Abstract
Background
Endoscopic resection (ER) with or without ablation is the first choice treatment for early Barrett’s neoplasia. Adequate staging is important to assure a good oncological outcome.
Objective
The purpose of this study was to investigate the diagnostic accuracy of pre-operative biopsies in patients who undergo ER for high-grade dysplasia (HGD) or early adenocarcinoma (EAC) in Barrett’s oesophagus (BE) and the cardia.
Methods
Between November 2005–May 2012, 142 ERs performed in 137 patients were obtained. Worst pre-ER and ER histology were compared. Upgrading/downgrading was defined as any more/less severe histological grading on the ER specimen.
Results
The accuracy of pre-ER biopsies in predicting final histology was 61%. ER changed the pre-treatment diagnosis in 55 of the 142 procedures (39%) with downgrading in 23 cases (16%) and upgrading from HGD to T1a or T1b in 32 cases (23%). In the majority of upgraded cases, a visible lesion according to the Paris classification could be detected (26/32, 81%).
Conclusion
The diagnostic accuracy of oesophageal biopsies alone in predicting final pathology in Barrett’s dysplasia is only 61%. The majority of upgraded lesions are detectable. When ablative therapy is considered in HGD Barrett's dysplasia a meticulous inspection for and removal of all small visible lesions is mandatory.
Introduction
The incidence of oesophageal adenocarcinoma (AC) has risen dramatically over the recent decades. 1 Barrett's metaplasia is a major risk factor in the development of AC, arising from intestinal metaplasia through a cascade of low-grade dysplasia (LGD) and high-grade dysplasia (HGD). Progression rates between 0.12–0.5% per year have been described. The outcome of advanced oesophageal AC is poor but uncontrolled surgical data suggest that endoscopic surveillance of Barrett’s oesophagus (BE) results in a better survival for patients when diagnosed with AC while under surveillance. Despite the progress in imaging techniques, the golden standard for diagnosis and risk stratification remains histology, obtained through four-quadrant biopsies every 2 cm and of endoscopic suspicious lesions.2–4 It has already been shown that greater interobserver agreement exists for the histological assessment of endoscopic resection (ER) specimens in comparison to biopsy samples. 5
Endoluminal treatment is now accepted as a first-line treatment option for early Barrett’s neoplasia that is limited to the mucosa, well to moderately differentiated and free of lymphovascular invasion.6–8 These criteria for patient selection are crucial to guarantee the excellent outcome of endoluminal treatment. 9 Recently, the therapeutic armamentarium has extended considerably with the introduction of new ablation techniques. Therapeutic algorithms are often based on the histological assessment of biopsies. 5
Radiofrequency ablation (RFA) has now been well established as a treatment option for flat HGD and LGD in Barrett's dysplasia. It is important to emphasise that in all controlled trials visible lesions were resected first, and that residual cancer was an exclusion criterion for RFA (EURO-II trial, surf trial, EURO I trial).10–12 Therefore a holistic endoluminal treatment combines ER with subsequent RFA. Indeed in contrast to ER, histological assessment of the treated area is not possible after RFA. Therefore, the major challenge in treating early Barrett’s neoplasia lies in detecting and excluding early AC (EAC) prior and during RFA treatment.
The aim of our study was to investigate the accuracy of pre-operative biopsies in patients who undergo ER for HGD or EAC in BE and the cardia, to predict final histology and to assess the relation to the endoscopic appearance of the targeted lesions.
Materials and methods
Between November 2005–May 2012, all patients undergoing ER for histologically confirmed HGD, EAC or endoscopic suspicious lesions in Barrett's metaplasia were prospectively enrolled in this database. This is a retrospective analysis of prospectively collected single centre data. A total of 176 consecutive ERs were performed in 149 patients in our institute. Six procedures were excluded from further analysis, due to non-lifting of the mucosa.
Synchronic lesions were defined as HGD/EAC found within six months after a previous ER in the same patient. In this case both ERs were regarded as a single diagnostic staging procedure. In case the interval was longer than six months and new pre-ER histology was available prior to a new resection, both ERs were regarded as separate procedures and included in this study. Therefore, a total of 142 procedures were analysed of which 121 were performed in a BE and 21 at the cardia.
Prior to endoluminal therapy, all patients were assessed endoscopically and biopsies were taken from suspicious sites in our centre or in referring hospitals. All pre-ER biopsies and ER specimens were reviewed by at least one expert pathologist, as part of our routine clinical practice. At endoscopy, visible lesions were classified according to the Paris classification system. 13 Patients with observed ulcerated lesions (0–III) were excluded from endoluminal treatment.
Pre-ER endoscopies were performed with high-resolution or high definition video endoscopes in combination with computer enhanced chromoendoscopy (Narrow Band Imaging, i-scan, Flexible spectral Imaging Color Enhancement) and/or chromo-endoscopy with methylene blue or acetic acid.
Oesophageal biopsies were taken using Boston Scientific Radial Jaw TM3 forceps. ER was performed using multi-band mucosectomy or the ER cap technique. In multi-band mucosectomy, the targeted lesion is sucked into the banding chamber and a ligation band is deployed. The resulting pseudopolyp is then resected by snare polypectomy. 14 In the ER cap technique, the mucosa is elevated by submucosal saline injection, and is sucked into the cap fitted to the tip of the endoscope. Next, a snare transects the captured tissue. 15
Small lesions were resected ‘en bloc’. In case of larger lesions piecemeal resection was performed. Patients were hospitalised for 24 h. They were advised to take a soft diet for the first week. A double dose of a proton pump inhibitor was prescribed for eight weeks (pantoprazole or esomeprazole 40 mg twice day). Follow-up endoscopy was repeated at three, six, nine and 12 months after ER and then yearly with biopsies from the ER site and four-quadrant biopsies from the residual BE, if no additional ablation was performed.
All ER specimens were retrieved and sent to the pathology lab in saline within 30 min after the ER procedure. Subsequently, they were marked with Indian ink along their lateral and deep margins and were then stretched and pinned to a wax block, photographed and fixed in 10% neutral buffered formalin for 24 h (Figure 1).
Endoscopic resection (ER) specimen marked with Indian ink along the lateral and deep margins, stretched and pinned to a wax block.
After fixation, the specimens were cut in parallel slices of 2 mm thickness according to their long axis. These slices were further processed routinely for histology. Serial 5 µm-thick sections of each slice were stained with haematoxylin-eosin for microscopic investigation. When indicated, step sections and/or immunohistochemical stains (pan-cytokeratin, CD31, alpha-smooth muscle actin and p53) were performed. The lesions were classified according to the Vienna classification system. 16 Following World Health Organisation (WHO) guidelines, we defined HGD as architectural and cytological changes confined to the Barrett’s glands, in T1a invasion is confined to the mucosal lamina propria, and in T1b it extended into the submucosa. 17
All data were prospectively collected in a database. Worst pre-ER and ER histology were compared. Diagnostic discrepancies were considered as ‘upgrade' when pre-ER biopsy showed lower grading in comparison to the ER specimen. ‘Downgraded lesions’ showed higher grading on pre-ER biopsy than in the ER specimen. Concordant specimens were considered as ‘correct’.
All patients signed informed consent for endoscopic therapy and prospective data collection, prior to the procedure. The study was approved by the local Ethical Committee, University Hospitals Leuven (s52432).
Results
Study population
A total of 142 ERs (including 21 at the cardia and 121 in BE) were performed in 137 patients. (108 men and 29 women; median age: 66 years; standard error of the mean 0.925). 120 patients underwent a single ER session for the eradication of neoplasia. Of these, 83 were piecemeal resections.
Four patients developed metachronic lesions (three patients with two lesions, one patient with three lesions) and underwent an ER for each lesion. Thirteen patients underwent multiple resections to eradicate HGD or EAC.
Accuracy of pre-ER biopsies to predict final histology
Histology.
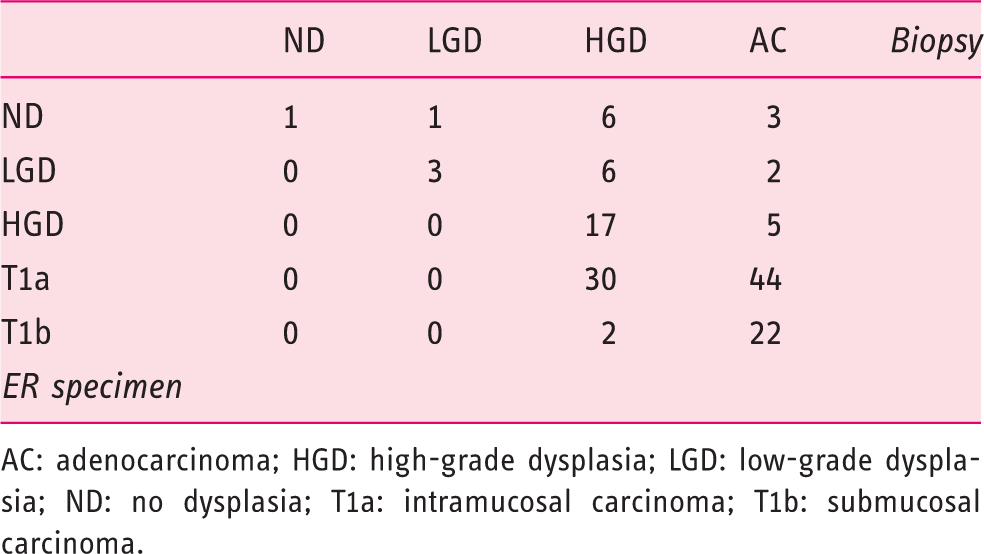
AC: adenocarcinoma; HGD: high-grade dysplasia; LGD: low-grade dysplasia; ND: no dysplasia; T1a: intramucosal carcinoma; T1b: submucosal carcinoma.
The accuracy of pre-ER biopsies in predicting final histology after ER was 61%. ER histology was different from pre-ER biopsies in 55 of the 142 procedures (39%) with downgrading in 23 cases (16%) and upgrading from HGD to T1a or T1b in 32 cases (23%) (Table 1).
Eighty-one percent (26/32) of the upgraded lesions were endoscopically visible with the following Paris classification: three 0–Is lesions, 16 0–IIa lesions, two 0–IIc lesions and five mixed lesions with visible component. Invisible or flat lesions (Paris 0–IIb) were found in six of the upgraded cases.
Sixteen pre-ER biopsies revealed an uncertain pathology and were classified as Vienna class 4.3, indicating the presence of HGD with suspicion of carcinoma. Interestingly, after ER a definitive histological classification was possible for all lesions: five lesions were classified as HGD and 11 as T1a. No diagnosis of Vienna class 4.3 was made after ER.
Procedure-related complications and follow-up
In four patients, ER was complicated with intraprocedural bleeding requiring additional treatment besides electrocoagulation with need for adrenaline injection and/or haemostatic clip placement. No perforations or delayed bleeding occurred. In 24 patients a final diagnosis of T1b was made after ER. Seventeen of these underwent oesophagectomy. In five patients a conservative approach was followed because of co-morbidity or advanced age. In two of these, favourable histological characteristics were present (no lymph or vascular infiltration and minimal submucosal invasion).
One patient obtained complete remission after ER and the second patient underwent subsequent RFA obtaining complete remission of Barrett’s disease.
For 91 patients follow-up data of at least 12 months after remission were available. Remission rates for dysplasia were 71/91 (78%). Seventeen patients have died. The cause was disease-related in four patients: one patient refused surgery and ER was performed in the remaining three patients without curative intent. Two patients died from cardiac disease., two patients suffered from pulmonary infections with respiratory insufficiency. One patient died after a complicated oesophagectomy. The cause of mortality is unknown in three patients.
Discussion
Studies with available pre- and post-endoscopic resection (ER) histology (2000–2015)
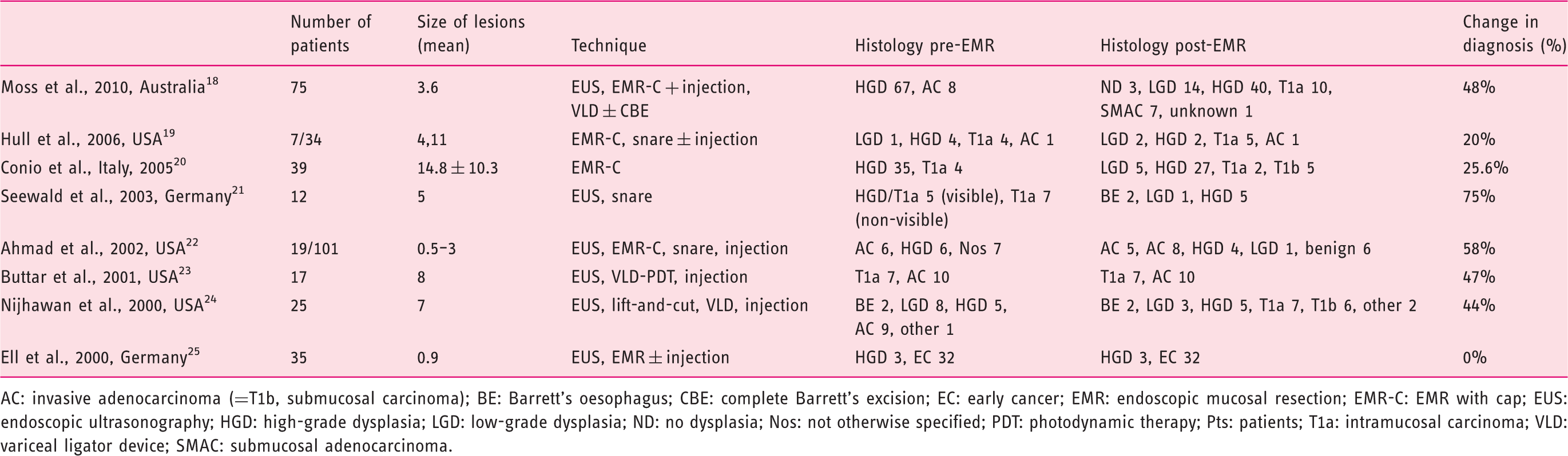
AC: invasive adenocarcinoma (=T1b, submucosal carcinoma); BE: Barrett’s oesophagus; CBE: complete Barrett’s excision; EC: early cancer; EMR: endoscopic mucosal resection; EMR-C: EMR with cap; EUS: endoscopic ultrasonography; HGD: high-grade dysplasia; LGD: low-grade dysplasia; ND: no dysplasia; Nos: not otherwise specified; PDT: photodynamic therapy; Pts: patients; T1a: intramucosal carcinoma; VLD: variceal ligator device; SMAC: submucosal adenocarcinoma.
These findings underline the importance of performing a staging ER to obtain a correct T-staging and provide the correct oncological treatment for the patient. Indeed, in the era of radiofrequency ablation, this issue is even more important. RFA is generally indicated for HGD or LGD (EURO II trial, surf trial, EURO I trial).10–12 However, if the indication is based only on biopsies, the risk of ablating cancer and providing the wrong treatment to the patient is considerable. In fact the high success rate reported in the well-controlled RFA trials, is to a large extend attributable to the strict patient selection, with ER being one of the hallmarks in the work-up of the patients.
Interobserver agreement for the diagnosis of HGD and LGD is suboptimal.26,27 However, interobserver variability between pathologists has been shown to be better for ER specimens in comparison to biopsies of BE. 5 In our study it was remarkable that ER allowed us to properly classify all lesions with a Vienna 4.3 classification on biopsy.
In a study by Hull et al. describing mainly gastric ERs (31 of 41 ERs), a histological upgrade after ER was found in 14 out of 16 discordant cases. In addition, the size of lesions and extent of biopsy sampling was evaluated. Discrepancies between the specimens occurred in larger lesions (>10 mm) with less extensive pre-ER biopsy sampling. 19 Multiple biopsies carry the potential risk of compromising and hampering subsequent ER due to fibrosis. Therefore the number of biopsies of visible lesions was kept to a strict minimum. In fact, if patients were referred for treatment and a visible lesion was detected during work-up, previous biopsies were revised by at least one of our expert pathologists for confirmation. So, to some extent, the limited number of biopsies may account for the low diagnostic accuracy. In view of the fact that our study confirms that ER is a low-risk procedure to treat HGD and EAC the abovementioned issue becomes redundant. ER safely provides tissue specimens for accurate histological assessment rendering multiple biopsies redundant.
Upgraded lesions in this study were mainly endoscopically visible. Identifying these lesions as such probably constitutes the biggest challenge in the endoluminal treatment of Barrett’s associated neoplasia. New techniques such as high-definition endoscopy with narrow band imaging may aid in the detection of visible lesions. 28 The most appealing technique that may serve as a red flag technique is the use of acetic acid chromoendoscopy. 29 Recently, acetic acid chromoendoscopy has been shown to increase the detection of neoplasia by 14 times. The authors calculated that loss of aceto-whitening before the cut-off of 142 s predicts the presence of neoplasia with a sensitivity of 98% and specificity of 84%. 30
In conclusion, our study showed only moderate diagnostic accuracy of biopsies to predict final T stage in early Barrett’s neoplasia. The study underlines the importance of a staging ER of the smallest visible lesion prior to ablation and confirms that the presence of a visible lesion is a contraindication for primary RFA.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
