’s t-test for normally distributed- and the Mann–Whitney U test for non-normally distributed continuous values. Categorical variables were expressed by percentage and were compared using the chi-square test. In cases where cells had an expected count of less than 5, Fisher’s exact test was performed. A two-tailed significance level was chosen. P less than 0.05 was considered statistically significant.
Results
Patient characteristics and endoscopic intervention
Baseline characteristics. Pancreatic ductal adenocarcinoma (PDAC), standard deviation (SD).

PDAC = pancreatic ductal adenocarcinomas.
Final diagnosis of the solid pancreatic lesions. Pancreatic ductal adenocarcinoma (PDAC).
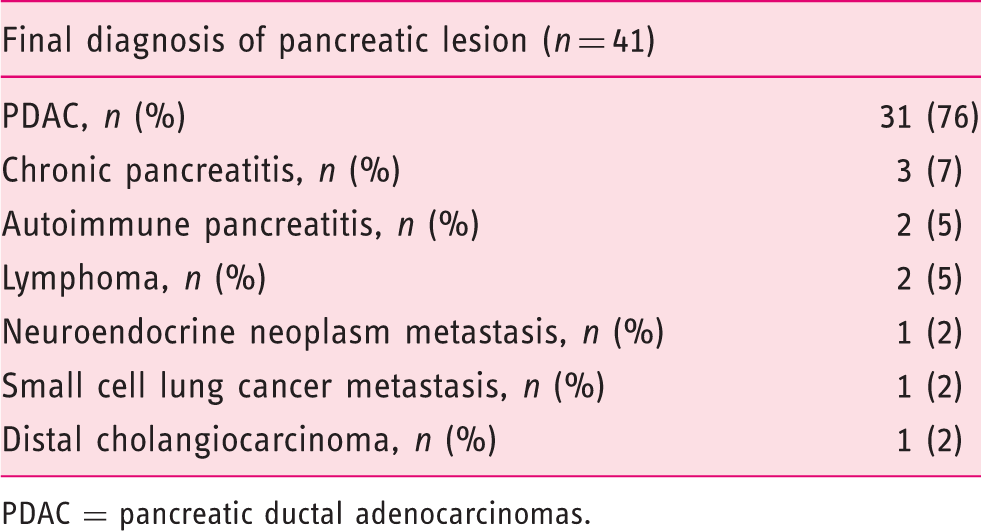
PDAC = pancreatic ductal adenocarcinomas.
Diagnostic yield
Diagnosis made by EUS-FNB corresponding with final diagnosis subdivided by puncture route.
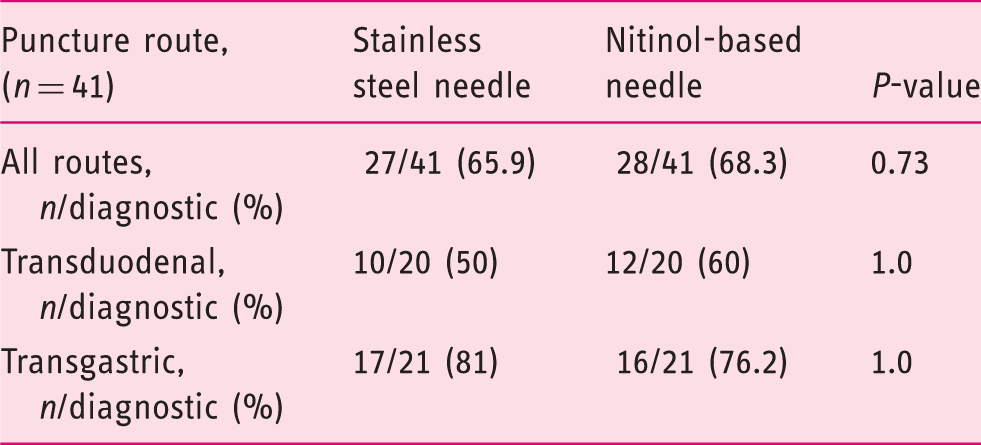
The overall diagnostic accuracy using both needles and thus using two needle passages per pancreatic mass was 87.8 % (Supplementary Table 3).
In seven cases, the pancreatic mass was surgically resected. In those seven cases, overall diagnostic accuracy using both needles was 71.4%. The final diagnosis was matched in three cases (42.9%) using the stainless steel and in four cases (57.1%) using the nitinol-based needle.
In order to evaluate the influence of lesion size we analyzed the 23 pancreatic masses smaller than 30 mm maximum diameter. Twelve (52.2%) were diagnosed correctly by using the stainless steel and 16 (69.6%) by using the nitinol-based needle. The difference was statistically not different.
We also evaluated whether the final PDAC diagnosis of 31 included patients had an influence on diagnostic accuracy. The overall diagnostic accuracy using both needles was 90.3%. The stainless steel needle was able to match the final diagnosis in 21 (67.7%) cases, the nitinol-based needle in 22 (71%) cases.
Other secondary endpoints
Time measured from insertion to extraction of the needle subdivided by puncture route.
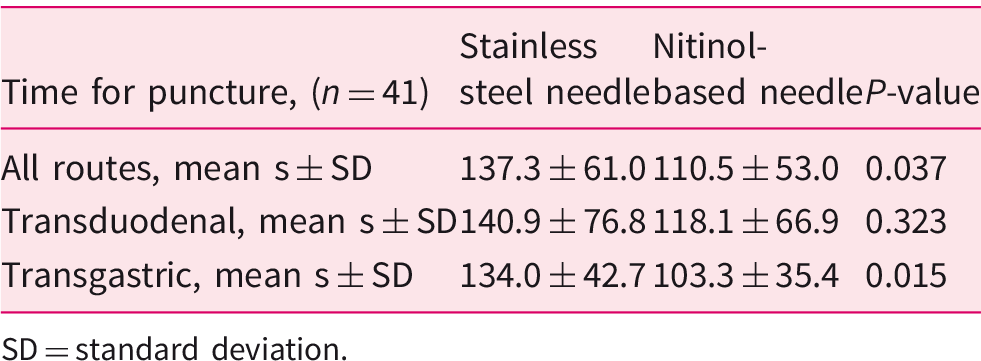
SD = standard deviation.
Technical failure represented by inability to advance needle of the endoscope.
Surface area of total tissue and tumour tissue. The number of measurable total tissue surfaces is related to the 41 analyzed patients. The number of measurable tumour tissue surfaces is related to the 36 patients with a malignant pancreatic mass.

Discussion
EUS-FNB is the standard procedure for obtaining histology, rather than cytology, from a suspected solid pancreatic lesion. Histologic analysis is increasingly becoming the diagnostic standard due to the emerging field of neoadjuvant treatment strategies for patients suspected of having a PDAC.13,14 Furthermore, larger tumour samples might be beneficial as the tissue can be used in routine clinical procedures and for additional molecular testing of newly recognized subtypes of the PDAC.15–17 Due to the angulation in cases of transduodenal biopsies, obtaining a histological sample using the 19-G needle is often difficult or even impossible. 7 The development of a nitinol-based needle promises to tackle such problems owing to the flexibility of the needle tip. A case series by Chavalitdhamrong et al. found that technical success using a nitinol-based needle was achievable in all 10 patients. 11 In this study the authors aimed to obtain cytological specimens; final diagnosis was possible in 71% of cases. Another single-arm study of 38 patients, analyzing a nitinol-based 19-G needle, was published by Varadarajulu et al. 9 Sufficient histological specimens were obtained from 36 (94.7%) patients using the new needle with a mean of 1.45 ± 0.79 needle passes. In addition, this group used ROSE to improve the diagnostic yield of the procedure. More recent studies have also used the single-arm design, which biopsied a variety of lesions adjacent to the gastrointestinal wall8,10,12; technical success rates were reported to be over 90% in all studies, similar to the results of our study.
To compare the diagnostic yield with a regular stainless steel 19-G needle we obtained the histological samples in a random order using both the stainless steel and nitinol-based needles. The pathologist was blinded to the type of needle used. A restriction in the procedure to one pass per needle led to a drop in diagnostic accuracy of between 50% and 60% for the transduodenal route and between 76% and 81% for the transgastric route. Cumulative diagnostic yield of the EUS-FNB procedure using both needle passes was 88%, which was similar to previously published studies that did not incorporate ROSE. 18 Hence, one might conclude that number of passes appears to be more relevant to diagnostic accuracy than the type of needle used.
The time from insertion to extraction of the needle was measured per puncture. Only one puncture per needle was evaluated. Unfortunately, no additional details about the contributing factors to time span were recorded.
A strength of this study was the measurement of the exact amount of tissue obtained in relation to the diagnostic area within the sample, in square millimetres, by a pathologist blinded to the needle type. To our knowledge, this has not been done before in similar studies. In one study, the length and width of the specimens (at their widest point) were measured 9 ; however, measurement of both the total surface area and the tumour’s proportion in square millimetres is presumably more accurate. As expected, the median amount of material obtained by the two needles did not differ.
Weaknesses of the present study include the relatively low number of patients participating per month in each centre and the inability to blind the examiner to the needle type used. The fact that some patients did not agree to participate in the study might have contributed to the low number of patients. Unfortunately, this was not prospectively evaluated in our study. We further used a relatively large diagnostic accuracy difference for the sample size calculation based on clinical assumption. The diagnostic yield of EUS-FNB using a 19-G needle is very limited for lesions located in the pancreatic head because of the angulation of the endoscope. To the best of our knowledge, no study has reported on the accuracy of the specific needle types used here with one puncture, thus the assumed difference in diagonostic accuracy was made based on clinical experience. Previous publications on the sensitivity of stainless steel needles report sensitivity rates as low as 60%.4,19 Single-arm studies using nitinol-based needles report rates of approximatively 85%.8,12 We assumed that puncturing with a nitinol-based needle would lead to a much better diagnostic yield in transduodenal punctures due to its greater flexibility and the fact that most pancreatic lesions are located in the pancreatic head. Furthermore, we elected only one puncture per needle to avoid harming the patient with a higher number of punctures.
In order to represent a real-world scenario, we included pancreatic head, corpus and tail lesions. Thus, 50% of our biopsies were taken via the transgastric route, where stainless steel needles perform well because of the non-angulated position of the endoscope. Nonetheless, the lack of a significant difference in the diagnosis of a solid pancreatic lesion using one or another needle somewhat diminishes the usefulness of the new nitinol-based needle.
The question whether for transduodenal punctures the newer nitinol needles are preferable, requires further comparative studies including a greater number of patients. We did not observe a superior performance from nitinol-based needles over stainless steel as we might have expected. Nevertheless, based on our study, the number of passes (two instead of only one) rather than the type of needle appears to be most clinically relevant to achieving sufficient diagnostic accuracy for further assessment of pancreatic masses.
Supplemental Material
UEG887580 Supplemetal Material - Supplemental material for Multicenter, randomized comparison of the diagnostic accuracy of 19-gauge stainless steel and nitinol-based needles for endoscopic ultrasound-guided fine-needle biopsy of solid pancreatic masses
Supplemental material, UEG887580 Supplemetal Material for Multicenter, randomized comparison of the diagnostic accuracy of 19-gauge stainless steel and nitinol-based needles for endoscopic ultrasound-guided fine-needle biopsy of solid pancreatic masses by Alexander Hann, Sonja Epp, Lothar Veits, Ulrich Rosien, Julian Siegel, Oliver Möschler, Wolfram Bohle and Alexander Meining in United European Gastroenterology Journal
Footnotes
Acknowledgement
The EZ Shot 3 Plus 19-G needles were kindly provided by Olympus for this study; however Olympus did not play a role in the planning, analysis or interpretation of this study.
Author contributions
Conception and design: AH and AM; provision of study materials: AM; performed the intervention and follow-up: UR, JS, OM, WB and AM; analyzed histology: LV; analysis and interpretation of the data: AH, SE, AM; drafting of the article: AH and AM; critical revision of the article for important intellectual content: all authors. All authors approved the final version of the article.
Ethics approval
The study, the protocol and patient informed consent forms were approved by the ethics committee of each participating centre before recruitment.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from each patient prior to inclusion in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
