Abstract
Background
Recent studies demonstrated the positive impact of neoadjuvant treatment in locally advanced gastric cancer.
Objective
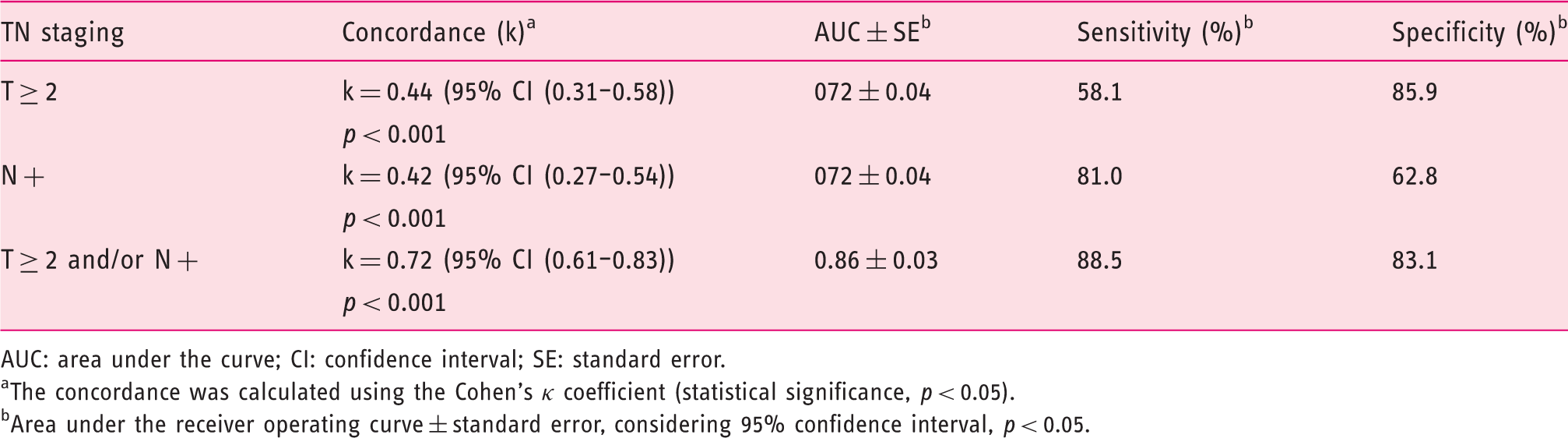
To assess the accuracy of endoscopic ultrasound (EUS) in the selection of patients with gastric adenocarcinoma for neoadjuvant therapy (T ≥ 2 and/or N+).
Methods
Retrospective analysis of patients with an anatomopathological diagnosis of gastric adenocarcinoma between January 2011 and June 2017, who had EUS for staging and underwent surgery as a first therapeutic attempt. The concordance (k) and accuracy (area under the curve (AUC)) of EUS for T ≥ 2 and/or N+ were assessed using the anatomopathological staging of the resected surgical specimen as the gold standard.
Results
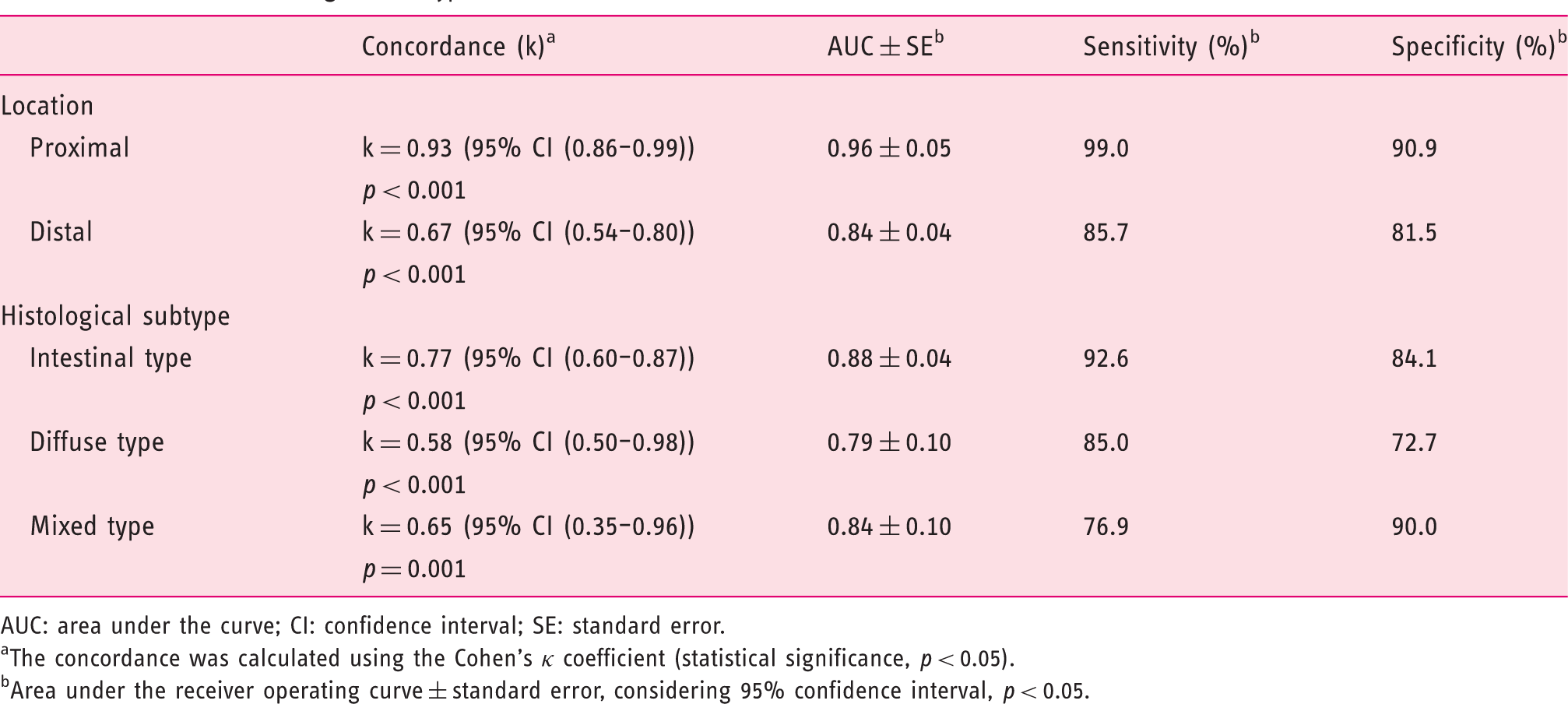
The final sample included 152 patients (66.4% male, 67.1 ± 12.2 years). The concordance, accuracy, sensitivity and specificity of the EUS for T ≥ 2 and/or N+ were 0.72, 0.86 ± 0.03, 88.5% and 83.1%, respectively. The results were higher in proximal (k = 0.93, AUC = 0.96 ± 0.05, sensitivity (S) = 99.0% and specificity (E) = 90.9%) compared with distal lesions (k = 0.67, AUC = 0.84 ± 0.04, S = 85.7% and E = 81.5%), and in intestinal subtype (k = 0.77, AUC = 0.88 ± 0.04, S = 92.6% and E = 84.1%) compared with diffuse (k = 0.58, AUC = 0.79 ± 0.10, S = 85.0% and E = 72.7%) or mixed-subtype tumours (k = 0.65, AUC = 0.84 ± 0.10, S = 76.9% and E = 90.0%).
Conclusion
In one of the largest series of patients, we showed that EUS has overall high agreement and accuracy in the selection of patients with gastric adenocarcinoma for neoadjuvant therapy, although the agreement and accuracy are greater for proximal and intestinal lesions.
Introduction
Gastric cancer continues to be a substantial global health problem, representing the fifth most common source of malignant disease and the third most common cause of cancer death worldwide, causing 723,000 deaths/year, and accounting for 8.8% of all cancer-related deaths. 1
In the last decade, a significant change in the approach to gastric cancer has been seen, and this is largely due to the positive impact of neoadjuvant treatment and preoperative imaging staging. 2 Although gastrectomy remains the only potentially curative therapeutic strategy in locally advanced gastric cancer, current available evidence suggests that neoadjuvant treatment improves overall survival and disease-free survival, reducing local recurrence rates.3–7
The therapeutic approach algorithm for gastric cancer currently contemplates neoadjuvant treatment according to established criteria of preoperative imaging staging (disease that extends to or beyond the muscularis propria (T ≥ 2) and/or the presence of malign lymph nodes (N+): stages IB–IIIC).8,9
Therefore, neoadjuvant therapy highlights the importance of locoregional staging of the gastric cancer, namely the distinction between T1 and T2–T4 stages, and N− and N+ stages. 10 Currently, endoscopic ultrasound (EUS) is considered to be the gold standard for locoregional gastric cancer staging, but few studies have focused on the distinction between early disease (T1N0) and locally advanced disease (T ≥ 2 and/or N+), which is more clinically useful than evaluating T and N stages globally or individually.11,12 Many studies have been conducted with evaluating the accuracy of EUS in gastric cancer staging and some of them have reported that EUS is useful in determining the invasion depth (T stage) with an accuracy higher than 90%. EUS performance for N stage evaluation seems to be less effective than T stage evaluation, although previous studies have shown excellent accuracy of nodal stage evaluation by EUS.13–27 These data suggest that EUS is an excellent adjunct to standard staging modalities in gastric cancer.
The main aim of the present study was to evaluate the performance of EUS in preoperative gastric cancer staging (T and N) and the selection of patients for preoperative treatment. Specifically, our aims were: to assess the concordance (k) between clinical staging by EUS and anatomopathological staging (resected specimen); to evaluate the accuracy (area under the curve (AUC)), sensitivity (S) and specificity (E) of the EUS in the selection of patients with gastric adenocarcinoma for neoadjuvant therapy (T ≥ 2 and/or N+); to analyse the differences according to tumour location (proximal versus distal) and histological subtype (Laurén Classification: intestinal, diffuse and mixed subtype); and to identify the overstaged and understaged groups, and compare them with the correctly staged group.
Materials and methods
Study design and population
We retrospectively analysed patients with anatomopathological diagnosis of gastric adenocarcinoma between January 2011 and June 2017 at Hospital de Braga (Portugal), who had EUS for staging and underwent surgery (without prior neoadjuvant treatment) as a first therapeutic attempt. The need for emergent treatment (bleeding or obstruction), and the presence of contraindication for chemo- and radiotherapy, were the main reasons for surgery as a first option in patients with stages T 3 and 4 and N+ gastric cancer. We evaluated the concordance and accuracy of the EUS for locoregional staging and selection for preoperative chemotherapy (T ≥ 2 and/or N+) globally, and according to location and histological subtype tumour, considering anatomopathological staging as the gold standard.
Patients were excluded according to the following criteria: ambiguous and incomplete anatomopathological staging, exploratory laparotomy as a first staging intervention, patients submitted to neoadjuvant or palliative therapy (chemo- and/or radiotherapy) as the first therapeutic option, incomplete or inconclusive preoperative staging by EUS, and patients who underwent EUS for re-staging or follow-up of gastric adenocarcinoma (Figure 1).
Flow diagram of the study. Patients with gastric cancer who had not had prior endoscopic ultrasound were patients with metastatic cancer and/or patients with contraindication for neoadjuvant therapy.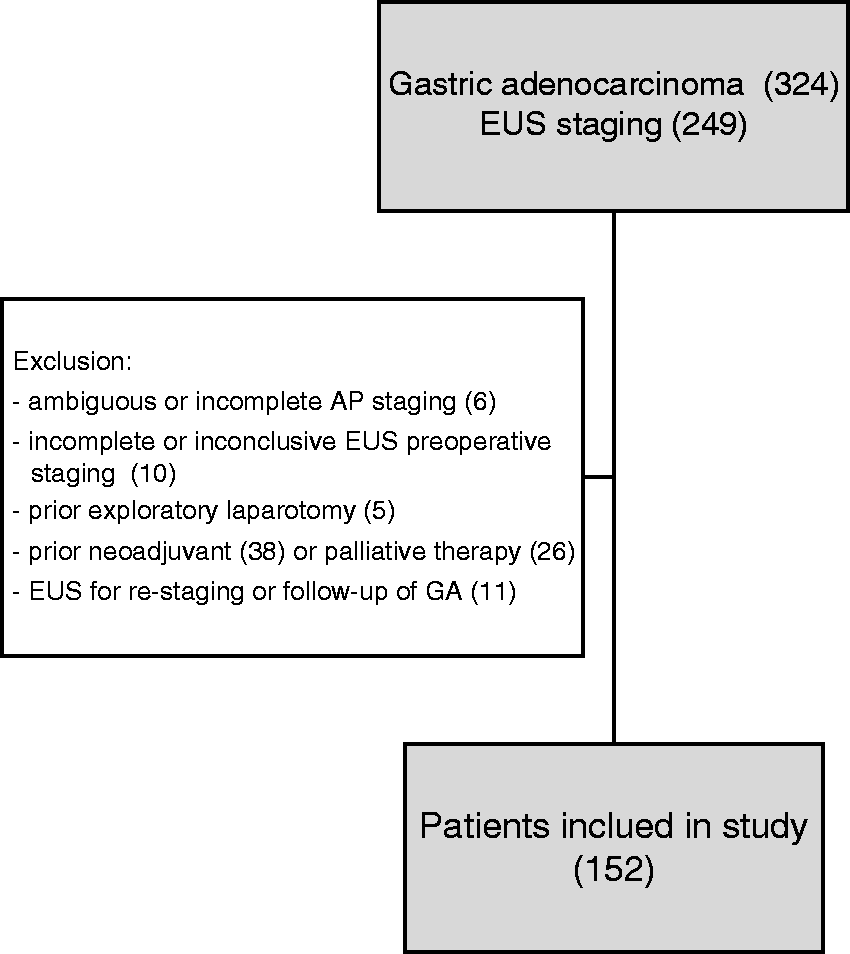
EUS staging
EUS was performed using a 7.5 MHz radial echo endoscope (EG3830UTK; Olympus®) connected to an endoscopic ultrasonic observation unit (Aloka Prosound α-5) under deep sedation, with propofol administration by anaesthetist. All examinations had been carried out by three experienced ultrasonographers (who perform more than 300 procedures per year).
The stage was determined according to American Joint Committee on Cancer (AJCC) 7th edition TNM (T: tumor, N: lymph nodes and M: mestastasis) staging system. Tumours arising in the antrum, incisura angularis and body were defined as distal tumours, and tumours arising in cardia (without involvement of the oesophagogastric junction) or fundus were defined as proximal tumours. Malignant lymph node was diagnosed if at least two or more of the following EUS criteria were found present: a hypoechoic pattern, a lymph node larger than 5 mm, and a lymph node with a round shape and a smooth border.
Taking into consideration the limitations of EUS in the detection of distant metastases (M stage), this TMN category was not evaluated in our study and was not considered for preoperative staging.
Anatomopathological tumour staging and grading were completed according to the AJCC 7th edition TNM staging system.
The median time between EUS and surgery was 16 ± 9.9 days.
Data collection
Data (demographic, anatomopathological and ultrasonographic findings) were collected by consulting electronic health records, while ensuring anonymity and confidentiality. Interventions were recorded, including the procedures performed for staging, neoadjuvant therapy and surgery.
Data analysis
Continuous variables were presented as mean and SD. Descriptive data was presented as absolute and relative frequencies for qualitative variables.
The concordance (k), accuracy (AUC), sensitivity (S) and specificity (E) for preoperative staging and selection for neoadjuvant therapy by EUS were calculated using standard definitions.
Cohen’s kappa coefficient (κ) was used to evaluate the consistency between the EUS and the anatomopathological staging of gastric cancers. Regarding the κ statistic, we assessed the agreement as follows: slight: k = 0.00 to 0.19; fair: k = 0.20–0.39; moderate: k = 0.40–0.59; substantial: k = 0.60–0.79; and almost perfect: k ≥ 0.80.
Receiver operating characteristic (ROC) curve analysis was performed to evaluate overall concordance between EUS and anatomopathological staging in patients selected for neoadjuvant therapy. Considering ROC curves, an AUC ranging between 0.5 and 1, and a maximum AUC = 1 means excellent accuracy.
We have identified the overstaged and understaged groups, which were compared with the correctly staged group.
The statistical analysis was carried out using IBM SPSS Statistics for Windows, Version 22.0 (Armonk, NY: IBM Corp.). The p value was set at 0.05.
Ethics approval
The study was carried out in accordance with the Declaration of Helsinki. Ethical approval was granted by the Ethics Subcommittee for Life and Health Sciences (SECVS) and the Ethics Committee for Health of the Hospital of Braga (CESHB) on 31 October 2017.
Informed consent
Patients provided written informed consent for all the procedures, and only then were they included in the database.
Results
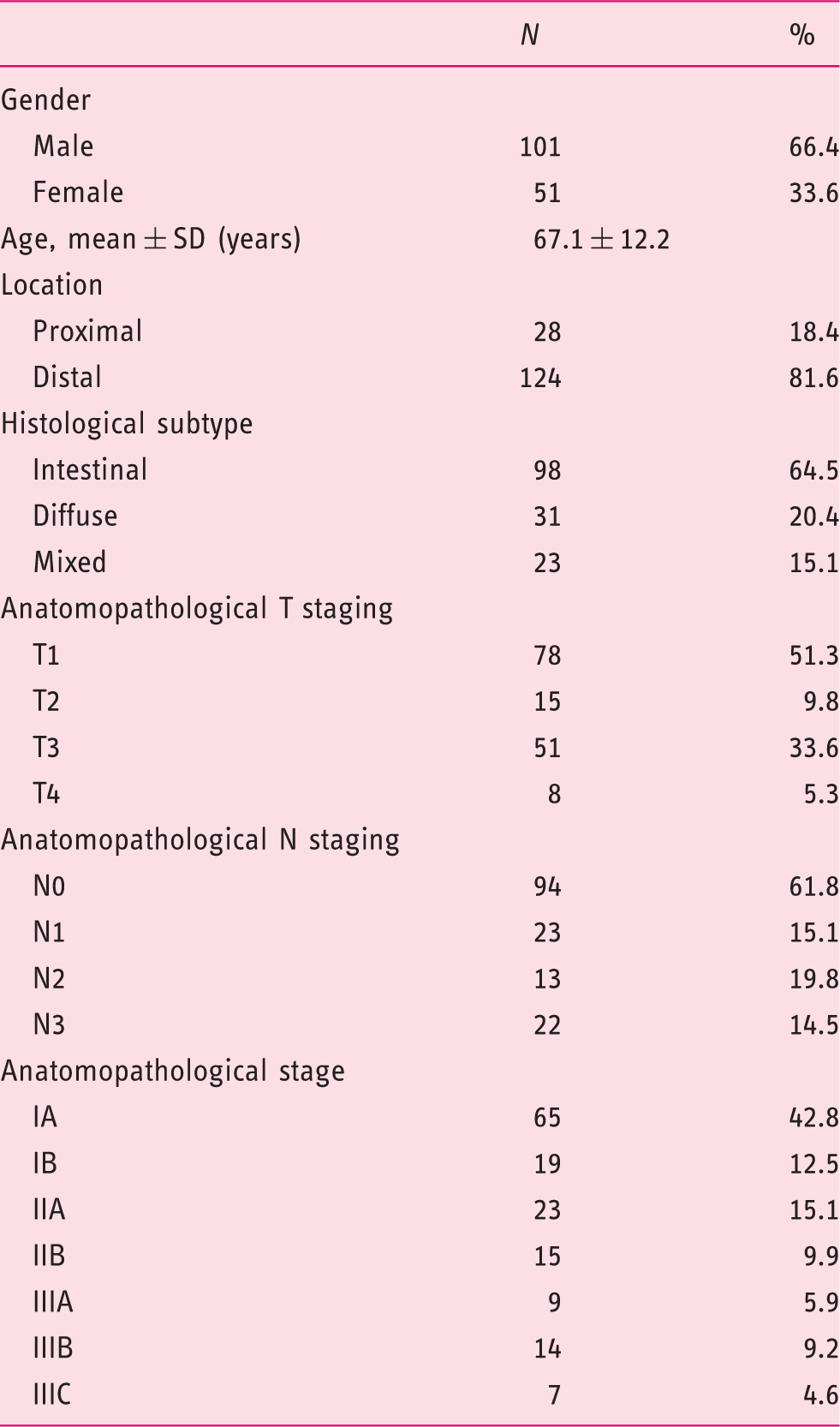
Patient characteristics
Clinical and pathological characteristics of the enrolled patients (N = 152).
Concordance and accuracy of the EUS for preoperative staging
The overall accuracy of preoperative T and N staging by EUS was 75.7 and 72.9%, respectively, when compared to the postoperative histopathological staging.
Concordance and accuracy of the endoscopic ultrasound for preoperative T (T1–4) and N (N0–3) staging versus anatomopathological staging.
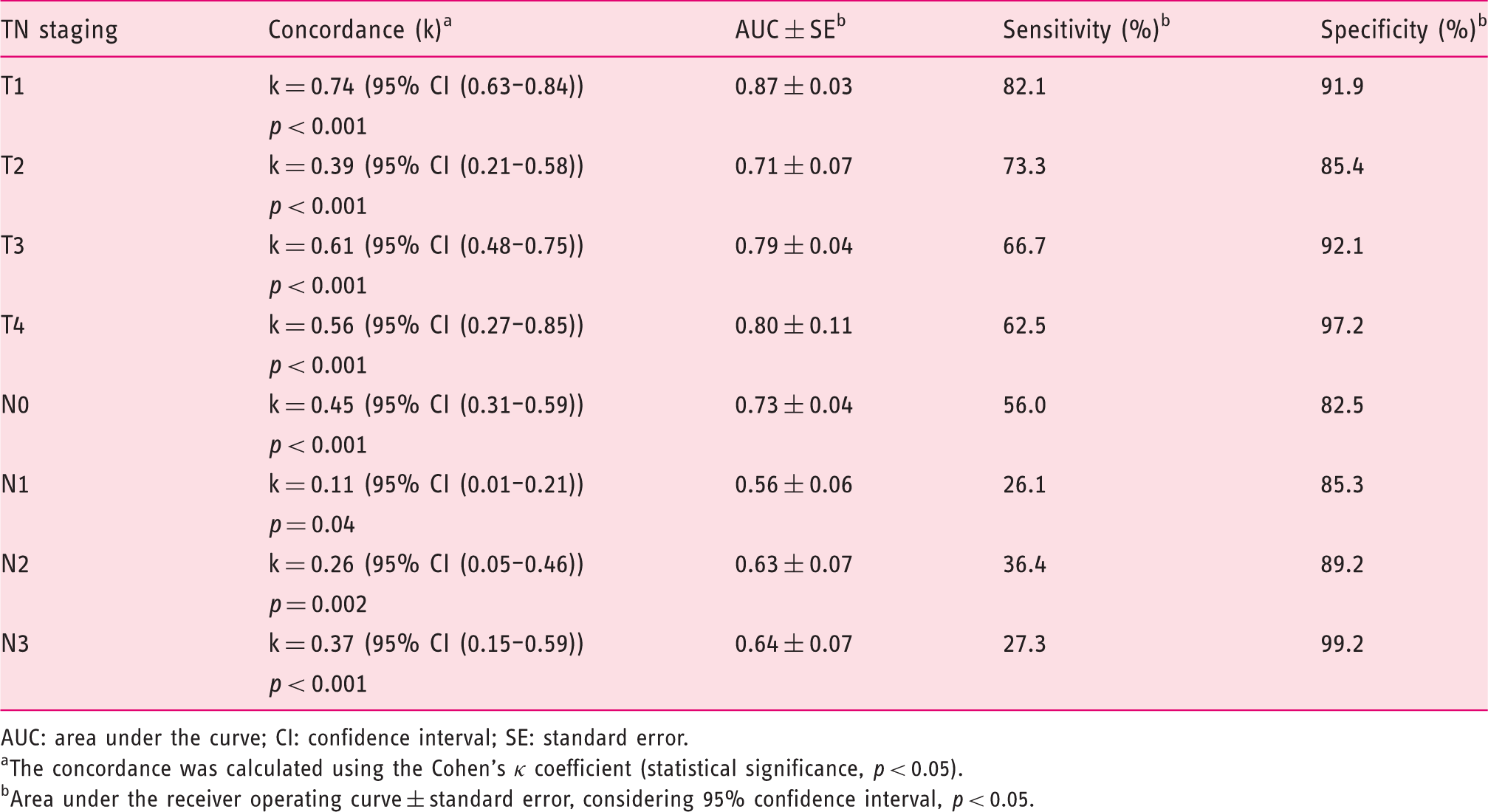
AUC: area under the curve; CI: confidence interval; SE: standard error.
The concordance was calculated using the Cohen’s κ coefficient (statistical significance, p < 0.05).
Area under the receiver operating curve ± standard error, considering 95% confidence interval, p < 0.05.
Concordance and overall accuracy of EUS were higher for T staging (k ranging from 0.39–0.74 and AUC ranging from 0.71–0.87) than for N staging (k ranging from 0.11–0.45 and AUC ranging from 0.56–0.73). Concerning T staging, the concordance and overall accuracy of EUS were higher for the T1 stage than for other stages. Concerning N staging, the concordance and overall accuracy of EUS were higher for the N0 stage than for other stages.
Concordance and accuracy of the EUS for the selection of patients with gastric adenocarcinoma for neoadjuvant therapy
Table 3 presents the results of concordance, accuracy, sensitivity and specificity of the EUS for the selection of candidates for neoadjuvant therapy (T ≥ 2 and/or N+). The ROC curve corresponding to overall accuracy of EUS in the group of patients selected for neoadjuvant treatment in gastric adenocarcinoma is shown in Figure 2. The concordance and overall accuracy of EUS for T ≥ 2 and N+ were very similar. Nonetheless, for T ≥ 2, the sensitivity of EUS was relatively low and the specificity was high. The inverse was true for N+ staging by EUS. The combination of the two criteria for neoadjuvant therapy increased the concordance, accuracy, sensitivity and specificity of the EUS.
Overall accuracy of the endoscopic ultrasound in the selection of patients for neoadjuvant treatment in gastric adenocarcinoma (area under the curve = 0.86 ± 0.03 (95% confidence interval p < 0.05), sensitivity = 0.885 (88.5%), specificity = 0.831 (83.1%)). Overall concordance and accuracy of the endoscopic ultrasound for the selection of patients for neoadjuvant therapy. AUC: area under the curve; CI: confidence interval; SE: standard error. The concordance was calculated using the Cohen’s κ coefficient (statistical significance, p < 0.05). Area under the receiver operating curve ± standard error, considering 95% confidence interval, p < 0.05.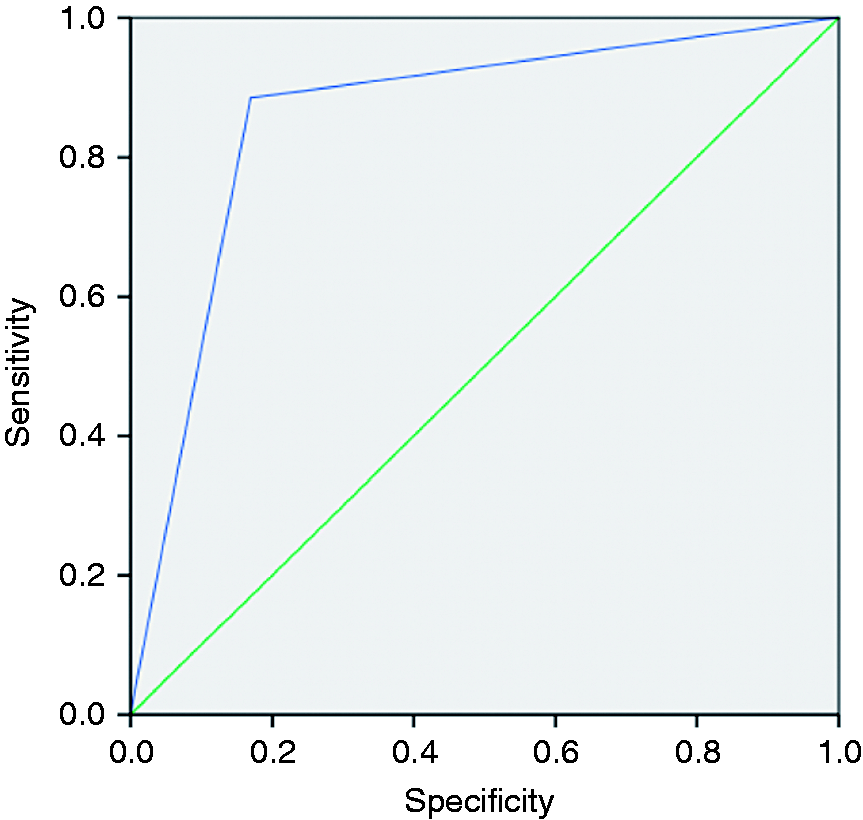
Concordance and accuracy of the EUS for the selection of patients with gastric adenocarcinoma for neoadjuvant therapy according to tumour location and histological subtype
Overall concordance and accuracy of the endoscopic ultrasound for the selection of patients for neoadjuvant therapy according to tumour location and histological subtype.
AUC: area under the curve; CI: confidence interval; SE: standard error.
The concordance was calculated using the Cohen’s κ coefficient (statistical significance, p < 0.05).
Area under the receiver operating curve ± standard error, considering 95% confidence interval, p < 0.05.
The results revealed good performance of the EUS in the group of patients selected for preoperative chemotherapy (k = 0.72, AUC = 0.86 ± 0.03, S = 88.5% and E = 83.1%). The concordance and accuracy of EUS was higher for proximal and intestinal histological subtype tumours when compared with distal tumours and other histological subtypes.
Misdiagnosed cases
After analysing the non-agreement cases for the selection of patients for neoadjuvant therapy by EUS, patients were stratified according to the T or N staging responsible for the under- or overstaging. The results are reported in Figure 3.
Non-agreement cases for neoadjuvant treatment according to preoperative staging by endoscopic ultrasound versus anatomopathological staging in gastric adenocarcinoma.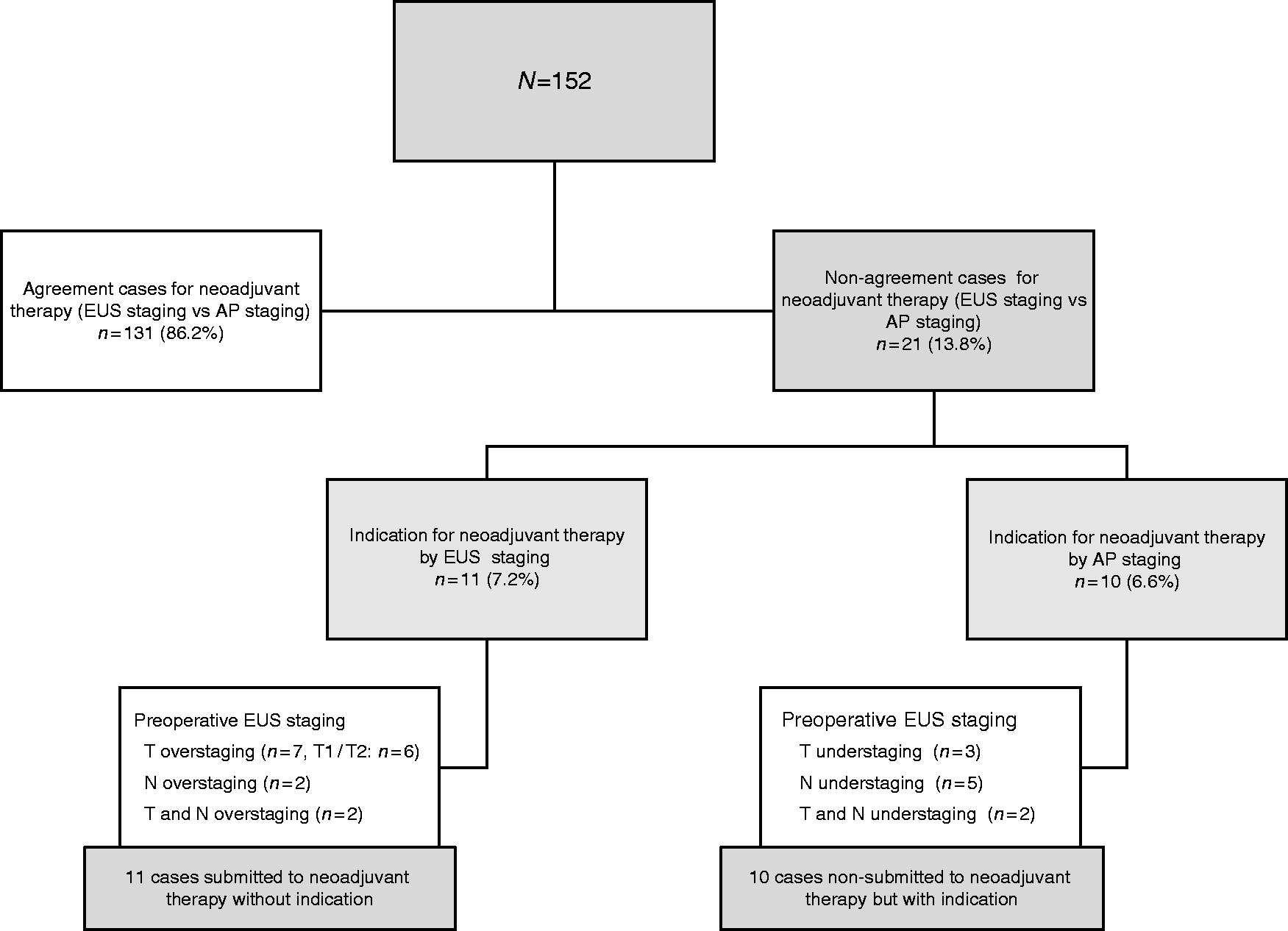
Our data showed that correct decisions (i.e. the selection of patients for neoadjuvant therapy) based on preoperative EUS staging would be made in 131 patients (86.2%). In 21 patients (13.8%), EUS staging was incongruent with anatomopathological staging: overstaging in 11 patients (7.2%) and understaging in 10 patients (6.6%). Overstaging was more frequent in patients with T1 stage (in six cases, the T staging was categorized as T2 by EUS but actually corresponded to T1 stage after resected surgical specimen analysis); the majority of the understaging cases were due to incorrect N staging by EUS (in seven cases the N staging was incorrectly categorized as N− by EUS).
According to EUS findings, 21 patients (13.8%) would be wrongly treated: 10 patients (6.6%) would be undertreated (would not receive the indicated preoperative therapy) and 11 patients (7.2%) would be overtreated (would receive unnecessary preoperative therapy) due to inaccurate EUS staging (T1/T2 or/and N−/N+).
Discussion
Neoadjuvant treatment is currently recommended as a standard treatment in locally advanced gastric cancer (stages IB–IIIC), underscoring the importance of preoperative imaging staging.8–9 EUS has been increasingly used as an image modality in this setting due to its superior ability to assess the depth of invasion (T stage) and lymph node involvement (N stage). 3 Nonetheless there is great heterogeneity in the accuracy rates of T and N stages, with reports ranging between 60–90% for T stage and 50–90% for N stage.3,11–13,15,17,19–20,24–27
In contrast with previous studies focusing on T and N stage evaluation separately, we have evaluated T and N in combination in this study, focusing on the distinction between T1N0 and T ≥ 2 and/or N+ tumours, which is more clinically applicable. This is the main question when we consider neoadjuvant chemotherapy.
Our study showed that EUS correlates strongly with anatomopathological staging (k = 0.72) and has high accuracy for the selection of patients for neoadjuvant therapy. The ability of EUS to accurately discriminate between T1N0 and T ≥ 2 and/or N+ is high with an AUC of 0.86 ± 0.03, and a sensitivity and specificity of 88.5 and 83.1%, respectively. The data are slightly better than those of Fairweather et al., which showed that EUS had moderate accuracy in distinguishing early from locally advanced disease, with an AUC of 0.76 in a sample of 49 patients. 23 These differences may be related to sample size (152 patients in our study versus 49 patients in the study of Fairweather et al.). 23 Interestingly, the combination of the two criteria for neoadjuvant therapy (T ≥ 2 and N+) increased the concordance, accuracy, sensitivity and specificity of EUS in selecting patients for neoadjuvant therapy, emphasizing the importance of considering T and N stage together in the analysis of EUS accuracy for gastric cancer staging. In our sample, 21 patients (13.8%) would be wrongly treated: 10 patients (6.6%) would be undertreated (would not receive the indicated preoperative therapy) and 11 patients (7.2%) would be overtreated (would receive unneeded preoperative therapy) due to inaccurate EUS staging. In the study by Fairweather et al., 10 patients (25.6%) with locally advanced disease were understaged and 2 (20%) patients with early disease were overstaged by EUS. 23 In the present study, most cases of overstaging (patients wrongly assigned to neoadjuvant therapy) were due to incorrect evaluation regarding of the depth of tumour invasion, namely, overstaging of T1 tumours. The main reason for the overstaging of T1 tumours may be related to thickening of the gastric wall due to perifocal inflammatory reaction, which is difficult to distinguish from tumoural tissue, mimicking the presence of T2 tumours.11–12,24 Regarding understaged cases, the majority were due to understaging of the nodal lymph status. The misdiagnosis of metastatic lymph nodes may be related to the presence of metastatic disease in normally size lymph nodes.11,20 This results in misdiagnosis of the N status, which can be overcome by EUS-guided fine needle aspiration of the perigastric lymph nodes; in our study, we did not perform EUS-guided fine needle aspiration of perigastric lymph nodes, which could explain our results. Another reason for inaccurate evaluation of tumour lymph nodes is related to the limited depth of the transducer and unsatisfactory visualization of distant lymph node by EUS, as EUS cannot access tissue beyond 5–6 cm. 17
In this study, we also tried to find factors that may influence the accuracy of EUS for the identification of locally advanced gastric cancer. We found that the performance of EUS for the selection of patients for neoadjuvant therapy is higher in proximal (k = 0.93, AUC = 0.96 ± 0.05, S = 99.0% and E = 90.9%) and intestinal histological subtype tumours (k = 0.77, AUC = 0.88 ± 0.04, S = 92.6% and E = 84.1%). The inferior performance of EUS in the locoregional staging for distal lesions may be justified as an inherent limitation of this image modality, since it is more difficult to obtain perpendicular images of the gastric wall in this part of the stomach. The inferior performance of EUS in staging of diffuse and mixed histological subtypes may be justified by their association with a higher rate of invasion and difficult delimitation of the tumour; an additional explanation for the low global accuracy of these subgroups seems to be the tumour behaviour, namely the presence of desmoplastic reaction (fibrous tissue reactive to the presence of neoplastic cells), which is common in gastric adenocarcinoma and leads to lower EUS staging performance in diffuse and mixed-subtype tumours. 17 Other studies have shown that the diameter and morphology (flat versus ulcerated versus vegetative) of the tumour, and ultrasonographer experience, may also influence the accuracy of EUS in gastric cancer staging; however, these were not evaluated in our study.21,26–27
Although we have focused on the accuracy of EUS to identify locally advanced gastric cancer, we have also evaluated the accuracy of EUS to stage T and N status separately. Here, we showed that EUS has an overall higher accuracy (T stage: 75.7% and N stage: 72.9%) in preoperative staging of the gastric adenocarcinoma. These results are consistent with previous literature, including a recent meta-analysis. 21 Similarly to most previous studies, in our study the concordance and overall accuracy of EUS were higher for T staging (k ranging from 0.39–0.74 and AUC ranging from 0.71–0.87) than for N staging (k ranging from 0.11–0.45 and AUC ranging from 0.56–0.73). The lower accuracy of EUS for N staging may be related to the limited depth of the transducer and unsatisfactory visualization of distant lymph nodes by EUS, as explained above. Concerning T staging, the concordance and overall accuracy of EUS was higher for the T1 stage than for other stages, particularly the T2 stage. Lower accuracy rates for the T2 stage are well documented in the literature, and may reflect the increased difficulty in delineating the margin between the muscularis propria and the subserosal connective tissue by EUS.19,22,28 Concerning N staging, the concordance and overall accuracy of EUS was higher for the N0 stage than for other stages. This may reflect the presence of metastatic disease in normal-sized lymph nodes, and the limited depth of the transducer and unsatisfactory visualization of distant lymph nodes by EUS, as explained above.11,16,18
Taken together, our data strongly suggest that, despite higher performance from EUS in patients selected for chemotherapy for gastric adenocarcinoma, EUS should still be combined with other imaging modalities to guide treatment plans. A combination of imaging methods (fine needle aspiration and computed tomography (CT) scan) seems to be an ideal modality in preoperative gastric cancer staging, as the diagnostic accuracy of imaging metastatic lymph nodes and distant metastases is greatly improved.15,17,23 This study has two main strengths: it is the first study primarily focusing on assessing the accuracy of EUS in patients with gastric adenocarcinoma selected for neoadjuvant therapy combining T and N stages, and it corresponds to one of the largest series published in the literature with 152 patients included in the final analysis. However, it has also some limitations. First, it was developed in a single academic centre with all the procedures performed by three experienced ultrasonographers, and thus results might not be applicable to other settings with less experienced ultrasonographers, due to the innate operator variability in performing EUS. Secondly, as with all retrospective and single-centre studies, selection bias is a possibility. Thirdly, due to the retrospective design of the study, ultrasonographers were not blind to the CT staging in most cases, and so EUS staging might have been influenced by CT staging. Fourthly, we did not perform EUS-guided fine needle aspiration of lymph nodes, a tool that may increase to the accuracy of nodal staging. The use of single-frequency scanning in the EUS evaluation could have contributed to the unsatisfactory accuracy of evaluating the N stage, and the absence of accuracy in comparison with the other image modality, namely CT scanning, both singly (EUS accuracy versus CT scan accuracy) and in combination (EUS plus CT scan accuracy), are other limitations of our study. The latter would be interesting to study in future investigations.
Conclusions
In summary, our results suggest that EUS is an accurate assessment tool for patients with gastric adenocarcinoma selected for neoadjuvant therapy, particularly in patients with proximal and intestinal histological subtype tumours. Nonetheless, in order to further reduce the number of misdiagnosed cases, especially concerning the N status, EUS should always be considered in combination with other tools such as CT and fine needle aspiration.
Declaration of conflicting interests
None declared
Ethics approval
The study was carried out in accordance with the Declaration of Helsinki. Ethical approval was granted by the SECVS and the CESHB on 31 October 2017.
Funding
None declared
Informed consent
Patients provided written informed consent for all the procedures and only then were they included in the database.
