Abstract
Intraductal papillary mucinous neoplasms are common lesions with the potential of harbouring/developing a pancreatic cancer. An accurate evaluation of intraductal papillary mucinous neoplasms with high-resolution imaging techniques and endoscopic ultrasound is mandatory in order to identify patients worthy either of surgical treatment or surveillance. In this review, the diagnosis and management of patients with intraductal papillary mucinous neoplasms are discussed with a specific focus on current guidelines. Areas of uncertainty are also discussed, as there are controversies related to the optimal indications for surgery, surveillance protocols and surveillance discontinuation.
Brief clinical case
A 68-year-old male with hepatitis C virus (HCV)-related liver cirrhosis underwent trans-abdominal ultrasound for screening of hepatocellular carcinoma. A 25 mm cystic lesion of the head of the pancreas was detected. The patient was asymptomatic, with neither history of diabetes nor of pancreatitis. Subsequent magnetic resonance imaging showed a 24 mm cystic lesion of the pancreatic head communicating with a non-dilated main pancreatic duct, with no solid components. A diagnosis of branch-duct intraductal papillary mucinous neoplasm (IPMN) without worrisome features was made.
How to diagnose this disease
The diagnosis of cystic neoplasms of the pancreas has markedly increased in recent years. The pooled prevalence of pancreatic cystic neoplasms was 8% in systematic review of 17 studies with 48,860 patients. 1 Most of these are IPMNs, although it is difficult to determine the exact nature of small cysts (IPMNs versus other cystic tumours versus non-neoplastic cysts). The prevalence of pancreatic cysts consistent with IPMNs in an adult population was 6.6%. 2 Of note, a recent study found a higher prevalence of pancreatic cysts, mainly IPMNs, in patients with liver cirrhosis (14% compared with 7% in matched controls). The mechanisms for this association are unknown, but a role for pro-inflammatory cytokines and for obesity has been hypothesised. 3 IPMNs occur more frequently during the sixth or seventh decades of life, (median age 65 years, range: 40–75) with a slightly more prevalent for male sex.4–6 The majority of patients with IPMN are asymptomatic, being IPMNs incidentally detected after abdominal imaging performed for unrelated reasons. Less frequently, patients can present with symptoms, including jaundice, abdominal pain, acute pancreatitis, new onset or worsening diabetes, weight loss and steatorrhoea.4–6
The diagnosis of IPMNs is based on high-resolution imaging techniques and on endoscopy. The diagnostic work-up has different objectives. Firstly, IPMNs should be differentiated from other pancreatic cystic lesions. Secondly, it is important to determine the type of IPMN. Thirdly, malignancy-related features should be identified. Since IPMNs frequently occur in elderly people, if a patient is considered unfit for surgery because of advanced age/comorbidities, diagnostic work-up should be interrupted. 6-8
In surgically fit patients, all pancreatic cysts greater than 10 mm should undergo either computed tomography (CT) or gadolinium-enhanced magnetic resonance imaging (MRI) with magnetic resonance cholangiopancreatography (MRCP).6–8 MRI/MRCP should be considered as the preferred diagnostic imaging technique. Based on its superior contrast resolution, MRI/MRCP is more sensitive than CT for identifying (a) communication between a cyst and the main pancreatic duct (MPD), (b) multiple cysts, (c) nodules and thickened walls and (d) size of MPD.6–10 Recent studies showed that diffusion-weighted MRI (DWI) can improve diagnostic accuracy with increased specificity compared to conventional MRI for differentiating malignant and benign IPMNs. Diffusion restriction showed at DWI has been proposed as a radiologic marker of malignancy and invasiveness in IPMNs.11,12
IPMNs are classified into main duct (MD) IPMN, branch duct (BD) IPMN and mixed IPMN. MD-IPMN is characterised by segmental or diffuse dilation of MPD ≥5 mm without other causes of obstruction (i.e. chronic pancreatitis). The threshold of 5 mm for MPD dilation was associated with increased sensitivity for radiologic diagnosis of MD-IPMN without losing specificity. 4 BD-IPMNs are defined as pancreatic cyst(s) communicating with a non-dilated (<5 mm) MPD. In the mixed type, cysts communicate with a dilated MPD. The 5 mm cut-off for MPD should not be considered as a ‘dogma’. The diagnostic accuracy for MD/mixed-IPMNs is between 74–80%.13,14 In IPMNs MPD can be dilated for reasons other than direct tumour involvement, including ductal hypertension caused by mucin hypersecretion/plugs, being MPD ≥5 mm observed also in patients with BD-IPMNs.
Endoscopic ultrasound (EUS) should be considered as a ‘second-level’ diagnostic modality after CT and MRI. 6 EUS is an invasive, operator-dependent imaging technique with the possibility of performing fine-needle aspiration (FNA) for cyst fluid analysis.6–8 EUS is useful in differentiating IPMNs from other pancreatic cysts, including macrocystic serous cystadenomas (SCAs), mucinous cystic neoplasms (MCNs) or pseudocysts. By analysing CEA level, amylase/lipase, glucose as well as cytology in the cyst fluid, it may be possible to increase the diagnostic yield in their differential diagnosis. A cut-off ≥192 ng/ml of cyst fluid carcinoembryonic antigen (CEA) has been suggested for distinguishing mucinous from non-mucinous cysts, with a sensitivity of 38–78% and specificity of 63–99%,7,15 but it is not helpful in distinguishing between benign and malignant mucinous cysts. Similar results are reported for cytological examination. 16 The identification of DNA alterations in cyst fluid by next generation sequencing (NGS) has recently be proposed to improve the differential diagnosis of pancreatic cysts. Particularly, mutations of KRAS and GNAS can differentiate mucinous from non-mucinous cysts.15,17 Needle-based confocal laser endomicroscopy (nCLE) performed during EUS-FNA can give real-time imaging of the internal structure of pancreatic cysts, and this may improve the differential diagnosis between mucinous and non-mucinous cysts, particularly serous cystadenomas.18,19
In patients with initial MPD dilatation, EUS may show thickened walls or minimal mural nodules along the MPD with higher accuracy compared with CT/MRI, leading to a definitive diagnosis of MD/mixed IPMNs. 6 Once an IPMN has been diagnosed, it is of paramount importance to evaluate with imaging and/or EUS the presence of malignancy-related parameters, that will be discussed later in this review.
Natural history, including outcomes
IPMNs are clinically relevant entities not only for their frequency but because they do represent possible precursors of pancreatic cancer. IPMNs may exhibit different degrees of dysplasia including low-grade dysplasia, high-grade dysplasia and invasive carcinoma. 20 IPMN follow a classic ‘adenoma-carcinoma sequence’ and time of progression from low-grade dysplasia to invasive cancer is about 4–6 years.4–6 Specific genetic alterations have been identified in IPMNs. Somatic mutations in the oncogenes KRAS and GNAS are found in up to 90% IPMNs and are recognised as early events for IPMN development.21,22 Other mutated genes include TP53, CDKN2A/p16, SMAD4 and less commonly PIK3CA, BRAF, PTEN and STK11. Inactivation of CDKN2A/p16, loss of SMAD4 and mutation in TP53 are important for IPMN progression to cancer, and are almost exclusively detected in malignant IPMNs.15,23
Although the natural history of IPMNs is still unknown, the risk of developing cancer is significantly different when comparing MD/mixed- versus BD-IPMNs. In resected patients, the mean rate of invasive cancer is 43% for MD/mixed-IPMNs compared with 16.5% in the BD type.6,7 Of note, because these data are based on surgical series, the malignant potential of BD-IPMNs is overestimated, since most BD-IPMNs are managed non-operatively. 6 As a consequence the rate of malignancy considering all BD-IPMNs patients is likely less than 5%.
The prognosis of resected IPMNs with low-grade or high-grade dysplasia is excellent, as their five-year disease-specific survival (DSS) approaches 100%.4–8 Methacronous IPMNs can develop after resection in the pancreatic remnant in 5–8% of cases of patients with non-invasive tumours, and for this reason surveillance following resection is required. 6 The prognosis of invasive IPMNs is similar to the prognosis of pancreatic adenocarcinoma, although some series have reported better survival for invasive IPMNs, and this is likely due to the inclusion of malignant IPMNs treated at an earlier stage.2,18,24
Patients with IPMNs are at an increased risk for IPMN-independent pancreatic ductal adenocarcinoma (PDAC), at a rate between 2–11%.6,7,25 This event can be related to a ‘field defect’ that potentially predisposes the development of multiple synchronous or methacronous IPMNs, but also PDAC. The risk of IPMN-distinct pancreatic cancer must be considered in the follow-up of both resected patients and of those undergoing surveillance.
Interestingly, even ‘low-risk’ IPMNs show a low but persistent risk of malignancy over time. In a study with 577 BD-IPMNs undergoing surveillance, the rate of malignancy was 4.3% in the first five years from diagnosis, 5.5% in patients followed for more than five years, and a risk of malignancy of 4% was still present after 10 years. 26
Current management including therapy with supporting evidence
Possible indications for surgery, diagnostic techniques and management according to the International, European and American Gastroenterological Association (AGA) guidelines.
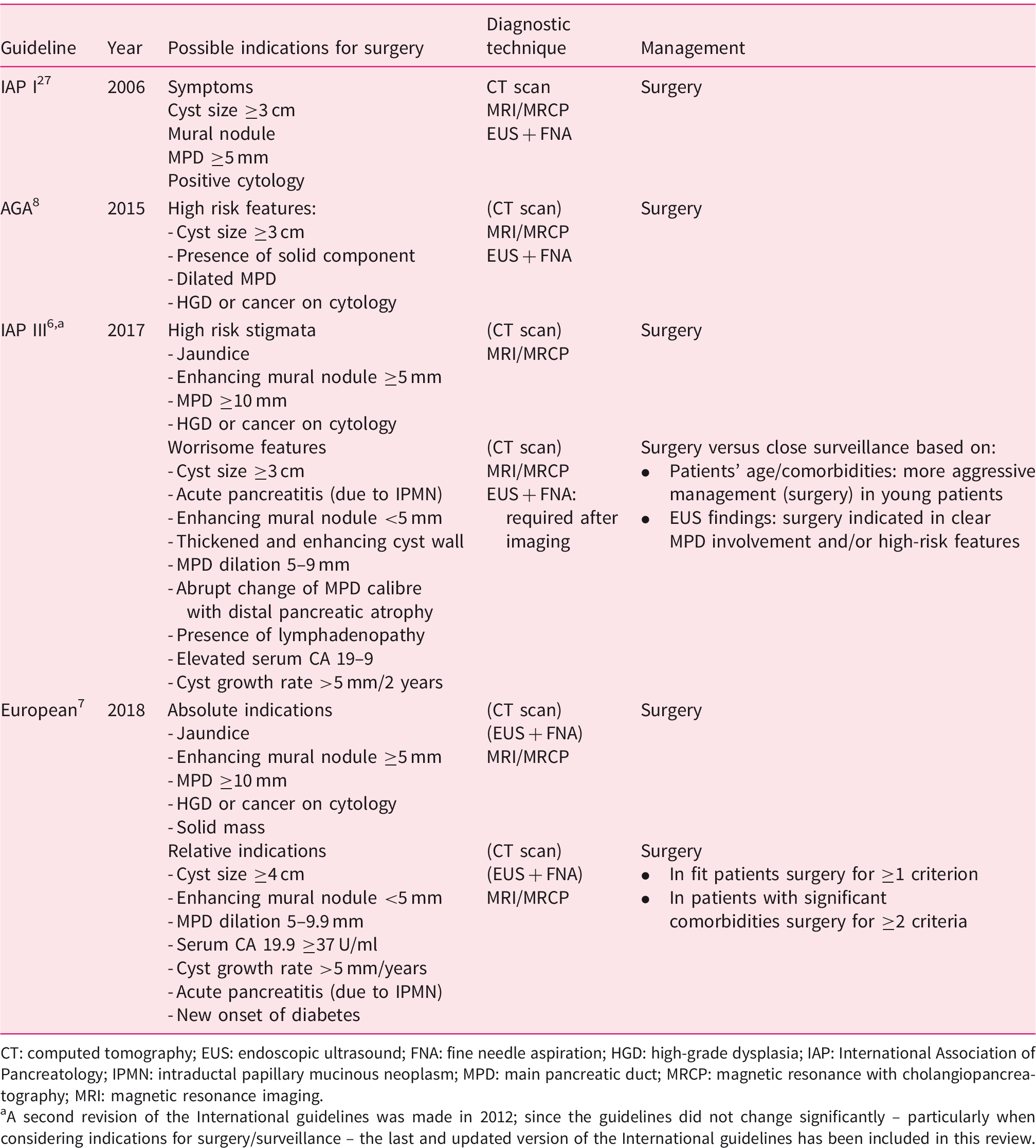
CT: computed tomography; EUS: endoscopic ultrasound; FNA: fine needle aspiration; HGD: high-grade dysplasia; IAP: International Association of Pancreatology; IPMN: intraductal papillary mucinous neoplasm; MPD: main pancreatic duct; MRCP: magnetic resonance with cholangiopancreatography; MRI: magnetic resonance imaging.
A second revision of the International guidelines was made in 2012; since the guidelines did not change significantly – particularly when considering indications for surgery/surveillance – the last and updated version of the International guidelines has been included in this review.
While using a different terminology (‘high-risk stigmata’ in IGs or ‘absolute indication for surgery’ in EGs), all guidelines identify some risk factors for malignancy that require immediate surgery, including jaundice, enhancing mural nodule >5 mm, solid mass in the pancreas, MPD ≥10 mm. Both IGs and EGs identify a second category of risk for malignancy called ‘worrisome features’ (WFs) or ‘relative indications’ (RIs) for surgery, respectively. Criteria included in WFs and RIs are similar, but their management is different according to the guidelines. IGs suggest that WFs should be evaluated by EUS + FNA, and surgery or close surveillance can be both considered. 6 Pancreatic resection is indicated when patients have a long-life expectancy or when EUS show a clear involvement of the MPD or high-risk features. On the other hand, EGs support a more aggressive policy. Surgical resection is recommended when a single RI is present in surgically fit patients, or when two or more RIs are identified in patients with significant comorbidities, without performing EUS. 7 However, cytological analysis may improve the diagnosis of malignancy in patients with WFs/RIs, at least in referral centres with experience in EUS.6,28 DNA-based testing of cyst fluid obtained during EUS showed promising results for the identification of malignancy. Singhi et al. found that the combination of KRAS/GNAS mutations and alterations in TP53/PIK3CA/PTEN had an 89% sensitivity and 100% specificity for the identification of malignant IPMN. 23 These authors also showed that MPD dilatation, mural nodules and malignant cytology had lower sensitivities (42%, 32% and 32%) and specificities (74%, 94% and 98%) for malignancy compared with molecular testing. 23
Among different WFs/RIs, the indication for surgery is particularly controversial for MPDs between 5–9 mm. In fact different studies have reported a risk of harboring malignancy of 37–90% for MPDs of size 5–9 mm.29,30 It should be noted that these studies include only surgically resected patients with pathologic diagnosis of MD/mixed-IPMNs, but they do not consider patients with an MPD of 5–9 mm under surveillance, as previously pointed out. The significance of cyst size is also debated. A meta-analysis identified cyst size >30 mm as a risk factor for malignancy, 31 but several studies showed that the exclusive presence of this parameter in the absence of high-risk features is not strictly indicative of a malignancy.6,32 Other possible indications include cyst growth rate over time and increased level of serum CA 19.9.6,7
Patients with WFs undergoing surveillance do represent an interesting opportunity to evaluate the actual risk of malignancy of WFs, although there is a lack of a certain histological diagnosis in this setting. In a cohort of 281 IPMN patients with WFs (n = 231, 82%) and HRS (n = 50, 18%) who did not undergo surgery because of advanced age/comorbidities, 33 five-year DSS was 96% for patients with WFs compared with 60% of those with high-risk stigmata (HRS). Interestingly, 83 patients had a MPD of 5–9 mm, and their five-year DSS was 94%. If a MPD of 5–9 mm was a strong risk factor for malignancy, a worse survival rate in this population would have been expected. In the same study no correlation among cyst size >30 mm or cyst size >50 mm and survival was found. 33 Taken together, these data suggest that patients with high-risk features should undergo immediate surgery, while WFs/RIs should be examined with EUS + FNA – especially if molecular tests on cyst fluids are available – and non-operative management can be considered at least in individuals with limited life expectancy.
In the presence of HRS or malignant cytology, pancreaticoduodenectomy or distal pancreatectomy are required. The intraoperative examination of the transection margin is mandatory, and resection should be extended up to total pancreatectomy if high-grade dysplasia or invasive cancer are present at the margin.6–8
Patients with IPMNs lacking HRS/absolute indications (AI) and WFs/RIs should undergo non-operative management.6–8 By definition, surveillance applies mainly to BD-IPMNs. A systematic review and meta-analysis of 20 studies including 2177 patients with BD-IPMNs undergoing non-operative management showed reassuring results. 34 The overall rate of malignancy was 3.7% including IPMNs with invasive cancer (2.4%) and IPMN-distinct PDAC (1.3%). This 3.7% rate of malignancy is comparable to the risk of postoperative mortality following pancreatectomy if one had operated on all of these patients ‘to prevent’ a possible cancer. It should be stressed that surveillance of ‘low-risk’ IPMNs should include not only the evaluation of cystic lesions, but also of the remaining parenchyma because of the possible risk of IPMN-independent PDAC.6,7,25
Different surveillance strategies according to International, European and American Gastroenterological Association (AGA) guidelines.
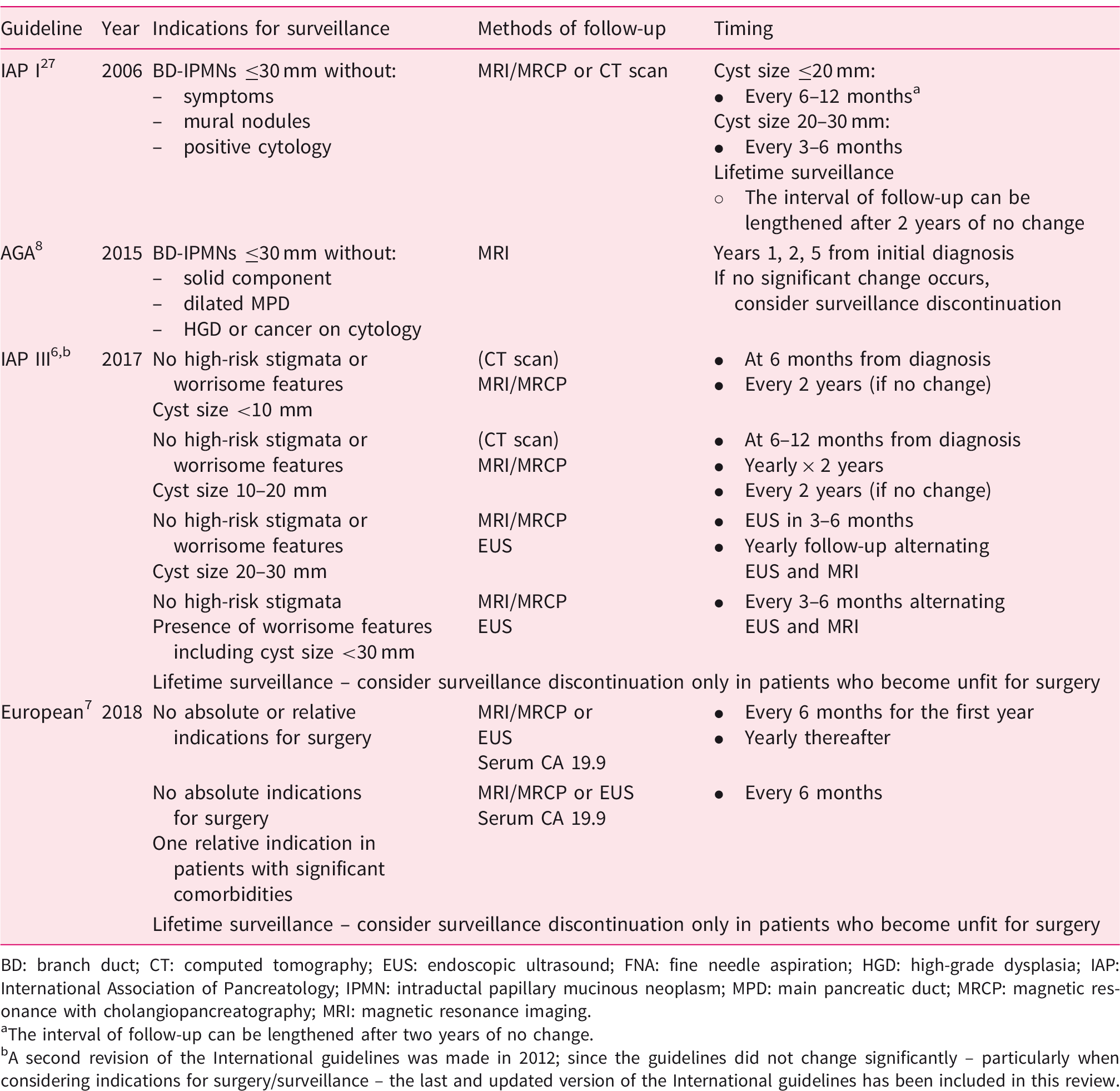
BD: branch duct; CT: computed tomography; EUS: endoscopic ultrasound; FNA: fine needle aspiration; HGD: high-grade dysplasia; IAP: International Association of Pancreatology; IPMN: intraductal papillary mucinous neoplasm; MPD: main pancreatic duct; MRCP: magnetic resonance with cholangiopancreatography; MRI: magnetic resonance imaging.
The interval of follow-up can be lengthened after two years of no change.
A second revision of the International guidelines was made in 2012; since the guidelines did not change significantly – particularly when considering indications for surgery/surveillance – the last and updated version of the International guidelines has been included in this review.
Final outcomes, areas of uncertainty, is stopping surveillance feasible?
The high number of patients with IPMNs requiring surveillance poses critical issues related to the significant costs associated with this strategy. AGA guidelines suggested in 2015 the discontinuation of surveillance after five years for patients with pancreatic cysts, including IPMNs, that did not show significant changes. 8 However, in a cohort of 144 low-risk BD-IPMNs undergoing surveillance with a minimum follow-up of five years, we reported a rate of new WFs/HRS of 18%. 35 Similarly, Pergolini et al. found that 22% of BD-IPMNs followed for more than five years developed WFs/HRS. 26 It is evident that discontinuation of surveillance should be applied selectively, for example to all patients who become unfit for surgery. Interestingly, in both of the studies, no significant changes were observed in BD-IPMNs <15 mm in size,26,35 and this parameter associated with patients’ age can be used to select a subgroup of patients that are unlikely to benefit for surveillance >5 years. In this setting, recently Sahora et al. 36 showed that the Charlson comorbidity index can be useful for identifying patients with IPMNs who have a high mortality risk for causes unrelated to the pancreas.
Further studies are needed to stratify patients with low risk in order to better define surveillance strategies (type and timing) as well as surveillance discontinuation in selected groups.
Other areas of uncertainty still remain. A critical point is represented by the proper identification of surgical candidates, especially among patients with WFs/RIs. It is likely that the definitive answer to this question will come by the development of molecular biomarkers, leading to sort of personalised medicine. A test that includes clinical, imaging, and molecular features (CompCyst test) has been developed, but its prospective evaluation is still required. 15 In this light, it will be important to ensure the identification not only of malignant IPMNs at diagnosis, but also of those apparently low-risk lesions at risk of developing as malignant over time. It is definitively time to move to prospective randomised or observational studies trying to address these pending controversial issues!
Footnotes
Declaration of conflicting of interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
