Abstract
Background
Obesity is a known risk factor of colorectal cancer (CRC). However, precise interconnections between excessive body fat and CRC are still vague. Therefore, the aim of this study was to assess whether stage of CRC detected in overweight and obese individuals differs from individuals with normal body mass index (BMI). A secondary aim of this study was to elucidate whether overweight and obesity influence the overall survival in CRC.
Methods
This study was a cross-sectional analysis of 163,129 individuals who underwent screening colonoscopy performed on data from a prospectively maintained database of the Polish Colonoscopy Screening Program.
Results
Overweight and obese individuals present with a less advanced CRC in screening setting (
Conclusions
Obese screenees present with a less advanced disease in comparison to non-obese. Moreover, survival stratified by clinical stage seems to not be influenced by BMI category. Therefore, a higher proportion of early diagnosed cancers can potentially create a survival benefit in this group.
Introduction
With colorectal cancer (CRC) being one of the most common and most deadly cancers, national screening programs have been established in many countries. However, only relatively recently it has been proven that screening lowers the incidence of CRC and improves survival. 1 In Poland, asymptomatic citizens between 50 and 66 years old are eligible for participation in the Polish Colonoscopy Screening Program (PCSP) – a CRC screening program utilizing colonoscopy as primary diagnostic tool. Non-symptomatic individuals participate in opportunistic or letter-invited screening. Data available from this program create unique opportunities to perform cross-sectional population-based analyses.
Obesity is a known risk factor for higher incidence of colorectal adenomas, advanced colorectal adenomas, and CRC.2,3 Most studies assessing the influence of obesity on CRC, as well as on other diseases, employ body mass index (BMI) as an indicator of body fat and obesity. However, precise interconnections between BMI and CRC are still vague. Studies have shown that obese individuals have higher incidence of adenomas, higher incidence of CRC, and lower compliance to screening programs. 4 Moreover, quality of bowel preparation in obese individuals is inferior to that in non-obese. 5 On the other hand, adenoma detection rates (ADR) and advanced adenoma detection rates (AADR) are higher in this group, which can be both due to screening performance and mentioned changes in the incidence.6,7 Survival seems to be influenced by body weight as well – with some studies reporting inferior results in obese individuals. 8 Thus, evidence is accumulating to support the hypothesis that people with obesity are a distinct subgroup of CRC patients. This distinction includes incidence, progression rates, as well as screening and treatment characteristics. An especially interesting observation, originating from a case-control study by Choe et al., 9 is that obesity enhances the rate of normal to adenoma sequence but not adenoma to early carcinoma sequence. It is possible that dynamics of CRC formation and CRC progression are both influenced by obesity, but do not follow the same pattern of change. This could lead to an altered distribution of CRC stages in obese individuals. Therefore, the aim of this study is to assess whether the stage of CRC detected in overweight and obese individuals in a screening setting differs from individuals with normal body weight. A secondary aim of this study is to elucidate whether overweight and obesity influence the overall survival (OS) of patients diagnosed with CRC.
Material and methods
Study design
We performed a cross-sectional analysis of database records of individuals who entered the national colonoscopy screening program for CRC in Poland, from January 2007 through December 2011. The database contained demographic data, colonoscopy results, self-reported data on weight and height, and colorectal findings from 114 screening centers throughout Poland. Patients diagnosed with CRC were identified and followed for OS through the National Cancer Registry. Follow-up time was censored on December 31, 2015.
The research proposal was reviewed by the Bioethical Committee at the Maria Sklodowska-Curie Memorial Cancer Centre and Institute of Oncology on October 3, 2014 and was judged to be exempt from oversight, as data was de-identified and could not be linked to specific subject. Study protocol conforms to the 1975 Declaration of Helsinki, as reflected by abovementioned exemption by Bioethical Committee. Written informed consent was obtained from all participants entering the National Colorectal Cancer Screening Program.
Study procedures and definitions
Study procedures were in line with Polish Screening Colonoscopy Program and have been previously described in detail. 10 Patients between the ages of 50 and 66 years (40 and 66 years in the case of positive family history of cancer of any type) were offered screening. Exclusion criteria were clinical suspicion of CRC, characteristics that met the criteria for Lynch syndrome, familial adenomatous polyposis, or inflammatory bowel disease, and colonoscopy within the preceding 10 years, according to PCSP regulations.10–12 On the day of colonoscopy (before the procedure) all participants were asked to fill in an epidemiological questionnaire including data on self-reported weight and height. Screening colonoscopy procedures were previously described in detail.10–12
Cancer stage was assessed using the American Joint Committee on Cancer (AJCC) TNM scoring system after histopathological examination of specimens. 13 Thereafter, for the purpose of some of the univariable and multivariable analyses, Stage 1 and Stage 2 were pooled together as early CRC. Stage 3 and Stage 4 were consequently pooled together as advanced CRC.
BMI was calculated using weight (kg(/(height (m)) 2 and stratified according to the World Health Organization (WHO) classification. 14 First, second, and third class of obesity were pooled together to achieve appropriate power of analysis. For the clarity of the manuscript, individuals within the range of normal BMI (18.5–24.99) will be referred to as “normal,” individuals with overweight (BMI 25–29.99) will be referred to as “overweight,” and individuals with class I, II, or III obesity (BMI > 30) will be referred to as “obese.”
Study population
Between January 2007 and December 2011, 163,129 participants who met eligibility criteria were screened within the program. Of those, 48,176 had normal BMI, 70,173 were overweight, and 34,891 were obese; 1561 were diagnosed with CRC. Of those, 76 had missing data on BMI and were excluded and 295 had missing data on TNM and were excluded. An additional seven patients had BMI below 18.5 (BMI 16.65–18.37; stage 1–2) and were excluded from analysis. Finally, 1183 patients remained and were stratified into BMI, according to WHO guidelines. Procedures for achieving study populations are presented as a flowchart in Figure 1.
Flowchart of patients' inclusion into analyses.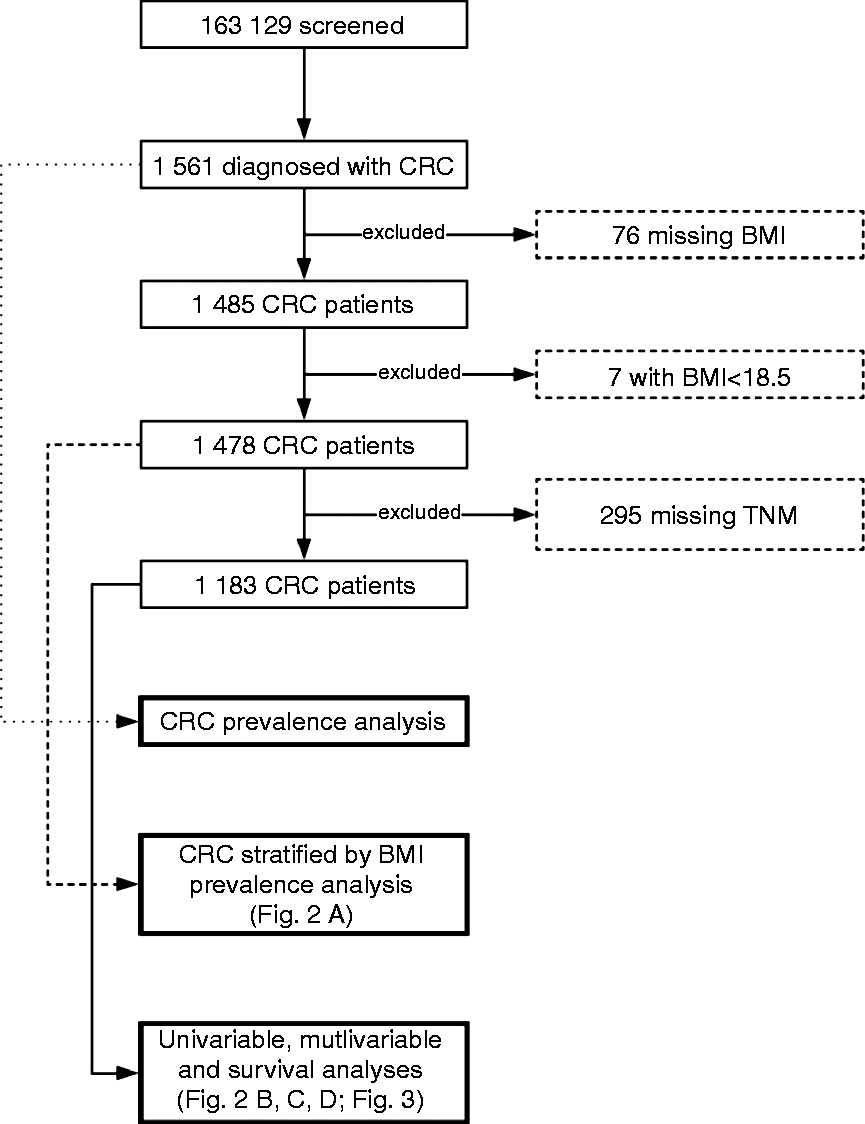
Statistical methods
Descriptive statistics were prepared with the use of contingency tables, means, and standard deviations depending on variables type. Categorical variables were compared with the use of chi-squared test and test for trend. Tests for trend were used to investigate differences between stage distribution stratified by BMI and by BMI and gender. Univariable and multivariable logistic regression models were used to investigate the associations between advanced CRC and patient's BMI, gender, age, and family history of CRC. Probability of OS stratified by stages and BMI was estimated using the Kaplan–Meier method. Survival curves were compared using log-rank test. Univariable and multivariable Cox proportional hazard models were used to estimate hazard ratios of death according to CRC stage, patient's BMI, gender, age, and family history of CRC. Forward stepwise selection at the 0.1 significance level was used for variable selection in multivariable models. BMI was included in all multivariable models regardless of its significance. All reported
Results
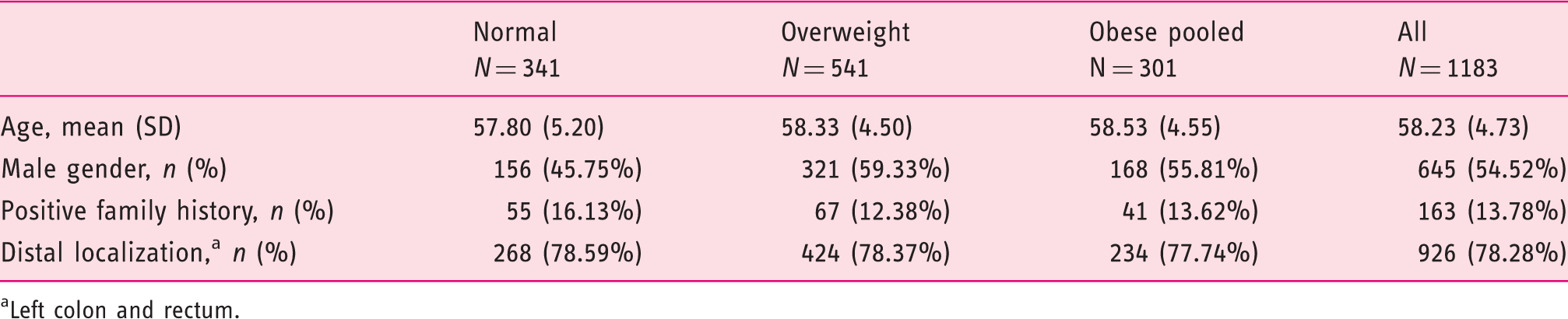
Between January 2007 and December 2011, 163,129 participants who met eligibility criteria were screened within the program. Of those, in 1561 (0.96%) CRC was diagnosed. Prevalence of CRC in the whole screened population was almost twofold higher in males than in females (855 (1.36%) vs 706 (0.70%), respectively, (a) Prevalence of CRC stratified by BMI categories with representation of distribution of stages. Stage 1 on the top, Stage 4 on the bottom. Significant Kaplan–Meier survival curve stratified by BMI and stage. Baseline characteristics stratified by BMI categories. Underweight BMI category is excluded from the table. Obesity class I, II, and III categories are pooled into “Obese pooled.” Left colon and rectum.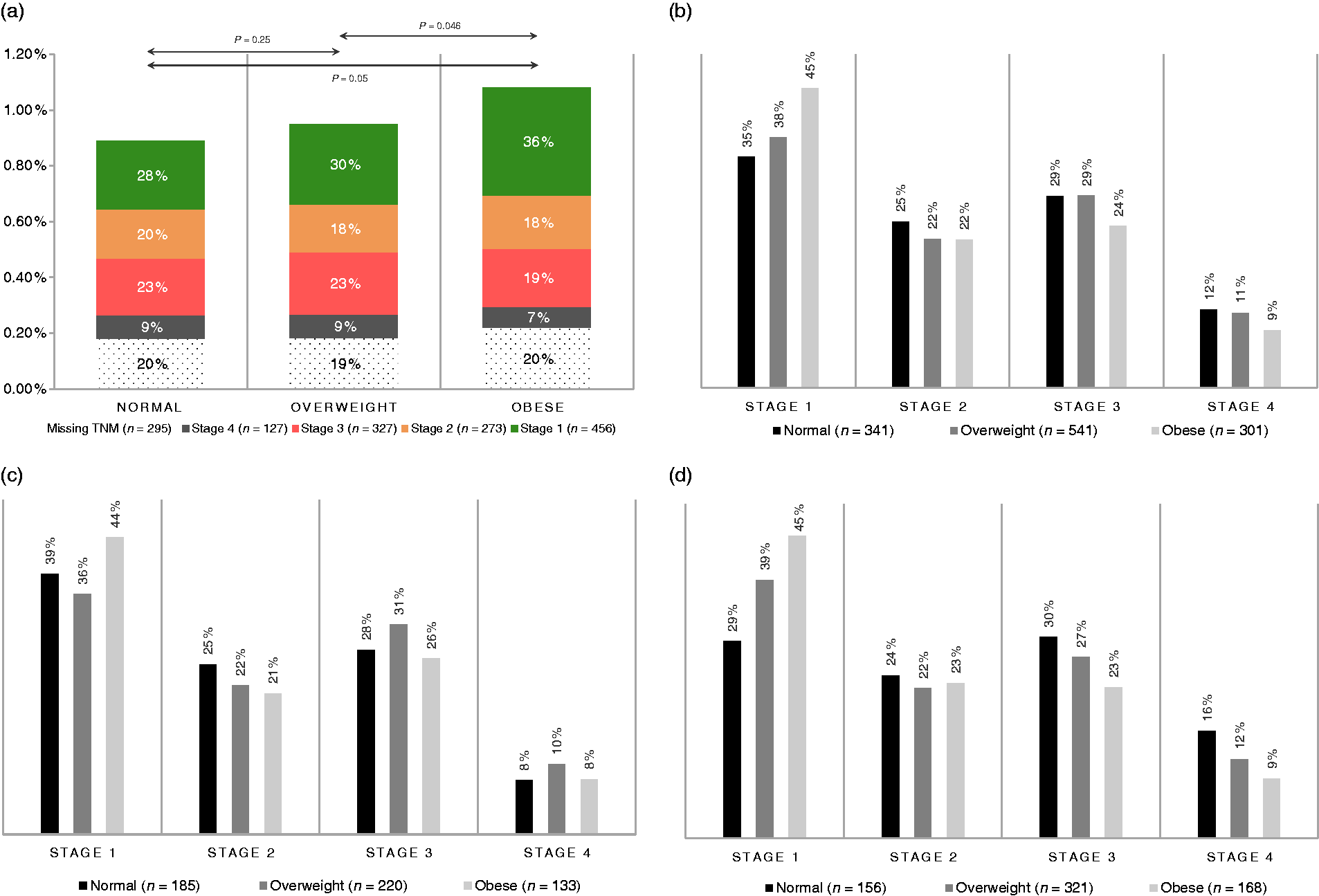
BMI and CRC stage
Figure 2(a) presents distribution of CRC prevalence with sub-analysis of stages in each BMI category. Significant
Gender subgroups analysis
There were no significant differences in the distribution of stages between genders (
Females
Furthermore, in the female subgroup there were no differences in stage distribution when stratified by BMI categories with
Males
In the male subgroup, analysis of trend for stage stratified by BMI categories revealed significant differences in distribution with higher frequency of Stage 1 CRC in group of patients with obesity, as shown on Figure 2(d) (
Risk factors of advanced CRC
Odds ratio (OR) for diagnosis of advanced CRC: univariable and multivariable model.
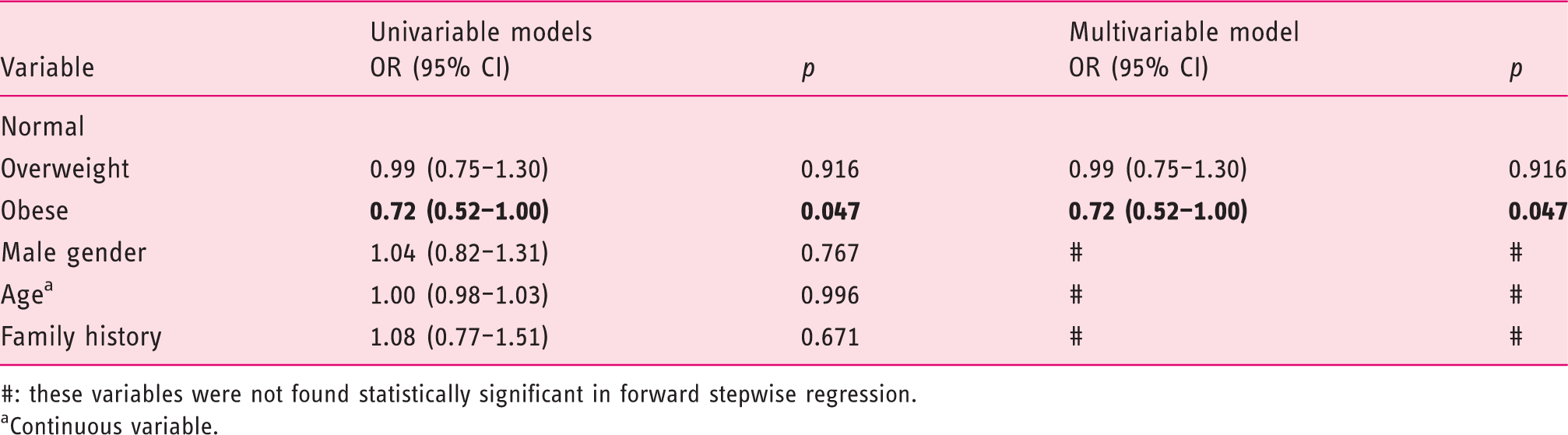
#: these variables were not found statistically significant in forward stepwise regression.
Continuous variable.
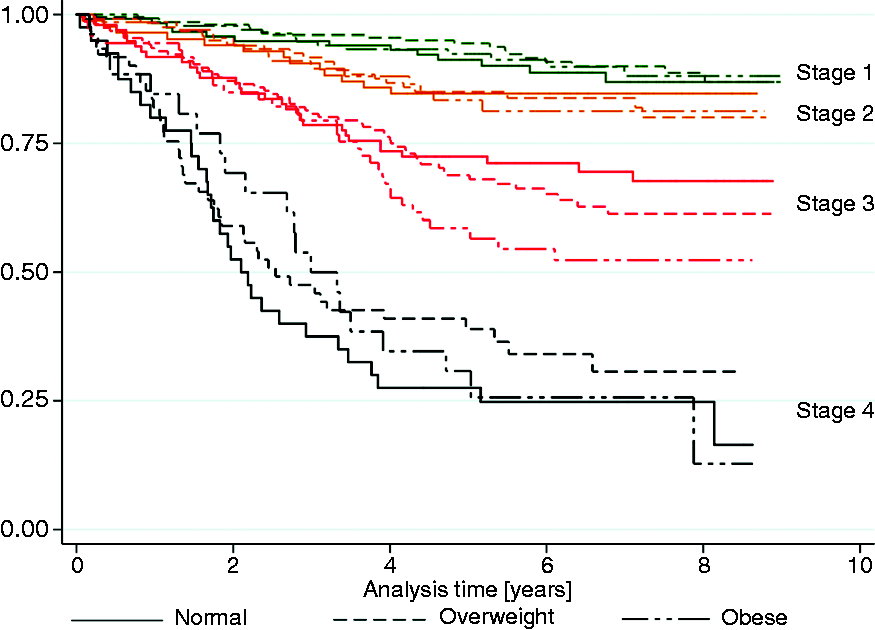
Survival analysis
Five-year survival rates for groups stratified by BMI and stage.

Univariable and multivariable analysis of overall survival (OS).
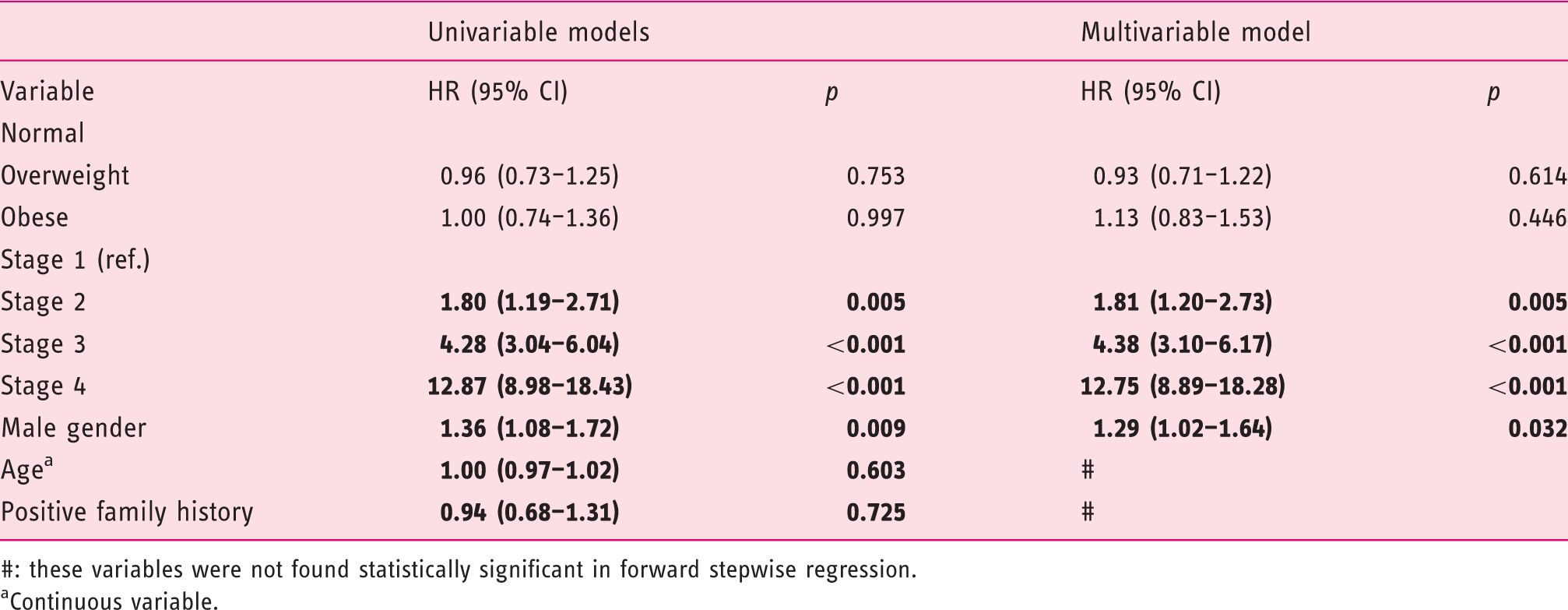
#: these variables were not found statistically significant in forward stepwise regression.
Continuous variable.
Discussion
To our knowledge this is the first study reporting increase of early stage CRC (Stage 1 and 2) in obese individuals in a screening setting. This finding is especially observed in the male subgroup. Below we will explore potential explanations of the detected phenomenon as well as limiting factors that have to be considered while interpreting the results of our study.
It is proven that obese individuals have higher risk of colorectal adenoma, advanced adenoma, and CRC.2,15,16 However, it is not fully understood whether this is due to acceleration of adenoma formation only (i.e. acceleration of normal to adenoma sequence) or acceleration of the whole carcinogenesis and progression process. The higher proportion of Stage 1 CRC observed in a screening setting could be explained by increased rate of adenoma formation without a proportional increase in CRC progression rate. Some authors observed a similar phenomenon, describing that only the rate of normal to adenoma sequence is accelerated but not the adenoma to carcinoma sequence. 9 However, enhanced formation of adenomas solely may not be a sufficient explanation for the observed phenomenon. In such a situation, we would see a higher proportion of adenoma prevalence to CRC prevalence in obese individuals. Nevertheless, there is no data demonstrating whether this proportion differs between BMI categories. The missing piece of the explanation could be a change in dynamics of CRC progression. It is possible that while obesity is a risk factor for the formation of adenomas and CRC, it does not influence the progression in the same way. This may be due to metabolic factors explored below.
The first explanation of potentially decreased progression rates may be that lymph nodes and liver in obese individuals are distinct, unfavorable microenvironments for CRC cells. Park et al. observed that in individuals with CRC visceral obesity was associated with lower rates of lymph node involvement and improved OS.
17
The same study did not demonstrate such association for BMI. This suggests that it is visceral obesity and not the BMI that actually influences epidemiology and clinical presentation of CRC. Furthermore, Murono et al. observed that hepatic steatosis is associated with lower incidence of liver metastases of CRC.
18
The authors of this study propose that fat deposition in the liver results in an unfavorable microenvironment for the invasion and growth of metastatic tumor cells and suppresses seeding and growth of cancer cells. While this was demonstrated only for the liver, and not for other organs, it has to be remembered that liver metastases contribute to almost 70% of all CRC metastases.
19
Therefore, this mechanism may substantially influence proportions of advanced CRC observed in obese patients. These explanations are in accordance with results presented in this study. In Figure 2(b) and (d) it can be noticed that overweight and obese individuals have higher proportion of Stage 1 CRC and lower proportions of Stage 3 and Stage 4 disease with significant
Another possible metabolic explanation is insulin resistance that is often caused by obesity. Yamaji et al. found that, even after adjustment for BMI, higher C-peptide serum concentration is associated with increased risk of colorectal adenoma and this effect is most potent in males. 20 These observations are in accordance with results from the present study, where discrepancies between stages of CRC were observed in males. This may be due to factors influencing the CRC carcinogenesis particularly concerning females, such as estrogen concentrations, which may exert a protective effect on CRC risk. 20
Lastly, the observed phenomenon may be elucidated by pre-diagnosis changes in body mass. Development of neoplastic disease is very energy consuming and causes loss of body mass, and finally cachexia. It is possible that observed higher frequency of Stage 1 CRC in our group, is due to the fact that individuals with higher stages of CRC lost enough weight before screening to be classified in lower BMI group. This would mean that only the least affected by the disease individuals remain in higher BMI categories. This last notion may be a part of a phenomenon called the obesity paradox, which has been previously described in numerous diseases, including cancer.
21
According to the obesity paradox, observation that obese individuals present with a less advanced cancer maybe altogether false due to a logical fallacy known as
Analysis of survival in our study did not reveal statistically significant differences between groups stratified by BMI. This suggests that BMI does not influence survival of CRC detected in screening setting. Some authors have reported that survival in obese individuals is inferior, available studies vary in methods of BMI acquisition (pre versus post diagnosis). 8 On the other hand, Aparicio et al. reported that survival in overweight men was superior in comparison to normal BMI patients. This study however acquired BMI prior to palliative chemotherapy. 22 In our group, BMI was acquired before CRC diagnosis, and therefore was less susceptible to change occurring due to cancer. This may explain why survival in our cohort was not influenced by BMI. In the present study the only factors that were predictors of inferior survival were: higher stage of the disease and male gender.
The main strength of our study is a prospectively maintained database that was used to perform analysis. It contains data on every screening colonoscopy performed in Poland between 2007 and 2011. This enabled us to gather a substantial group of 163,129 individuals. Furthermore, screening setting of diagnosis creates unique opportunities. Firstly, it enabled gathering data on BMI that was possibly the least biased by obesity paradox. Secondly, only asymptomatic individuals are eligible for screening, and therefore analysis of a homogeneous group was possible. It has been reported that obese individuals tend to not attend screening, 23 which may cause underrepresentation of obese in study sample. However, comparison of data from epidemiological study WOBASZ II on BMI structure of polish society to data from our study confirms good compliance of obese individuals in the study sample. In our study 46% of diagnosed patients were overweight and 26% were obese, while in WOBASZ II overweight rates were between 34–45% and obesity rates were 26–37%. 24
There are several limiting factors that have to be considered when analyzing results of our study. Those especially include selection bias (healthy screenee bias), response bias (BMI was based on a self-reported survey), as well as missing data on BMI and CRC stage. To explore impact of those limitations we have performed post-hoc analyses which are described and discussed in detail in appendix. These analyses shown that despite mentioned limitations conclusions can be drawn and our study adds to the current state of knowledge.
Conclusions
Our study provides new insight into the relationship between obesity and CRC – that screenees, especially males, present with a less advanced disease. Moreover, survival stratified by clinical stage seems to not be influenced by BMI category. More studies are warranted to explore possible causes of this phenomenon. Especially studies exploring dynamics of CRC development and progression are needed. However, regardless of origin of the present observation it is already clear that obese individuals especially benefit from CRC screening. Higher proportion of early diagnosed cancers can potentially create a survival benefit in this group. This should be taken into consideration when planning public health campaigns.
Supplemental Material
Supplemental material for Clinical stages of colorectal cancer diagnosed in obese and overweight individuals in the Polish Colonoscopy Screening Program
Supplemental Material for Clinical stages of colorectal cancer diagnosed in obese and overweight individuals in the Polish Colonoscopy Screening Program by Piotr Spychalski, Jarek Kobiela, Paulina Wieszczy, Michał F Kamiński and Jarosław Reguła in United European Gastroenterology Journal
Footnotes
Declaration of Conflicting Interests
The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
Waived from ethics board oversight.
Informed consent
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
